J. Cent. South Univ. Technol. (2010) 17: 40-44
DOI: 10.1007/s11771-010-0008-4 
Preparation of Ni nanoparticles plating by electrodeposition using reverse microemulsion as template
ZHOU Hai-hui(周海晖)1, 2, PENG Chun-yu(彭春玉)1, FU Chao-peng(付超鹏)1, AN Jing(安静)1,
ZOU He(邹贺)1, WANG Yi-dong(王一栋)1, XU Yan(许岩)1, KUANG Ya-fei(旷亚非)1, 2
1. College of Chemistry and Chemical Engineering, Hunan University, Changsha 410082, China;
2. State Key Laboratory for Chemo/Biosensing and Chemometrics, Hunan University,
Changsha 410082, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2010
Abstract: Ni nanoparticles plating was prepared in reverse microemulsion. The deposition was carried out through the Brownian motion of water pools in the reverse microemulsion and the adsorption of water pools on the electrode surface. Effects of electrolytic parameters on the size of Ni particles were studied. The performances of hydrogen evolution and hydrogen storage of the Ni nanoparticles plating electrode were also investigated. The results indicate that the size of Ni nanoparticles decreases with the increase of Ni2+ concentration and the decrease of current density. The electrochemical activity of Ni nanoparticles plating electrode is much higher than that of bulk Ni electrode.
Key words: Ni nanoparticles; reverse microemulsion; electrodeposition; electrolytic parameters
1 Introduction
Nanoparticles show good application perspective in electricity, optics, catalysis and biosensor due to their unique physical and chemical properties [1-3]. Reverse microemulsion is defined as a transparent, thermodynamically stable system where water pools stabilized by surfactant disperse in continuous oil phase [4-6]. The diameter of dispersive phase in reverse microemulsion is 10-100 nm. The nano-water pools in reverse microemulsion provide a good microenvironment for preparation of nanoparticles. The size and morphology of the resultant nanoparticles can be controlled by adjusting the size of nano-water pools [7-8]. In electrochemical reaction system, aqueous solution, organic solution or melted salt is often used as electrolyte. And the obtained plating is usually bulk metal or alloy layer. However, it is difficult for reverse microemulsion to construct electrochemical system due to very low electric conductivity of reverse microemulsion whose continuous phase is oil. ZHOU et al [9-10] presented that the electric conductivity of reverse microemulsion can be improved by adding high concentration HCl into the water phase. In this work, reverse microemulsion with high electric conductivity was used as the electrolyte, and Ni nanoparticles plating was prepared by electrodeposition. Moreover, influences of electrolytic parameters on the morphology and performance of Ni nanoparticles plating were investigated.
2 Experimental
2.1 Reagents
Ni sulfate, n-hexane, n-hexanol, p-octyl polyethylene glycol phenyl ether (TritonX-100), hydrochloric acid, sodium hydroxide, sulfuric acid, ethanol and acetone were used. All chemical reagents used are analytical grade. All solutions were prepared with double distilled water.
2.2 Preparation and characterization of reverse microemulsion
After mixing 10 mL TritonX-100, 20 mL n-hexanol and 20 mL n-hexane as the oil phase, 3.2 mL aqueous solution (9.375 mol/L HCl + 0.065-0.286 mol/L NiSO4) was added under stirring, and a transparent micro- emulsion was obtained. Then the microemulsion was centrifuged by a high-speed centrifuge (Eppondorf Centrifuge 5804 R, Germany) with a speed of 1×104 r/min at 5 ℃ for 5 min. The microemulsion was still transparent without any phase separation, indicating that the microemulsion used was quite stable. The structure of microemulsion was characterized by a dynamic light scattering equipment (ALV/CGS-5022F, ALV/Laser Vertriebsgesellschaft m.b.H Company, Germany).
2.3 Preparation of Ni nanoparticles plating electrode
Electrodepositon of Ni nanoparticles was carried out in a two-electrode cell from a reverse microemulsion with a direct current electrical source. The working electrode was a Ni electrode with a surface area of 0.2 cm2. A platinum foil was used as the counter electrode. Prior to use, the working electrode was first polished with alumina emery paper. Then, it was washed with alcohol, acetone and double distilled water, respectively.
