
Bioleaching of pyrite by A. ferrooxidans and L. ferriphilum
ZHANG Lin(张 麟), QIU Guan-zhou(邱冠周), HU Yue-hua(胡岳华),
SUN Xiao-jun(孙小俊), LI Jian-hua(李建华), GU Guo-hua(顾帼华)
School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China
Received 20 September 2008; accepted 5 November 2008
Abstract: Pyrite oxidation rates were examined under various conditions in the presence of A. ferrooxidans and L. ferriphilum, in which different pulp concentration, inoculation amount, external addition of Fe3+ and initial pH value were performed. It is found that A. ferrooxidans and L. ferriphilum show similar behaviors in the bioleaching process. The increasing pulp concentration decreases the leaching rate of iron, and external addition of high concentration Fe3+ is also adverse to leaching pyrite. The increased inoculation amount and high initial pH value are beneficial to leaching pyrite, and these changed conditions bring more obvious effects on leaching pyrite by L. ferriphilum than by A. ferrooxidans. The results also show that adjusting the pH values in leaching process baffles leaching pyrite due to the formed jarosite. Jarosite formed in leaching process was observed using XRD, SEM and energy spectrum analysis, and a considerable amount of debris with a crystalline morphology is present on the surface of pyrite. The results imply that the indirect action is more important for bioleaching pyrite.
Key words: pyrite; A. ferrooxidans; L. ferriphilum; bioleaching; jarosite; redox potential
1 Introduction
The oxidation of pyrite is often necessary as pre-treatment for refractory ores before valuable metals can be released from the mineral matrix[1-2]. The oxidation of pyrite results in acidic metal-rich dissolution, known as acid mine drainage(AMD), causing serious environmental pollution[3-4]. Therefore, research on dissolution of pyrite in bioleaching process has an important guiding significance for hydrometallurgy and environmental protection.
Many researches on pyrite have been done. BOON et al[5-6] analyzed air composition before and after reaction and found that oxygen was mainly consumed in solution, rather than on the surface of pyrite. The role of bacteria is to oxidize Fe2+ to Fe3+. That is to say, the indirect mechanism plays a major role. MAGDALENA et al[7] studied pyrite oxidation by A. ferrooxidans at various concentrations of dissolved oxygen. The results of his study supported the indirect mechanism, too. JIANG et al[8-9] observed the surface of pyrite bioleached residues using scanning electron microscopy (SEM) and found there were bacteria adsorbed on the temper surface of pyrite. And shapes of corrosion were similar to those of the bacteria. This showed that the direct mechanism might exist. Therefore, whether direct mechanism exists or not is inconclusive. Currently, there are few the reports about leaching pyrite by L. ferriphilum. In this study, the relationship between the redox potential and extraction of iron was evaluated. The effects of pulp concentration, inoculation amount, external addition of ferric ion and the initial pH value, on the bioleaching of pyrite were examined using the selected strains of bacteria. The pyrite bioleaching mechanism was looked at.
2 Materials and methods
2.1 Pyrite preparation
The sample of pyrite used in this study was from Yunfu Sulfur Iron Ore (Guangdong Province, China). The samples were splintered into small fragments with a geological hammer and dry ground in a porcelain ball milling. The product was sieved to obtain suitable size less than 0.045 mm for bioleaching experiments. The XRD pattern (Fig.1) of the crushed sample was typical of that expected for pyrite and showed it included trace of quartz. The chemical analyses showed the pyrite contained 46.19% Fe and 48.99% S. The purity of pyrite sample reached more than 92%.
