
Spectroscopic properties of vacancies and trap levels in
Lu3Al5O12:Ce3+ crystals
ZHONG Jiu-ping(钟玖平)1, LIANG Hong-bin(梁宏斌)1, SU Qiang(苏 锵)1,
ZHOU Jian-ying(周建英)1, WANG Ji-yang(王继扬)2
1. State Key Laboratory of Optoelectronic Materials and Technologies, Sun Yat-sen University,
Guangzhou 510275, China;
2. State Key Laboratory of Crystal Materials, Shandong University, Ji’nan 250100, China
Received 10 August 2009; accepted 15 September 2009
Abstract: In order to study the spectroscopic properties of vacancies and trap levels in Lu3Al5O12?Ce3+ (LuAG?Ce3+) crystal, the <111>-oriented LuAG?Ce3+ crystal grown in pure nitrogen atmosphere by Czochralski method was annealed in oxidizing atmosphere (air) and reducing atmosphere (H2+N2), respectively. The excitation and emission spectra of LuAG?Ce3+ crystal after different thermal annealing treatments were measured in the temperature range of 8-450 K, and the thermally stimulated luminescence curves of LuAG?Ce3+ crystal were characterized. It is found that the oxygen vacancies in LuAG?Ce3+ crystal are effectively eliminated through the annealing treatment in air and four trap levels are observed in as-grown LuAG?Ce3+ crystal with temperature position peaking at 110, 210, 325 and 475 ℃, respectively.
Key words: Lu3Al5O12?Ce3+ crystal; oxygen vacancy; thermoluminescence
1 Introduction
Scintillator material is a kind of optical material converting the energy of high energy radiation (X-ray, γ-ray) or particle (proton, electron, neutron, α-particle, etc.) into UV/visible photons, which are easily detected with photosensitive devices such as photomultiplier tubes (PMTs), charge-coupled devices (CCDs), or photodiodes (for example, silicon diodes). Inorganic scintillator materials have increasing scientific and commercial applications in diverse fields such as medical imaging, safety inspection, industrial measuring systems, and calorimeters in high-energy physics. The search for appropriate inorganic scintillator materials has been stimulated over a period of more than 100 years and a great number of inorganic scintillator materials have been found and some of the materials were successfully industrialized[1].
Lutetium-aluminium garnet (Lu3Al5O12, LuAG) crystal, due to its high density (6.695 g/cm3), shock resistivity, non-hygroscopicity, chemical radiation stability, and large absorption coefficient for high energy radiation[2], is known to be quite promising host crystal for scintillator materials. Trivalent cerium activated lutetium-aluminium garnet (LuAG?Ce3+) single crystal is effective inorganic scintillator, which shows extremely high mechanical stability, high light yield and short luminescence decay time, resulting from the allowed 5d→4f transitions within Ce3+ ions[3]. However, some LuAG?Ce3+ crystals prepared by Czochralski method often show poor luminescence efficiency. Usually, the quenching centers and structure defects formed during crystal growth are regarded to be the main factors for the luminescence efficiency restraining[4]. Therefore, the thermal annealing treatment is often used as an effective method to study the effects of vacancies or defects on the luminescence properties and to improve the scintillation properties of inorganic scintillator single crystal[5-6]. In the present work, the spectroscopic properties of oxygen vacancies in LuAG?Ce3+ crystal and its thermo- luminescence properties were investigated through different thermal annealing treatments.
2 Experimental
The LuAG?Ce3+ single crystal was grown from melt by Czochralski method using iridium crucible under highly pure nitrogen (99.999%) atmosphere. The initial cerium content in the melt was 0.5% (molar fraction) with respect to total rare-earth sites. The detailed process about single crystal growth was described in Ref.[7]. The samples with dimensions of 2.0 mm×2.0 mm×10.0 mm were cut from the LuAG?Ce3+ boule and both surfaces perpendicular to the <111> direction were polished.
Two sets of annealing procedure were used to treat the LuAG?Ce3+ single crystal samples in the present study. In the first procedure, the crystal samples were continuously heated at 1 550 ℃ for 6 h in a reducing mixture gas (10%H2+90%N2) flux and cooled to room temperature. The second annealing treatment was sequentially performed at 1 200 ℃ for 40 h in oxidizing atmosphere (air) and cooled to room temperature. Therefore, after aforementioned two annealing procedures, there were three sets of LuAG?Ce3+ crystal samples in present study: as-grown crystal, crystal annealed in H2+N2 and crystal annealed in air.
