Power production enhancement with polyaniline composite anode in benthic microbial fuel cells
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2018���3��
�������ߣ��Ⱥ� ����� ������
����ҳ�룺499 - 505
Key words��benthic microbial fuel cell; anode modification; polyaniline; high power output
Abstract: In this study, conductive polymer polyaniline (PANI) is employed to modify the anodes of benthic microbial fuel cells (BMFC). Four electrochemical methods are used to synthesize the polyaniline anodes; the results show that the PANI modification, especially the pulse potential method for PANI synthesis could obviously improve the cell energy output and reduce the anode internal resistance. The anode is modified by PANI doped with Fe or Mn to further improve the BMFC performance. A maximum power density of 17.51 mW/m2 is obtained by PANI-Fe anode BMFC, which is 8.1 times higher than that of control. The PANI-Mn anode BMFC also gives a favorable maximum power density (16.78 mW/m2). Fe or Mn modification has better effect in improving the conductivity of polyaniline, thus improving the energy output of BMFCs. This work applying PANI composite anode into BMFC brings new development prospect and could promote the practical application of BMFC.
Cite this article as: JIA Yu-hong, QI Zhen-lian, YOU Hong. Power production enhancement with polyaniline composite anode in benthic microbial fuel cells [J]. Journal of Central South University, 2018, 25(3): 499�C505. DOI: https://doi.org/10.1007/s11771-018-3754-3.

J. Cent. South Univ. (2018) 25: 499-505
DOI: https://doi.org/10.1007/s11771-018-3754-3

JIA Yu-hong(�����)1, 2, QI Zhen-lian(������)2, YOU Hong(�Ⱥ�)1, 2
1. State Key Laboratory of Urban Water Resource and Environment, Harbin Institute of Technology,Harbin 150090, China;
2. School of Marine Science and Technology, Harbin Institute of Technology at Weihai, Weihai 264209, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2018
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2018
Abstract: In this study, conductive polymer polyaniline (PANI) is employed to modify the anodes of benthic microbial fuel cells (BMFC). Four electrochemical methods are used to synthesize the polyaniline anodes; the results show that the PANI modification, especially the pulse potential method for PANI synthesis could obviously improve the cell energy output and reduce the anode internal resistance. The anode is modified by PANI doped with Fe or Mn to further improve the BMFC performance. A maximum power density of 17.51 mW/m2 is obtained by PANI-Fe anode BMFC, which is 8.1 times higher than that of control. The PANI-Mn anode BMFC also gives a favorable maximum power density (16.78 mW/m2). Fe or Mn modification has better effect in improving the conductivity of polyaniline, thus improving the energy output of BMFCs. This work applying PANI composite anode into BMFC brings new development prospect and could promote the practical application of BMFC.
Key words: benthic microbial fuel cell; anode modification; polyaniline; high power output
Cite this article as: JIA Yu-hong, QI Zhen-lian, YOU Hong. Power production enhancement with polyaniline composite anode in benthic microbial fuel cells [J]. Journal of Central South University, 2018, 25(3): 499�C505. DOI: https://doi.org/10.1007/s11771-018-3754-3.
1 Introduction
Benthic microbial fuel cell (BMFC) converts chemical energy into electrical energy directly via electro-active micro-organisms, degrading organic matter in marine sedimentary mud. A BMFC consists of an anode embedded in the anoxic sediment and a cathode suspended in the aerobic water column and it has gotten considerable attention to serve as a remote source of power to drive pelagic instruments work for a long term. However, the low power density of BMFC in current technologies limits its practical application [1].
Anode material is known to be an important factor for BMFC since it plays a key role on the biofilm formation and power generation [2�C4]. Therefore, to find superior anode materials and good modification methods is a crucial problem, and is attracting more attention [5]. Various materials for anode modification had been investigated. Conductive polymer polyaniline has been used as electrode modifier for the performance improvement of biosensors and MFCs, due to its high conductivity at room temperature, easy for synthesis and environmental stability [6]. ZHAO et al [7] synthesized hydroxylated and aminated polyaniline nanowire networks and used it as anode materials in MFCs. The results showed that the cell electrical outputs were enhanced. LAI et al [6] reported that charge transfer was facilitated by using a HSO4- doped polyaniline modified carbon cloth anode, and the MFC power generation was improved.
