����ϩ��ˮ����������ȥ��ˮ��ͭ�Ķ���ѧ�ͻ���
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2014���10��
�������ߣ�Tongsai JAMNONGKAN Krittiya KANTAROT Kitiya NIEMTANG P. Pungboon PANSILA Amnuay WATTANAKORNSIRI
����ҳ�룺3386 - 3393
Key words��heavy metals removal; hydrogel; wastewater; adsorption; diffusion mechanism
ժ Ҫ���о�����ϩ��ˮ�����Է�ˮ��ͭ�������������ֱ�ʹ�ú������������ˮ���ⶨ�����о�ˮ�����Ļ�ѧ�ṹ��������Ϊ���������������ϩ�������ȩ�����γɾ���ϩ��ˮ������������������ˮ�Զ����ֳ��ߵ�ƽ�����ͱȡ����⣬�������������̷�����������ѧ����ɢ�����Ӽ���ɢ��Ĥ��ɢ���ơ�
Abstract: Recently, a renewed interest in techniques for heavy metal removal of wastewater has been growing because of embarking opportunities for industrial applications. We investigated the adsorption capacity of the copper on the poly (vinyl alcohol) hydrogel from the aqueous solution. Chemical structure and water absorption of the hydrogel were studied using FTIR and water uptake measurement, respectively. The results showed that the poly (vinyl alcohol) was crosslinked with glutaraldehyde, and the hydrogel highly exhibited the equilibrium swelling ratio because of its hydrophilicity property. Additionally, it was found that the adsorption process followed the pseudo-second-order kinetics and the mechanism diffusion was controlled by particle and film diffusions.
Trans. Nonferrous Met. Soc. China 24(2014) 3386-3393
Tongsai JAMNONGKAN1, Krittiya KANTAROT1, Kitiya NIEMTANG1, P. Pungboon PANSILA1, Amnuay WATTANAKORNSIRI2
1. Department of Fundamental Science and Physical Education, Faculty of Science at Siracha, Kasetsart University, Chonburi 20230, Thailand;
2. Department of Agriculture and Environment, Faculty of Science and Technology, Surindra Rajabhat University, Surin 32000, Thailand
Received 1 November 2013; accepted 16 January 2014
Abstract: Recently, a renewed interest in techniques for heavy metal removal of wastewater has been growing because of embarking opportunities for industrial applications. We investigated the adsorption capacity of the copper on the poly (vinyl alcohol) hydrogel from the aqueous solution. Chemical structure and water absorption of the hydrogel were studied using FTIR and water uptake measurement, respectively. The results showed that the poly (vinyl alcohol) was crosslinked with glutaraldehyde, and the hydrogel highly exhibited the equilibrium swelling ratio because of its hydrophilicity property. Additionally, it was found that the adsorption process followed the pseudo-second-order kinetics and the mechanism diffusion was controlled by particle and film diffusions.
Key words: heavy metals removal; hydrogel; wastewater; adsorption; diffusion mechanism
1 Introduction
Heavy metals are serious causes of environmental pollutions affecting living microorganism and human with regards to their non-biodegradation and then accumulation in environmental compartments (air- water-soil-biota). Particularly, water is very important to organisms and is a dissolving medium of heavy metals. Many toxic heavy metals to human, such as copper (Cu), lead (Pb), cadmium (Cd), and chromium (Cr) [1,2], should be removed from wastewater before discharging to environments. Especially, Cu is an essential element in trace amount but at a higher level it is toxic to plants, animals, and humans [3]. Recently, Cu is widely used in various important industrial applications, so its removal and recovery from wastewater are significant for the environmental protection and human health.
Nowadays, numerous processes exist for removing dissolved heavy metals, including chemical precipitation [4,5], photocatalytic degradation [6], membrane filtration [7], electrochemical technology [8], reverse osmosis [9,10], and adsorption [11]. Among these processes, adsorption is one of the most effective and economical methods for removing the heavy metals from aqueous solutions [12-15].
Considering environmental concerns, poly (vinyl alcohol) (PVA) is a water soluble polymer with characteristics of non-toxicity, biocompatibility and biodegradability [16,17]. Another crucial factor enhancing the suitability of PVA is hydrophilicity owing to its regular linear structure with a large number of side hydroxyls on the molecular chain [18,19]. Hence, PVA has been manufactured into a variety of adsorbent types, such as ion-exchange film [20] and hydrogel [21,22]. PVA is a semi-crystal hydrophilic polymer consisting of one hydroxyl group in each repeat unit with being crosslinkable representing desirable adsorbent structure. Its mechanical strength and chemical stability can be enhanced through crosslinking by chemical reaction, which can be defined as three-dimensional polymeric network [23]. Like other hydrogels, the PVA hydrogel shows faster adsorption kinetics for removing heavy metals from aqueous solution [24]. Recently, several research groups have investigated the removal of Cu with different types of adsorbent materials such as PVA/chitosan [22,25], PVA semi-IPN poly (acrylic acid)/ tourmaline [26], and PVA/SiO2 [27].