The excitation and emission spectra of above three sets of LuAG?Ce3+ single crystal samples were determined at different temperatures on an Edinburgh FLS 920 combined fluorescence lifetime and steady state spectrometer, which was equipped with a CTI-Cryogenics temperature controlling system (Helix Technology Corporation, USA). A 450 W xenon lamp was used as the excitation source for the UV-visible spectra and a blue-sensitive photomultiplier tube (R1527 PMT) was used for the emission spectra recording.
The three-dimensionally thermally stimulated luminescence curves (3D TSL, the thermoluminescence as a function of both temperature and wavelength) of LuAG?Ce3+ crystal were determined with a 3D thermal spectrometer made by the Division of Nuclear Physics and Technology in Sun Yat-sen University, China. Prior to each 3D TSL experiment, the samples were exposed to 90Sr β-ray for 30 min and kept in black box for 24 h. The TSL signal was detected in the wavelength region of 300-800 nm with a heating rate of 1 ℃/s from 30 to 500 ℃.
3 Results and discussion
3.1 Photoluminescence properties
The excitation and emission spectra of all three sets of LuAG?Ce3+ single crystal samples were measured at 8, 110, 210, 310, and 450 K, respectively. Fig.1(a) shows the excitation spectra of as-grown LuAG?Ce3+ crystal. Two broad excitation bands at around 345 nm and 448 nm are characteristic absorption of Ce3+ ions doped in LuAG host due to the transition from ground state 4f1 to excited 5d1 configuration. Fig.1(b) shows the emission spectra of as-grown LuAG?Ce3+ crystal under 345 nm excitation. The broad band from 450 nm to 640 nm is the typical double emission of Ce3+ ions due to the allowed transition from the lowest crystal field component of the excited 5d1 configuration to the two levels of the ground states, 2F5/2 and 2F7/2.
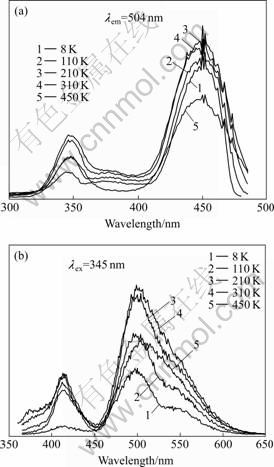
Fig.1 Luminescence spectra of as-grown LuAG?Ce3+ crystal: (a) Excitation spectra; (b) Emission spectra
In order to obtain clear double emission bands of Ce3+ ions doped in LuAG host, the emission spectrum of as-grown LuAG?Ce3+ crystal at low temperature was measured under 448 nm excitation, as shown in Fig.2. The broad emission band from 450 nm to 640 nm can be well fitted by two Gaussian bands peaking at 500 and 555 nm, respectively. Then, it can be calculated that two ground levels 2F5/2 and 2F7/2 within Ce3+ ions are separated by 1 950 cm-1 due to spin-orbit coupling in LuAG host.
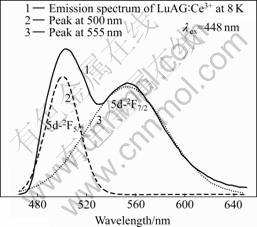
Fig.2 Emission spectrum of as-grown LuAG?Ce3+ crystal at 8 K
Besides the Ce3+ double emission, there is another broad emission band from 375 nm to 450 nm in Fig. 1(b). In order to ascribe this emission band, the excitation spectrum of as-grown LuAG?Ce3+ crystal monitoring at 415 nm was measured, as shown in Fig.3(a). From Fig.3(a), a maximum position at 373 nm can be observed. And under 373 nm excitation, two broad emission bands peaking at 415 and 475 nm are obtained, as shown in Fig.3(b). These two emission bands are obviously different from the emission band of Ce3+ ions shown in Fig. 2. In Ref.[7], the excitation band peaking at 373 nm was ascribed to the absorption of oxygen vacancies formed during crystal growth in pure nitrogen atmosphere. Herein, these two broad emission bands peaking at 415 and 475 nm are assumed to originate from oxygen vacancies.
