Oxidation behavior of CVI, MSI and CVI+MSI C/SiC composites
来源期刊:中国有色金属学报(英文版)2010年第4期
论文作者:闫志巧 陈峰 熊翔 肖鹏 张红波 黄伯云
文章页码:590 - 596
Key words:chemical vapor infiltration (CVI); molten silicon infiltration (MSI); C/SiC composites; oxidation
Abstract:
The oxidation behavior of chemical vapor infiltration (CVI), molten silicon infiltration (MSI) and CVI+MSI C/SiC composites at 500-1 400 ℃ was studied. The oxidation below 900 ℃ increased successively for CVI, CVI+MSI and MSI composites. However, the oxidation of CVI composite above 1 000 ℃ was much faster than that of MSI and CVI+MSI composites. As active carbon atoms produced by siliconization of fibers during MSI process were oxidized first and decreased initial oxidation temperature. The initial oxidation temperature of MSI, MSI+CVI and CVI composites was 526, 552 and 710 ℃, respectively. New active carbon atoms were generated due to the breaking of 2D molecular chains during oxidation, so the activation energy of three C/SiC composites was decreased gradually at 500-800 ℃ with oxidation process, exhibiting a self-catalytic characteristic.
基金信息:the National Basic Research Program of China

YAN Zhi-qiao(闫志巧)1, 2, CHEN Feng(陈 峰)1, 2, XIONG Xiang(熊 翔)2,
XIAO Peng(肖 鹏)2, ZHANG Hong-bo(张红波)2, HUANG Bai-yun(黄伯云)2
1. Guangzhou Research Institute of Nonferrous Metals, Guangzhou 510650, China;
2. State Key Laboratory for Powder Metallurgy, Central South University, Changsha 410083, China
Received 25 May 2009; accepted 17 September 2009
Abstract: The oxidation behavior of chemical vapor infiltration (CVI), molten silicon infiltration (MSI) and CVI+MSI C/SiC composites at 500-1 400 ℃ was studied. The oxidation below 900 ℃ increased successively for CVI, CVI+MSI and MSI composites. However, the oxidation of CVI composite above 1 000 ℃ was much faster than that of MSI and CVI+MSI composites. As active carbon atoms produced by siliconization of fibers during MSI process were oxidized first and decreased initial oxidation temperature. The initial oxidation temperature of MSI, MSI+CVI and CVI composites was 526, 552 and 710 ℃, respectively. New active carbon atoms were generated due to the breaking of 2D molecular chains during oxidation, so the activation energy of three C/SiC composites was decreased gradually at 500-800 ℃ with oxidation process, exhibiting a self-catalytic characteristic.
Key words: chemical vapor infiltration (CVI); molten silicon infiltration (MSI); C/SiC composites; oxidation
1 Introduction
Continuous carbon fiber-reinforced silicon carbide matrix composites (C/SiC) possess many attractive properties, such as low density, high strength, good thermal shock resistance and excellent mechanical properties at elevated temperatures. They have been widely used as parts in aerospace industry and civil engineering[1-2]. However, when exposed to oxidizing environments above 400 ℃, the composites can be easily oxidized and burn out, leading to deterioration or even failure during service[3]. Poor oxidation resistance becomes a major drawback of C/SiC composites. It is significant to study their oxidation behavior in depth for the sake of safety.
Three basic techniques are applied to manufacture C/SiC composites, i.e. chemical vapor infiltration (CVI) [4], polymer impregnation and pyrolysis (PIP)[5] and molten silicon infiltration (MSI)[6]. Oxidation behavior of PIP and CVI C/SiC composites has been studied extensively[7-8], whereas that of C/SiC composite prepared by MSI method has been quite neglected up to date. It is therefore interesting and meaningful to study and compare the oxidation characteristics of C/SiC composites prepared by different techniques. In this work, the oxidation behavior of C/SiC composites prepared by CVI, MSI and CVI combined with MSI technique, respectively, has been investigated by using thermogravimetric (TG) analysis and other surface analysis techniques.