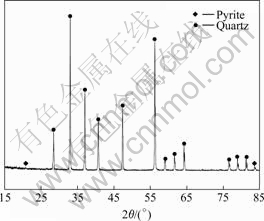
Fig.1 XRD pattern of mineral sample
2.2 Microorganisms and culture media
A. ferrooxidans and L. ferriphilum used in the experiments were obtained from the Key Laboratory of Biological Metallurgy of Central South University. They were grown in medium 9K with the initial pH values of 2.0 and 1.6, the temperature of the shaking table 30 ℃ and 40℃, respectively, rotating speed 170 r/min. The 9K medium: 3 g/L (NH4)2SO4, 0.1 g/L KCl), 0.5 g/L K2HPO4, 0.5 g/L MgSO4・7H2O, 0.01 g/L Ca(NO3)2, 44.7 g/L FeSO4・7H2O. The strains were gradually adapted to the 9K medium supplemented with 4% FeS2 concentrate. Before the bioleaching experiment, the pyrite-grown bacteria transferred at least 5 times in pyrite medium in order to adapt the experimental conditions. Cells were harvested by centrifugation and washed twice in distilled water adjusted to pH 2.0 or 1.6. The cells were then suspended in basal salts medium without energy sources. The concentration of the soliquid was 2×108 cell/mL.
2.3 Bioleaching experiments
Bioleaching tests were carried out in 250 mL flasks containing 100 mL medium with different amounts of ferric addition. The 9K basal salt medium without iron was used in the sulfide mineral bioleaching experiments. The inoculation amount was 5-15 mL and mineral concentration was 2%-8% (w/v). The initial pH was 2.0 for A. ferrooxidans and 1.6 for L. ferriphilum. The pH was adjusted to initial pH with 20% H2SO4 every 3 d except the test of changing the initial pH value. The flasks were shaken in rotary shakers for 18 d, controlled at growth temperature 30 ℃ for A. ferrooxidans, 40 ℃ for L. ferriphilum, and the rotating speed was 170 r/min.
In all leaching tests, every flask was sampled every 3 d by removing a 2 mL aliquot of the leaching solution, which was used for the analysis of total iron by the titration method. The redox potentials were measured using a platinum electrode combined with a Hg/HgCl2 reference electrode. The bioleaching residues were examined using X-ray diffractograms(XRD), scanning electron microscopy(SEM) and energy dispersion spectrum(EDS).
3 Results and discussion
3.1 Effect of pulp concentration
The leaching rate of iron and the redox potential evolutions by A. ferrooxidans(A. f) and L. ferriphilum(L. f) with 10% of the inoculation under different pulp concentrations are shown in Fig.2 and 3, respectively.

Fig.2 Leaching rate of pyrite as function of time at different pulp concentrations containing A. ferrooxidans or L. ferriphilum
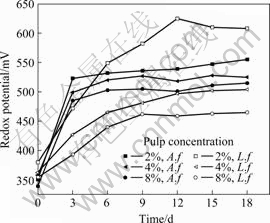
Fig.3 Redox potential as function of time at different pulp concentrations containing A. ferrooxidans or L. ferriphilum
It is found in Fig.2 that the increasing pulp concentration results in leaching rate of pyrite decreasing. This is because the high solid content of pulp hinders oxygen and carbon dioxide to transfer to strains and shear stress of mineral is harmful to bacteria, which leads to lag phase of strains longer. Fig.2 also shows that the effect of pulp concentration on the corrosive oxidation action of L. ferriphilum to pyrite is more obvious than that of A. ferrooxidans, and the leaching rate of pyrite with pulp concentration of 2% containing L. ferriphilum reaches the highest value at the 12th day and then decreases. This suggests that the ferric ion forms precipitation of jarosite (reaction (1)) and the iron concentration in solution decreases[10], which is confirmed by XRD analysis.
K++3Fe3++2SO42-+6H2O→KFe3(SO4)2(OH)6+6H+ (1)
Fig.3 shows that the pulp redox potential inoculated with A. ferrooxidans increases rapidly at the initial stage and then remains at a high level about 500 mV (SCE). The phenomenon related to physiological characteristics of A. ferrooxidans. A. ferrooxidans needs to grow in appropriate range of redox potential. But the effect of pulp concentration on the redox potential is unapparent in A. ferrooxidans leaching process. For pulp inoculated with L. ferriphilum, the redox potential changes remarkably at different pulp concentrations. The pulp redox potential of which containing 2% FeS2 reaches above 600 mV (SCE) after 10 d and it is much higher than pulp containing 8% FeS2. Linking to Fig.2, it can be found that the high redox potential value is beneficial to the bioleaching of pyrite.