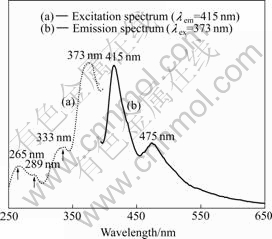
Fig.3 Excitation and emission spectra of oxygen vacancies in as-grown LuAG?Ce3+ crystal
The excitation and emission spectra of LuAG?Ce3+ crystal annealed in reducing atmosphere are shown in Fig.4. Compared with the excitation spectra of as-grown LuAG?Ce3+ crystal in Fig.1(a), the absorption band at around 373 nm and the emission band peaking at 415 nm are more obvious when LuAG?Ce3+ crystal is thermally treated in reducing atmosphere. And the emission band peaking at 415 nm significantly decreases as the temperature increases.
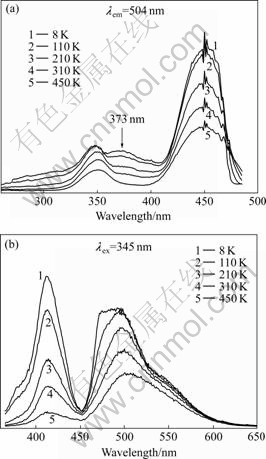
Fig.4 Luminescence spectra of LuAG?Ce3+ crystal annealed in reducing atmosphere: (a) Excitation spectra; (b) Emission spectra
Fig.5 shows the Gaussian fitting spectra of LuAG?Ce3+ crystal annealed in reducing atmosphere in the wavelength range of 450-630 nm measured at 8 K. Besides the typical 5d→ 2F5/2 and 2F7/2 transition bands of Ce3+ ions, there is a small band peaking at 475 nm. This small band is ascribed to the emission of oxygen vacancies, which is agreement with the emission spectrum shown in Fig.3(b).
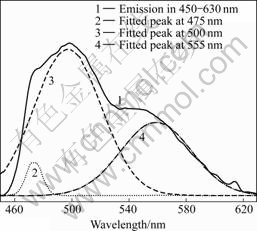
Fig.5 Gaussian fitting spectra of LuAG?Ce3+ crystal annealed in reducing atmosphere in wavelength range of 450-630 nm
The excitation and emission spectra of LuAG?Ce3+ crystal annealed in air are shown in Fig.6. Both the excitation band at around 373 nm and the emission band at 415 nm disappear completely, which confirms that the emission bands at 415 and 475 nm in as-grown LuAG?Ce3+ crystal and the crystal annealed in reducing atmosphere come from oxygen vacancies, because the oxygen vacancies are eliminated when LuAG?Ce3+ crystal is annealed in air. And the oxygen vacancies compete with Ce3+ ions in the processes of energy transfer from the host lattice, which has a negative influence on the scintillation characteristics of LuAG?Ce3+ crystals.
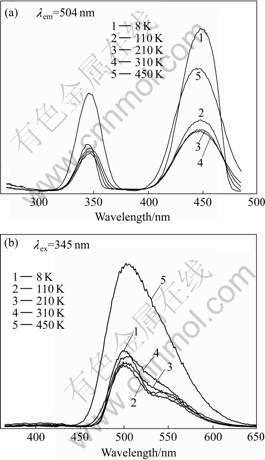
Fig.6 Luminescence spectra of LuAG?Ce3+ crystal annealed in air: (a) Excitation spectra; (b) Emission spectra
By comparing the excitation spectra in Fig.6(a) with those in Fig.1(a) and Fig.4(a), it can be concluded that the sharp peaks on the shoulder of the excitation band of Ce3+ in the wavelength range of 440-470 nm must be related to the oxygen vacancies, because these superposed components also disappear when the LuAG?Ce3+ crystal sample is treated under oxidizing atmosphere at 1 200 ℃ for 40 h.
3.2 Thermoluminescence properties
Thermoluminescence is a useful tool for studying the role of charge carriers in the scintillation process. It has been proposed that deep traps, revealed by high-temperature thermo-luminescence (measured at temperatures higher than 300 K), can reduce the scintillation light yield[8-9], and shallow traps are usually responsible for slow components in scintillation decays[10-11]. The model in which the charge carrier traps actively participate in the scintillation mechanism was first proposed by WOJTOWICZ et al[12] in the case of the well known scintillator materials LuAP?Ce3+ (LuAlO3?Ce3+) and YAP?Ce3+ (YAlO3?Ce3+). To further investigate the effects of annealing treatments on the luminescence properties of LuAG?Ce3+ crystal, the point defects acting as trap levels were characterized on present three sets of LuAG?Ce3+ crystals by 3D TSL measurements.