Surprisingly, there is less report about treatment of Cu(II) from aqueous solution using only PVA so far. Therefore, in the present study, we investigated the kinetics of Cu(II) ion adsorption with the neat PVA hydrogel as a novel adsorbent. The PVA hydrogel was prepared from PVA with glutaraldehyde as a crosslinking agent. The synthesized PVA hydrogel was investigated by Fourier transform infrared spectroscopy and water uptake capacity test, as well as batch adsorption equilibrium in order to study the effect of pH, contact time and temperature. Additionally, the kinetics and mechanism of adsorption were studied by using pseudo-first-order, pseudo-second-order and intraparticle diffusion models.
2 Experimental
2.1 Materials
Poly(vinyl alcohol) (Mw=140000 g/mol, Mw/Mn= 3.0, 87%-89% hydrolyzed) was purchased from Sigma�C Aldrich (Germany). Glutaraldehyde was obtained from Fluka (Switzerland) and CuSO4��5H2O was from Merck (Germany). All other chemicals were of analytical grade, and were used in as-received state without further purification.
2.2 Preparation of PVA hydrogel
PVA aqueous solution (10% in mass fraction) was prepared by dissolving PVA powder in distilled water at 90 ��C with constant stirring for 2 h, and then cooled to room temperature. Crosslinking solution (5.71 mL) prepared from 50% (mass fraction) methanol, 10% acetic acid, 1.25% glutaraldehyde and 10% sulfuric acid with solution mass ratio of 3:2:1:1 was added into PVA solution (10.0 mL) under constant stirring. After that, the mixture was poured into a Petri disk and the gel was then formed within 30 min. The prepared PVA hydrogel was dried at 40 ��C in a vacuum oven overnight.
2.3 FTIR spectroscopy
The PVA hydrogel was characterized by a Fourier transform infrared (FTIR) spectrophotometer (FT-710, Horiba FTIR) to verify its composition and chemical structure from functional groups. The samples were prepared by film casting techniques, and then the spectrum for each sample was recorded in the wavenumber range of 4000-400 cm-1 at room temperature.
2.4 Water uptake capacity of PVA hydrogel
The ability of the PVA hydrogel to absorb medium was studied to understand the diffusion of medium and heavy metals into the PVA hydrogel, which is essential for heavy metal removal application. The water uptake capacity (Wc) of the PVA hydrogel was evaluated using the following equation.
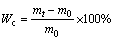 (1)
(1)
where mt and m0 refer to the mass of swollen adsorbent hydrogel at time t and dry state, respectively. Pre-weighed dry PVA hydrogel sample, defined as m0, was immersed into a certain amount of deionized water. After immersion in water, the samples were taken out from water at certain time intervals (every 10 min), gently rapidly dried with filter paper to remove the excess water, and subsequently weighed to define mt. All the tests were performed in triplicate (n=3) under identical conditions.
2.5 Adsorption capacity
The adsorption capacity of Cu(II) ions onto the PVA hydrogel was carried out at ambient condition. Dry PVA hydrogel (1 g) was introduced into the Erlenmeyer flask with solution (30 mL) containing 50 mg/L Cu(II) ions. The flasks were placed in a thermostatic stirrer and agitated at the fixed speed (100 r/min). Samples were taken periodically for measurement of aqueous phase at each time interval. The concentration of remaining Cu(II) ions in solution was estimated by an atomic absorption spectrophotometer (AAS) (AAnalyst 200, Perkin Elmer) [28]. The effect of contact time, pH and temperature was studied. For each condition, three samples (n=3) were investigated. The adsorption capacity (qe) of the Cu(II) ions onto the PVA hydrogel was calculated using the following equation:
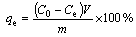 (2)
(2)
where C0 and Ce refer respectively to the initial and the equilibrium concentrations of Cu(II) ions solution; V is the volume of the Cu(II) ions solution; and m is the mass of the PVA hydrogel. The adsorption of Cu(II) ions was determined by
 (3)
(3)
where C0 and Ct are the concentrations at initial and time t of Cu(II) ions solution, respectively.
2.6 Kinetics of adsorption and mechanism
The kinetic parameters are useful in predicting the adsorption rate used as important information in designing and modeling of the adsorption process. Therefore, we investigated the kinetic rate of the Cu(II) ion adsorption on the PVA hydrogel using pseudo- first-order and pseudo-second-order models. Additionally, the intraparticle diffusion or Webber and Morris model was used to investigate the mechanism of Cu(II) ion adsorption.
