J. Cent. South Univ. Technol. (2009) 16: 0763-0767
DOI: 10.1007/s11771-009-0127-y
Overcharge performance of LiMn2O4/graphite battery with large capacity
LIU Yun-jian(刘云建), LI Xin-hai(李新海), GUO Hua-jun(郭华军),
WANG Zhi-xing(王志兴), HU Qi-yang(胡启阳), PENG Wen-jie(彭文杰)
(School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China)
Abstract: The LiMn2O4/graphite battery was fabricated and its 3 C/10 V overcharge performance was studied. Spinel LiMn2O4 was synthesized by solid-state method and 325680-type size full battery was fabricated. The structure and morphology of the powders were characterized by XRD and SEM technique, respectively. The battery explodes after 3 C/10 V overcharged test, and surface temperature of the battery case arrives at 290 ℃ in 12 s after exploding. Black air is given out with blast. Carbon, MnO, and Li2CO3 are observed in the exploded powders. The cathode electrode remains spinel structure with 5.0 V charged. Cracks in the cathode electrode particles are detected with the increase of voltage by SEM technique. The 5.0 V charged electrode can decompose into Mn3O4 at 400 ℃. It is demonstrated that the decomposition of 5.0 V charged electrode can be promoted and Mn4+ can be deoxidized to Mn2+ by carbon and electrolyte through the simulation of blast process.
Key words: LiMn2O4; MnO; battery; overcharge; blast
1 Introduction
The demand for high energy rechargeable battery for electric vehicle application has accelerated the development of lithium ion battery. However, large-size lithium ion battery is not yet applicable because of safety concerns primarily. Extensive applications of lithium ion batteries generate increasing safety concerns and many researchers are focused on the safety of lithium ion battery [1-5]. A main reason for the unsafe behavior is known to be the thermal runaway due to the chemical reactions of highly active electrode and electrolyte materials at elevated temperatures [6-8].
Spinel type LiMn2O4 has attracted particular interest for electric vehicle application because it is safety, environmentally benign, inexpensive and abundant in its raw material [9-12]. MACNEIL and DAHN [13-14] reported that the thermal stability between LixMn2O4 and solvent was better than that between Li0.5CoO2 and solvent. However, the manganese spinel battery with large capacity is still unsafe, such as 3 C/10 V over- charge in our recent experiments. A few researches have focused on the overcharge performance of LiMn2O4/ graphite battery although the safety performance of LiCoO2/graphite battery has been studied [15].
In order to develop adequate safety measures, it is important to analyze the thermal runaway mechanism of LiMn2O4 battery in detail. In this work, spinel LiMn2O4 was synthesized by a solid-state method, and manganese spinel battery with large capacity (10 A?h) was fabricated. The overcharge (3 C/10 V) performance of large capacity battery (10 A?h) was studied. And the reactions of cathode during the overcharge test were discussed.
2 Experimental
Spinel LiMn2O4 was synthesized by a solid-state method. Stoichiometric amounts of lithium carbonate (Li2CO3) and electrolytic manganese dioxide (MnO2, Aldrich) were firstly mixed together and ground in an agate mortar for 30 min, and then the mixture was preheated at 500 ℃ for 5 h before being recalcined at 750 ℃ for 10 h in air.
A 325680-type size battery was introduced for this study. LiMn2O4, carbon black, polyvinylidene fluoride (PVDF) and N′-methyl pyrrolidinone (NMP) were mixed together in a high-speed mixer at a certain ratio, and then the viscous slurry was coated onto an aluminum foil current-collector and dried at 120 ℃ under vacuum for 24 h. The cathode was pressed and divided into small patch. Graphite was chosen for anode. The cathode and anode were assembled through convoluting, and then enclosed in stainless steel can, and dried under vacuum for 36 h. Celglard 2400 microporous membrane and 1.0 mol/L LiPF6 in mixture of EC, EMC, DMC (1:1:1, in volume ratio) were chosen as separator and electrolyte, respectively.
The capacity test of battery was charged to 4.2 V at 0.5 C rate and 25 ℃, and then discharged to 3.0 V at 1 C rate and 25 ℃. For the cycling test, current rates for charge and discharge were held at 1 C.
The overcharge performance of 325680-type battery was tested by the DC power supply. Current limiting and temperature trip safety devices (e.g. PTC) were not used in the experimental batteries.