Fig.7 shows the 3D thermoluminescence spectra and their corresponding contour plots of the LuAG?Ce3+ crystal. It is obvious that the spectral composition of all peaks in the 3D TSL curves is governed by the characteristic emission from 450 to 640 nm related to the radiative 5d→ 4f transition of Ce3+ doped in LuAG host. So, it can be ascertained that the Ce3+ ions are the recombination centers for the detrapped charge carriers and that carriers thermally freed from the traps undergo delocalization to the conduction band before their radiative recombination. Fig.7(a2) shows the contour plot of as-grown LuAG?Ce3+ crystal relative to Fig.7(a1). The contour plot is dominated by four TSL components which are applicable for the traps corresponding to temperature position peaking at 110, 210, 325 and 475 ℃, respectively. But after annealing in a reducing mixture gas (10%H2+90%N2) flux for 6 h at 1 550 ℃, the TSL peak at around 200 ℃ disappears, as shown in Fig.7(b2), and the component locating at around 110 ℃ becomes weaker than that of as-grown LuAG?Ce3+ crystal. Whereas, from Fig.7(c2), it can be found that the component at around 110 ℃ still remains in the LuAG?Ce3+ crystal annealed in air, although three components peaking at 110, 210 and 475 ℃ become much weaker than those of as-grown LuAG?Ce3+ crystal.
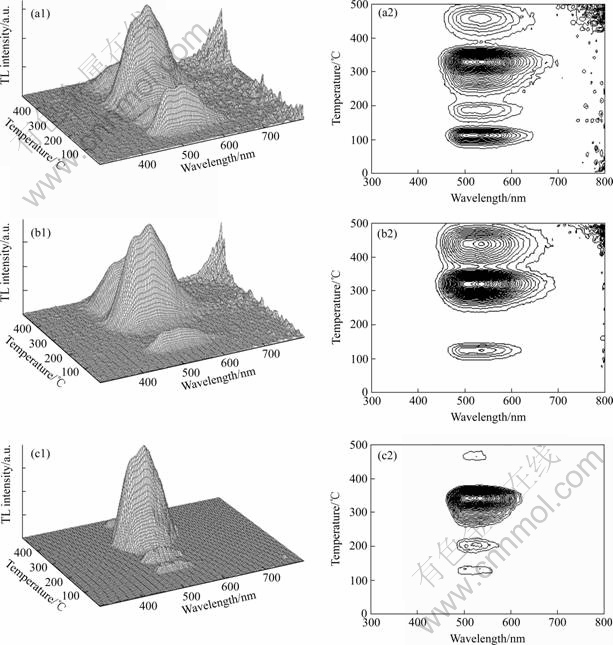
Fig.7 3D thermo-luminescence spectra and corresponding contour plots of LuAG?Ce3+ crystal: (a) As-grown; (b) Annealed in H2+N2; (c) Annealed in air
Due to the very good purity of the raw materials used to grow the LuAG?Ce3+ crystal samples, an intrinsic nature of the defects responsible for such localized trap levels can be suggested. Such a deep trap is surely harmful for the intended application of the crystal as scintillator, since it can trap carriers in a stable way so competing with prompt radiative recombination pathway. And these deep traps are mostly the reason for the radiation damage phenomena and coloring of scintillators causing the long term instabilities of their optical parameters[13]. The physical origin of these traps still remains unknown, therefore additional studies are needed to identify them.
4 Conclusions
1) The oxygen vacancies are formed during crystal growth and the oxygen vacancies in LuAG?Ce3+ crystal can be effectively eliminated through the thermal annealing treatment in air.
2) The TSL measurements performed on present three sets of LuAG?Ce3+ crystal samples prove the existence of several localized trap levels with temperature position peaking at 110, 210, 325 and 475 ℃ in LuAG?Ce3+ crystals grown by Czochralski method in pure nitrogen atmosphere.
3) The emission of oxygen vacancies in LuAG?Ce3+ single crystals indicates a strong competition between the vacancies and Ce3+ ions in the energy transfer processes from the host lattice and points to a negative effect on the scintillation characteristics of these materials.