2 Experimental
2.1 Specimen preparation
2.5D bulk needled carbon fiber felts (0.56 g/cm3 bulk density) were used as preforms. A CVD process was used to deposit pyrocarbon in the felts in a gas mixture of C3H6 and N2 at 980 ℃. After 2 h and 100 h deposition, respectively, two types of C/C porous preforms were prepared, whose bulk density was about 0.6 and 1.3 g/cm3, respectively. Then a high temperature treatment (HTT) at 2 300 ℃ was conducted on them. The above mentioned three techniques were used to prepare SiC matrix. For the first CVI composite, Methyltrichloro- silane (MTS)-H2-Ar system was adopted to densify C/C porous preforms (0.6 g/cm3 bulk density). The deposition temperature was 1 000 ℃ and H2/MTS ratio was set to 10:1. The CVI process was interrupted several times for surface machining to open closed pores and ended until further densification became quite difficult after 350 h. For the second MSI composite, C/C porous preforms (1.3 g/cm3 bulk density) were placed inside a graphite vessel covered with compact Si powder in the bottom and treated at 1 650 ℃ for 2 h. For the third CVI+MSI composite, C/C porous preforms (0.6 g/cm3 bulk density) were densified by CVI process for 60 h and C/SiC porous preforms with 1.3 g/cm3 bulk density were obtained. The C/SiC porous preforms were then impregnated with furan resin, cured and carbonized. As a result, resin carbon was formed and ready to react with Si. Finally, MSI technique was conducted again as a finial step of composite synthesis. Fig.1 shows detailed manufacturing processes of these three types of C/SiC composites, where IC represents impregnation and carbonization of the resin. These three C/SiC composites were cut into small pieces with a size of 20 mm×20 mm×5 mm.
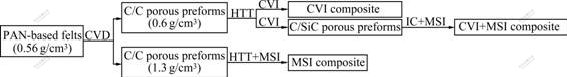
Fig.1 Manufacturing techniques of three C/SiC composites
2.2 Microstructure characterization and oxidation tests
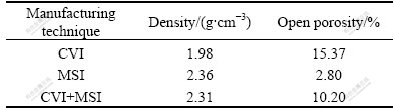
The density and open porosity of these three C/SiC composites were measured and calculated using Archimedes method and the results are listed in Table 1. Compositional and microstructural analyses were conducted through X-ray diffractometry (XRD) and scanning electron microscope (SEM) equipped with energy dispersive X-ray (EDX) analysis.
Table 1 Density and open porosity of three C/SiC composites
Isothermal oxidation was conducted at 500-1 400 ℃ in a corundum tube furnace in air. The samples were put into the furnace heated to a desired temperature. After holding for 30 min, they were taken out of the furnace, cooled in air to room temperature. Based on the mass of sample before and after oxidation test, the mass loss of the samples is calculated.
Simultaneous thermal analyzer (SDT-Q600, USA) was employed to study the non-isothermal oxidation behavior. An air flow of 100 mL/min and a heating rate of 10 ℃/min were used. The mass of sample as a function of temperature was recorded continuously.
3 Results and discussion
3.1 Microstructure and composition
The morphologies of three as-prepared C/SiC composites were observed by SEM, and shown in Fig.2. For the composite made by CVI method (Fig.2(a)), small pores inside the fiber bundles were well infiltrated, while large pores between the fiber bundles were still partially empty. The open porosity was measured to be 15.37%, as shown in Table 1. The infiltration of SiC into the pores is mainly dependent on the diffusion of gaseous species, and the diffusion resistance increases with the decrease of pore size. It is therefore difficult to make the pores infiltrated completely. In addition, the bottleneck effect hampers the densification of the composite. The increase of infiltration time has no obvious effect on further densification. As shown in Fig.2(b), the MSI composite was much denser than the CVI composite. Large pores between the fiber bundles had been filled with SiC (in gray) and Si (in white), as identified by EDX analyses. In MSI process, the densification is controlled by the flow of molten Si. The infiltration rate is mainly dependent on the wettability between Si(l)―C(s) and Si(l)―SiC(s). As the wetting angle is between 0?-20?, Si(l) is driven by capillary force and infiltrate spontaneously into pores in C/C porous preforms. Massive Si(s) evaporate as Si(g) to diffuse into fiber/pyrocarbon interface to form SiC. The molar volume of SiC and carbon in graphite is 12.49 cm3/mol and 5.31 cm3/mol respectively. There is 2.35 times volume expansion accompanying the transformation of C to SiC. Due to density change, 2.53 g/cm3 for Si(l) and 2.34 g/cm3 for Si(s), the freeze of unreacted Si (l) to Si (s) leads to 8.1% volume expansion. These factors are in favor of densification. The open porosity of MSI composite was less than 3%. Compared with C/SiC composites prepared by single CVI or MSI technique, CVI+MSI composite has moderate densification (Fig.2(c)), and has fibers less corroded. The open porosity of CVI+MSI composite was 10.20%, lower than that of CVI composite.

