Trans. Nonferrous Met. Soc. China 22(2012) s760-s763
Corrosion resistance of composite coating on magnesium alloy using combined microarc oxidation and inorganic sealing
YANG Wei1, WANG Ai-ying1, JIANG Bai-ling2
1. Ningbo Institute of Materials Technology and Engineering, Chinese Academy of Sciences, Ningbo 315201, China;
2. School of Material Science and Engineering, Xi��an University of Technology, Xi��an 710048, China
Received 21 May 2012; accepted 5 November 2012
Abstract: The combined microarc oxidation (MAO) and inorganic sealing process was used to deposit a composite coating to improve the corrosion resistance of AZ31 magnesium alloy. The surface morphologies of the resulting duplex coatings were studied by SEM. Furthermore, the corrosion resistance of the coated Mg alloy substrates was investigated using electrochemical workstation and dropping corrosion test. The results show that the composite coating surface consists of Mg, Si, O and Na. It is difficult to deposit inorganic coating on a thick MAO coating surface. As the composite coating was solidified by CO2 under 175 ��C, it exhibits a better corrosion resistance than the MAO monolayer, owing to the thick and compact inorganic coating.
Key words: AZ31 magnesium alloy; microarc oxidation; composite coating; microstructure; corrosion resistance
1 Introduction
Magnesium and its a alloys are increasingly used in the automotive, aerospace and communication fields because of the flexibility in fabrication and high specific strength. However, the corrosion resistance of magnesium limits its application to these fields [1-3]. Microarc oxidation (MAO) is a simple and clean technique to fabricate ceramic coatings on the metal surface such as aluminum alloy, magnesium alloy, and titanium alloy [4-6]. However, the corrosion resistance of the MAO-coated specimens is limited for the micropores on the ceramic coating surface, which is necessary to use different techniques to seal, as a subsequent treatment for further improving the material performance [7-9]. Generally, the sealing techniques contain heat sealing, cold sealing and polymer topcoat, etc [10,11]. It is found that all these techniques have some deficiencies, such as high cost, low speed or environment pollution. In this experiment, inorganic soluble glass is used to fill into the micropores of ceramic coating formed by MAO due to its low cost and contamination.
This research will focus on the effect of the thickness of ceramic coating and CO2 solidifying treatment on the corrosion resistance of composite coating formed by MAO combined with inorganic sealing. The results will be helpful to improving the corrosion resistance of magnesium alloy significantly and expand its application.
2 Experimental
The substrate material was a commercial grade AZ31magnesium alloy with composition of Al 3.10%, Mn 0.3%, Zn 0.90%, Ca 0.04%, Si 0.05%, Cu 0.01%, Mg balance (mass fraction). Each substrate was shaped as a plate of 25 mm��25 mm��5 mm. The substrates were finished with 1200# aluminum oxide waterproof abrasive paper. The MAO process was conducted by a constant voltage mode on a direct-current (DC) pulsed electrical source, with frequency of 400 Hz and duty cycle of 10%. The MAO coating with different thicknesses was formed by controlling the oxidation time (3, 5 and 10 min) in an aqueous electrolyte of sodium silicate (10.0 g/L) in distilled water with potassium hydroxide (8.0 g/L). The temperature of the solution was kept at 35 ��C during the oxidation. The soluble glass with the modulus of 3 was used for sealing the micropores of ceramic coating. The compositions of soluble glass are shown in Table 1. The temperature of inorganic sealing process was 100 ��C, and the time was 15 min. Furthermore, the composite coating was treated by CO2 solidifying under 175 ��C for 30 min to improve the corrosion resistance of Mg alloy substrate.
Table 1 Properties of soluble glass

The thickness of the coatings was measured using the TT260 eddy-current coating-thickness measurement gauge, the microstructure of composite coatings was observed using a JEOL JSM-6700F scanning electron microscope (SEM) and an energy dispersive spectroscope (EDS) attachment (Oxford INCA) operated at an acceleration potential of 20 kV was used for chemical analysis. The corrosion resistance of the coated samples was measured using CHI660B electrochemical workstation in 5% NaCl (mass fraction) solution at 25 ��C, in which the samples with an exposed area of 1 cm2 were immersed. A three electrode cells with the coated samples as a working electrode, the saturated calomel electrode (SCE) as a reference electrode, and a platinum rod as a counter electrode were used in the corrosion resistance tests. After 10 min of initial delay, the potentiodynamic polarization curves were measured with a scan rate of 4 mV/s during the above electrochemical measurements. The acid resistance of the coated samples was evaluated by observing the required time of changing green of the corrosive liquid. The corrosive solution consisted of 25 mL hydrochloric acid (l.19 g/cm3), 3 g potassium dichromate and 75 mL distilled water. The temperature of the solutions was set at 20 ��C during the corrosion test.
3 Results and discussion
3.1 Microstructure of coatings
Figure 1 shows the surface morphologies of composite coatings with different thicknesses of the ceramic coatings. With increasing the thickness of MAO coating, there are more micropores exposed on the coating surface and the sealing effect gets worse. It is observed that the micropores on the ceramic coating with thickness of 5 ��m completely are covered by inorganic coating. It is supposed that the effect of inorganic sealing to Mg alloy substrate is strongly related to the adsorption character of the micropores. EDS analysis was conducted on different composite coatings, and the results are shown in Table 2. It illustrates that the composite coatings are mainly composed of O. Silicon content from the soluble glass decreases with increasing the thickness of ceramic coating. Mg from the substrate is also discovered in the coating, which indicates that the organic coating is thin.