The X-ray diffractometer (XRD, Rint-2000, Rigaku) with Cu Kα radiation was employed to identify the crystalline phase of the synthesized LiMn2O4 and exploded powders. The particle size and morphological property of the prepared LiMn2O4 exploded powders were measured by scanning electron microscope (JEOL, JSM-5600LV) at an accelerating voltage of 20 kV.
3 Results and discussion
3.1 XRD and SEM analysis
Fig.1 shows the X-ray diffraction (XRD) pattern of LiMn2O4 powders. The diffraction peaks of the sample can be indexed as spinel LiMn2O4 phases with a space group Fd3m (JCPDS cards, No.35-0782). Because of no diffraction peaks of impurity phases, we obtained single-phase products of LiMn2O4 by solid-state method from Li2CO3 and electrolytic manganese dioxide. The lattice constant c of as-prepared LiMn2O4 powders is 0.824 1 nm, which is in a good agreement with the standard value (0.824 7 nm).
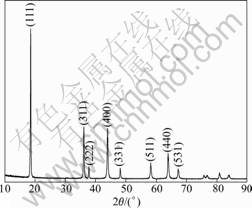
Fig.1 XRD pattern of LiMn2O4
The surface morphology of the LiMn2O4 powders is shown in Fig.2. The sample reveals secondary particle sizes of 8-12 μm and regular primary particles, and possesses crystalline morphology.
3.2 Electrochemical characterization
Fig.3 shows the discharge curve of the 325680-type LiMn2O4 battery performed at 3.0-4.2 V and 25 ℃. The initial discharge capacity of 325680-type battery is 10.45 A?h. The capacity of LiMn2O4 is 106 mA?h/g. The

Fig.2 SEM image of LiMn2O4

Fig.3 Discharge curve of LiMn2O4 battery
discharge curve of the LiMn2O4 electrode presents two undistinguished plateaus. The result shows that the as-synthesized LiMn2O4 product is expected to have better cycle performance as a cathode electrode material than the normal one. The capacity retains 84.2% after 300 cycles.
3.3 Voltage and temperature change at overcharge
Fig.4 shows the battery voltage and battery case temperature of a 325680-type lithium-ion battery overcharged at a 3 C rate. The battery voltage increases gradually to 5.1 V, and then rises sharply to 10 V because of the separator shutdown. The battery explodes when the voltage arrives at 10 V and lots of black gas is given out. The amount of gas increases with the increase of the battery temperature.
The battery case temperature remains low for the first 15 min, and then rises to 44 ℃ gradually while the voltage increases to 5.1 V. But the temperature of battery case surface ascends to 123 ℃ quickly with the voltage reaching 10 V. Finally, the battery case surface temperature arrives at 290 ℃ in 12 s, with exploding the battery.
According to Ref.[15], the evolved gas consisted of CO2, CO, H2, CH4, C2H6 and C2H4. It is reasonable to
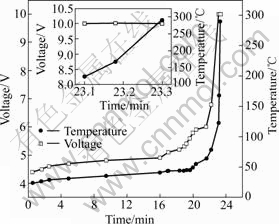
Fig.4 Overcharge curves of LiMn2O4 325680-type battery
consider that CO2, CH4, C2H6 and C2H4 are generated by the oxidation of the electrolyte at the cathode. The anode graphite can also be oxidized during the blast process because the heat and oxygen are released. But the blast process is finished in several seconds. The anode graphite cannot be oxidized completely. Furthermore, the gas is black. So, it can be concluded that the carbon powders should be evolved in the gas.
Fig.5 shows the SEM image of electrode after 3 C/ 10 V test. Compared with Fig.2, the original particles disappear, and the amount of small particles reuniting together is observed. This indicates that original LiMn2O4 particles react with electrolyte during the overcharge process. And the original large particles decompose into small particles and new substances form.

Fig.5 SEM image of electrode after overcharge
The structural change of the cathode powders after overcharge test is shown in Fig.6. Compared with Fig.1, the spinel phase of LiMn2O4 disappears. The peaks of carbon, MnO and Li2CO3 are indexed in the diffraction pattern. This indicates that the cathode electrode reacts with the electrolyte, and Mn4+ is deoxidized to Mn2+.
The structures of the cathode material in different charging states are shown in Fig.7. The results indicate that the cathode electrode retains spinel structure with 5.0 V overcharged. This shows that the spinel MnO2 has stable structure and thermal property at 5.0 V. But the overcharge results in the breakdown of the crystal structure of the cathode material due to the reaction between the electrolyte and the highly oxidized cathode.