2.4 Measurements of morphology and electro- chemical property of Ni nanoparticles plating electrode
The morphology of Ni nanoparticles plating was observed using scanning electron microscopy (SEM, JSM-5600LV, JEOL Company, Japan). The electro- chemical property of Ni nanoparticles plating was investigated by linear polarization (scan rate: 10 mV/s) and cyclic voltammetry (scan rate: 50 mV/s) using a CHI model 660B electrochemical workstation (Shanghai Chenhua Instrument Factory, China). In the electrochemical measurement, the working electrode was a Ni nanoparticles plating electrode. A platinum foil and a saturated calomel electrode (SCE) were used as the counter electrode and the reference electrode, respectively.
3 Results and discussion
3.1 Preparation of Ni nanoparticles plating
Fig.1 shows SEM image of the Ni nanoparticles plating obtained from a reverse microemulsion containing 0.125 mol/L NiSO4 in water phase. It can be observed from Fig.1 that the plating is composed of Ni particles with diameter of about 50 nm, and the nanoparticles on the electrode surface accumulate together. This presents that Ni nanoparticles plating obtained from reverse microemulsion is incontinuous, and hence the electrodeposition process in reverse microemulsion is obviously different from that in conventional homogeneous solution.

Fig.1 SEM image of Ni nanoparticles plating obtained from reverse microemulsion at Ni2+ concentration of 0.125 mol/L and current density of 10 mA/cm2
The surfactant-stabilized nano-water pools in reverse microemulsion are unique nanosized reactors. The average hydrodynamic radius of the reverse microemulsion is 17.1 nm with a distribution from 3.2 to 87.1 nm. Fig.2 shows the cathodic reduction process of Ni2+ at the electrode/reverse microemulsion interface. The nano-water pools containing Ni2+ collide with the electrode continuously because of their Brownian motion. Moreover, due to the interaction between the surfactant film and the electrode/reverse microemulsion interface, some water pools also adsorb at the interface. When the equilibrium between the adsorbed water pools and those colliding with the electrode is set up, a layer of homeostatic adsorbed water pools forms. Because Ni2+ can only dissolve in the water phase, the distribution of Ni2+ in the reverse microemulsion is incontinuous and ununiform. Thus, only Ni2+ in the water pools adsorbed at the interface can be reduced to Ni, which exhibits nanosized particle due to the size effect of nano-water pool [11-12]. The organic compound especially surfactant around the nano-water pool adsorbs on the surface of Ni nanoparticles and inhibits further growth of Ni particles. Subsequently, the reduction of Ni2+ in another nano-water pool occurs. Go round and round, Ni nanoparticles plating forms on the electrode surface eventually.
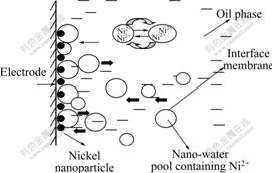
Fig.2 Sketch map of cathodic reduction process of Ni2+ at electrode/reverse microemulsion interface
3.2 Influences of NiSO4 concentration on morphology and electrochemical activity of Ni nanoparticles plating
Fig.3 shows SEM images of the Ni nanoparticles plating obtained from reverse microemulsion containing different Ni2+ concentrations. It can be observed from Fig.3 that when Ni2+ concentration of the water phase is 0.065 mol/L, the diameter of the Ni nanoparticles is about 90 nm. When Ni2+ concentration increases to 0.286 mol/L, the diameter of the Ni nanoparticles decreases to about 30 nm. This presents that Ni2+ concentration of the water phase markedly affects the size of Ni nanoparticles. For the electrode reaction of Ni2++2e→Ni, the change of Ni2+ concentration will lead to the change of exchange current density and limit diffusion current density of the electrode reaction. On account of the special structure of reverse microemulsion, Ni2+ ions participating in the electrodeposition process are almost from the water pools. When Ni2+ concentration of the water pools is low, Ni2+ concentration will decrease constantly with the consumption of Ni2+ during the electrodeposition process. The controlling step of the electrode reaction will convert to concentration polarization step from electron exchange step and the overvoltage will mainly result from concentration polarization. The crystals of the Ni nanoparticle plating formed are large. So, the diameter of Ni nanoparticles obtained from the microemulsion containing 0.065 mol/L Ni2+ is larger than that obtained from the microemulsion containing 0.286 mol/L Ni2+.