Fig.2 SEM images of three C/SiC composites prepared by different methods: (a) CVI; (b) MSI; (c) CVI+MSI
Fig.3 shows the XRD patterns of three C/SiC composites. It can be seen that carbon in fibers and matrices exhibited quasi-graphite structure[9]. The sharp diffraction peaks of SiC in MSI and CVI+MSI composites indicate the high crystallization of these two materials, and the broadened peaks obtained on CVI composite suggest low crystallization. The strong peaks of Si in MSI composite indicate high residual Si content. While in CVI+MSI composite, there is almost no residual Si.
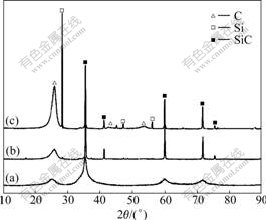
Fig.3 XRD patterns of three C/SiC composites prepared by different methods: (a) CVI; (b) CVI+MSI; (c) MSI
3.2 Isothermal and non-isothermal oxidation process
Fig.4 shows the curves of mass loss of three C/SiC composites during isothermal oxidation at 500-1 400 ℃ for 30 min. The mass loss of MSI composite increased quickly from 600 ℃ to 900 ℃ and reached the maximum at 900 ℃, while that of CVI+MSI composites was relatively small below 900 ℃ and reached the maxima at 1 200 ℃. Above 1 000 ℃, CVI composite had the largest mass loss. When the temperature exceeded 1 200 ℃, mass loss decreased slightly due to mass gain during rapid oxidation of SiC and Si into SiO2.

Fig.4 Curves of mass loss of three C/SiC composites after oxidation for 30 min
Fig.5 exhibits the SEM images of three C/SiC composites after oxidation at 800 ℃ for 5 min. In CVI composite shown in Fig.5(a), intact pyrocarbon layer (about 0.2 μm) around fibers can be observed clearly. Bonding between fibers and matrices kept close with no obvious oxidation. In comparison, distinct tile-like gaps between fibers and matrices can be observed in MSI composite (Fig.5(b)), which provided further diffusion tunnels for oxygen. Oxidation occurred firstly at gaps between fibers/matrices and matrices/matrices, and then continued along cracks[10]. For CVI+MSI composite, pyrocarbon layer parallel to axis of fiber was mostly oxidized (Fig.5(c)) and gaps were formed, which also accelerated oxidation. It can be concluded that the oxidation degree at 800 ℃ was the smallest for CVI composite and was the biggest for MSI composite, which was in good accordance with the changes of mass loss shown in Fig.4.
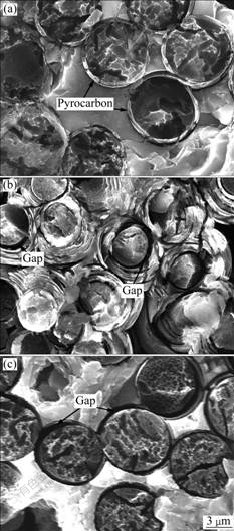
Fig.5 SEM images of three C/SiC composites prepared by different methods after oxidation at 800 ℃ for 5 min: (a) CVI; (b) MSI; (c) CVI+MSI
The morphologies of three C/SiC composites oxidized at 1 200 ℃ for 5 min are shown in Fig.6. In CVI composite, the fibers were oxidized completely and only CVI SiC around the fibers was left (Fig.6(a)). In MSI composite, the outer fibers were severely oxidized and only impurity remnants were left, while the internal fibers were kept relatively intact (Fig.6(b)). Fibers in CVI+MSI composite appeared as needles due to oxidation (Fig.6(c)). Obviously, the fibers in CVI composite were most severely oxidized at 1 200 ℃, which was consistent with the maximum mass loss at 1 200 ℃ as shown in Fig.4. The fibers in MSI and CVI+ MSI composites were less oxidized.