3 Results and discussion
3.1 FTIR spectrum
FTIR spectroscopy was carried out to elucidate the presence of reaction of crosslink in the PVA molecules. Figure 1 shows the FTIR spectra of the PVA and PVA hydrogel. The FTIR spectrum of PVA shows a predominant broad band around 3338 cm-1 relating to the stretching O��H group, of which frequency has been modified by the intermolecular and intramolecular hydrogen bonds [29]. Peaks around at 2939 cm-1 and at 1450 cm-1 correspond to the C��H stretching and ��CH2�� bending from alkyl groups, respectively. The sharp peak at 1735 cm-1 indicates the presence of acetate groups of poly(vinyl acetate) [29]. This is related to the partial (87%-89%) hydrolyzation of the PVA mentioned earlier. An important peak is verified at a wavenumber of 1098 cm-1 that is mostly attributed to the crystallization of PVA, related to carboxyl stretching group [30,31]. For the IR spectrum of PVA hydrogel, all of the characteristic peaks can be still observed in the PVA, but essentially expresses a new peak at 1231 cm-1, corresponding to the C��O��C groups. Therefore, we tentatively suggested that the FTIR results indicated the possibility that the PVA could be successfully crosslinked with glutaraldehyde by a formation of acetal bridges within PVA structure, as presented in our previous work [32].
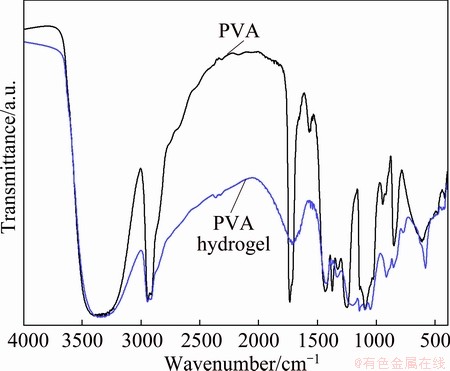
Fig. 1 FTIR spectra of PVA and PVA hydrogel
3.2 Water uptake capacity of PVA hydrogel
Figure 2 shows the swelling behavior of the PVA hydrogel in distilled water as a function of time. The PVA hydrogel absorbed water readily upon soaking during which a rapid swelling rate was observed at the beginning within 10 min, and a saturation value, approximately 250%, was gradually achieved within approximately 40 min. This result revealed that the PVA hydrogel can be used as adsorbent for removing heavy metals from the aqueous solution as mentioned earlier.
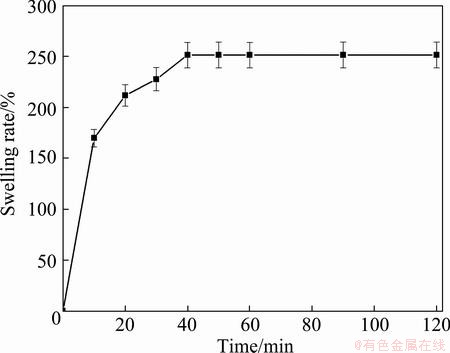
Fig. 2 Swelling rate of PVA hydrogel as function of time
3.3 Adsorption capacity
Basically, the removal efficiency of the system depends on the contact time, pH and temperature. Therefore, they were investigated to understand their effects on adsorption capacity. The experimental results will be presented in the following sub-sections.
3.3.1 Effect of contact time
Figure 3 shows the adsorption rate of Cu(II) ion with the time. The removal rate of Cu(II) ion increased with the contact time increasing. A sharp increase was observed at the initial time within 20 min and attained an optimum at the time of 40 min. The results could be explained by following the time duration of adsorption process. First, the Cu(II) ion rapidly reached the boundary layer of the adsorbent by mass transfer. Second, it slowly diffused from boundary layer film to the adsorbent surface. This behavior is probably explained that the availably external sites of the adsorbent were occupied. Finally, it diffused into the porous structure of the adsorbent. This is in agreement with the results of SENTHILKUMAAR et al [33].
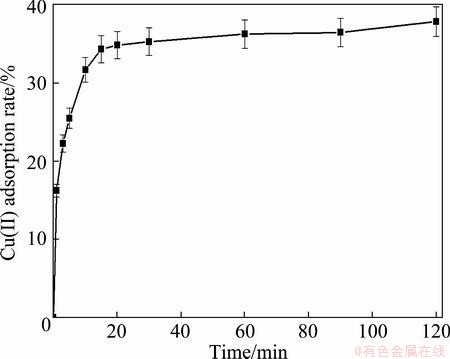
Fig. 3 Adsorption rate of Cu(II) ion as function of contact time
3.3.2 Effect of pH
pH plays a vital role in adsorption of Cu(II) ion because hydrogen ions in the solution compete with the positive charge interacted with the negative charge on active sites of adsorbent [34,35]. Effect of pH on the adsorption characteristics was determined in the pH range of 4-7. The study showed that the system was strongly pH dependent. As shown in Fig. 4, the equilibrium adsorption capacity increased with higher pH of the Cu(II) solution. The maximum value of the equilibrium adsorption capacity of the Cu(II) ion on the PVA hydrogel occurs at pH 5, and then declines at pH value higher than 5. To our best knowledge, this behavior can be explained as the following three steps. First, at low pH the positive charge density is high due to many protons adsorbed on surface; thus, the electrostatic repulsion is also high during uptaking of metal ions, resulting in lower removal efficiency. Second, with increasing pH value, electrostatic repulsion decreases because of the reduction of positive charge density on sorption sites, resulting in an enhancement of metal ions adsorption [36]. Finally, at higher optimized pH value (pH>5), OH- ions completely react with metal ions at the active sites on absorbent surface; then, the adsorption capacity decreases [37].