In addition, anode modified with iron or manganese compound has been reported to enhance the power output from MFC and BMFC [8]. FU et al [9] employed manganese oxide (MnO2)/ multiwall carbon nanotubes composites as anodes in BMFCs and results indicate that the modified anode has a better wettability, greater kinetic activity and higher power density than that of the plain graphite anode. Meanwhile, Fe/ferric oxide modified anode was fabricated by electrolytic deposition [10]; the modified anode has a lower surface contact angle and high wettability, which favors the adhesion of bacteria and presents 17.4-fold higher output power than plain graphite. An electrodeposited hybrid film of polyaniline and manganese oxide in nanofibrous structures was developed by SUN et al [11] for electrochemical supercapacitor and results showed that the hybrid films have larger effective areas than plain PANI, and display higher electrochemical activities.
Based on these observations, we hypothesized that increase in the anode performance could be made by PANI, PANI-Fe or PANI-Mn modified anode. PANI anode was synthesized by 4 electrochemical methods. The fabricated anodes were then tested by electrochemical impedance spectroscopy analysis and in BMFC. The PANI anodes prepared by pulse potential method had the best electrochemical performance, thus this method was further used to synthesize PANI-Fe and PANI-Mn anode. The electrochemical activity of these fabricated anodes was tested, and the BMFC performance with these anodes was examined.
2 Materials and methods
2.1 Electrode preparation
Graphite felt (Sanye Carbon Co., Ltd. China) served as electrode material with a dimension of 5��50��37.5 mm. All graphite felts used in this study were pretreated by 1 mol/L HCl for 15 min, washed with deionized water and dried in air. The PANI was synthesized in a three-electrode electrochemical cell, with an Ag/AgCl reference electrode, a platinum-wire counter electrode, and a graphite-felt working electrode (on which polymers were synthesized). The electrolyte contained 1 mol/L aniline and 0.5 mol/L HClO4. Four electrochemical methods for polyaniline anodes synthesis were conducted: potentiostatic method (PM), pulse potential method (PPM), cyclic voltammetry (CV) and linear sweep voltammetry (LSV). The synthesis conditions are shown in Table 1. The PANI synthesis was carried out under room temperature. After treatment, the anodes were washed with deionized water 3 times, and then placed in a drying oven drying.
Table 1 Synthesis methods of PANI anodes

The PANI-Fe and PANI-Mn anodes were prepared by PPM method as deduced before. The electrolyte used and the synthesis condition are shown in Table 2.
Table 2 Synthesis methods of PANI-Fe and PANI-Mn anodes

2.2 BMFCs construction and operation
Graphite felt electrodes loaded with PANI, PANI-Fe, PANI-Mn were used as anodes and graphite felt electrodes without electrocatalyst were used as comparisons and cathodes. In this setup, the cell was of cylindrical shape (15 cm in diameter,15 cm in height) and made of polypropylene. The natural seawater and marine sediment utilized here were obtained from Mazi Port, Weihai, China (37��32.5��N, 122��4.4��E). The anode was buried 4 cm below the sediment surface, and the cathode was 11 cm above the anode. Each cell experiment was conducted in three parallels to obtain average values. The reactor was operated at room temperature during the whole experiment.
PANI anode was synthesized by four electrochemical methods. The fabricated anodes were then tested by electrochemical impedance spectroscopy analysis and in BMFC. PANI-Fe and PANI-Mn anodes were further synthesized by pulse potential method. The electrochemical activity of these fabricated anodes was tested, and the BMFC performance (open circuit voltage and polarization test) with these anodes was examined.
2.3 Analysis and calculation
BMFC potentials were measured using a digital multimeter (KEITHLEY, American Tektronix corporations). Current (I) was calculated as I=cell voltage/external resistance, and power (P) was calculated as P=I��cell voltage. The current and power were normalized to the projected area of anode surface to get power and current density. Electrochemical activities were tested by CV and LSV using an electrochemical workstation (CHI660E, Chenhua Instruments Co., Ltd, China). CV and LSV were carried out with anode as working electrode, cathode as counter electrode, and an Ag/AgCl electrode as reference electrode [12]. For all tests, triplicate runs were carried out and the relative standard deviation was generally less than 10%. The PANI, PANI-Fe and PANI-Mn anode samples were structurally characterized by X-ray diffraction (XRD, DX-2700, Dandong Haoyuan Instrument Co., LTD). The morphology of anode samples was examined with a scanning electron microscope (SEM, Thermo Electron Nicolet 380).
