FeCoNiCrCu0.5Alx高熵合金的结构和性能
来源期刊:中国有色金属学报(英文版)2013年第3期
论文作者:李宝玉 彭 坤 胡爱平 周灵平 朱家俊 李德意
文章页码:735 - 741
Key words:high-entropy alloy; microstructure; heat treatment; stable phase; hardness; corrosion resistance
摘 要:研究Al含量和热处理对FeCoNiCrCu0.5Alx多主元高熵合金的相结构、硬度和电化学性能的影响规律。随着Al含量的增加,铸态合金的相结构由FCC相向BCC相转变。当x从0.5增加到1.5时,FeCoNiCrCu0.5Alx高熵合金的稳定结构由FCC结构向FCC+BCC双相结构转变。BCC相的硬度高于FCC相的,在氯离子及酸性介质中BCC相的耐腐蚀性均优于FCC相的。FeCoNiCrCu0.5Al1.0铸态合金具有高硬度和良好的抗腐蚀性能。
Abstract: Effects of Al content and heat treatment on the structure, hardness and electrochemical properties of FeCoNiCrCu0.5Alx high-entropy alloys were investigated. The phase structure of as-cast alloys evolves from FCC phase to BCC phase with the increase of Al content. The stable phase of FeCoNiCrCu0.5Alx high-entropy alloys will transform from FCC phase to FCC+BCC duplex phases when x value increases from 0.5 to 1.5. The hardness of BCC phase is higher than that of FCC phase, and the corrosion resistance of BCC phase is better than FCC phase in chlorine ion and acid medium. High hardness and good corrosion resistance can be obtained in as-cast FeCoNiCrCu0.5Al1.0 alloy.
Trans. Nonferrous Met. Soc. China 23(2013) 735-741
Bao-yu LI, Kun PENG, Ai-ping HU, Ling-ping ZHOU, Jia-jun ZHU, De-yi LI
College of Materials Science and Engineering, Hunan University, Changsha 410082, China
Received 6 December 2011; accepted 28 April 2012
Abstract: Effects of Al content and heat treatment on the structure, hardness and electrochemical properties of FeCoNiCrCu0.5Alx high-entropy alloys were investigated. The phase structure of as-cast alloys evolves from FCC phase to BCC phase with the increase of Al content. The stable phase of FeCoNiCrCu0.5Alx high-entropy alloys will transform from FCC phase to FCC+BCC duplex phases when x value increases from 0.5 to 1.5. The hardness of BCC phase is higher than that of FCC phase, and the corrosion resistance of BCC phase is better than FCC phase in chlorine ion and acid medium. High hardness and good corrosion resistance can be obtained in as-cast FeCoNiCrCu0.5Al1.0 alloy.
Key words: high-entropy alloy; microstructure; heat treatment; stable phase; hardness; corrosion resistance
1 Introduction
The design of traditional alloy is selected as a principal component (the content is greater than 50% generally) based on some main properties to improve primary properties and satisfy the secondary performance requirement through the alloying process with iron-based steel and Al-based alloy. YEH et al [1,2] proposed a new design concept calling high-entropy alloy with multiprincipal elements, which contains at least five major elements and each element content is greater than 5% (mole fraction) and less than 35%. High-entropy alloy is a new material with great development potential, due to its good thermal stability at high temperatures [3], excellent corrosion resistance [4,5], high strength [6,7], high hardness [8,9], high oxidation resistance [10,11], and excellent magnetic properties [12,13].
According to the Gibbs phase rule, the number of equilibrium state phase in alloy will increase with increase of the number of element and some intermetallic compounds or intermediate phases will be formed in traditional alloys. High-entropy alloys have a large mixing entropy because of a variety of elements, which makes the alloy systems tend to be stable easily and a simple structure of solid solution and even amorphous phase are formed in high-entropy alloy system. Only a simple BCC phase, FCC phase or BCC+FCC phases and amorphous phase will form in high-entropy alloy due to its unique high entropy effects [14,15]. CoCrCuFeNi alloy exhibited exceptional ductility so that the sample did not fracture even under true strain about 50% [12]. The corrosion resistance of FeCoNiCrCux alloys reduced with the increase of copper content [16]. The FeCoNiCrCu0.5 alloy was a failover at significantly corrosion resistance in 3.5% NaCl solution and the corrosion resistance was not influenced by annealing at different temperatures due to the fact that the Cu-rich phase can degrade its corrosion resistance [17]. The addition of aluminum element could improve the corrosion resistance of the AlxFeCoNiCrTi high-entropy alloys [18]. And it was also found that the hardness of AlxCoCrCuFeNi alloy increased from HV153 to HV735 with the addition of Al from x=0 to 3.0 due to solid solution strengthening and dispersion strengthening [7]. Up to now, the study of high-entropy alloy is mainly focused on the material system, synthesis, microstructure and mechanical properties, but its electrochemical performance has been investigated rarely. The electrochemical performance of FeCoNiCrCu0.5Alx high-entropy alloys has not been reported.