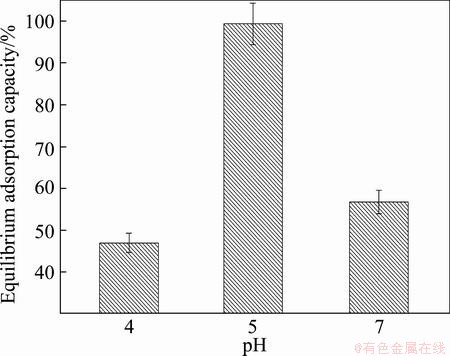
Fig. 4 Relationship between equilibrium adsorption capacity of PVA hydrogel and pH
3.3.3 Effect of temperature
The influence of temperature on the adsorption capacity of the PVA hydrogel for Cu(II) ion was investigated at 30 ��C and 60 ��C. The results obtained are represented in Fig. 5. The equilibrium adsorption capacity was significantly affected by temperature. The result showed that the efficiency of Cu(II) adsorption was favorable at high temperature. This is probably due to the adsorption expressing an endothermic in nature, in agreement with the results of ALMEIDA et al [38] and BULUT and AYDIN [35].
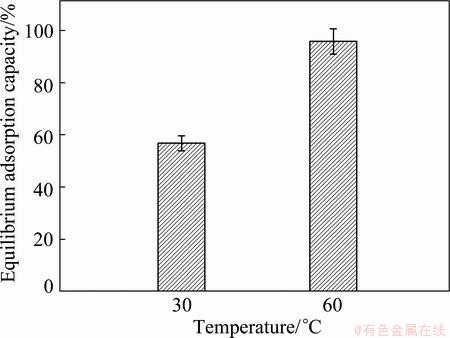
Fig. 5 Equilibrium adsorption capacity of Cu(II) ion as function of temperature
3.4 Kinetics and mechanism
Common sorption kinetic models, i.e. pseudo- first-order and pseudo-second-order, were considered to fit the obtained adsorption data.
3.4.1 Pseudo-first-order model
The pseudo-first-order equation or Lagergren��s equation is given by
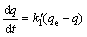 (4)
(4)
where 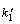 is the rate constant of pseudo-first-order adsorption; q is the amount of metal ions adsorbed at time t; t is the contact time; and qe is the amount of metal ion adsorbed onto adsorbent at equilibrium state. After integrating with the initial conditions, the equation becomes
is the rate constant of pseudo-first-order adsorption; q is the amount of metal ions adsorbed at time t; t is the contact time; and qe is the amount of metal ion adsorbed onto adsorbent at equilibrium state. After integrating with the initial conditions, the equation becomes
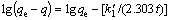 (5)
(5)
The rate constant, 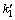 , can be determined from the slope of linear plots between lg(qe�Cq) and t. Figure 6 shows the pseudo-first-order kinetic plot for the Cu(II) ion adsorption with temperature. At 30 ��C, the rate constant of pseudo-first-order model exhibits very low value of 0.048 min-1, and R2=0.912, as summarized in Table 1. At 60 ��C, the results of the rate constant and correlation coefficient were low values, similar to those from the temperature of 30 ��C. Therefore, we can conclude that this model was unsatisfied for use as a model for prediction of the kinetic adsorption of Cu(II) ion onto the PVA hydrogel.
, can be determined from the slope of linear plots between lg(qe�Cq) and t. Figure 6 shows the pseudo-first-order kinetic plot for the Cu(II) ion adsorption with temperature. At 30 ��C, the rate constant of pseudo-first-order model exhibits very low value of 0.048 min-1, and R2=0.912, as summarized in Table 1. At 60 ��C, the results of the rate constant and correlation coefficient were low values, similar to those from the temperature of 30 ��C. Therefore, we can conclude that this model was unsatisfied for use as a model for prediction of the kinetic adsorption of Cu(II) ion onto the PVA hydrogel.
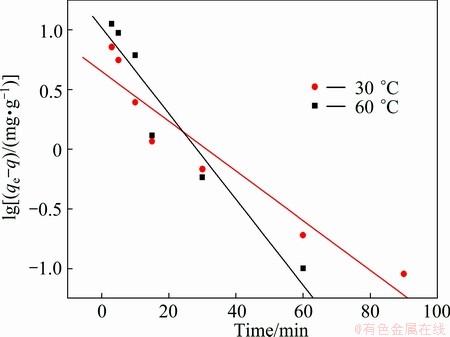
Fig. 6 Pseudo-first-order kinetic plot for Cu(II) ion adsorption
Table 1 Pseudo-first-order and pseudo-second-order kinetic models for adsorption of Cu(II) ion on PVA hydrogel at temperatures of 30 ��C and 60 ��C

3.4.2 Pseudo-second-order model
The pseudo-second-order model can be represented by
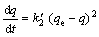 (6)
(6)
where 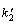 is the rate constant of pseudo second order model. After integrating with the initial conditions and rearrangement, the obtained equation is shown by Eq. (7).
is the rate constant of pseudo second order model. After integrating with the initial conditions and rearrangement, the obtained equation is shown by Eq. (7).
