
Anodization of AZ91 magnesium alloy in alkaline solution containing silicate and corrosion properties of anodized films
LI Ling-ling(李玲玲)1, CHENG Ying-liang(程英亮)1, WANG Hui-min(王慧敏)1, ZHANG Zhao(张 昭)2
1. College of Materials Science and Engineering, Hunan University, Changsha 410082, China;
2. Department of Chemistry, Zhejiang University, Hangzhou 310027, China
Received 9 July 2007; accepted 9 October 2007
Abstract: The anodization of AZ91 magnesium alloy in an alkaline electrolyte of 100 g/L NaOH+20 g/L Na2B4O7?10H2O+50 g/L C6H5Na3O7?2H2O+60 g/L Na2SiO3?9H2O was studied. The corrosion resistance of the anodized films was studied by electrochemical impedance spectroscopy(EIS) and potentiodynamic polarization techniques. The microstructure of the films was examined with scanning electronic microscope(SEM) and X-ray diffractometer(XRD). The results show that, under the experimental conditions, the optimum anodizing time and the optimum anodizing current density are 40 min and 20 mA/cm2 respectively for obtaining the anodic film with high corrosion resistance. The XRD pattern shows that the components of the anodized film consist of MgO and Mg2(SiO4).
Key words: AZ91 magnesium alloy; anodized coating; corrosion
1 Introduction
Magnesium alloys are light structural metals, which makes them one of the most favorite materials to minimize the mass of vehicle and therefore to reduce exhaust gas emissions in transport applications[1-5]. Despite good mechanical properties, such as high specific strength, high specific stiffness, damping characteristic and being easy to be recycled, their applications are limited severely for their low corrosion resistance. So, it is quite urgent and significant to improve the anti-corrosion property of magnesium alloys in their industrial application[6-8].
Surface treatments can provide corrosion protection for magnesium alloys. A number of surface modification and coating methods have been developed and used. Typical surface modification techniques include physical surface modification, chemical conversion, electro- chemical anodizing, chemical and electroplating and organic coatings. Among these techniques, anodizing treatment is one of the most promising methods for magnesium alloys[9-14]. Although an anodizing treatment is usually more expensive than a chemical surface treatment, it is always selected for applications requiring high performance, such as wear resistance and heavy duty paint preparation.
In this work, the anodization of AZ91 magnesium alloy in an environment-friendly electrolyte that contains 100 g/L sodium hydroxide+20 g/L borate+50 g/L citrate+60 g/L sodium silicate was investigated and the corresponding corrosion resistance of the films was evaluated. The structure and property of the anodized film were studied by SEM, XRD, EIS and potentiodynamic polarization curves.
2 Experimental
2.1 Specimens
AZ91 Mg alloy with a nominal composition (mass fraction, %) of 9.0 Al, 1.0 Zn and balance Mg was chosen. In order to ensure the same surface roughness of different tested samples (10 mm×10 mm×3 mm), the working electrodes were successively polished using 280-grit, 600-grit, 1200-grit and 2000-grit abrasive paper, then carefully degreased with acetone and rinsed with distilled water.
2.2 Anodization
All anodizing experiments were performed in 500 mL alkaline solutions containing 100 g/L NaOH, 60 g/L Na2SiO3・9H2O, 50 g/L C6H5Na3O7・2H2O and 20 g/L Na2B4O7・10H2O. The anodizing processes were carried out at a constant current density on a DYY-6C (600 V, 1 000 mA) power supplier. The specimen was the anode and the steel plate acted as the cathode. The experiment was performed at room temperature. To maintain a uniform distribution of temperature, the bath solution was stirred by a magnetic stirrer in the container. The anodized specimen was washed with flowing tap water and distilled water, then dried for the following tests. The cell voltages were recorded during anodization. The effects of anodizing time and current density were studied in this work.
2.3 Evaluation of corrosion performance
The corrosion performance of the anodic film was measured in neural 3.5% (mass fraction) NaCl solutions using electrochemical impedance spectroscopy(EIS) and potentiodynamic polarization technique which were carried out using a commercial Mode CHI660C (CH Instrument, Inc.3700 Tennison Hill Driver Austin, TX 78738-5012 USA) electrochemical alanalyzer/ workstation at ambient temperature. A three-electrode cell with anodized samples as working electrode, saturated calomel electrode(SCE) as reference electrode and platinum sheet as counter electrode was employed in the test. The ratio of volume of neutral 3.5% (mass fraction) NaCl solutions to sample area is 500 mL/cm2. Before the EIS measurement, the working electrodes were immersed in the NaCl solution for 10 min. EIS was measured at the open circuit potential(OCP) from 10 000 to 0.005 Hz with a perturbative voltage amplitude of 5 mV. After the EIS measurement, the sample was also measured by potentiodynamic test with a scan rate of 0.5 mV/s from -0.1 V vs open circuit potential to -0.7 V. All potentials reported in this paper are referred to the SCE.
2.4 Microstructure and chemical composition of anodized film
The surface microstructures of the anodized coatings on the specimen after anodization were examined with a JEOL 6700F scanning electron microscope(SEM).
The chemical composition of each anodized film was determined by X-ray diffractometer with Cu Kα radiation (SIMENS D5000).
3 Results and discussion
3.1 Effect of anodizing time
The voltage―time response for the anodization of AZ91 magnesium alloy at 20 mA/cm2 in an alkaline silicate solution is highly reproducible, and three regions are identified, as shown in Fig.1. In the first region (0-60 V), the voltage increases sharply and no apparent sparks are found on the metal surface. In the second region (60-100 V), the cell voltage continues to increase at a moderately fast rate. The inflection point on the voltage―time curve at 60 V corresponds to the breakdown voltage, beyond which a large number of small size sparks are observed on the electrode surface. In the third region (beyond 100 V), a steady state is established on the anode surface and the cell voltage reaches a relatively stable value. A bright spark appears only at localized location on anode surface and moves slowly across the surface.