In this work, the effects of Al content on structure, hardness, thermal stability and electrochemical properties of FeCoNiCrCu0.5Alx high-entropy alloys were studied. And the effects of cooling rate after heat treatment on structure and properties of alloy were also investigated.
2 Experimental
High purity Fe, Co, Ni, Cr, Cu and Al elements were used as raw materials. The FeCoNiCrCu0.5Alx high-entropy alloys (x is molar ratio, x=0.5, 1.0, 1.5, denoted by Al0.5, Al1.0, Al1.5, respectively.) were prepared by vacuum arc melt casting in a Ti-gettered high-purity argon atmosphere with a water-cooled copper mould. The alloys were repeatedly melted and solidified to improve the chemical composition homogeneity. In order to study the effects of cooling rate after heat treatment on structure and properties of alloys, the as-cast alloys were solution treated at 1100 °C for 4 h and then cooled to room temperature by furnace cooling and water quenching, respectively.
The crystal structure analysis was performed on the SIEMENS XRD diffractometer D5000 using Cu Kα radiation, tube voltage was 35 kV, and tube current was 30 mA. The DSC curves of alloys were measured by a STA-449C type thermal analyzer. The microstructure of alloys was characterized by FEI type QUANTA-200 environmental scanning electron microscope, and the micro-composition was analyzed by EDS. MHV-2000 type hardness tester was used to measure the Vickers hardness of alloys. The electrochemical properties of alloys were characterized by CHI660C electrochemical workstation. The initial potential was open circuit potential minus 0.5 V, and final potential was about 1.2 V, scanning speed was 1 mV/s. The used electrolyte solution was 0.5 mol/L H2SO4+0.5 mol/L NaCl solution, the reference electrode was saturated KCl solution, and the auxiliary electrode was platinum sheet.
3 Results and discussion
3.1 Effect of Al content on phase structure of alloys
Figure 1 shows the DSC curves of the Al0.5 and Al1.5 alloys. The DSC curves of alloys only have one obvious exothermic peak. The peak value of DSC curve of the Al0.5 is at 850 °C and the temperature range of phase transformation is very wide. The phase transition of alloy is nearly completed at 1000 °C. In order to make phase transition process fully complete, the heat treatment temperature was selected as 1100 °C.
Figure 2(a) shows the XRD patterns of as-cast FeCoNiCrCu0.5Alx alloys. Figures 2(b) and (c) show the XRD patterns of water-quenched alloys and furnace-cooled alloys after heat treatment at 1100 °C for 4 h, respectively. According to the results of systematic extinction, the crystal structure of alloys is simple FCC and BCC.