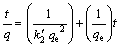 (7)
(7)
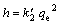 (8)
(8)
where h is the initial adsorption rate. The 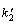 and h can be calculated from the slope and intercept of the linear plot of t/q versus t, according to Eq. (7). The plots for PVA hydrogel at temperature of 30 ��C and 60 ��C for Cu(II) ion adsorption are presented in Fig. 7. The values for
and h can be calculated from the slope and intercept of the linear plot of t/q versus t, according to Eq. (7). The plots for PVA hydrogel at temperature of 30 ��C and 60 ��C for Cu(II) ion adsorption are presented in Fig. 7. The values for 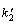 and h were calculated and concluded in Table 1. The correlation coefficient, R2, of PVA hydrogel for this model were very high (0.999), close to unity. Hence, it can be established that the adsorption of Cu(II) ion on the PVA hydrogel perfectly followed the pseudo- second-order kinetic model. It can be concluded from the value of
and h were calculated and concluded in Table 1. The correlation coefficient, R2, of PVA hydrogel for this model were very high (0.999), close to unity. Hence, it can be established that the adsorption of Cu(II) ion on the PVA hydrogel perfectly followed the pseudo- second-order kinetic model. It can be concluded from the value of 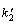 of Cu(II) ion that the reaction-taking place was of the pseudo-second-order. Additionally, the temperature also affected the rate constant of the Cu(II) ion adsorption, as illustrated in Table 1.
of Cu(II) ion that the reaction-taking place was of the pseudo-second-order. Additionally, the temperature also affected the rate constant of the Cu(II) ion adsorption, as illustrated in Table 1.
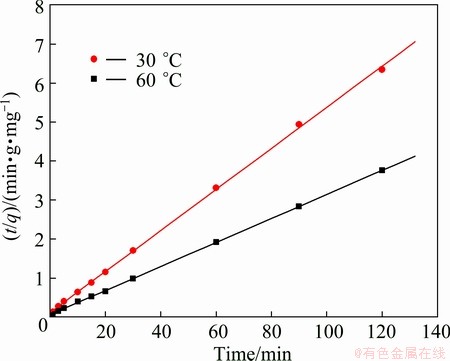
Fig. 7 Pseudo-second-order kinetic plots for adsorption of Cu(II) ion
3.4.3 Adsorption mechanism
The kinetics of the Cu(II) ion adsorption on the PVA hydrogel was further studied according to mass transfer or intraparticle diffusion model. It is well known that the overall rate of the adsorption process is controlled by the slowest step, called the rate-limiting step. This step in a batch system can be determined from the properties of the solute and adsorbent. To our best knowledge, the rate of adsorption process depends on the structural properties of adsorbent (i.e., porosity, specific area and particle size), the properties of metal ions (i.e., ionic radius and number of coordinations), the concentration of metal ions and the interactions between metal ions and active sites of adsorbent [39]. In adsorption systems, the intraparticle diffusion or rate-limiting step can also be expressed using Weber and Morris model. This equation has been described by several groups such as CABAL et al [40] and SIVARAJ et al [41].
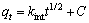 (9)
(9)
where kint is the initial rate of intraparticle diffusion; qt is the amount of solute adsorbed at time t. According to Eq. (9), kint can be determined from linear plot qt as a function of t1/2. According to the intraparticle diffusion model, if a plot of qt versus t1/2 gives a linear line, it indicates that intraparticle/pore diffusion is the rate-limiting step in the adsorption process [42]. The obtained result of the plots is shown in Fig. 8, which is contrast with the prediction of the intraparticle diffusion model, illustrating a nonlinear distribution of points with two distinct portions (curved and linear). This probably indicated that the intraparticle diffusion in the processes is not the singular rate-limiting step in the adsorption process [43,44]. The initial curvature is attributed to film diffusion, and in the linear range to the intraparticle diffusion [45]. Therefore, the assumption addressed that both the film diffusion and the diffusion within the PVA hydrogel could be controlled by the intraparticle diffusion process.
According to URANO and TACHIKAWA model [46], owing to the multi-step nature of this plot, the linear portions were linearized by the plot of -lg[1-(q/qe)2] against initial time. If the plot is linear and pass through origin, then the slowest step or rate-limiting step in the adsorption process is the internal diffusion [47]. From Fig. 9, it was observed that the plot is linear but does not pass through the origin, suggesting that the adsorption process was controlled not only by intraparticle diffusion, but also by film diffusion. This is in accordance with the research reported by HAMEED et al [36].