Fig.1 Cell voltage curves of anodizing process at 20 mA/cm2
To study the effect of anodizing time on the properties of anodized films, samples with different anodizing time are first prepared and the applied current density of the anodizing process is 20 mA/cm2. Then the corrosion resistant properties of the anodized AZ91 magnesium alloy are evaluated by the EIS and potentiodynamic polarization tests. Fig.2 shows the Nyquist plots and Bode-phase plots of AZ91 magnesium alloy after being anodized with various time.

Fig.2 EIS of anodized films with different treatment time at 20 mA/cm2: (a) Nyquist plots; (b) Bode-phase plots
The EIS results of anodized samples at different time display one capacitive loop (at the high and middle frequency range) and a low-frequency inductive component. The high and middle frequency capacitive loop may arise from the anodic film, while in the low-frequency region some discrete points occur. The diameter of the capacitive semicircle of a measured Nyquist spectrum is closely related to the corrosion rate [15], the larger the semicircle is, the better the corrosion resistance is. It can be seen in Fig.2, with the processing time increasing, the corrosion resistance of the anodized films becomes better. When the treatment time is 40 min, there is an optimum value. However, the corrosion resistance of the anodic films declines instead when the treatment time exceeds 40 min.
Fig.3 shows the potentiodynamic polarization results, and the free corrosion current densities(Jcorr) are calculated and listed in Table 1.
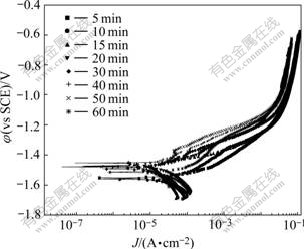
Fig.3 Potentiodynamic polarization results of anodized films with different treatment time
Table 1 Fitted result of free corrosion current density in Fig.3
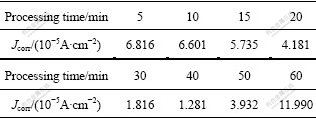
Table 1 shows that Jcorr of the AZ91 sample with anodizing time of 40 min is the lowest of those processing time. The free corrosion current density is directly related to the corrosion resistance of the anodized anodized film. The less the free corrosion current density, the higher the corrosion resistance of the coatings. Then it can be seen that the results of the potentiodynamic tests are consistent with the EIS results. This proves that the two techniques are nearly equivalent in predicting the corrosion resistance of the anodic specimens.
3.2 Effect of current density
The applied anodic current density is also an important factor of the anodizing process. In this study, the anodizing processes were carried out at constant current density of 5, 10, 15, 20, 25 and 30 mA/cm2, respectively, and the treatment time was chosen as 40 min. After anodization, the samples were also examined by electrochemical tests in 3.5% (mass fraction) NaCl solutions.
The EIS and potentiodynamic polarization tests of the samples with different anodizing current densities are shown in Figs.4 and 5, respectively. The EIS features are similar to the EIS behavior in Fig.2, and the Jcorr values can also be acquired from the potentiodynamic polarization curves in Fig.5 and the data are listed in Table 2.

