


Trans. Nonferrous Met. Soc. China 22(2012) 1711-1716
Phase field calculation of interface mobility in a ternary alloy
WANG Gang, ZENG De-chang, LIU Zhong-wu
School of Materials Science and Engineering, South China University of Technology, Guangzhou 510640, China
Received 23 September 2011; accepted 29 October 2011
Abstract: A novel approach based on the quantitative phase field model was proposed to calculate the interface mobility and applied to the α/β interface of a ternary Ti-6Al-4V alloy. Phase field simulations indicate that the higher interface mobility leads to the faster transformation rate, but only a unique value of interface mobility matches the diffusion equation under the diffusion-controlled condition. By comparing the transformation kinetics from phase field simulations with that from classical diffusion equation, the interface mobility at different temperatures can be obtained. The results show that the calculated interface mobility increases with increasing temperature and accords with Arrhenius equation very well.
Key words: phase transformation; diffusion; phase field modeling; interface mobility
1 Introduction
The kinetics of phase transformation in alloys may be controlled by the solute diffusion or by the interfacial reaction or by both mechanisms [1]. It is also possible that the transformation mechanism changes during the transformation process [2]. In general, the different transformation mechanisms are closely related to the interface mobility because it is the motion of interfaces that sets the evolution rate and final form of the interfacial microstructure. However, it is very hard to measure directly the interface mobility by means of experimental methods, especially for multicomponent multiphase systems. In most cases, the interface mobility only serves as an assumed parameter.
Many efforts have been made to measure interface mobility. In 1975, HILLERT [3] estimated the interface mobility from experiments of grain growth and recrystallization in pure iron, but his procedure was just a crude attempt. Later on, sophisticated in situ techniques, such as hot stage transmission electron microscopy, were applied to measuring the interface velocity directly and, thereby, for a known driving pressure, the interface mobility could be obtained [4]. Unfortunately, these experiments are usually limited to a very small observation range. Recently, molecular dynamics (MD) method has been applied to computing interface mobility [5-8], but it cannot handle the complicated microstructures in multicomponent alloys. Moreover, quantitative comparison between experiments and atomic-scale computer simulations revealed large discrepancies which were difficult to reconcile, even for the relatively simple case of grain boundaries [5-7] and solid-liquid interface in pure metals [8].
In the last several years, the quantitative phase field method has been well established and widely applied to studying the phase transformation kinetics [9-12]. It provides an indirect approach to evaluate the interface mobility because it is directly proportional to the phase-field mobility in quantitative phase field model. Once the phase-field mobility is determined, the intrinsic interface mobility can be readily obtained. However, the value of phase-field mobility is usually unknown in phase field model. In Ref. [13], an estimated value of interface mobility was given and the phase-field mobility was calibrated accordingly. In the case of solidification, under the assumptions of negligible diffusivity in solid and constant diffusivity within the interfacial region, a formulation was proposed to estimate the phase-field mobility at a thin-interface limit [9], but it is restricted within very narrow application limits due to the assumptions.
Therefore, the evaluation of interface mobility is still in suspense. The aim of this work is to propose a novel approach to calculate the interface mobility based upon the quantitative phase field simulation. The approach is then applied to calculating the mobility of α/β interface in a ternary Ti-6Al-4V alloy as a function of temperature.
2 Computational methodology
In this work we propose a new approach to calculate the interface mobility without the conditions advanced in Ref. [9]. It is noted that the quantitative phase field model should coincide with Fick’s diffusion equation if the phase transition is diffusion controlled. Therefore, the phase-field mobility in phase field model can be adjusted to ensure a diffusion-controlled process until the obtained transformation kinetics is identical with that calculated from diffusion equation. By comparing the kinetics from both methods, the phase-field mobility at different temperatures can be gained, thus the interface mobility as a function of temperature is promising.
It is well known that, according to Fick’s second law, a diffusion process can be described accurately by the classical diffusion equation:
 (1)
(1)
where ci (or ck) is the concentration of the ith (or kth) component; t is the time; Dik is the diffusivity. In order to maintain the mass balance at the interface, the amount of a solute removed from α phase and partitioned into β phase per unit time must equal the flux of the solute atoms diffusing away from the interface, thus the following boundary condition must be obeyed [14]:
 (2)
(2)
where v is the velocity of the interface; cα and cβ are the concentrations at the interface, respectively. For a diffusion-controlled phase transformation, the concentrations at the interface approximate to the equilibrium values due to a very high interface mobility [15]. Under these circumstances, there is effectively local equilibrium at the interface.