Fig.8 shows the SEM image of LiMn2O4 electrode with 5.0 V charged. Compared with Fig.2, we can see that the surface of electrode is eroded by the electrolyte.
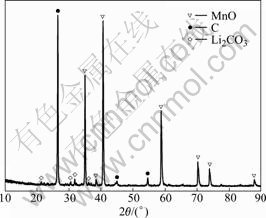
Fig.6 XRD pattern of exploded powders
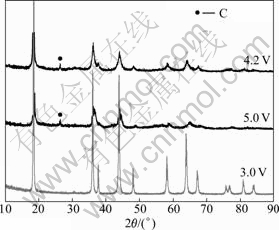
Fig.7 XRD patterns of cathode electrode with different charged states

Fig.8 SEM image of cathode electrode with 5.0 V charged
And small particles on the surface which are similar with Fig.5 can be observed. According to Fig.5, the small particles may be MnO and Li2CO3. The result suggests that the reaction between LiMn2O4 electrode and electrolyte takes place on the surface.
Furthermore, cracks in the particle can be observed clearly. It can be concluded that the appearance of crack may be the stress released from the breakdown of spinel structure. We can deduce that the number of cracks in the cathode electrode particle will be increased with increasing voltage. The crack is deleterious to the battery safety. The crack increases the contact area between the cathode particle and electrolyte and results in accelerating the react ion velocity between the cathode electrode and electrolyte. The crack can also result in the reaction between the cathode electrode and electrolyte entering into the interior of cathode electrode particle from the surface. So the crack is responsible for the energy release which is the direct reason for blast during the overcharge. This should be an important reason for the temperature increasing fleetly and blast finally.
In order to investigate the origin of MnO more precisely, the cathode electrode with 5.0 V charged was washed with EC+DEC (1:1 in volume ratio) mixed solution to remove the electrolyte salt. It was heated to 400 ℃ with graphite or electrolyte added under argon gas, respectively, and compared with that heated to 400 ℃ solely. Fig.9 shows the XRD patterns of cathode electrode with different additives. The phases of Mn3O4 and spinel MnO2 are observed under different additives. The phase of spinel MnO2 in the heated powders solely is obvious and the intensity of (111) peak for spinel phase is the highest. These results show that the 5.0 V charged electrode can decompose into Mn3O4 at 400 ℃.

Fig.9 XRD patterns of cathode electrode heated with different additives
But the reaction is not complete. The intensity of (111) peak for spinel MnO2 in the heated powders with electrolyte or graphite added is lower, which shows that the cathode electrode can be deoxidized by graphite and electrolyte, and the decomposition of cathode electrode is promoted. Although the phase of MnO is not observed, the reaction in which Mn4+ is deoxidized to Mn2+ is demonstrated. This may be that the heating process can not simulate the blast process commendably. MACNEIL and DAHN [14] just assumed that the cathode decomposed in the presence of solvent (i.e. EC) according to the following equation:
Mn2O4+2/5C3H4O3→2MnO+6/5CO2+4/5H2O (1)
In our experiment, we have demonstrated the validity of the reaction. Furthermore, we conclude that Mn2O4 can also be deoxidized by the graphite anode. The reaction can be expressed as
3MnO2+C→Mn3O4+CO2 (2)
MnO2 can decompose into Mn3O4 and release O2 at high temperature. The reaction is as follows:
3MnO2→Mn3O4+O2 (3)
In the overcharged powders, Li2CO3 can be easily thinkable because of the SEI film. And the solvent might react with lithium (either metallic or intercalated) as follows [1]:
2Li+C3H4O3(EC)→Li2CO3+C2H4 (4)
MACNEIL and DANH [14] found that the thermal stability of LixMn2O4 was decreased with the increase of the concentration of LiPF6. They speculated that the increase of the amount of HF made the increase of the dissolution of Mn into the heated electrolyte, which might keep the surface of the particles of MnO product clean and allowed the solvent oxidation to proceed more rapidly. In our experiment, we fabricated the battery using the electrolyte (V(EC):V(EMC):V(DMC)=1:1:1) with 0.25 mol/L LiPF6. Unfortunately, the battery still exploded after 3 C/10 V test. This result indicates that the reason for battery blast may be the amount of energy accumulation and the increase of inner temperature of battery, but not the speed of energy accumulation. So the safety problem should be solved by other methods, such as surface coating or safety electrolyte, to reduce the amount of energy. And this research will be studied in the future.