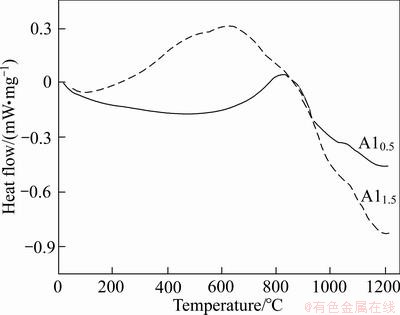
Fig. 1 DSC curves of Al0.5 and Al1.5 alloys
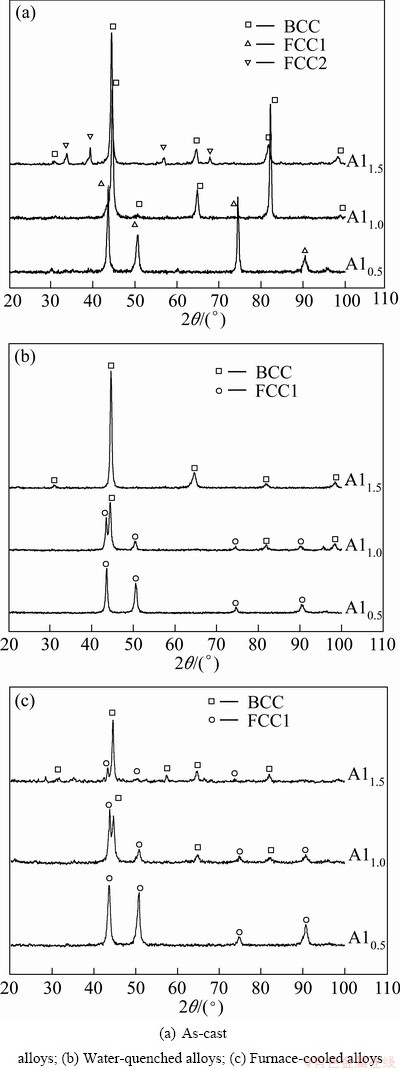
Fig. 2 XRD patterns of FeCoNiCrCu0.5Alx alloys
The lattice constants can be calculated, and the results are shown in Table 1. According to the XRD patterns and DSC curves of alloys, the as-cast and heat treated Al0.5 alloys are both FCC1 phase. The FCC phase in Table 1 whose lattice constant is about 0.359 nm is defined as FCC1 phase and another FCC phase whose lattice constant is about 0.458 nm is defined as FCC2 phase. The heat treatment has no influence on the crystal structure and lattice constant for Al0.5 alloy. For Al1.0 alloy, the as-cast structure is intermediate state BCC phase maybe due to the incomplete atomic diffusion. Element segregation occurred in the process of heat treatment by atomic diffusion, BCC phase was formed in Al-rich region, and steady state FCC1 phase was formed in Al-poor region. Therefore, both water-quenching and furnace-cooling after heat treatment make the alloy transform into FCC1+BCC duplex structures, indicating that the stable structure of Al1.0 alloy is FCC1+BCC duplex structures. For Al1.5 as-cast alloy, BCC phase crystallizes first and then exists in metastable state. FCC2 phase precipitates along the grain boundary of the BCC phase in the crystallization caused by difficult atomic diffusion due to its high entropy effects, therefore, the structure of as-cast Al1.0 alloy is FCC2+BCC duplex structures. The metastable FCC2 phase transforms into BCC structure by atomic diffusion at high temperatures, and the BCC structure is retained to room temperature by water quenching with high cooling rate inhibiting the formation of the FCC1 phase. The steady state FCC1 phase precipitates from the BCC phase by furnace cooling, the BCC structure decomposes into FCC1+BCC duplex structures, and lattice constant increases slightly, due to more Al atoms whose atomic radius is larger in solid solution.
It can be known that the FCC2 phase with large lattice constant is an unstable phase and has intrinsic difference with the steady state FCC1 phase. Figure 3 shows the EDS results of the as-cast Al1.5 high-entropy alloy, because BCC phase crystallizes first and then FCC2 phase precipitates along the grain boundary of the BCC phase, therefore, the cast-dendrite structure is BCC phase and the interdendrite structure is FCC2 phase for the as-cast Al1.5 alloy. According to Fig. 3, the FCC2 phase is Al-rich phase. The lattice constant of FCC2 is very large which results from the larger lattice constant and atomic radius of Al. The Al0.5 high-entropy alloy has high thermal stability and FCC1 structure. The thermal stability of high-entropy alloy decreases when the Al content increases. Coexistence of FCC1+BCC duplex phases is the stable structure when x≥1.0. In XRD patterns, the intensity of diffraction peak of BCC phase increases and the intensity of diffraction peak of FCC phase decreases with the increase of Al content. Therefore, it is more conducive to the formation of BCC phase in high-entropy alloy with the increase of Al content, because the packing factor of BCC cell is much lower than that of FCC cell, so BCC structure can ease the lattice distortion caused by large atomic radius of Al atoms.