In a phase field model, a multicomponent multiphase system is characterized by the structural field parameter ηi and concentration field parameter cj, and their temporal evolutions are governed by Allen-Cahn equation [16] and Cahn-Hilliard equation [17], respectively:
 (3)
(3)
 (4)
(4)
The total free energy of the system is given by:
 (5)
(5)
where V is the system volume; κ is the gradient coefficient; f is the local free energy density. A function of field parameters and temperature can be expressed by:
 (6)
(6)
with the constraint that the summation of ηi everywhere is always unity. Here fi is the free energy density of the ith phase that can be extracted from the thermodynamic databases, and ω is the energy barrier coefficient which along with the gradient coefficient κ in Eq. (5), can be determined from interfacial energy and interface thickness [9].
The parameter Mij is the so-called chemical mobility in the volume-fixed frame of reference. In a structurally and compositionally non-uniform system, the chemical mobility can be expressed by:
 (7)
(7)
where Mijp is the chemical mobility in a single phase p and it is quantitatively related to the atomic mobility [18]:
 (8)
(8)
where δjk and δki are the Kronecker delta; Vm is the molar volume; the composition dependence of the atomic mobility in the single phase p, Mkp, can be modeled in a CALPHAD type fashion [19].
The chemical mobility is also related to the diffusivity in Eqs. (1) and (2) by the following relationship [20]:
 (9)
(9)
Thus, the diffusivity can be calculated according to Eq. (9) after the chemical mobility is obtained.
The kinetic coefficient L, also called as phase-field mobility, is directly correlated to the intrinsic interface mobility MI in the sharp interface approach by the following equation [13]:
 (10)
(10)
where σ is the interfacial energy.
Therefore, for a diffusion-controlled phase transformation, one can solve numerically the diffusion equation (Eq. (1)) with its boundary condition (Eq. (2)) firstly, and then solve the phase field equations (Eqs. (3) and (4)) with different values of L, until the kinetic curves from both methods achieve a perfect agreement. Finally, the interface mobility may be derived from Eq. (10).
3 Results
3.1 Computational conditions
A novel approach is proposed above to estimate the interface mobility. To take an example, this method is applied to the β-α transformation in Ti-6Al-4V ternary alloy. The supersaturated alloy is heat-treated in α+β two-phase zone and the different heat-treatment temperatures are set. The Gibbs free energy of the single phase and the atomic mobility are extracted from the thermodynamic and kinetic databases, respectively. The chemical mobility is calculated by Eqs. (7) and (8). The diffusivity used in Fick’s diffusion equation is calibrated from the chemical mobility according to Eq. (9). The phase-field mobility acts as an adjustable parameter. And the coefficients κ and ω are determined according to the interface thickness and the interfacial energy.
The simulations are implemented in one-dimension. The Ti-6Al-4V alloy is initialized to the equilibrium state at 1213 K, and then is heat-treated at different temperatures to obtain the corresponding transformation kinetics. According to the phase field simulation, the equilibrium state of Ti-6Al-4V at 1213 K is composed of 2.413% α phase (volume fraction) and 97.587% β phase; the mole fractions of aluminum in the matrix and precipitate are 0.101492 and 0.121614, and the mole fractions of vanadium in the matrix and precipitate are 0.0365953 and 0.0122996, respectively. The system size is chosen to be 82.9 μm, initially comprising α phase of 2 μm and β phase of 80.9 μm to satisfy the equilibrium volume fraction of the phases at 1213 K. The equilibrium concentrations at 1213 K are set as the initial concentrations of the system. In both computational models, the same mesh size of 0.1 μm is adopted during the numerical discretization. In the phase field model, the interfacial energy and interface thickness are assumed to be independent of the temperature, and their values are set as 0.5 J/m2 (a typical value for incoherent interface at elevated temperature) and 0.5 μm (five times the mesh size to ensure a diffused interface), respectively.