3 Results and discussion
3.1 Electrocatalytic characterizations of PANI anodes
The PANI anodes synthesized by four different electrochemical methods previously were applied to BMFCs. The maximum open circuit voltages (OCV) of 0.73 V (control), 0.83 V (PM), 0.84 V (PPM), 0.75 V (CV) and 0.85 V (LSV) are achieved after about 10 d inoculation (data not shown). Electrochemical impedance spectroscopy (EIS) analysis was then carried out to investigate the charge transfer resistance of different anodes. The EIS Nyquist plot is the superimposition of a preceding frequency-dependent semi-circle (high frequency region) and a subsequent straight line (low frequency region), the diameter of the former representing the charge transfer resistance (Rct), which usually represents the resistance of electrochemical reactions on the electrode [13]. The anode resistance is also affected by the solution resistance (Rs, representing the resistance from electrolyte, electrode materials and the contract resistance) [14]. EIS of the PANI anodes is shown in Figure 1. Rs of the PANI modified anodes and the control is all about 1 ��, which might be due to the same sediment environment for all anodes. Rct of the anodes is 16.2 �� (PM), 11.1 �� (PPM), 3.5 �� (LSV), 11.1 �� (CV) and 16.0 �� (control), respectively. Obviously, the PANI synthesized by different electrochemical methods reduces the charge transfer resistance at various degrees, confirming that the electron transfer efficiency of the PANI anode is higher than that of control [15]. Similar results were obtained in previous study. An Rct of 18 �� was achieved by HOU et al [15] with a PANI modified carbon cloth anode MFC as compared with the 47 �� obtained with the unmodified carbon cloth.
Additionally, the straight line over low frequency region is characteristic of the diffusion- limiting step. The straight line region of the PANI anodes is significantly smaller than the control anode, especially for the PANI anode synthesized by PPM method. The narrow region of the diffusion-controlled process indicates that the PANI anodes, especially the PANI anode synthesized by PPM method, have a good structure for reactants to access the reaction centers without a diffusion limit over a wide frequency range [14], thus improving the diffusion of electrolyte toward the electrode surface [15].
Polarization test was carried with the PANI anode BMFCs and results are shown in Figure 2.
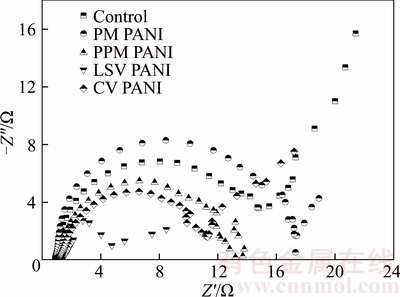
Figure 1 EIS of PANI anodes synthesized by different methods

Figure 2 Polarization curve and power density curve of PANI anodes BMFCs
Bare control exhibits a maximum power density of 2.16 mW/m2, which is the lowest among the studied anodes. A maximum power density of 9.93 mW/m2 is obtained from the BMFC using PANI anode (synthesized by PPM method), and the corresponding current density is 33.8 mA/m2. With the anode modification, conductive PANI was attached to the graphite surface, which could enhance the charge transfer efficiency and cell-material interaction [16], and thus improve the power output. The maximum power density achieved in this study is 9.0 times the 1.1 mW/m2 reported by HONG et al [17] with sediment MFC.
The surface morphologies of PANI anodes and control were examined by SEM (Figure 3). For the control and the PANI anode synthesized by PPM method, the image magnification is 1000, 2000, 5000 times according to order from left to right. The PPM PANI anode gives rise to a rough surface and a compact structure. In contrast, the plain graphite felt anode consists of carbon fibers which construct a much looser network structure, and each fiber features a relatively smooth surface. The surface morphologies of PANI anodes synthesized by PM, LSV and CV method were given with 5000 times magnification. The results indicate that PANI was successfully deposited to all of the modified anodes.
Previous research [18, 19] showed that the morphology and properties of polyaniline prepared by different methods were different. Using pulse position method [20], the consumption of deposition ions in electrode-solution interface can be replenished within pulse interval. So, high maximum current density could be applied in the synthesis process, and the formed grain size of PANI is smaller than the direct current deposit [21]. Synthesis of PANI using PPM is not easy to form thick crystals as this method could change the grain growth trend with the existence of pulse interval, thus hampering the crystals growth and reducing the epitaxial growth of crystals. It could be concluded that the small PANI grain synthesized by the PPM method has high specific surface area and enhanced conductivity, and thus could obviously improve the BMFC performance.