Table 1 Lattice constants of as-cast and heat treated FeCoNiCrCu0.5Alx high-entropy alloys
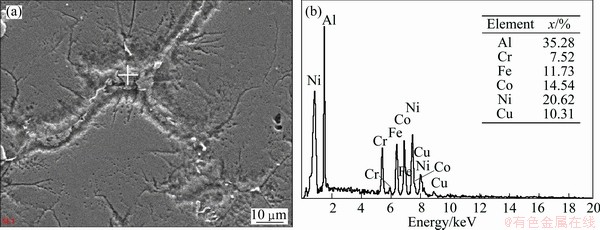
Fig. 3 SEM image (a) and EDS result of as-cast FeCoNiCrCu0.5Al1.5 high-entropy alloys
3.2 Effect of Al content on microscopic structure
The as-cast alloys were grinded, polished, and corroded by aqua regia. Finally, the morphology of alloys was characterized by a FEI QUANTA-200 scanning electron microscope. The SEM images of as-cast FeCoNiCrCu0.5Alx alloys are shown in Fig. 4 and the cast structure is uniform. Both the Al0.5 and Al1.0 alloys are single phase, and the Al1.5 alloy is double phase. This is consistent with the XRD result. The cast structure of Al1.5 alloy is dendrite structure. Combined with the XRD pattern, the phase in Fig. 4(a) is the FCC1 phase, the phase in Fig. 4(b) is BCC phase, and the white phase and the gray phase in Fig. 4(c) are FCC2 and BCC phases, respectively.
3.3 Effect of Al content and heat treatment on hardness of alloys
Seven positions of the as-cast and heat treated
FeCoNiCrCu0.5Alx alloys were selected for the hardness test and the average hardness was taken as the alloy hardness. Table 2 shows the Vickers hardness test results. The Al content has significant effects on hardness of alloys, and the hardness of Al0.5 as-cast alloy is low, but that of as-cast Al1.0 or Al1.5 alloy improves obviously. From structure analysis of as-cast alloys, Al0.5 alloy is of single FCC1 phase, Al1.0 alloy is of single BCC phase, Al1.5 alloy is of BCC+FCC2 duplex phase, and the hardness of BCC structure is much higher than that of FCC structure.

Fig. 4 SEM images of as-cast FeCoNiCrCu0.5Alx alloys
Table 2 Hardness of FeCoNiCrCu0.5Alx high-entropy alloys
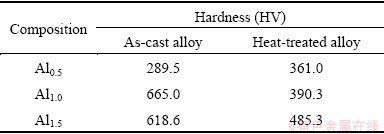
For Al0.5 alloy, the hardness increases after heat treatment. For Al1.0 and Al1.5 alloys, the hardness in heat- treated state is much lower than that in as-cast state. The phase structure decomposed into the equilibrium state with FCC+BCC duplex phase after heat treatment, and the hardness of FCC phase is lower than that of BCC phase, therefore, the hardness of alloys decreases.
3.4 Effect of Al content on electrochemical properties of alloys
Electrochemical test method as a modern research method to study the corrosion properties of materials is fast and accurate. The as-cast Al0.5, Al1.0, Al1.5 alloys and 321 stainless steel were made into electrodes and the working faces should be grinded and polished. Electrodes were immersed in 0.5 mol/L NaCl solution and characterized by CHI660C electrochemical workstation, and then the polarization curves of as-cast FeCoNiCrCu0.5Alx alloys and 321 stainless steel were obtained. The polarization curves are shown in Fig. 5. The corrosion potential (φcorr) and corrosion current density (Jcorr) could be acquired by use of extrapolation method of Tafel curve and the results are shown in Table 3. The corrosion resistance of the alloys was analyzed according to the corrosion current density compared with 321 stainless steel.