4) The luminescence mechanism of oxygen vacancies and the physical origin of the traps in LuAG?Ce3+ crystals still remains unknown. The effect of thermal treatments should be further verified in the future, in order to eliminate the oxygen vacancies and intrinsic point defects in optimizing scintillator crystals.
References
[1] WEBER M J. Inorganic scintillators: Today and Tomorrow [J]. J Lumin, 2002, 100: 35-45.
[2] KRASNIKOV A, SAVIKHINA T, ZAZUBOVICH S, NIKLB M, MARES J A, BLAZEK K, NEJEZCHLEB K. Luminescence and defects creation in Ce3+-doped aluminium and lutetium perovskites and garnets [J]. Nucl Instrum Methods A, 2005, 537:130-133.
[3] MARES J A, NIKL M, BEITLEROVA A, SOLOVIEVA N, D’AMBROSIO C, BLAZEK K, MALY P, NEJEZCHLEB K, FABENI P, PAZZI G P. Ce3+-doped scintillators: Status and properties of (Y, Lu) aluminium perovskites and garnets [J]. Nucl Instrum Methods A, 2005, 537: 271-275.
[4] BLAZEK K, KRASNIKOV A., NEJEZCHLEB K, NIKL M, SAVIKHINA T, ZAZUBOBICH S. Luminescence and defects creation in Ce3+-doped Lu3Al5O12 crystals [J]. Phys Stat Sol (b), 2004, 241:1134-1140.
[5] ZENG X H, ZHAO G J, XU J, LI H J, HE X M, PANG H Y, JIE M Y. Effect of air annealing on the spectral properties of Ce?Y3Al5O12 single crystals grown by the temperature gradient technique [J]. J Cryst Growth, 2005, 274: 495-499.
[6] FASOLI M, FONTANA I, MORETTI F, MIHOKOVA E, NIKL M, VEDDA A, ZOREKO Y, GORBENKO V. Shallow traps in YAlO3?Ce single crystal perovskites [J]. IEEE Trans Nucl Sci, 2008, 55(3): 1114-1117.
[7] ZHONG J P, LIANG H B, SU Q, ZHANG G B, DORENBOS P, BIROWOSUTO M D. Effects of annealing treatments on the luminescence and scintillation properties of Ce?Lu3Al5O12 crystal grown by Czochralski method [J]. J Rare Earths, 2007, 25(5): 568-572.
[8] MEIJVOGEL K, BOS A J J, DORENBOS P, VAN EIJK C W E. On the relation between prompt luminescence and thermo-luminescence (TL) properties of some materials [C]// Proc SCINT 95 Int Conf Inorganic Scintillators and Their Applications, Delft, 1996:159-161.
[9] BARTRAM R H, HAMILTON D S, KAPPERS L A, LEMPICKI A. Electron traps and transfer efficiency of cerium-doped aluminate scintillators [J]. J Lumin, 1997, 75: 183-192.
[10] WOJTOWICZ A J, GLODO J, LEMPICKI A, BRECHER C. Recombination and scintillation processes in YAlO3?Ce [J]. J Phys Condens Mater, 1998, 10: 8401-8415.
[11] WOJTOWICZ A J, GLODO J, DROZDOWSKI W, PRZEGIETKA K R. Electron traps and scintillation mechanism in YAlO3?Ce and LuAlO3?Ce scintillators [J]. J Lumin, 1998, 79: 275-291.
[12] WOJTOWICZ A J, DROZDOWSKI W, WISNIEWSKI D, PRZEGIETKA K R, OCAKOWSKI H L, PITERS T M. Thermoluminescence and scintillation of LuAlO3?Ce [J]. Rad Meas, 1998, 29: 323-326.
[13] NIKL M, MIHOKOVA E, MARES J A, VEDDA A, MARTINI M, NEJEZCHLEB K, BLAZEK K. Traps and timing characteristics of LuAG?Ce3+ scintillator [J]. Phys Stat Sol (a), 2000, 181: R10-R12.
Foundation item: Project(2006B14801001) supported by the Science and Technology Program of Guangdong Province, China; Project(20080440804) supported by China Postdoctoral Science Foundation; Project(KF0810) supported by the Open Foundation from the State Key Laboratory of Crystal Materials, China
Corresponding author: ZHONG Jiu-ping; Tel: +86-20-84112112; E-mail: zhongjp@mail.sysu.edu.cn
DOI: 10.1016/S1003-6326(09)60080-7
(Edited by YANG Bing)