Fig.4 EIS of anodic film with different current densities anodized for 40 min: (a) Nyquist plots; (b) Bode-phase plots

Fig.5 Potentiodynamic polarization tests of samples with different current densities anodized for 40 min
Table 2 Fitted result of free corrosion current density in Fig.5

It is clearly seen in Fig.4 that, the applied current density of 15 mA/cm2 and 20 mA/cm2 shows nearly the same EIS value of the anodized films for AZ91 magnesium alloy and the best corrosion resistance in our study. However, in Table 2, the optimum anodizing current density of the anodized film for AZ91 magnesium alloy with best corrosion resistance is 20 mA/cm2. When current density is less than 20 mA/cm2 in this experiment, the corrosion resistance of the films increases with current density due to the fact that the film growth rate is higher at larger current density. The film formed at higher current density with the same anodizing time will be thicker and thus is more protective. However, when the current density exceeds 20 mA/cm2, the corrosion resistance of the anodized films begins to decrease with current density. It can be concluded that although the high current density can increase the growth rate of the film and shorten the time to a requested film thickness, there exists a contrary effect. High current density would make the growth of the film too fast, which would result in coarser oxide grain and the internal stress of the anodized film will increase, resulting in micro-cracks on the films. As a result, the corrosion resistance of the anodized films will decrease.
These mentioned above indicate that the optimal anodizing current density for AZ91 magnesium alloy is 20 mA/cm2.
3.3 Morphology and XRD pattern of anodized coating
The surface microstructures of the anodized coatings with different treatment time at 20 mA/cm2 on AZ91 magnesium alloy are shown in Fig.6. All the ceramic-like anodized coatings are porous. The diameters of the pores are from several micrometers to more than ten micrometers. When the processing time is short, the surface of the film is rather smooth and the pores are small. With the time increasing, rough surface appears and contains large pores. Besides, when the processing time is too long, some cracks appear, which can be seen in Fig.6(d). The corrosion of the anodic film probably starts with these pores and cracks.

Fig.6 SEM images of surface morphology of samples anodized with 20 mA/cm2 at different time: (a) 5 min; (b) 20 min; (c) 40 min; (d) 60 min
The cross-sectional morphologies of AZ91 magnesium alloy with different treatment time after anodizing at 20 mA/cm-2 are shown in Fig.7. As demonstrated in Fig.7(a), with 5 min treatment, it has a film with a thickness of about several micrometers. With increasing the processing time, the film thickness increases to about 20 ?m, as seen in Fig.7(b). The treatment time continues to increase, and the film thickness also increases, which can be seen in Figs.7(c) and (d). However, for the anodic film is not compact, the corrosion resistance of them is not good.