Fig.6 SEM images of three C/SiC composites after oxidation at 1 200 ℃ for 5 min: (a) CVI; (b) MSI; (c) CVI+MSI
Fig.7 and Fig.8 show TG and differential thermogravimetric (DTG) curves of three C/SiC composites, respectively. It can be seen from TG curves that the initial oxidation temperature of MSI, CVI+MSI and CVI composites increased successively, i.e. the lowest 526 ℃ for MSI, the moderate 552 ℃ for CVI+MSI and the highest 710 ℃ for CVI. The initial oxidation temperature of CVI composite was about 180 ℃ higher than that of MSI composite, but that of MSI and CVI+MSI composites was approximately the same. DTG curves show at the initial stage of oxidation, oxidation rate decreased successively for MSI, CVI+MSI and CVI composites. The largest oxidation rate was 0.50 %/℃ at 681 ℃ for MSI composite and 0.12%/℃ at 744 ℃ for CVI+MSI composite, respectively. Though the CVI composite had the lowest initial oxidation rate, the rate increased quickly above 850 ℃ and reached the maximum at 0.19 %/℃ at 928 ℃. This tendency of oxidation rate was in good agreement with oxidation- mass loss below 900 ℃ shown in Fig.4.
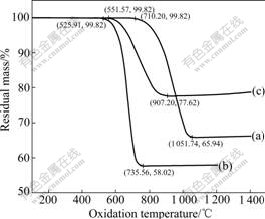
Fig.7 TG curves of three C/SiC composite: (a) CVI; (b) MSI; (c) CVI+MSI

Fig.8 DTG curves of three C/SiC composites: (a) CVI; (b) MSI; (c) CVI+MSI
3.3 Oxidation mechanism
The above results showed that at the initial stage of oxidation, MSI composite was most likely to oxidize and reached the largest oxidation rate at only 681 ℃. While for CVI composite, the initial oxidation temperature was 710 ℃ and the largest oxidation rate was reached at 928 ℃. As for CVI+MSI composite, these two aspects were moderate. All these phenomena were related closely to the manufacturing technique.
Below 900 ℃, Si and SiC are oxidized very slowly [11], while naked carbon fibers exposed to air could be oxidized easily. Oxidation mass loss of the composites after a short time treatment at temperatures below 900 ℃ should result from oxidation of carbon phase. As fibers in the three C/SiC composites had experienced HTT at 2 300 ℃, which was a graphitization treatment and the graphitization effect was mainly determined by temperature[12], it can be concluded that the fibers were the same in all the composites and the difference in the oxidation behavior of the composites was related to the different manufacturing techniques.
During the preparation of MSI composite, pyrocarbon was deposited to prepare C/C porous preforms. When pyrocarbon layer uniformly covered the surface of the fibers, its thickness was simply calculated to be 1.83 μm through the following equation[13]:
![]()
where x is the relative increase in mass. In MSI process, once SiC formed between Si(l) and C(s), the growth of its thickness was further controlled by diffusion of Si(l) through the as-formed SiC layer[14]. The diffusion coefficient (Deffc) at 1 650 ℃ was 5.19×10-10 cm2/s, which was calculated through[15]:

where the activation energy (ED) and the diffusion coefficient constant (D0) are 132 kJ/mol and 2.0×10-6 cm2/s[14], respectively. According to the relationship between the reaction time (t) and the thickness of SiC (dSiC)[15],
![]()
the thickness of SiC formed for 2.5 min was 2.79 μm, which was equal to that of SiC transformed from 2.10 μm carbon with 33% line expansion. These results indicated that pyrocarbon could be fully converted into SiC under suitable conditions. MSI process at 1 650 ℃ lasted for 2 h to achieve high density, which was sufficient for pyrocarbon to be converted into SiC. Then the surplus Si(l) further reacted with inner fibers. Moreover, owing to its diffusion, Si(g) inevitably reacted with fibers. These two factors led to siliconization of the fibers and the composite were deteriorated. Microcracks and siliconization of fibers in MSI composite can be clearly seen in Fig.9. The microcracks may result from two causes. One is the expansion when Si(l) freezes, and the other is the mismatch between SiC and fibers in thermal expansion. These microcracks as diffusion tunnels for oxygen accelerated the oxidation.