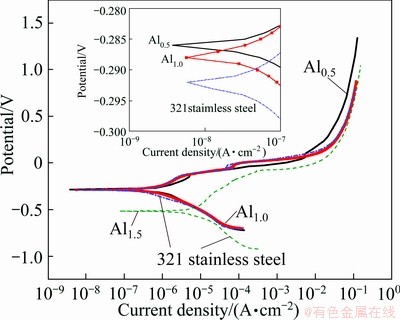
Fig. 5 Polarization curves of FeCoNiCrCu0.5Alx alloys and 321 stainless steel in 0.5 mol/L NaCl solution
Table 3 φcorr and Jcorr of as-cast FeCoNiCrCu0.5Alx high-entropy alloys and 321 stainless steel in 0.5 mol/L NaCl solution

The Jcorr values of as-cast Al0.5, Al1.0, Al1.5 alloys and 321 stainless steel are 1.306×10-6, 8.214×10-7, 1.868×10-5 and 6.822×10-7 A/cm2, respectively. In the chlorine ion medium, the corrosion resistance of Al0.5 alloy is between Al1.0 and Al1.5 alloys. Al1.0 alloy has the best corrosion resistance which is close to that of 321 stainless steel. The as-cast Al1.5 alloy has the lowest corrosion potential and the highest corrosion current density, so its corrosion resistance is the worst. Among as-cast alloys, Al0.5 alloy is of single FCC1 phase, Al1.0 alloy is of single BCC phase but Al1.5 alloy is of BCC+FCC2 duplex phase. Due to the heterogeneous composition, microstructure and stress, micro-anode region and micro-cathode region are formed in the electrolyte solution. Anode region and cathode region constitute the corrosion cell. Therefore, the alloys with two-phase structures are corroded easily and the difference of electrode potentials is more, the Jcorr between micro anode and micro cathode regions is larger. From the phase structure analysis, FCC2 phase is metastable, so FCC2 phase can reduce the corrosion resistance of Al1.5 alloy. The corrosion resistance of BCC solid solution is better than that of FCC solid solution in chlorine ion medium.
Electrodes were immersed in 0.5 mol/L H2SO4 solution and characterized by electrochemical workstation, the polarization curves of as-cast FeCoNiCrCu0.5Alx alloys and 321 stainless steel are shown in Fig. 6. The φcorr and Jcorr are shown in Table 4. All of the as-cast high entropy alloys and 321 stainless steel have an obvious passive region. However, the passivation potential of Al1.0 alloy is the lowest and the passive region is the widest. Therefore, the passivation ability of as-cast Al1.0 alloy is the strongest. The corrosion resistances of as-cast Al0.5, Al1.0 and Al1.5 alloys were compared in acid medium. The corrosion resistance of Al0.5 alloy with FCC phase is also between as-cast Al1.0 and Al1.5 alloys. The as-cast Al1.0 alloy with single BCC phase has the lowest Jcorr which is lower than that of 321 stainless steel. As a result, the corrosion resistance of Al1.0 alloy is the best. The as-cast Al1.5 alloy with BCC+FCC2 duplex phase has the highest Jcorr, so its corrosion resistance is the worst. Therefore, the conclusions of as-cast alloys in acid medium and chlorine ion medium are consistent. The corrosion resistance of as-cast Al1.0 alloy is the best and stronger than that of 321 stainless steel, the corrosion resistance of BCC phase is better than that of FCC phase.
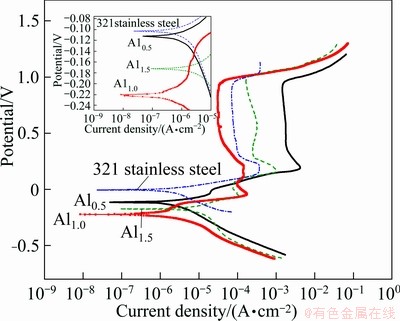
Fig. 6 Polarization curves of FeCoNiCrCu0.5Alx alloys and 321 stainless steel in 0.5 mol/L H2SO4 solution
Table 4 φorr and Jcorr of as-cast FeCoNiCrCu0.5Alx alloys and 321 stainless steel in 0.5 mol/L H2SO4 solution

3.5 Effect of heat treatment on electrochemical properties of alloys
The heat-treated alloys were made into electrodes and were tested by electrochemical workstation in 0.5 mol/L NaCl solution. Figure 7 shows the polarization curves of heat-treated FeCoNiCrCu0.5Alx alloys in 0.5 mol/L NaCl solution, and the φcorr and Jcorr of heat-treated high-entropy alloys are shown in Table 5. The corrosion resistances of Al0.5 and Al1.0 alloys decrease after heat treatment. However, the corrosion resistance of Al1.5 alloy increases after heat treatment. The passivation ability of water-quenched alloys improves slightly.