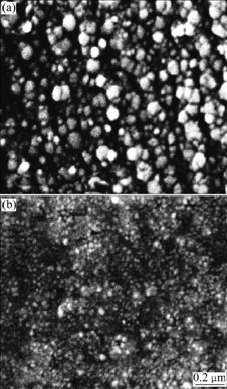
Fig.3 SEM images of Ni nanoparticles platings obtained from reverse microemulsion containing different Ni2+ concentrations at current density of 10 mA/cm2: (a) 0.065 mol/L; (b) 0.286 mol/L
Fig.4 shows the anodic polarization curves of bulk Ni electrode and the Ni nanoparticles plating electrodes obtained from reverse microemulsion containing different Ni2+ concentrations in 0.5 mol/L H2SO4. The passive peaks of Ni can be observed at about 0.2 V. Because the apparent areas of the three electrodes are equal, the real areas of the electrodes can be evaluated according to the value of the passive current density. It can be deduced from Fig.4 that the electrochemical activity of the Ni nanoparticles plating electrodes is higher than that of the bulk Ni electrode. Moreover, the activity of the Ni nanoparticles plating increases with the decrease of the size of Ni nanoparticles. This is attributed to the fact that the Ni nanoparticles plating electrode has larger specific surface areas than the bulk Ni electrode. And the smaller the size of Ni nanoparticles, the larger the specific surface area of the electrode.
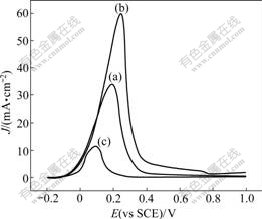
Fig.4 Anodic polarization curves of bulk Ni electrode and Ni nanoparticles plating electrodes obtained from reverse microemulsion containing different Ni2+ concentrations in 0.5 mol/L H2SO4: (a) 0.065 mol/L; (b) 0.286 mol/L; (c) Bulk Ni
3.3 Influences of current density on morphology and electrochemical activity of Ni nanoparticles plating
The electrochemical kinetic rule of Ni deposition can also be changed by changing the deposition current density under the same Ni2+ concentration. Fig.5 shows SEM images of Ni nanoparticles platings prepared at 5, 15 and 25 mA/cm2. It can be seen that the smaller the current density, the smaller the size of Ni nanoparticles and the more uniform of the Ni nanoparticles plating. When the current density is 25 mA/cm2, the diameter of Ni nanoparticles reaches about 70 nm. This may result from the mass transfer characteristic of reverse microemulsion. In reverse microemulsion, the water pools are very small and hence convection transfer is approximately zero. The reaction particles in the water pools reach the electrode surface only by diffusion, liquid phase transfer is the controlling step at large current density, and hence the crystals of the plating become large.

Fig.5 SEM images of Ni nanoparticles platings obtained at Ni2+ concentration of 0.125 mol/L and different current densities: (a) 5 mA/cm2; (b) 15 mA/cm2; (c) 25 mA/cm2
3.4 Hydrogen evolution and hydrogen storage performances of Ni nanoparticles plating
The cyclic voltammetric curves of the Ni nanoparticles plating electrode and bulk Ni electrode in7 mol/L NaOH are shown in Fig.6. Hydrogen evolution can be observed obviously at potential from -0.90 to -0.95 V during the negative sweeping. Anodic oxidation peaks emerge at potential from -0.70 to -0.60 V during the positive sweeping, which may be corresponding to the anodic oxidation of hydrogen atoms adsorbing on the electrode surface during the negative sweeping [13-14]. Comparing curve (a) with curve (b), it can be seen that both the cathodic reduction peak current of H+ and the anodic oxidation peak current of absorbed H on the Ni nanoparticles plating electrode are almost 9 times higher than those on the bulk Ni electrode. This reveals that the electrochemical activity of the Ni nanoparticles plating electrode for redox of hydrogen is much higher than that of the bulk Ni electrode. The electrochemical reaction of the Ni nanoparticles plating electrode in 7 mol/L NaOH is controlled by adsorption/oxidation processes. Moreover, it can be deduced that the Ni nanoparticles plating electrode has potential value in hydrogen storage.
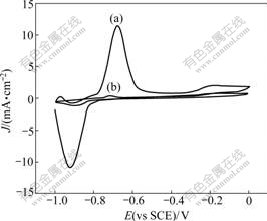
Fig.6 Cyclic voltammetric curves of Ni nanoparticles plating electrode and bulk Ni electrode in 7 mol/L NaOH: (a) Ni nanoparticles plating electrode; (b) Bulk Ni electrode
4 Conclusions
(1) Ni nanoparticles plating is prepared using reverse microemulsion as template.