3.2 Effect of inoculation amount
Fig.4 shows the leaching rate of pyrite as function of time with different inoculation amounts containing A. ferrooxidans or L. ferriphilum with 4% pulp density.

Fig.4 Leaching rate of pyrite as function of time in different inoculation amounts containing A. ferrooxidans or L. ferriphilum
It can be seen from Fig.4 that the leaching rate of iron increases fast at the initial stage and then rises slowly. Compared with A. ferrooxidans, the inoculation amounts have obvious effect on the action of of L. ferriphilum to pyrite. The leaching rate of iron by L. ferriphilum with 15% inoculums reaches above 33% that is much higher than that in 5% and 10% inoculums. Moreover, it is higher than that by A. ferrooxidans after 12 d. Under this solution condition, the redox potential mensurated is above 600 mV (SCE). From Fig.4, it also can be seen that the leaching rates by L. ferriphilum with 5% and 10% inoculums are lower than those by A. ferrooxidans. This might be caused by slow growth rate of L. ferriphilum. According to the related literatures reports, the growth rate of L. ferriphilum is only half of A. ferrooxidans[11].
3.3 Effect of initial pH value
The leaching rate of iron and changes of pH as function of time at various initial pH values are shown in Fig.5 and 6, respectively. The inoculation amount is 10% and pulp concentration is 4%. The pH value is not adjusted in the leaching process.
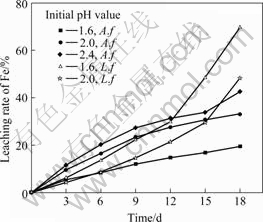
Fig.5 Leaching rate of iron as function of time at various initial pH values containing A. ferrooxidans or L. ferriphilum
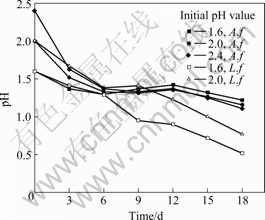
Fig.6 Changes of pH as function of time at various initial pH values containing A. ferrooxidans or L. ferriphilum.
It is found from Fig.5 that the increasing initial pH value is beneficial to leaching pyrite with A. ferrooxidans and L. ferriphilum, and it is more efficient to L. ferriphilum than A. ferrooxidans. From the change of pH during bioleaching process in Fig.6, it can be seen that the leaching of pyrite is an acid production process. The pH value in solution decreases rapidly, especially at early stage of leaching, and at 18th day, the pH values of pulp inoculated with A. ferrooxidans and L. ferriphilum reduce to about 1.2 and 0.5, respectively. Generally, when the pH value decreases, the activity of bacteria weakens [12-13], but it is found that leaching of pyrite increases at all time. This indicates that the main reactions in solution are Reactions (2)-(4)[14], and when pH is below 1.0, the Reaction (5) is included yet[15]. At low pH, the iron ion is mainly present as Fe3+. Therefore, the significantly high Fe3+ concentration is likely the major factor responsible for the high oxidation rates[7]. That is to say, the chemical leaching and indirect way play a major role in the bioleaching process of pyrite.
FeS2+(7/2)O2+H2O Fe2++2SO42-+2H+ (2)
Fe2++2SO42-+2H+ (2)
Fe2++(1/4)O2+H+ Fe3++(1/2)H2O (3)
Fe3++(1/2)H2O (3)
FeS2+14Fe3++8H2O→15Fe2++2SO42-+16H+ (4)
FeS2+(15/4)O2+(1/2)H2O→Fe3++2SO42-+H+ (5)
The results also show that the leaching rate of iron is higher not adjusting the pH value than that adjusting the pH value in leaching process because low pH hindered jarosite forms, which was confirmed by XRD SEM and EDS analysis of bioleaching residues.
3.4 Effect of external addition of Fe3+
The effects of external addition of Fe3+ on the leaching process and the redox potential are shown in Figs.7 and 8, respectively. The pulp concentration is 4% and the inoculation amount is 10%.