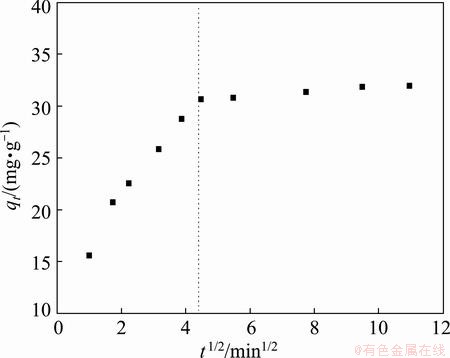
Fig. 8 Amount of Cu(II) ion adsorbed as function of square root of time for PVA hydrogel
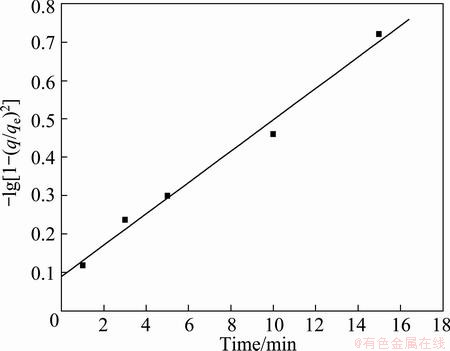
Fig. 9 Plot of -lg[1-(q/qe)2] against initial time of Cu(II) ion adsorption on PVA hydrogel
4 Conclusions
The results shown here provide the possibility of producing a new adsorbent hydrogel from PVA molecules. Interestingly, the PVA hydrogel could effectively remove Cu(II) from wastewater and the cost of manufacturing PVA adsorbent was cheap. The results showed that the contact time, pH and temperature affected the adsorption capacity of the Cu(II) ion on the PVA hydrogel. The kinetics of the adsorption of the Cu(II) ion on the PVA hydrogel was best described by the pseudo-second-order model. In addition, the mechanism of adsorption of the Cu(II) ion on the PVA hydrogel was controlled by particle and film diffusion.
Acknowledgements
This work was financially supported by Special Computer Science Project, Faculty of Science at Siracha, Kasetsart University (Grant number 012554). Acknowledgements also go to Faculty of Agriculture and Natural Resource, Rajamangala University of Technology Tawan-Ok, Bangpra Campus, for providing AAS experiment.
References
[1] DEMIRBAS A. Heavy metal adsorption onto agro-based waste materials: A review [J]. Journal of Hazardous Materials, 2008, 157: 220-229.
[2] FU F, WANG Q. Removal of heavy metal ions from wastewaters: A review [J]. Journal of Environmental Management, 2011, 92: 407-418.
[3] NGAH W S W, FATINATHAN S. Adsorption of Cu(II) ions in aqueous solution using chitosan beads, chitosan-GLA beads and chitosan-alginate beads [J]. Chemical Engineering Journal, 2008, 143: 62-72.
[4] CHENG H. Cu(II) removal from lithium bromide refrigerant by chemical precipitation and electrocoagulation [J]. Separation and Purification Technology, 2006, 52: 191-195.
[5] SAKAI H, MATSUOKA S, ZINCHENKO A A, MURATA S. Removal of heavy metal ions from aqueous solutions by complexation with DNA and precipitation with cationic surfactant [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2009, 347: 210-214.
[6] GUO Z, MA R X, LI G J. Degradation of phenol by nanomaterial TiO2 in wastewater [J]. Chemical Engineering Journal, 2006, 119: 55-59.
[7] OZAKI H, SHARMA K, SAKTAYWIN W. Performance of an ultra-low-pressure reverse osmosis membrane (ULPROM) for separating heavy metal: Effects of interference parameters [J]. Desalination, 2002, 144: 287-294.
[8] RODGERS J D, JEDRAL W, BUNCE N J. Electrochemical oxidation of chlorinated phenols [J]. Environmental Science and Technology, 1999, 33: 1453-1457.
[9] NIA M M, MONTAZERI P, MODARRESS H. Removal of Cu2+ and Ni2+ from wastewater with a chelating agent and reverse osmosis processes [J]. Desalination, 2007, 217: 276-281.
[10] CHAN B K C, DUDENEY A W L. Reverse osmosis removal of arsenic residues from bioleaching of refractory gold concentrates [J]. Minerals Engineering, 2008, 21: 272-278.
[11] YAVUZ  , ALTUNKAYNAK Y, GUZEL F. Removal of copper, nickel, cobalt and manganese from aqueous solution by kaolinite [J]. Water Research, 2003, 37: 948-952.
, ALTUNKAYNAK Y, GUZEL F. Removal of copper, nickel, cobalt and manganese from aqueous solution by kaolinite [J]. Water Research, 2003, 37: 948-952.
[12] ZHANG S Q, HOU W G. Adsorption behavior of Pb(II) on montmorillonite [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2008, 320: 92-97.