3.2 Interface mobility at different temperatures
The isothermal transformation kinetics at 1193 K in Ti-6Al-4V alloy obtained from diffusion equation and phase field simulations with different phase-field mobilities are shown in Fig. 1. For convenience, a dimensionless parameter Lre, called reduced mobility, is introduced, and it holds that L=Lre×10-9 m3/(J・s). It can be seen that the length of alpha phase increases with time which means the occurrence of the β-α transformation, and the diffusion equation gives the unique kinetic solution while the phase field simulations present the various kinetics when the different values of L are adopted. The larger L results in the faster transformation rate and only a proper L can bring out the same kinetics with that from diffusion equation. Figure 2 displays the temporal evolution of mole fraction of aluminum and vanadium in the system at this temperature obtained from diffusion equation and phase field simulation with Lre=6.4. It is shown that, with the value of phase-field mobility, the profiles of mole fractions of solute atoms at different time obtained from phase field simulation agree very well with those from diffusion equation. There is a jump of the concentration at the interface in the case of diffusion equation because it is a sharp-interface model. While the interface diffuses in phase field model, there are several data points of the concentration at the interface, and the sparse data points are adopted outside the interface. Therefore, it is concluded that the phase-field mobility of Ti-6Al-4V alloy at 1193 K is 6.4×10-9 m3/(J・s). Then the intrinsic interface mobility of α/β interface in Ti-6Al-4V alloy at 1193 K is calculated to be about 2.6×10-15 m4/(J・s) according to Eq. (10).
The interface mobility is obviously dependent upon the temperature. Following the same procedures above, the isothermal phase transformations in Ti-6Al-4V alloy at different heat-treatment temperatures (from 1183 K to 1203 K) are simulated by diffusion equation and quantitative phase field model. By choosing a proper phase-field mobility in the phase field model, the kinetics from the two methods can accord perfectly with each other, as shown in Fig. 3. Table 1 gives the values of phase-field mobility at different temperatures according to the kinetic comparison. The relationship between ln Lre and 1/T is plotted in Fig. 4, and it can be seen readily that ln Lre is directly proportional to 1/T within the studied temperature range. The linear fitting indicates that:
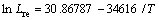 (11)
(11)

Fig. 1 Isothermal transformation kinetics at 1193 K in Ti-6Al-4V alloy obtained from diffusion equation and phase field simulations with different phase-field mobility values
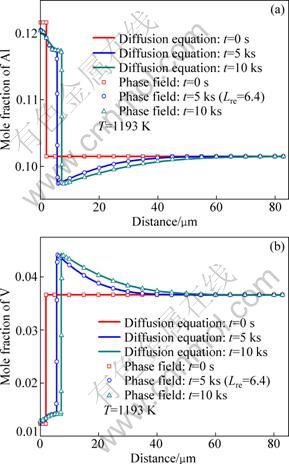
Fig. 2 Temporal evolution (color online) of mole fractions of Al (a) and V (b) at T=1193 K in Ti-6Al-4V alloy obtained from diffusion equation and phase field simulation with Lre=6.4

Fig. 3 Isothermal phase transformation kinetics in Ti-6Al-4V alloy obtained from diffusion equation (solid lines) and phase field simulation (discrete symbols) at different temperatures by choosing proper phase-field mobility shown in Table 1
Therefore, the intrinsic interface mobility can be obtained as follows:
 (12)
(12)
As shown in Eq. (12), the intrinsic interface mobility obeys the Arrhenius law very satisfactorily, and the activation energy is calculated to be 287.8 kJ/mol.
Table 1 Reduced phase-field mobility at different temperatures obtained from kinetic comparison between diffusion equation and phase field simulation

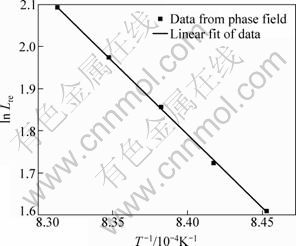
Fig. 4 Plot of ln Lre versus 1/T and its linear fit
4 Discussion
Although the present results in our work may not be validated at first by comparing them with experimental measurements, due to the actual interface mobility very difficult to be determined experimentally, especially at high temperatures, the numerically calculated results can still be evaluated indirectly.
Firstly, it is found that the interface mobility accords with Arrhenius equation very well, which bears out that the interface motion is controlled by the thermal activation. For the β-α transformation in Ti-6Al-4V alloy, the interface motion is coupled to long-range diffusional fluxes of aluminum and vanadium atoms in the system. The α/β interface acts as a source or a sink of the fluxes. To accomplish the motion of the interface, aluminum and vanadium atoms must be added to, or removed from both of the crystals adjoining the interface. In such case, the long-range diffusion process of aluminum and vanadium atoms relies on the thermal activation. Therefore, it is reasonable that our calculated interface mobility follows the Arrhenius law. Actually, the same rule is always observed in various alloys. For example, the intrinsic mobility of γ/α interface in steels was determined as a thermally activated quantity following an Arrhenius relation [3,21,22]; SINGER et al [23] estimated the interface mobility of Ti-Al alloy based on the Burke-Turnbull equation and found that the expression of the mobility follows the Arrhenius law.