(2) The higher the Ni2+ concentration of the water phase and the smaller the deposition current density, the smaller the size of Ni nanoparticles.
(3) The Ni nanoparticles plating electrode has higher electrochemical activity than the bulk Ni electrode. There are two redox peaks for the Ni nanoparticles plating electrode in 7 mol/L NaOH, indicating good hydrogen evolution and hydrogen storage performances of the electrode.
References
[1] JOZEFCZAK A, LABOWSKI A S. Effects of biocompatible coating of nanoparticles on acoustics property of the magnetic fluid [J]. Journal of Magnetism and Magnetic Materials, 2005, 290/291(1): 265-268.
[2] YANG J, HIDAJAT K, KAWI S. Synthesis of nano-SnO2/SBA-15 composite as a highly sensitive semiconductor oxide gas sensor [J]. Materials Letters, 2008, 62(8/9): 1441-1443.
[3] ZHANG X, ZHANG F, GUAN R F. Preparation of Pt-Ru-Ni ternary nanoparticles by microemulsion and electrocatalytic activity for methanol oxidation [J]. Materials Research Bulletin, 2007, 42(2): 327-333.
[4] FU C P, ZHOU H H, KUANG Y F. Research on electrochemical properties of nonaqueous ionic liquid microemulsions [J]. Colloid Polymer Science, 2008, 286(13): 1499-1504.
[5] GULLON J, RODES A, MONTIEL V. Electrochemical characterization of platinum/palladium nanoparticles prepared in a water-in-oil microemulsion [J]. Journal of Electroanalytical Chemistry, 2003, 554/555(15): 273-284.
[6] LI H, LIU J, XIE S H, LI H X. Highly active Co-B amorphous alloy catalyst with uniform nanoparticles prepared in oil-in-water microemulsion [J]. Journal of Catalysis, 2008, 259(1): 104-110.
[7] XIONG L, MANTHIRAM A. Catalytic activity of Pt-Ru alloys synthesized by a microemulsion method in direct methanol fuel cells [J]. Solid State Ionics, 2005, 176(3/4): 385-392.
[8] ZHANG X, CHAN K Y. Water-in-oil microemulsion synthesis of platinum ruthenium nanoparticles, their characterization and electrocatalytic properties [J]. Chemistry Material, 2003, 15(2): 451-459.
[9] ZHOU H H, PENG C Y, JIAO S Q, KUANG Y F. Electrodeposition of nanoscaled Ni in a reverse microemulsion [J]. Electrochemistry Communications, 2006, 8(7): 1142-1146.
[10] PENG Chun-yu, ZHOU Hai-hui, ZENG Wei, KUANG Ya-fei. Studies on the conductivity of reverse microemulsion [J]. Acta Physico-Chimica Sinica, 2006, 22(4): 409-413. (in Chinese)
[11] CAPEK I. Preparation of metal nanoparticles in water-in-oil microemulsions [J]. Advances in Colloid and Interface Science, 2004, 110(1/2): 49-74.
[12] FU C P, ZHOU H H, PENG W C. KUANG Y F. Comparison of electrodeposition of silver in ionic liquid microemulsions [J]. Electrochemistry Communications, 2008, 10(5): 806-809.
[13] GUO X P, LIU J, YE S H. Hydrogen adsorption of metal Ni and hydrogen storage alloy electrodes [J]. Journal of Alloys and Compounds, 1997, 253/254(20): 515-519.
[14] DENG B H, LIY F, WANG R Q. Two reduction processes for hydrogen adsorption and absorption at MmNi5-type alloy electrodes [J]. Electrochimica Acta, 1999, 44(17): 2853-2857.
Foundation item: Projects(20673036, J0830415) supported by the National Natural Science Foundation of China; Project(09JJ3025) supported by Hunan Provincial Natural Science Foundation of China; Project(09GK3173) supported by the Planned Science and Technology Project of Hunan Province, China
Received date: 2009-03-14; Accepted date: 2009-07-18
Corresponding author: ZHOU Hai-hui, Professor; Tel: +86-731-88822286; E-mail: haihuizh@163.com
(Edited by CHEN Wei-ping)