Figure 3 SEM images of PANI and control anodes:
3.2 Electrocatalytic characterizations of PANI composite anodes
The PANI-Fe and PANI-Mn modified anodes were inoculated in BMFCs and after stable voltage production of the cells, EIS analysis was carried out to investigate the anode resistance. EIS results are shown in Figure 4. Rs values of all anodes are about 1.0 ��. The charge transfer resistance of anodes is 15.8 �� (control), 11.3 �� (PANI), 6.6 �� (PANI-Fe) and 7.2 �� (PANI-Mn), respectively. Obviously, the electron transfer efficiency of the PANI-Fe and PANI-Mn anode is much higher than that of control (plain graphite felt) or plain PANI anode [15]. The EIS of PANI-Mn anode shows a short diffusion control process at low frequency region, and the EIS of PANI-Fe anode displays almost no diffusion control process, indicating that the PANI-metal composite modification could reduce the diffusion limit in significant measure. The results demonstrate that the modified anode could improve the anodic reaction rate and reduce the anode internal resistance of BMFC.
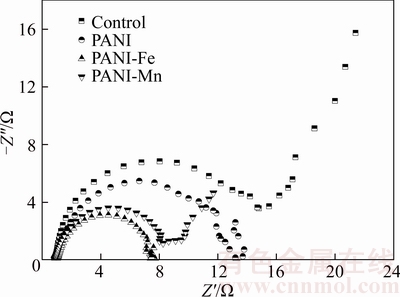
Figure 4 EIS of PANI-Fe and PANI-Mn anodes in BMFCs
Polarization and power density curve of BMFCs using various anodes are shown in Figure 5. A maximum power density of 17.51 mW/m2 is achieved with PANI-Fe anode in BMFC, which is 8.1 times higher than that of control, and the corresponding current density is 56.8 mA/m2. The PANI-Mn anode in BMFC also gives a favorable maximum power density (16.78 mW/m2), which is much higher than the 10.12 mW/m2 obtained from the plain PANI anode in BMFC. For Fe and Mn ions have strong metal and catalytic activity, PANI-Fe and PANI-Mn anodes in BMFCs give better performance in energy production. The essence of doping protons and metal ions into PANI main chain could deduce that the dopant seizes electrons from �� orbit, or adds electrons to �� empty electronic track, thus making the energy level difference between adjacent orbital decrease, and improving the electrical conductivity [22]. Iron and manganese ion, with more positive activity than proton, are relatively easy to seize electrons from �� orbit, or add electrons to �� empty electronic track, thus have better effect on improving the conductivity of polyaniline [19], and then improve the power output of BMFCs.
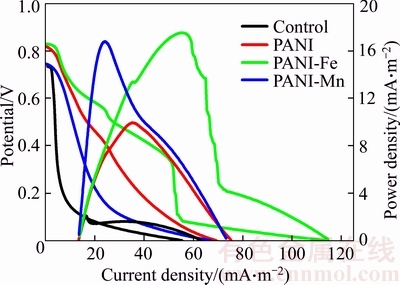
Figure 5 Polarization curve and power density curve of PANI-Fe and PANI-Mn anodes in BMFCs
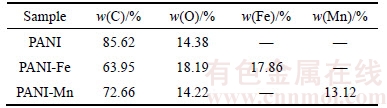
Figure 6 presents the SEM images of PANI-Fe and PANI-Mn anodes. As shown in the figure, the control is in the form of smooth surface and no attachments. However, a few of nanofibers are attached to the carbon fiber of PANI anode, and give rise to a rough surface. In contrast, the polyaniline nanofibers on PANI-Fe anode surface condense into scale and generate fine particles concomitantly, while the surface of PANI-Mn anode has plentiful particles with no nanofibers. It proves that the present method is effective for forming PANI-Fe or PANI-Mn on graphite felt in this study. The elements contents of the modified anodes were determined by EDX. As SEM coupled EDX microprobe analysis was semi-quantitative, analysis of several spots was carried out and an average value for each sample was considered. The results of the representative samples are summarized in Table 3. The results indicate that Fe/Mn was successfully doped into the PANI-Fe/PANI-Mn anodes.

Figure 6 SEM images of PANI-Fe and PANI-Mn anodes
Table 3 Elements content of PANI, PANI-Fe and PANI-Mn anodes
X-ray diffraction (XRD) was employed to confirm the successful fabrication of the PANI- Metal composite (Figure 7). In the pattern of the pristine PANI sample, one well-defined peak is observed at around 25��, which is the amorphous peak of the polyaniline; other impurity peak probably is the side reaction of polymerization process, which can be ignored for its faint peak intensity. The patterns of the PANI-Fe and PANI-Mn display almost the same peak with the pattern of PANI, indicating that Fe and Mn are doped into polyaniline chain in the shape of metal ions without metal crystals forming. Results obtained in this study demonstrate that the PANI-Fe and PANI-Mn anode exhibit a favorable and stable electrochemical behavior. It could be concluded that the iron or manganese doping into the PANI main chain could increasing the conductivity of polyaniline, thus increase the electron transfer function on the electrode surface.