Fig. 7 Polarization curves of FeCoNiCrCu0.5Alx alloys in 0.5 mol/L NaCl solution
Table 5 φcorr and Jcorr of heat-treated FeCoNiCrCu0.5Alx high-entropy alloys in 0.5 mol/L NaCl solution
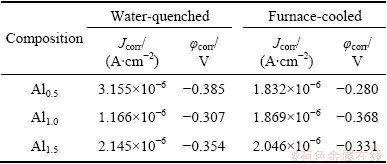
In the chlorine ion medium, heat treatment has no obvious effect on Jcorr for Al0.5 alloy, due to the unchanged phase structure after heat treatment. For Al1.0 alloy, the phase transforms from single BCC phase to FCC1+BCC double structure after heat treatment, the formation of corrosion micro-cell reduces the corrosion resistance of alloys. Although both the as-cast Al1.5 alloy and heat-treated Al1.0 alloys are BCC + FCC duplex phase, the corrosion resistance of the heat-treated Al1.0 alloy is stronger than that of as-cast Al1.5 alloy, because FCC2 phase in as-cast Al1.5 alloy is metastable and can be corroded more easier compared with the steady-state FCC1 phase. For Al1.5 alloy, its corrosion resistance is improved after heat treatment, because the phase structure transforms from BCC + FCC2 structures to single BCC or BCC plus a small amount of FCC1 structure, the metastable state changes into stable state and the structure becomes uniform relatively after heat treatment. The corrosion resistance of steady-state alloys reduces with the increase of Al content, because the stable structure of alloy changes from FCC structure to BCC+FCC duplex structures with the increase of Al content. On the other hand, aluminum has a poor corrosion resistance in the solution of F-, Cl- and other anions contained. It will accelerate the corrosion of aluminum due to the secondary precipitation effect [16], when the solution also contains others metal ions with more positive potential, such as Fe2+, Ni2+ and Cu2+ especially.
4 Conclusions
1) The phase structure of as-cast FeCoNiCrCu0.5Alx alloys evolves from FCC phase to BCC phase with the increase of Al content. The stable phase of FeCoNiCrCu0.5Alx high-entropy alloys is FCC phase when the Al content is lower and the stable phase is FCC+BCC duplex phase when the Al content is higher.
2) In the FeCoNiCrCu0.5Alx high-entropy alloys, the hardness of BCC phase is higher than that of FCC phase, and the corrosion resistance of BCC phase is better than that of FCC phase in both chlorine ion and acid medium.
3) The as-cast Al1.0 alloy has high hardness and excellent corrosion resistance. The corrosion resistance of as-cast Al1.0 alloy is close to that of 321 stainless steel in chlorine ion medium. However, its corrosion resistance is better compared with 321 stainless steel and has stronger passivation ability in acid medium.
References
[1] YEH J W, CHEN S K, LIN S J, GAN J Y, CHIN T S, SHUN T T, TSAU C H, CHANG S Y. Nanostructured high-entropy alloys with multiple principal elements: Novel alloy design concepts and outcomes [J]. Advanced Engineering Materials, 2004, 6(5): 299-303.
[2] YEH J W, CHEN S K. High-entropy alloy [J]. Science Development, 2004, 377: 16-21. (in Chinese)
[3] LIN C M, TSAI H L, BOR H Y. Effect of aging treatment on microstructure and properties of high-entropy Cu0.5CoCrFeNi alloy [J]. Intermetallics, 2010, 18: 1244-1250.
[4] LIN C M, TSAI H L. Equilibrium phase of high-entropy FeCoNiCrCu0.5 alloy at elevated temperature [J]. Journal of Alloys and Compounds, 2010, 489: 30-35.
[5] LI Wei, LIU Gui-zhong, GUO Jing-jie. Microstructure and electrochemical properties of AlFeCuCoNiCrTix high-entropy alloys [J]. Special Casting and Nonferrous Alloys, 2009, 29(10): 941-944. (in Chinese)
[6] TONG C J, CHEN M R, CHEN S K, YEH J W, SHUN T T, LIN S J, CHANG S Y. Mechanical performance of the AlxCoCrCuFeNi high-entropy alloy system with multiprincipal elements [J]. Metallurgical and Materials Transactions A, 2005, 36(5): 1263-1271.