Fig.7 Leaching rate of pyrite as function of time in different amounts of external addition of Fe3+containing A. ferrooxidans or L. ferriphilum

Fig.8 Redox potential as function of time in different amounts of external addition of Fe3+containing A. ferrooxidans or L. ferriphilum
From Figs.7 and 8, it can be seen that external addition of Fe3+ is adverse to leaching pyrite and the leaching rate of iron is always lower than no Fe3+ addition for both A. ferrooxidans and L. ferriphilum. And the external addition of Fe3+ can produce a high initial redox potential but cannot maintain it. At the initial stage of leaching process, the redox potential decreases, after 3 d, the redox potential begins to increase, but it is lower than no Fe3+ addition and below 500 mV (SCE). This explains that high concentration ferric ion can inhibit the growth of A. ferrooxidans and L. ferriphilum. And the ability of oxidation leaching of cells decreases. This phenomenon is consistent with Refs.[16-17].
3.5 XRD, SEM and EDS results of bioleaching residues
X-ray diffractograms for residues by A. ferrooxidans and L. ferriphilum contacting with the pyrite sample after 18 d show that pyrite peaks weaken and jarosite is present as a secondary phase, as shown in Fig.9. This proves that the formed iron precipitation in leaching process causes the leaching rate decreasing. From Fig.9, it is found that jarosite peaks is more in residues with L. ferriphilum than in residues leached by A. ferrooxidans. And diffraction lines for S0 are not found, which shows the intermediate during oxidation of pyrite is not element sulphur.
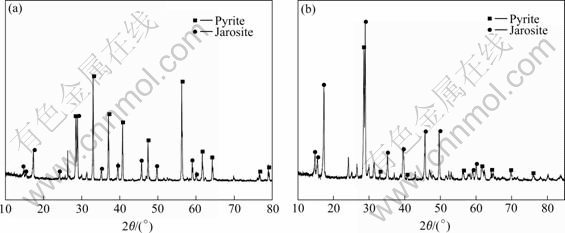
Fig.9 XRD patterns of leached pyrite residues by A. ferrooxidans (a) and L. ferriphilum (b) with pulp concentration of 2%
The SEM images of the above leached residues are shown in Fig.10(a) and Fig.10(b), respectively. Residues leached by A. ferrooxidans and L. ferriphilum are both covered with oxidation products. A considerable amount of debris, with a crystalline morphology is present on the surface.

Fig.10 SEM images of leached residues by A. ferrooxidans (a) and L. ferriphilum (b) with pulp concentration of 2%
Table 1 lists the results of energy dispersion spectrum(EDS). Before leaching, the mass fractions of elements in pyrite are Fe 46.19% and S 48.99%. The atomic ratio of Fe and S is 1?1.86, which is very close to the theoretical atomic ratio of pyrite. In residues leached by A. ferrooxidans and L. ferriphilum, the atomic ratios are 1?1.12 and 1?0.79, respectively. The results indicate that the surface iron element relatively riches after bioleaching of pyrite and sulphur deficient due to the formation of sulphate. This further implies that the indirect action is more important in bioleaching of pyrite.
Table 1 Results of energy dispersion spectrum (EDS)
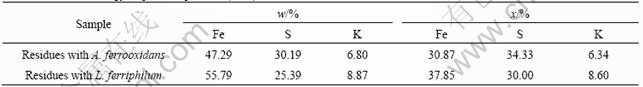
From Table 1, it also can be seen that the element K is present in residues. This proves that jarosite forms in the leaching process, which is consistent with the leaching experiments and XRD results.
4 Conclusions
1) The effect of pulp concentration and inoculation amount on the corrosive oxidation action of L. ferriphilum to pyrite is more obvious than that of A. ferrooxidans. The leaching speed of pyrite by L. ferriphilum is faster at low pulp concentration than that at high pulp concentration, but A. ferrooxidans is reverse. The increasing inoculation amount is beneficial to leaching pyrite.
2) The high redox potential and high initial pH value are beneficial to leaching pyrite, but adjusting the pH value in leaching process is against leaching pyrite due to jarosite forms, which is confirmed by XRD, SEM and EDS analysis of leached residues.
3) The pyrite bioleaching mechanism is dominated by the indirect action through our study.