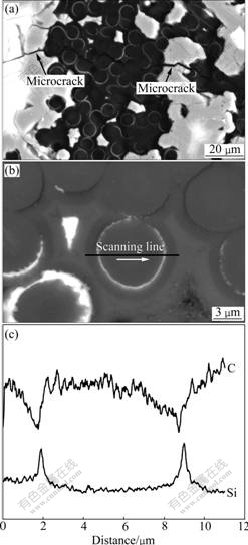
Fig.9 Microstructures in MSI C/SiC composites: (a) Micro- cracks; (b) Siliconization of fibers; (c) Profiles of C and Si along scanning line in (b)
During the preparation of CVI+MSI composite, C/SiC porous preforms were prepared by CVI method. CVI SiC layer with certain thickness played a barrier role. In the following MSI process, the corrosion degree of the fibers was weakened. However, due to the inherent uneven characteristic of CVI process and the diffusion activity of Si(l), Si(l) still penetrated CVI SiC layer and even inner pyrocarbon in some places and reacted with the fibers. For CVI composite, CVI SiC filled micropores between the fibers first and provided good protection.
The flexural strength of CVI, MSI and CVI+MSI composites was 310, 120 and 131 MPa, respectively. The value of CVI composite was as 2.58 times as that of MSI composite, which further confirmed severe siliconization of fibers during MSI while good protection during CVI. The close values between MSI and CVI+MSI composites verified insufficient protection of CVI SiC barrier layer.
All carbon in these three C/SiC composites exhibited quasi-graphite structure and the molecular structure was stretching 2D chains. Siliconization reaction led to breaking of these 2D chains and many unsaturated carbon atoms, i.e. active atoms, were produced. At the initial stage of oxidation, active carbon atoms produced by siliconization of fibers during MSI oxidized first. Therefore, the initial oxidation temperature of MSI composite with most severe siliconization was the lowest, and that of CVI+MSI composite was only 26 ℃ higher. At the same time, intact 2D chains also broke during oxidation and new active atoms were formed. With the increase of temperature, the mass loss increased continuously and more and more 2D chains were broken, which produced more active atoms and accelerated the oxidation rate. That is to say, the oxidation rate increased with the increase of mass loss, which was a typical self-catalytic reaction characteristic, so MSI composite had the largest oxidation rate.
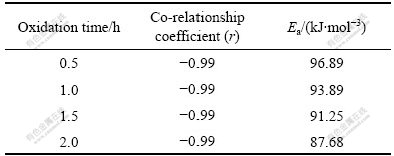
Fig.10 shows the Arrhenius curves of MSI composite at 500-800 ℃. The activation energy (Ea) with oxidation time are listed in Table 2. It shows that Ea gradually decreased with prolonging oxidation time, which further proved that the oxidation exhibited a self-catalytic characteristic. The same tendency for CVI and CVI+MSI composites suggested a similar oxidation mechanism.
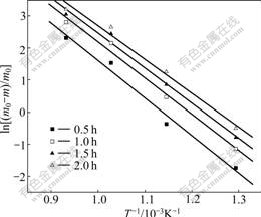
Fig.10 Arrhenius curves of MSI C/SiC composite
Table 2 Ea and r data of MSI C/SiC composite
In MSI composite, active carbon atoms were generated by siliconization of fibers, so its largest oxidation rate was reached at as a low temperature as 681 ℃. In CVI composite, active carbon atoms were produced only through oxidation, so its largest oxidation rate was reached at 928 ℃, much higher than that of MSI composite. The maximum mass loss for MSI composite was realized at 900 ℃, while that of CVI and CVI+MSI composite was at 1 200 ℃ due to their relatively slow oxidation rate.
4 Conclusions
1) The oxidation behavior of C/SiC composites made by three techniques was different. During MSI process, active carbon atoms were produced by siliconization of fibers and they would be first oxidized, which obviously decreased the initial oxidation temperature. The initial oxidation temperature of MSI and CVI+MSI composites was 526 and 552 ℃, respectively. For CVI composite, fibers were well protected and the initial oxidation temperature was 710 ℃. Below 900 ℃, the oxidation increased successively for CVI, CVI+MSI and MSI composites. Above 1 000 ℃, however, CVI composite was most easily oxidized.