Figure 7 XRD patterns of PANI, PANI-Fe and PANI-Mn anode
4 Conclusions
1) A relative low charge transfer resistance and a short diffusion control process are examined from the PANI anode, proving that polyaniline could reduce the anode internal resistance in significant measure. The PANI anode also promotes the BMFC power output.
2) The maximum power densities of 17.51 mW/m2 and 16.78 mW/m2 are obtained from the PANI-Fe and PANI-Mn anode BMFCs, which are 8.1 and 7.7 times higher than that of control. With the PANI-metal modification, Fe and Mn serving as both dopant and oxidant, have better effect on improving the conductivity of polyaniline, thus improving the cell power output.
References
[1] DONOVAN C, DEWAN A, PENG H, HEO D, BEYENAL H. Power management system for a 2.5 W remote sensor powered by a sediment microbial fuel cell [J]. Journal of Power Sources, 2011, 196(3): 1171�C1177.
[2] LIU B, WEINSTEIN A, KOLLN M, GARRETT C, WANG L, BAGTZOGLOU A, KARRA U, LI Y, LI B. Distributed multiple-anodes benthic microbial fuel cell as reliable power source for subsea sensors [J]. Journal of Power Sources, 2015, 286: 210�C216.
[3] LIU B, WILLIAMS I, LI Y, WANG L, BAGTZOGLOU A, MCCUTCHEON J, LI B. Towards high power output of scaled-up benthic microbial fuel cells (BMFCs) using multiple electron collectors [J]. Biosensors and Bioelectronics, 2016, 79: 435�C441.
[4] JIANG Hai-ming, LUO Sheng-jun, SHI Xiao-shuang, DAI Meng, GUO Rong-bo. A system combining microbial fuel cell with photobioreactor for continuous domestic wastewater treatment and bioelectricity generation [J]. Journal of Central South University, 2013, 20(2): 488�C494.
[5] HAO Li-mei, YAN Xiao-le, XIE You, ZHANG Tao, CHEN Zhi. A rapid one-step electrodeposition process for fabrication of superhydrobic surfaces on anode and cathode [J]. Journal of Central South University, 2016, 23(7): 1576�C1583.
[6] LAI Bin, TANG Xing-hua, LI Hao-ran, DU Zhu-wei, LIU Xin-wei, ZHANG Qian. Power production enhancement with a polyaniline modified anode in microbial fuel cells [J]. Biosensors and Bioelectronics, 2011, 28(1): 373�C377.
[7] ZHAO Y, NAKANISHI S, WATANABE K, HASHIMOTO K. Hydroxylated and aminated polyaniline nanowire networks for improving anode performance in microbial fuel cells [J]. Journal of Bioscience and Bioengineering, 2011, 112(1): 63�C66.
[8] SUN Min, SONG Wei, ZHAI Lin-feng, RU Xiao-rui, CUI Yu-zhi. Elucidating electro-oxidation kinetics of Fe(II) in the anode of air�Ccathode fuel cells from an Fe(II) speciation perspective [J]. Chemical Engineering Journal, 2013, 228: 781�C789.
[9] FU Yu-bin, YU Jian, ZHANG Ye-long, MENG Yao. Graphite coated with manganese oxide/multiwall carbon nanotubes composites as anodes in marine benthic microbial fuel cells [J]. Applied Surface Science, 2014, 317: 84�C89.
[10] FU Yu-bin, XU Qian, ZAI Xue-rong, LIU Yuan-yuan, LU Zhi-kai. Low electrical potential anode modified with Fe/ferric oxide and its application in marine benthic microbial fuel cell with higher voltage and power output [J]. Applied Surface Science, 2014, 289: 472�C477.
[11] SUN L, LIU X, LAU K K T, CHEN L, GU W. Electrodeposited hybrid films of polyaniline and manganese oxide in nanofibrous structures for electrochemical supercapacitor [J]. Electrochimica Acta, 2008, 53(7): 3036�C3042.
[12] JIA Yu-hong, YE Long, KANG Xi, YOU Hong, WANG Shu-tao, YAO Jie. Photoelectrocatalytic reduction of perchlorate in aqueous solutions over Ag doped TiO2 nanotube arrays [J]. Journal of Photochemistry and Photobiology A: Chemistry, 2016, 328: 225�C232.