On the other hand, the activation energy of interface motion can be compared with that of long-range diffusion of solution atoms. The observations by transmission electron microscopy and the results of energy-dispersive spectrometry analysis indicate that the β-α transformation in Ti-6Al-4V alloy is mainly controlled by vanadium diffusion [24]. Therefore, although the rate at which an interface moves depends on the intrinsic mobility (which is related to the process of vanadium atoms transfer across the interface) and the rate at which diffusion can remove the excess of vanadium atoms ahead of the interface, the transfer of atoms through the interface should be much easier than the diffusion process. A value of 97 kJ/mol for the activation energy of β/β interface in Ti-6Al-4V alloy has been reported [25]. Because of the lower temperature range of α+β phase zone and the structural difference between two phases, it is expected that the activation energy of α/β interface is much higher than 97 kJ/mol. In addition, MALINOV et al [26] studied the β-α phase transformation in Ti-6Al-4V alloy and estimated that the activation energy may achieve beyond 400 kJ/mol. In order to ensure a diffusion-controlled phase transformation, the activation energy of α/β interface must be lower than that of the β-α transition. Therefore, it is quite reasonable that our calculated result (287.8 kJ/mol) falls in the range between 97 kJ/mol to 400 kJ/mol.
It should be noted that, if the heat-treatment temperature is lower than a certain value, the phase field simulation results cannot agree with diffusion equation any more. Figure 5 gives the phase transformation kinetics from diffusion equation and phase field simulations at 1113 K where the reduced phase-field mobility is calculated to be 0.79 according to Eq. (11). It shows that the phase field simulation predicts a little slower kinetics than diffusion equation. This is because the β-α phase transition is not completely controlled by diffusion of solute atoms at the relatively low temperatures. For instance, the transfer process of atoms across the interface at 1113 K needs more time than at higher temperature, and this period of time cannot be neglected totally compared with the long-range diffusion process of solute atoms. But the diffusion equation can only deal with the fully diffusion-controlled process, and it aggrandizes the transfer rate of atoms cross the interface at the relatively low temperatures, thus predicts artificial transition kinetics.

Fig. 5 Isothermal transformation kinetics at 1113 K in Ti-6Al-4V alloy obtained from diffusion equation and phase field simulations with Lre=0.79 calculated by Eq. (11)
5 Conclusions
A novel approach based on the quantitative phase field model is proposed to calculate interface mobility. Phase field simulations indicate that the higher interface mobility will bring on the faster transformation rate, but only a unique value of interface mobility matches the diffusion equation under the diffusion-controlled condition. By comparing the transformation kinetics from phase field simulation with that from classical diffusion equation, the interface mobility at different temperatures can be obtained. The results show that the calculated mobility of α/β interface in Ti-6Al-4V alloy increases with temperature and accords with Arrhenius equation very well. This method may be applied to other ternary and multicomponent alloy systems.
References
[1] CHRISTIAN J W. The theory of transformations in metals and alloys (Part I) [M]. Oxford: Pergamon, 2002.
[2] SIETSMA J, van der ZWAAG S. A concise model for mixed-mode phase transformations in the solid state [J]. Acta Mater, 2004, 52: 4143-4152.
[3] HILLERT M. Diffusion and surface control of reactions in alloys [J]. Metall Trans A, 1975, 6: 5-19.
[4] ONINK M, TICHELAAR F D, BRAKMAN C M, MITTEMEIJER E J, van der ZWAAG S. An in situ hot stage transmission electron microscopy study of the decomposition of Fe-C austenites [J]. J Mater Sci, 1995, 30: 6223-6234.
[5] ZHANG H, MENDELEV M I, SROLOVITZ D J. Computer simulation of the elastically driven migration of a flat grain boundary [J]. Acta Mater, 2004, 52: 2569-2576.
[6] ZHANG H, UPMANYU N, SROLOVITZ D J. Curvature driven grain boundary migration in aluminum: Molecular dynamics simulations [J]. Acta Mater, 2005, 53: 79-86.
[7] JANSSENS K G F, OLMSTED D, HOLM E A, FOILES S M, PLIMPTON S J, DERLET P M. Computing the mobility of grain boundaries [J]. Nat Mater, 2006, 5: 124-127.
[8] GAO Y F, YANG Y, SUN D Y, ASTA M, HOYT J J. Molecular dynamics simulations of the crystal-melt interface mobility in HCP Mg and BCC Fe [J]. J Cryst Growth, 2010, 312: 3238-3242.
[9] KIM S G, KIM W T, SUZUKI T. Phase-field model for binary alloys [J]. Phys Rev E, 1999, 60: 7186-7197.