2) During oxidation, new active carbon atoms were produced by the breaking of 2D molecular chains, and the oxidation rate increased. The activation energy of three C/SiC composites at 500-800 ℃ decreased with the increase in oxidation time and the oxidation exhibited a self-acceleration characteristic.
References
[1] NASLAIN R. Design, preparation and properties of non-oxide CMCs for application in engines and nuclear reactors: An overview [J]. Compos Sci Technol, 2004, 64(2): 155-170.
[2] XIAO Peng, XIONG Xiang, ZHANG Hong-bo, HUANG Bai-yun. Process and application of C/C-SiC ceramic braking materials [J]. The Chinese Journal of Nonferrous Metals, 2005, 15(5): 667-674. (in Chinese)
[3] WEBSTER J D, WESTWOOD M E, HAYES F H, DAY R J, TAYLOR R, DURAN A, APARICIO M, REBSTOCK K, VOGEL W D. Oxidation protection coatings for C/SiC based on yttrium silicate [J]. J Eur Ceram Soc, 1998, 18(16): 2345-2350.
[4] TANG S F, DENG J Y, DU H F, LIU W C, YANG K. Fabrication and microstructure of C/SiC composites using a novel heaterless chemical vapor infiltration technique [J]. J Am Ceram Soc, 2005, 88(11): 3253-3255.
[5] de OMENA PINA S R, PARDINI L C, YOSHIDA. Carbon fiber/ceramic matrix composites: Processing, oxidation and mechanical properties [J]. J Mater Sci, 2007, 42(12): 4245-4253.
[6] KRENKEL W. Cost effective processing of composites by melt infiltration (LSI-process) [J]. Ceram Eng Sci Proc, 2001, 22(3): 442-444.
[7] XU Y D, CHENG L F, ZHANG L T, YIN X W. Effect of carbon interlayer on oxidation behavior of C/SiC composites with a coating from room temperature to 1 500 ℃ [J]. Mater Sci Eng A, 2001, 300(1/2): 219-225.
[8] DENG J Y, LIU W C, DU H F, CHENG H M, LI Y Y. Oxidation behavior of C/C-SiC gradient matrix composites [J]. J Mater Sci Technol, 2001, 17(5): 543-546.
[9] ZHAO L R, JIAN B Z. The oxidation behavior of low-temperature heat-treated carbon fibers [J]. J Mater Sci, 1997, 32(11): 2811-2819.
[10] LIAO J Q, HUANG B Y, SHI G, CHENG T F, XIONG X. Influence of porosity and total surface area on the oxidation resistance of C/C composites [J]. Carbon, 2002, 40(13): 2483-2488.
[11] KIM Y K, LEE J Y. The effect of SiC codeposition on the oxidation behavior of carbon/carbon composites prepared by chemical vapor deposition [J]. Carbon, 1993, 31(7): 1031-1038.
[12] ZUO Jin-lv, ZHANG Hong-bo, HUANG Qi-zhong, XIAO Peng, XU Hui-juan. Influence of bulk density and graphitization degree on hardness of C/C composites [J]. J Cent South Univ Technol, 2003, 34(3): 225-227. (in Chinese)
[13] YIN Hong-feng, XU Yong-dong, CHENG Lai-fei, ZHANG Li-tong. Effect of interphases on the properties of 3-D Cf/SiCm composites [J]. J Chin Ceram Soc, 2000, 28(1): 1-5. (in Chinese)
[14] LI J G, HAUSNER H. Reactive wetting in the liquid-silicon/solid carbon system [J]. J Am Ceram Soc, 1996, 79(4): 873-880.
[15] FITER E, GADOW R. Fiber-reinforced silicon carbide [J]. Am Ceram Soc Bull, 1986, 65(2): 326-335.
Foundation item: Project(2006CB600908) supported by the National Basic Research Program of China
Corresponding author: YAN Zhi-qiao; Tel: +86-20-61086627; E-mail: zhiqiaoyan@sina.com
DOI: 10.1016/S1003-6326(09)60183-7