Fig. 1 Surface morphologies of composite coatings with different thicknesses of ceramic coatings
Table 2 Composition of surface of coatings (mass fraction, %)
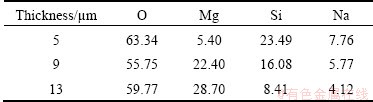
Figure 2 illustrates the surface features of the coatings formed by different techniques. It can be seen that a large number of discharge micropores ranged from 0.1 ��m to 3 ��m appear on the ceramic coating surface (Fig. 2(a)), which is related to the growth mechanism of MAO coating. As the micropores are sealed by the soluble glass, it shows that the inorganic coating is thin and the surface profile of ceramic coating is clear (Fig. 2(b)). While the composite coating is treated by CO2 solidifying under 175 ��C for 30 min, the inorganic coating becomes thick and compact, and the surface profile of ceramic coating can not be observed (Fig. 2(c)). It is believed that this compact coating is helpful to improving the corrosion resistance of the coated sample.

Fig. 2 Surface morphologies of coatings prepared by different techniques
3.2 Corrosion resistance of coatings
The corrosion behavior of the coatings formed by different techniques was evaluated by the electro- chemical potentiodynamic polarization in a 5% NaCl solution. Figure 3 demonstrates the potentio-dynamic polarization curves of the coated samples. The corrosion current density (Jcorr) derived from the potentiodynamic polarization curves are shown in Table 3. The data clearly shows that both of the composite coatings exhibit a higher positive corrosion potential and a lower corrosion current density than the MAO monolayer. Compared with the ceramic coating, the corrosion current densities of the composite coatings decrease nearly four orders of magnitude. This experiment illustrates that the composite coatings formed by MAO and inorganic sealing to magnesium alloy provide an effective corrosion protection in the solutions containing Cl-, which can be attributed to the chemical inertness of the outer inorganic coating [12,13].

Fig. 3 Polarization behaviors of magnesium alloy treated by different techniques
Table 3 Corrosion current density (Jcorr) of magnesium alloy treated by different techniques