[13] WAN M W, KAN C C, ROGEL B D, DALIDA M L P. Adsorption of copper (II) and lead (II) ions from aqueous solution on chitosan- coated sand [J]. Carbohydrate Polymer, 2010, 80: 891-899.
[14] FUTALAN C M, KAN C C, DALIDA M L, HSIEN K J, PASCUA C, WAN M W. Comparative and competitive adsorption of copper, lead, and nickel using chitosan immobilized on bentonite [J]. Carbohydrate Polymer, 2011, 83: 528-536.
[15] NAJAFI M, YOUSEFI Y, RAFATI A A. Synthesis, characterization and adsorption studies of several heavy metal ions on amino- functionalized silica nano hollow sphere and silica gel [J]. Separation and Purification Technology, 2012, 85: 193-205.
[16] KANG Y O, YOON I S, LEE S Y, KIM D D, LEE S J, PARK W H, HUDSON S M. Chitosan-coated poly(vinyl alcohol) nanofibers for wound dressings [J]. Journal of Biomedical Materials Research Part B: Applied Biomaterials B, 2010, 92: 568-576.
[17] JAMNONGKAN T, WATTANAKORNSIRI A, PANSILA P P, MIGLIARESI C, KAEWPIROM S. Effect of poly (vinyl alcohol)/chitosan ratio on electrospun-nanofiber morphologies [J]. Advanced Material Research, 2012, 463-464: 734-738.
[18] MATSUMURA S, TOMIZAWA N, TOKI A, NISHIKAWA K, TOSHIMA K. Novel poly(vinyl alcohol)-degrading enzyme and the degradation mechanism [J]. Macromolecules, 1999, 32: 7753-7761.
[19] MANSUR H S, COSTA H S. Nanostructured poly(vinyl alcohol)/ bioactive glass and poly(vinyl alcohol)/chitosan/bioactive glass hybrid scaffolds for biomedical applications [J]. Chemical Engineering Journal, 2008, 137: 72-83.
[20] STREET K W, HILL C M, PHILIPP W H. Properties of a novel ion-exchange film [J]. Industrial and Engineering Chemistry Research, 2004, 43: 7600-7607.
[21] JIN L, BAI R. Mechanisms of lead adsorption on chitosan/PVA hydrogel beads [J]. Langmuir, 2002, 18: 9765-9770.
[22] LI X, LI Y, YE Z. Preparation of macroporous bead adsorbents based on poly(vinyl alcohol)/chitosan and their adsorption properties for heavy metals from aqueous solution [J]. Chemical Engineering Journal, 2011, 178: 60-68.
[23] STAMMEN J A, WILLIAMS S, KU D N, GULDBERG R E. Mechanical properties of a novel PVA hydrogel in shear and unconfined compression [J]. Biomaterials, 2001, 22: 799-806.
[24] WANG X, ZHENG Y, WANG A. Fast removal of copper ions from aqueous solution by chitosan-g-poly(acrylic acid)/attapulgite composites [J]. Journal of Hazardous Materials, 2009, 168: 970-977.
[25] NGAH W S W, KAMARI A, KOAY Y J. Equilibrium and kinetics studies of adsorption of copper(II) on chitosan and chitosan/PVA beads [J]. International Journal of Biological Macromolecules, 2004, 34: 155-161.
[26] ZHENG Y, WANG A. Removal of heavy metals using polyvinyl alcohol semi-IPN poly(acrylic acid)/tourmaline composite optimized with response surface methodology [J]. Chemical Engineering Journal, 2010, 162: 186-193.
[27] WU S, LI F, WU Y, XU R, LI G. Preparation of novel poly(vinyl alcohol)/SiO2 composite nanofiber membranes with mesostructure and their application for removal of Cu2+ from waste water [J]. Chemical Communications, 2010, 46: 1694-1696.
[28] Association of Analytical Chemistry. Official methods of analysis [S]. 15th ed. Washington DC, USA, 1990.
[29] MANSUR H S, SADAHIRA C M, SOUZA A N, MANSUR A A P. FTIR spectroscopy characterization of poly (vinyl alcohol) hydrogel with different hydrolysis degree and chemically crosslinked with glutaraldehyde [J]. Materials Science and Engineering C, 2008, 28: 539-548.
[30] MANSUR H S, OREFICE R L, MANSUR A A P. Characterization of poly(vinyl alcohol)/poly(ethylene glycol) hydrogels and PVA-derived hybrids by small-angle X-ray scattering and FTIR spectroscopy [J]. Polymer, 2004, 45: 7193-7202.
[31] ANDRADE G I, STANCIOLI E F B, MANSUR A A P, VASCONCELOS W L, MANSUR H S. Small-angle X-ray scattering and FTIR characterization of nanostructured poly (vinyl alcohol)/ silicate hybrids for immunoassay applications [J]. Journal of Material Science, 2008, 43: 450-463.
[32] JAMNONGKAN T, KAEWPIROM S. Potassium release kinetics and water retention of controlled-release fertilizers based on chitosan hydrogels [J]. Journal of Polymer and the Environment, 2010, 18: 413-421.