[10] CHEN Q, MA N, WU K S, WANG Y. Quantitative phase field modeling of diffusion-controlled precipitate growth and dissolution in Ti-Al-V [J]. Scripta Mater, 2004, 50: 471-476.
[11] WANG G, XU D S, MA N, ZHOU N, PAYTON E J, YANG R, MILLS M J, WANG Y. Simulation study of effects of initial particle size distribution on dissolution [J]. Acta Mater, 2009, 57: 316-325.
[12] OHNO M, MATSUURA K. Quantitative phase-field modeling for two-phase solidification process involving diffusion in the solid [J]. Acta Mater, 2010, 58: 5749-5758.
[13] HUANG C J, BROWNE D J, MCFADDEN S. A phase field simulation of austenite to ferrite transformation kinetics in low carbon steels [J]. Acta Mater, 2006, 54: 11-21.
[14] PORTER D A, EASTERLING K E. Phase transformations in metals and alloys [M]. Oxford: Van Nostr and Reinhold Co Ltd, 1981.
[15] KATZAROV I, MALINOV S, SHA W. Finite element modeling of the morphology of β to α phase transformation in Ti-6Al-4V alloy [J]. Metall Mater Trans A, 2002, 33: 1027-1040.
[16] ALLEN S M, CAHN J W. A microscopic theory for antiphase boundary motion and its application to antiphase domain coarsening [J]. Acta Metall, 1979, 27: 1085-1095.
[17] CAHN J W. On spinodal decomposition [J]. Acta Metall, 1961, 9: 795-801.
[18] ANDERSSON J O, ?GREN J. Models for numerical treatment of multicomponent diffusion in simple phases [J]. J Appl Phys, 1992, 72: 1350-1355.
[19] BORGENSTAM A, ENGSTR?M A, H?GLUND L, ?GREN J. DICTRA, a tool for simulation of diffusional transformations in alloys [J]. J Phase Equil, 2000, 21: 269-280.
[20] ODE M, LEE J S, KIM S G, KIM W T, SUZUKI T. Phase-field model for solidification of ternary alloys [J]. ISIJ Int, 2000, 40: 870-876.
[21] WITS J J, KOP T A, van LEEUWEN Y, van der ZWAAG S. A study on the austenite-to-ferrite phase transformation in binary substitutional iron alloys [J]. Mater Sci Eng A, 2000, 283: 234-241.
[22] GAMSJ?GER E, MILITZER M, FAZELI F, SVOBODA J, FISCHER F D. Interface mobility in case of the austenite-to-ferrite phase transformation [J]. Comput Mater Sci, 2006, 37: 94-100.
[23] SINGER H M, SINGER I, JACOT A. Phase-field simulations of α-γ precipitations and transition to massive transformation in the Ti-Al alloy [J]. Acta Mater, 2009, 57: 116-124.
[24] GINEBRA M P, GILL F X, MANERO J M, PLANELL J A[C]//BLENKINSOP P A, EVANS W J, FLOWER H M. Titanium ’95: Science and Technology. London: Institute of Materials, 1996: 2563-2570.
[25] GIL F J, PLANELL J A. Behaviour of normal grain growth kinetics in single phase titanium and titanium alloys [J]. Mater Sci Eng A, 2000, 283: 17-24.
[26] MALINOV S, GUO Z, SHA W, WILSON A. Differential scanning calorimetry study and computer modeling of β-α phase transformation in a Ti-6Al-4V alloy [J]. Metall Mater Trans A, 2001, 32: 879-887.
三元合金中界面迁移率的相场法计算
王 刚,曾德长,刘仲武
华南理工大学 材料科学与工程学院,广州 510640
摘 要:基于定量相场模型,提出一种新方法来计算界面迁移率,并将该方法应用于Ti-6Al-4V合金的α/β界面。相场模拟表明,更高的界面迁移率将导致更快的相转变速率,但在扩散控制的条件下,只有唯一的界面迁移率能匹配扩散方程。通过比较采用相场模拟和经典扩散方程所得到的相变动力学,可以得到不同温度下的界面迁移率。结果表明,计算所得的界面迁移率随着温度的升高而增加,且与Arrhenius方程吻合得很好。
关键词:相变;扩散;相场模拟;界面迁移率
(Edited by YANG Hua)
Foundation item: Project (51101059) supported by the National Natural Science Foundation of China; Project (20110490874) supported by the China Postdoctoral Science Foundation
Corresponding author: ZENG De-chang; Tel: +86-20-87111278; E-mail: medczeng@scut.edu.cn
DOI: 10.1016/S1003-6326(11)61377-0