[13] HE Z, MANSFELD F. Exploring the use of electrochemical impedance spectroscopy (EIS) in microbial fuel cell studies [J]. Energy & Environmental Science, 2009, 2(2): 215�C219.
[14] QIAO Yan, LI Chang-ming, BAO Shu-juan, BAO Qiao-liang. Carbon nanotube/polyaniline composite as anode material for microbial fuel cells [J]. Journal of Power Sources, 2007, 170(1): 79�C84.
[15] HOU Jun-xian, LIU Zhong-liang, ZHANG Pei-yuan. A new method for fabrication of graphene/polyaniline nanocomplex modified microbial fuel cell anodes [J]. Journal of Power Sources, 2013, 224: 139�C144.
[16] WANG Ya-qiong, LI Bin, ZENG Li-zhen, CUI Dan, XIANG Xing-de, LI Wei-shan. Polyaniline/mesoporous tungsten trioxide composite as anode electrocatalyst for high- performance microbial fuel cells [J]. Biosensors and Bioelectronics, 2013, 41: 582�C588.
[17] HONG S W, CHANG I S, CHOI Y S, CHUNG T H. Experimental evaluation of influential factors for electricity harvesting from sediment using microbial fuel cell [J]. Bioresource Technology, 2009, 100(12): 3029�C3035.
[18] WEI X L, WANG Y Z, LONG S M, BOBECZKO C, EPSTEIN A J. Synthesis and physical properties of highly sulfonated polyaniline [J]. Journal of the American Chemical Society, 1996, 118(11): 2545�C2555.
[19] MACDIARMID A G, CHIANG J C, RICHTER A F, EPSTEIN A J. Polyaniline: A new concept in conducting polymers [J]. Synthetic Metals, 1987, 18(1�C3): 285�C290.
[20] WANG Hong-zhi, LIU Wei-hong, LI Jian, YAO Su-wei, ZHANG Wei-guo. Preparation and characterization of polyaniline film by potentiostatic pulse method [J]. Chemical Journal of Chinese Universities, 2012, 33(2): 421�C425.
[21] YOO J E, BUCHOLZ T L, JUNG S, LOO Y L. Narrowing the size distribution of the polymer acid improves PANI conductivity [J]. Journal of Materials Chemistry, 2008, 18(26): 3129�C3135.
[22] RAY A, ASTURIAS G E, KERSHNER D L, RICHTER A F, MACDIARMID A G, EPSTEIN A J. Polyaniline: Doping, structure and derivatives [J]. Synthetic Metals, 1989, 29(1): 141�C150.
(Edited by YANG Hua)
���ĵ���
�۱������������Ժ�������ȼ�ϵ�صIJ���Ӱ��
ժҪ�����о����õ绯ѧ����̼ձ�����ı���ԭλ�ϳɾ۱�����۱����C��/�̸������Ը��������ĵ绯ѧ���ܣ�������ߺ�������ȼ�ϵ�صIJ��ܡ��ֱ����4�ַ����ϳɾ۱������������λ���������λ����ѭ�������������Է����������������������������Ĵ��ڣ����������λ���ϳɵľ۱������п���С���ȱ��������ŵ㣬����������ܹ�����Ч�ؽ�����������ͬʱ��ߺ�������ȼ�ϵ�صĵ�����������������λ���ϳɾ۱����C��/�̸���������̼ձ�����������������/�̽����IJ��ӽ�һ����ǿ�˸��Ե缫�ĵ������ܣ�����ʹ��صĹ����������Ϊδ���Ե缫��8.1��7.7�������о��״ν��۱������临�����������Ӧ�õ���������ȼ�ϵ���У����ƶ���������ȼ�ϵ�ص�ʵ�û�����һ�����塣
�ؼ��ʣ���������ȼ�ϵ�أ��������ԣ��۱�����ǿ������
Foundation item: Project(HIT.NSRIF.2014128) supported by the Fundamental Research Funds for the Central Universities, China; Project(2014M551257) supported by the China Postdoctoral Science Foundation; Project(WH20150208) supported by the Subject Development Foundation of Harbin Institute of Technology at Weihai, China
Received date: 2016-06-07; Accepted date: 2017-12-20
Corresponding author: YOU Hong, Professor, PhD; Tel: +86�C631�C5687243; E-mail: youhong@hit.edu.cn; ORCID: 0000-0001-9454- 1681