[33] SENTHILKUMAAR S, VARADARAJAN P R, PORKODI K, SUBBHURAAM C V. Adsorption of methylene blue onto jute fiber carbon: Kinetics and equilibrium studies [J]. Journal of Colloid and Interface Science, 2005, 284: 78-82.
[34] WANG S, BOYJOO Y, CHOUEIB A. A comparative study of dye removal using fly ash treated by different methods [J]. Chemosphere, 2005, 60: 1401-1407.
[35] BULUT Y, AYDIN H. A kinetics and thermodynamics study of methylene blue adsorption on wheat shells [J]. Desalination, 2006, 194: 259-267.
[36] HAMEED B H, MAHMOUD D K, AHMAD A L. Equilibrium modeling and kinetic studies on the adsorption of basic dye by a low-cost adsorbent: Coconut (Cocos nucifera) bunch waste [J]. Journal of Hazardous Materials, 2008, 158: 65-72.
[37] AKLIL A, MOUFLIH M, SEBTI S. Removal of heavy metal ions from water by using calcined phosphate as a new adsorbent [J]. Journal of Hazardous Materials, 2004, 112: 183-190.
[38] ALMEIDA C A P, DEBACHER N A, DOWNS A J, COTTET L, MELLO C A D. Removal of methylene blue from colored effluents by adsorption on montmorillonite clay [J]. Journal of Colloid and Interface Science, 2009, 332: 46-53.
[39] IJAGBEMI C O, BAEK M H, KIM D S. Montmorillonite surface properties and sorption characteristics for heavy metal removal from aqueous solutions [J]. Journal of Hazardous Materials, 2009, 166: 538-546.
[40] CABAL B, ANIA C O, PARRA J B, PIS J J. Kinetics of naphthalene adsorption on an activated carbon: Comparison between aqueous and organic media [J]. Chemosphere, 2009, 76: 433-438.
[41] SIVARAJ R, NAMASIVAYAM C, KADIRVELU K. Orange peel as an adsorbent in the removal of Acid violet 17 (acid dye) from aqueous solutions [J]. Waste Management, 2001, 21: 105-110.
[42] MCKAY G, OTTERBURN M S, AGA J A. Intraparticle diffusion process occurring during adsorption of dyestuffs [J]. Water Air and Soil Pollution, 1987, 36: 381-390.
[43]  Investigation kinetics mechanisms of adsorption malachite green onto activated carbon [J]. Journal of Hazardous Materials, 2007, 146: 194-203.
Investigation kinetics mechanisms of adsorption malachite green onto activated carbon [J]. Journal of Hazardous Materials, 2007, 146: 194-203.
[44] SANKAR M, SEKARAN G, SADULLA S, RAMASAMI T. Removal of diazo and triphenylmethane dyes from aqueous solutions through an adsorption process [J]. Journal of Chemical Technology and Biotechnology, 1999, 74: 337-344.
[45] COSTODES V C T, FAUDUET H, PORTE C, DELACROIX A. Removal of Cd(II) and Pb(II) ions, from aqueous solutions, by adsorption onto sawdust of Pinus sylvestris [J]. Journal of Hazardous Materials, 2003, 105: 121-142.
[46] URANO K, TACHIKAWA H. Process-development for removal and recovery of phosphorus from waste-water by a new adsorbent 2: Adsorption rates and breakthrough curves [J]. Industrial and Engineering Chemistry Research, 1991, 30: 1897-1899.
[47] WANG B E, HU Y Y, XIE L, PENG K. Biosorption behavior of azo dye by inactive CMC immobilized Aspergillus fumigatus beads [J]. Bioresource Technology, 2008, 99: 794-800.
Tongsai JAMNONGKAN1, Krittiya KANTAROT1, Kitiya NIEMTANG1, P. Pungboon PANSILA1, Amnuay WATTANAKORNSIRI2
1. Department of Fundamental Science and Physical Education, Faculty of Science at Siracha, Kasetsart University, Chonburi 20230, Thailand;
2. Department of Agriculture and Environment, Faculty of Science and Technology, Surindra Rajabhat University, Surin 32000, Thailand
ժ Ҫ���о�����ϩ��ˮ�����Է�ˮ��ͭ�������������ֱ�ʹ�ú������������ˮ���ⶨ�����о�ˮ�����Ļ�ѧ�ṹ��������Ϊ���������������ϩ�������ȩ�����γɾ���ϩ��ˮ������������������ˮ�Զ����ֳ��ߵ�ƽ�����ͱȡ����⣬�������������̷�����������ѧ����ɢ�����Ӽ���ɢ��Ĥ��ɢ���ơ�
�ؼ��ʣ��ؽ���ȥ������������ˮ����������ɢ����
(Edited by Hua YANG)
Corresponding author: Tongsai JAMNONGKAN; Tel: +66-38-354587; E-mail: sfscitsj@src.ku.ac.th
DOI: 10.1016/S1003-6326(14)63481-6