References
[1] BLIGHT K, RALPH D E, THURGATE S. Pyrite surfaces after bio-leaching: A mechanism for bio-oxidation [J]. Hydrometallurgy, 2000, 58: 227-237.
[2] WU Biao, RUAN Ren-man, WEN Jian-kang, ZHOU Gui-ying. Electrochemical oxidative behavior of pyrite during ore bioleahing [J]. Metal Mine, 2007, 10: 64-67.
[3] JIANG Lei, ZHOU Huai-yang, PENG Xiao-tong. Phases in pyrite bio-oxidation process [J]. Metal Mine, 2007, 10: 59-63.
[4] GARCIA O Jr, BIGHAM J M, TUOVINEN O H. Oxidation of isochemical FeS2 (marcasite-pyrite) by Acidithiobacillus thiooxidans and Acidithiobacillus ferrooxidans [J]. Minnerals Engineering, 2007, 20: 98-101.
[5] BOON M, HEIJNEN J J. Chemical oxidation kinetics of pyrite in bioleaching processes [J]. Hydrometallurgy, 1998, 48: 27-41.
[6] BOON M. The use of off-gas analyses and stoichiometry in the biooxidation kinetics of sulphide minerals [J]. Hydrometallurgy, 1998, 48: 1-26.
[7] GLEISNER M, HERBERT R B, FROGNER KOCKUM P C. Pyrite oxidation by Acidithiobacillus ferrooxidans at various concentrations of dissolved oxygen [J]. Chemical Geology, 2006, 225: 16-19.
[8] JIANG Lei, ZHOU Huai-yang, PENG Xiao-tong. Oxidization effect of Thiobacillus ferroxidans on pyrite, chalcopyrite and pyrrhotine [J]. Chinese Science Bulletin, 2007, 15: 1802-1812.
[9] JIANG Lei, ZHOU Huai-yang, PENG Xiao-tong. Preliminary study on bio-oxidation of pyrite by A. ferrooxidans [J]. Acta Mingeralogica Sinica, 2007, 27: 25-29.
[10] DEVECI H, AKCIL A, ALP I. Bioleaching of complex zinc sulphides using mesophilic and thermophilic bacteria: Comparative importance of pH and iron [J]. Hydrometallurgy, 2004, 73: 293-303.
[11] LIU Fu-liang, LIANG Hua-zheng, LIU Cheng-zuo, JIN Wei-gen. Research on high density fermentation of Leptospirillums ferrooxidans [J]. Uranium Mining and Metallurgy, 2006, 1: 43-46.
[12] LU Dian-qun, LIU Da-xing, JIANG Kai-xi, WEI Xu-jun, YANG Li. The research progress about mechanism of bacteria oxidizing sulfide minerals [J]. Nonferrous Metals (Extractive Metallurgy), 2001, 1: 2-5.
[13] HANSFORD G S, VARGAS T. Chemical and electrochemical basis of bioleaching processes [J]. Hydrometallurgy, 2001, 59(2/3): 135-145.
[14] LIU Jian-she. Bioextraction and corrosion electrochemistry of sulfide minerals [D]. Changsha: Central South University, 2002: 1-146.
[15] WANG Dian-zuo, HU Yue-hua. Solution chemistry of flotation [M]. Changsha: Hunan Science and Technology Press, 1988: 1-343.
[16] BOON M, HEIJNEN J J. Solid-liquid mass transfer limitation of ferrous iron in the chemical oxidation of FeS2 at high redox potential [J]. Hydrometallurgy, 2007, 62: 57-66.
[17] SHARMA P K, DASB A, HANUMANTHA RAOA K, FORSSBERGA K S E. Surface characterization of Acidithiobacillus ferrooxidans cells grown under different conditions [J]. Hydrometallurgy, 2003, 71: 285-292.
Foundation item: Project(50621063) supported by the National Natural Science Foundation of China; Project(2004CB619204) supported by the National Basic Research Program of China
Corresponding authors: SUN Xiao-jun; Tel: +86-731-8877867; E-mail: sxj547636@126.com
(Edited by LI Xiang-qun)