[7] LIU Yuan, CHEN Min, LI Yan-xiang, CHEN Xiang. Microstructure and mechanical performance of AlxCoCrCuFeNi high-entropy alloys [J]. Rare Metal Materials and Engineering, 2009, 38(9): 1602-1607. (in Chinese)
[8] CHEN M R, LIN S J, YEH J W, CHEN S K, HUANG Y S, TU C P. Microstructure and properties of Al0.5CoCrCuFeNiTix (x=0-2.0) high-entropy alloys [J]. Materials Transactions, 2006, 47(5): 1395-1401.
[9] CHEN M R, LIN S J, YEH J W, CHEN S K, HUANG Y S, CHUANG M H. Effect of vanadium addition on the microstructure, hardness and wear resistance of Al0.5CoCrCuFeNi high-entropy alloy [J]. Metallurgical and Materials Transactions A, 2006, 37(5): 1363-1369.
[10] HUANG P K, YEH J W, SHUN T T, CHEN S K. Multi-principal element alloys with improved oxidation and wear resistance for thermal spray coating [J]. Advanced Engineering Materials, 2004, 6(1-2): 74-78.
[11] GAO Jia-cheng, LI Rui. The microstructure and heat-resistance of AlZnMnSnSbPbMg high-entropy alloy [J]. Functional Materials, 2009, 40(4): 602-603. (in Chinese)
[12] WANG Xue-fei, ZHANG Yong, QIAO Yi, CHEN Guo-liang. Novel microstructure and properties of multicomponent CoCrCuFeNiTix alloys [J]. Intermetallics, 2007, 15(3): 357-362.
[13] ZHANG Kui-bao, FU Zheng-yi, ZHANG Jin-yong, SHI Jing, WANG Wei-min, WANG Hao, WANG Yu-cheng, ZHANG Qing-jie. Annealing on the structure and properties evolution of the CoCrFeNiCuAl high-entropy alloy [J]. Journal of Alloys and Compounds, 2010, 502: 295-299.
[14] CANTOR B, CHANG I T H, KNIGHT P, VINCENT A J B. Microstructural development in equiatomic multicomponent alloys [J]. Materials Science and Engineering A, 2004, 375-377: 213-218.
[15] TONG C J, CHEN Y L, CHEN S K, YEH J W, SHUN T T, TSAU C H, LIN S J, CHANG S Y. Microstructure characterization of AlxCoCrCuFeNi high-entropy alloy system with multiprincipal elements [J]. Metallurgical and Materials Transactions A, 2005, 36(4): 881-893.
[16] HSU Y J, CHIANG W C, WU J K. Corrosion behavior of FeCoNiCrCux high-entropy alloys in 3.5% sodium chloride solution [J]. Materials Chemistry and Physics, 2005, 92: 112-117.
[17] LIN C M, TSAI H L. Effect of annealing treatment on microstructure and properties of high-entropy FeCoNiCrCu0.5 alloy [J]. Materials Chemistry and Physics, 2011, 128: 50-56.
[18] LI Wei, LIU Gui-zhong, GUO Jing-jie. Microstructure and electrochemical properties of AlxFeCoNiCrTi high-entropy alloys [J]. Foundry, 2009, 58(5): 431-435. (in Chinese).
李宝玉,彭 坤,胡爱平,周灵平,朱家俊,李德意
湖南大学 材料科学与工程学院,长沙 410082
摘 要:研究Al含量和热处理对FeCoNiCrCu0.5Alx多主元高熵合金的相结构、硬度和电化学性能的影响规律。随着Al含量的增加,铸态合金的相结构由FCC相向BCC相转变。当x从0.5增加到1.5时,FeCoNiCrCu0.5Alx高熵合金的稳定结构由FCC结构向FCC+BCC双相结构转变。BCC相的硬度高于FCC相的,在氯离子及酸性介质中BCC相的耐腐蚀性均优于FCC相的。FeCoNiCrCu0.5Al1.0铸态合金具有高硬度和良好的抗腐蚀性能。
关键词:高熵合金;微观结构;热处理;稳定相;硬度;抗腐蚀性
(Edited by Xiang-qun LI)
Foundation item: Project (NCET-11-0127) supported by the Program for New Century Excellent Talents in University, China; Project supported by the Fundamental Research Funds for the Central Universities, China
Corresponding author: Kun PENG; Tel: +86-731-88822663; E-mail: kpeng@hnu.edu.cn
DOI: 10.1016/S1003-6326(13)62523-6