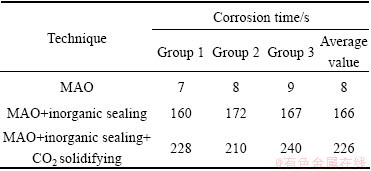
The results of the coated samples treated by three techniques in dropping corrosion test are listed in Table 4. Corrosion spot on the ceramic coating surface will be observed after only 8 s and the MAO coated sample has been destroyed. When the micropores of ceramic coating are sealed by the organic coating, the time can reach 166 s in the acid resistance test. In the experiment, the composite coating is solidified by CO2 and the time can further increase to 226 s. So, it also shows that the inorganic sealing is an effective method to improve the corrosion resistance of MAO coating.
Table 4 Acid resistance of magnesium alloy treated by different techniques
3.3 Discussion
The surface morphologies of the coatings formed by MAO and inorganic sealing to magnesium alloy indicated that the inorganic coating can be easily deposited on the ceramic coating surface. It has been proved that the MAO process is a balance of growth and dissolution [14,15], and lots of Mg2+ ions dispersed into the solution. During the process of soluble glass sealing to magnesium alloy, 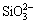 ions are adsorbed by the micropores of ceramic coating, which results in the combination of Mg2+ and
ions are adsorbed by the micropores of ceramic coating, which results in the combination of Mg2+ and 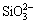 (shown in Eq. 1). It is known that the diameter of micropores increases with the increasing thickness of ceramic coating. So it may be difficult for soluble glass to combine with ceramic coating.
(shown in Eq. 1). It is known that the diameter of micropores increases with the increasing thickness of ceramic coating. So it may be difficult for soluble glass to combine with ceramic coating.
Mg2+ +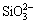 =MgSiO3 �� (1)
=MgSiO3 �� (1)
It can also be seen that the inorganic coating is so thin that the surface profile of ceramic coating surface is visible, and the protective effect of this composite coating is limited. In this experiment, the composite coating is further treated by CO2 solidifying, and the inorganic coating is thick and compact (Fig. 3(c)). It is considered that the reaction between 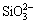 ions and CO2 takes place under 175 ��C, and the phase of silicon oxide has been produced on the ceramic coating surface, which insulates the substrate from the corrosive ions.
ions and CO2 takes place under 175 ��C, and the phase of silicon oxide has been produced on the ceramic coating surface, which insulates the substrate from the corrosive ions.
4 Conclusions
1) A composite coating can be prepared on magnesium alloy by MAO and inorganic sealing, and the effect of inorganic sealing gets worse with increasing the thickness of ceramic coating.
2) The composite coating is composed of Mg, Si, O and Na, and the trends of Si and Mg with the thickness of ceramic coating are opposite. The inorganic coating solidified by CO2 can is thick and compact.
3) The corrosion test shows that the acid resistance of the composite coating increases strongly compared with the MAO monolayer, and it is helpful to further improving the corrosion resistance of the composite coating solidified by CO2.
References
[1] CHEN F, ZHOU H, YAO B, QIN Z, ZHANG Q F. Corrosion resistance property of the ceramic coating obtained through microarc oxidation on the AZ31 magnesium alloy surfaces [J]. Surf Coat Technol, 2007, 201: 4905-4908.
[2] BARCHICHEE C E, ROCCA E, HAZAN J. Corrosion behaviour of Sn-containing oxide layer on AZ91D alloy formed by plasma electrolytic oxidation [J]. Surf Coat Technol, 2008, 202: 4145-4152.
[3] ZHANG Y J, YAN C W, WANG F H, LOU H Y, CAO C N. Study on the environmentally friendly anodizing of AZ91D magnesium alloy [J]. Surf Coat Technol, 2002, 161: 36-43.
[4] WANG Y H, WANG J, ZHANG J B, ZHANG Z. Effects of spark discharge on the anodic coatings on magnesium alloy [J]. Mater Lett, 2006, 60: 474-478.
[5] GUO H F, AN M Z, HUO H B, XU S, WU L J. Microstructure characteristic of ceramic coatings fabricated on magnesium alloys by micro-arc oxidation in alkaline silicate solutions [J]. Appl Surf Sci, 2006, 252: 7911-7916.
[6] LIANG J, HU L T, HAO J C. Characterization of microarc oxidation coatings formed on AM60B magnesium alloy in silicate and phosphate electrolytes [J]. Appl Surf Sci, 2007, 253: 4490-4496.
[7] LIANG J, HU L T, HAO J C. Improvement of corrosion properties of microarc oxidation coating on magnesium alloy by optimizing current density parameters [J]. Appl Surf Sci, 2007, 253: 6939-6945.
[8] WANG Y Q, WU K, ZHENG M Y. Effects of reinforcement phases in magnesium matrix composites on microarc discharge behavior and characteristics of microarc oxidation coatings [J]. Surf Coat Technol, 2006, 201: 353-360.
[9] GUO H F, AN M Z. Growth of ceramic coatings on AZ91D magnesium alloys by micro-arc oxidation in aluminate�Cfluoride solutions and evaluation of corrosion resistance [J]. Appl Surf Sci, 2005, 246: 229-238.
[10] DUAN H P, DU K Q, YAN C W, WANG F H. Electrochemical corrosion behavior of composite coatings of sealed MAO film on magnesium alloy AZ91D [J]. Electrochimica Acta, 2006, 51: 2898-2908.
[11] LIANG J, WANG P, HU L T, HAO J C. Tribological properties of duplex MAO/DLC coatings on magnesium alloy using combined microarc oxidation and filtered cathodic arc deposition [J]. Mater Sci Eng A, 2007, 454-455: 164-169.
[12] LEI W, WANG Y M, ZHOU Y, GUO L X, OUYANG J H. Microstructure and corrosion resistance of modified 2024 Al alloy using surface mechanical attrition treatment combined with microarc oxidation process [J]. Corros Sci, 2011, 53: 473-480.
[13] LEI W, WANG Y M, ZHOU Y, OUYANG J H, GUO L X, JIA D C. Corrosion evaluation of microarc oxidation coatings formed on 2024 aluminium alloy [J]. Corros Sci, 2010, 52: 2687-2696.
[14] LI J M, CAI H, XUE X N, JIANG B L. Growth mechanism of black ceramic layers formed by microarc oxidation [J]. Mater Lett, 2007, 201: 8702-8708.
[15] XUE W B, DENG Z W, CHEN R Y, ZHANG T H. Growth regularity of ceramic coatings formed by microarc oxidation on Al-Cu-Mg alloy [J]. Thin Solid Films, 2000, 372: 114-117.
(Edited by LONG Huai-zhong)
Foundation item: Project (51201176) supported by the National Natural Science Foundation of China; Project (2006BAE04B05-1) supported by the National Key Technologies of Research and Development Program of China
Corresponding author: YANG Wei; Tel: +86-574-86685036; Fax: +86-574-86685159; E-mail: yangwei_smx@nimte.ac.cn
DOI: 10.1016/S1003-6326(12)61800-7