4 Conclusions
(1) Spinel LiMn2O4 is synthesized by solid-state method and 325680-type full battery is fabricated. The battery explodes after 3 C/10 V overcharge test, and the battery case surface temperature arrives at 290 ℃ in 12 s after blast and gives out lots of black gas. The evolved black gas consists of CO2, CO, H2, CH4, C2H6, C2H4 and carbon particles. Carbon, MnO, and Li2CO3 are observed in the exploded powders.
(2) SEM result shows that crack in LiMn2O4 particle appears with the increase of the voltage and the reaction velocity between the cathode electrode and electrolyte is accelerated by the crack. The 5.0 V charged electrode can decompose into Mn3O4 at 400 ℃. It is demonstrated that Mn4+ can deoxidize to Mn2+ by graphite and electrolyte and the decomposition of cathode electrode can be promoted through the simulation of exploded process.
References
[1] SPOTNITZ R, FRANKLIN J. Abuse behavior of high-power, lithium-ion batteries [J]. J Power Sources, 2003, 113(1): 81-100.
[2] LEE K H, SONG E H, LEE J Y, JUNG B K, LIM H S. Mechanism of gas build-up in a Li-ion battery at elevated temperature [J]. J Power Sources, 2004, 132(1): 201-205.
[3] YAMAUCHI T, MIZUSHIMA K, SATOH Y, YAMADA S. Development of a simulator for both property and safety of a lithium secondary battery [J]. J Power Sources, 2004, 136(1): 99-107.
[4] ZAGHIB K, CHAREST P, GUERFI A, SHIM J, PERRIER M, STRIEBEL K. Safe Li-ion polymer batteries for HEV applications [J]. J Power Sources, 2004, 134(1): 124-129.
[5] BALAKRISHNAN P G, RAMESH R, KUMAR T P. Safety mechanisms in lithium-ion batteries [J]. J Power Sources, 2006, 155(1): 401-414.
[6] ZHANG Z, FOUCHARD D, REA J R. Differential scanning calorimetry material studies: Implications for the safety of lithium-ion batteries [J]. J Power Sources, 1998, 70(1): 16-20.
[7] BIENSAN P, SIMON B, PERES J P, GUIBERT A, BROUSSELY M, BODET J M. On safety of lithium-ion batteries [J]. J Power Sources, 1999, 81/82(2): 906-912.
[8] IMHOF R, NOVAK P. Oxidative electrolyte solvent degradation in lithium-ion batteries: An in situ differential electrochemical mass spectrometry investigation [J]. J Electrochem Soc, 1999, 146(4): 1702-1711.
[9] XIA Y, ZHOU Y, YOSHIO M. Capacity fading on cycling of 4 V Li/LiMn2O4 batteries [J]. J Electrochem Soc, 1997, 144(6): 2593-2600.
[10] JANINA M, MARIUSZ Z, JACEK M, ZAJAC W, MOLENDA, BUCKO M. Electrochemical and high temperature physicochemical properties of orthorhombic LiMnO2 [J]. J Power Sources, 2007, 173(1): 707-711.
[11] LU W, BELHAROUAK I, LIU J, AMINE K. Effect of mixed LiBOB and LiPF6 salts on electrochemical and thermal properties in LiMn2O4 batteries [J]. J Power Sources, 2007, 174(2): 673-677.
[12] LI X F, XU Y L. Spinel LiMn2O4 active material with high capacity retention [J]. Applied Surface Science, 2007, 253(21): 8592-8596.
[13] MACNEIL D D, DAHN J R. The reaction of charged cathodes with nonaqueous solvents and electrolytes (I) [J]. J Electrochem Soc, 2001, 148(11): 1206-1210.
[14] MACNEIL D D, DAHN J R. The reaction of charged cathodes with nonaqueous solvents and electrolytes (II) [J]. J Electrochem Soc, 2001, 148(11): 1211-1215.
[15] TAKAHISA O, TAKASHI K, TAKAMI N, SHIMURA N, SATO Y, SEKINO M, SATOH A. Overcharge reaction of lithium-ion batteries [J]. J Power Sources, 2005, 146(1): 97-100.
(Edited by YANG You-ping)
Foundation item: Project(2007CB613607) supported by the National Basic Research Program of China
Received date: 2008-10-28; Accepted date: 2009-12-20
Corresponding author: LI Xin-hai, Professor; Tel: +86-731-88836633; E-mail: 13973168798@mail.hnmcc.com