Fig.7 Cross-sectional morphologies of samples anodized with 20 mA/cm2 at different time: (a) 5min; (b) 20 min; (c) 40 min; (d) 60 min
Fig.8 shows the XRD pattern of the film anodized for 60 min at 20 mA/cm2 on AZ91 magnesium alloy. The film was scraped off from the AZ91 substrate by a glass cutter with diamond tip, thus there is no interfering signal from the magnesium substrate. It can be seen form the XRD pattern that MgO, not hydroxide, is the main compound existing in the anodic film. Mg2SiO4 is also identified in the XRD pattern. However, it is noticed that no peaks associated with borate are seen in the XRD pattern. It is concluded that the content of borate is very small or exists in amorphous form and cannot be detected by XRD technique.
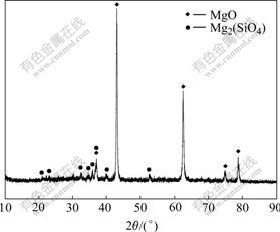
Fig.8 XRD pattern of film anodized for 60 min at 20 mA/cm2
4 Conclusions
1) The anodization of AZ91 magnesium alloy was studied in an alkaline solution of 100 g/L NaOH+20 g/L Na2B4O7?10H2O+50 g/L C6H5Na3O7?2H2O+60 g/L Na2SiO3?9H2O. The influence of treatment time was investigated at a fixed current density of 20 mA/cm2. The results show that the corrosion resistance of the anodic film first increases with the increase of treatment time and then decreases after an optimum time, and the optimal treatment time is 40 min.
2) The effect of applied anodic current densities was also studied. The results show that the optimum current density is about 20 mA/cm2.
3) The compositions of the anodized film of AZ91 magnesium alloy in this electrolyte solution are mainly MgO and Mg2SiO4, which results in a uniform and corrosion resistive film.
References
[1] GRAY J E, LUAN B. Protective coatings on magnesium and its alloys―A critical review [J]. J Alloys Compd, 2002, 336: 88-113.
[2] AVEDESIAN M M, BAKER H. Magnesium and magnesium alloys [M]. USA: ASM International, 1999.
[3] ABBAS G, LIU Z, SKELON P. Corrosion behavior of laser-melted magnesium alloys [J]. Applied Surface Science, 2005, 247(1/4): 347-353.
[4] ROSALBINO F, ANGELINI E, DE NEGRI S. Effect of erbium addition on the corrosion behavior of Mg-Al alloys [J]. Intermetallics, 2005, 13(1): 55-60.
[5] CHENG Ying-liang, WU Hai-lan, CHEN Zhen-hua, WANG Hui-min, LI Ling-ling. Phosphating process of AZ31 magnesium alloy and corrosion resistance of coatings [J]. Trans Nonferrous Met Soc China, 2006, 16(5):1086-1091.
[6] SHI Zhi-ming, SONG Guang-ling, ATRENS A. The corrosion performance of anodized magnesium alloys [J]. Corrosion Science, 2006, 48(11): 3531-3546.
[7] WU Hai-lan, CHENG Ying-liang, LI Ling-ling, CHEN Zhen-hua, WANG Hui-min, ZHANG Zhao. The anodization of ZK60 magnesium alloy in alkaline solution containing silicate and the corrosion properties of the anodized films [J]. Applied Surface Science, 2007, 253(24): 9387-9394.
[8] SHI Zhi-ming, SONG Guang-ling, ATRENS A. Influence of anodizing current on the corrosion resistance of anodized AZ91D magnesium alloy [J]. Corrosion Science, 2006, 48(8): 1939-1959.
[9] HSIAO H Y, TSAI W T. Characterization of anodic films formed on AZ91 magnesium alloy [J]. Surface & Coatings Technology, 2005, 190: 299-308.
[10] HSIAO H Y, TSUNG H C, TSAI W T. Anodization of AZ91D magnesium alloy in silicate-containing electrolytes [J]. Surface & Coatings Technology, 2005, 199(2/3): 127-134.
[11] MIZUTANI Y, KIM S J, ICHINO R, OKIDO M. Anodizing of Mg alloys in alkaline solutions [J]. Surface & Coatings Technology, 2003, 169/170: 143-146.
[12] ZHANG L J, FAN J J, ZHANG Z, CAO F H, ZHANG J Q, CAO C N. Study on the anodic film formation process of AZ91D magnesium alloy [J]. Electrochimica Acta, 2007, 52(7): 5325-5333.
[13] WU C S, ZHANG Z, CAO F H, ZHANG L J, ZHANG J Q, CAO C N. Study on the anodizing of AZ31 magnesium alloys in alkaline borate solutions [J]. Applied Surface Science, 2007, 253(8): 3893-3898.
[14] ZHANG Y J, YAN C W, WANG F H, LOU H Y, CAO C N. Study on the environmentally friendly anodizing of AZ91D magnesium alloy [J]. Surface and Coating Technology, 2002, 161(1): 36-43.
[15] SONG Guang-ling, BOWLES A L, STJOHN D H. Corrosion resistance of aged die cast magnesium alloy AZ91D [J]. Material Science and Engineering A, 2004, 366(1): 74-86.
Foundation item: Project(50471043) supported by the National Natural Science Foundation of China
Corresponding author: CHENG Ying-liang; Tel: +86-13036798588; E-mail: chengyl @ hnu.cn; deepblacksea@163.com
(Edited by LI Xiang-qun)