
Synthesis of nonlinear optical fluorinated polyimide/inorganic composites for photonic devices
LI Guo-yuan(李国元)1, REN Li(任 力)1,2
1. School of Materials Science and Engineering, Nanyang Technological University, Singapore 639798;
2. Key Laboratory of Specially Functional Materials and Advanced Manufacturing Technology, Institute of Materials Science and Engineering, South China University of Technology, Guangzhou 510640, China
Received 10 April 2006; accepted 25 April 2006
Abstract: A nonlinear optical (NLO) active alkoxysilane chromophore (SGDR1) was synthesized. A fluorinated polyimide/SGDR1 composite was prepared to improve the poor temporal stability of second-order NLO effects of the reported poled sol-gel film. The poled composite film was characterized by FTIR, DSC, TGA and UV-Vis. The composite displays good hydrophobic properties, high glass transition temperature (266 ℃), and high decomposition temperature (433 ℃). The second harmonic coefficient d33 of the composite was measured to be 16.77 pm/V by using maker fringe technique. The new NLO composite exhibits 85 % of the original d33 over 720 h at 100 ℃ and possesses much better stability than the reported sol-gel film.
Key words: nonlinear optical material; fluorinated polyimide; chromphore; silicon; composite
1 Introduction
NLO materials have attracted steady attention for many years due to their potential applications in integrated optical devices such as optical switching and high-speed electro-optic devices[1,2]. The device-quality NLO materials must possess large optical nonlinearity, high thermal stability and low optical loss[3]. NLO sol-gel materials are promising materials because they have excellent optical quality, refractive index control of films and ease of device fabrication[4,5]. However, one of its major disadvantages is the low temporal stability of nonlinearity at elevated temperature[6]. Recently, organic-inorganic sol-gel materials have received significant attention because of higher thermal stability [7,8]. By incorporation of a high performance organic polymer into an inorganic sol-gel material, the resulting organic-inorganic composite may display the desired properties of both components, which may be a potential approach to enhance long-term NLO stability. Aromatic polyimide, especially fluorinated polyimide, have been widely used in the advanced microelectronic and optoelectronic devices manufacturing because they possess a combination of properties, such as high thermal stability, low dielectric constants, high optical transparency and good hydrophobic property[9]. Furthermore, it has been found that the internal production of water due to the curing of precursor of polyimide, poly(amic acid), aids the hydrolysis of the alkoxysilane, and the carboxylic acid group of the poly(amic acid), being a Bronsted acid, might have a catalytic effect on hydrolysis and condensation of the alkoxysilane[10]. This will lead to no addition of catalyst and water and decrease phase separation between the inorganic and organic phase. In this study, a fluorinated polyimide/NLO alkoxysilane dye composites was prepared and characterized in order to obtain a NLO materials with better comprehensive properties especially higher stability.
2 Experimental
2.1 Materials
1,3-Phenylenediamine (mPDA) was purified by Disperse red 1 (DR1) was purified by recrystallization from ethanol. Tetrahydrofuran (THF)and Hexane were purified by distillation over sodium chips and benzophenone ketal. 4,4’-(Hexafluoroisopropylidene)diphthalic anhydride (6FDA) was dried under a vacuum at 150 ℃ overnight before use. The other materials were purchased from Aldrich, Merk and Clariant and used without further purification.
2.2 Synthesis of alkoxysilane chromophore (SGDR1)
The schematic of synthesis of SGDR1 is shown in Fig. 1. At a two-necked round-bottomed Schlenk flask, the ICTES (2.2 mL, 8.08 mmol) and DR1 (1.27 g, 4.04 mmol) were dissolved in freshly dried THF (30 mL). The mixture was then stirred at room temperature for 10 min. After that, it was refluxed for 4 h with a catalytic amount (25 μL) of dibutyltin dilaurate (DBTDL). The solution was poured in distilled hexane and the resulting red precipitated powder was collected and washed again with hexane/THF until no DR1 was detected. The product (SGDR1) was finally dried under vacuum at 60 ℃ for 24 h in 80% yield and then stored in a vacuum desiccator.
Synthesis of fluorinated poly(amic acid): m-PDA (2.0 mmol) was dissolved in 10 mL anhydrous NMP at 0 ℃, followed by the addition of 6FDA (2.08 mmol) at once. After stirring at a temperature between 0 and 5 ℃ for 4 h, the solution was then stirred at room temperature for 20 h. The resulting fluorinated poly(amic aicd) shown in Fig.1 was obtained.
2.3 Preparation of NLO fluorinated polyimide/
SGDR1 composites
The solution of SGDR1 (0.15 g) in THF (1 mL) and NMP (1 mL) was added into 4 mL of fluorinated poly(amic acid) solution. The mixed solution was then stirred at room temperature for more than 4 h to be homogeneous. The resulting solution was filtered through a 0.45 μm Teflon membrane and was then spin-coated on a substrate (glass slide or indium-tin oxide glass) at various speeds ranging from 500 r/min to 4 000 r/min for different durations from 5 s to 30 s under clean-room conditions. After dried in a vacuum oven at 65 ℃ for 12 h to remove residual solvent, the films were subjected to concurrent poling and cure. Composite films were poled in a temperature-controlled oven under a corona discharge field using 75 mm diameter tungsten wire in a wire-to-plane geometry of 1.5 cm above a Teflon protected, grounded copper block. A two-step corona poling process was employed to orient the NLO chromophores. The sample was heated to 200 ℃ at a heating rate of 5 ℃/min and successively held at this temperature for 1 h, while a 6 kV of poling voltage was applied to a corona wire simultaneously. Finally, the sample was cooled to room temperature in the presence of the poling field. The composite films had thicknesses ranging from 1 to 4 mm.
2.4 Measurements
1H NMR spectrum was measured in dimethyl sulfoxide-d6 (DMSO-d6) using Bruker 400 MHz instrument. Elemental analysis was performed by EURO EA elemental analyzer. FTIR spectroscopy was carried out with Perkin Elmer System 2000 FTIR. Glass transition temperatures (tg) of the polyimide and compo-
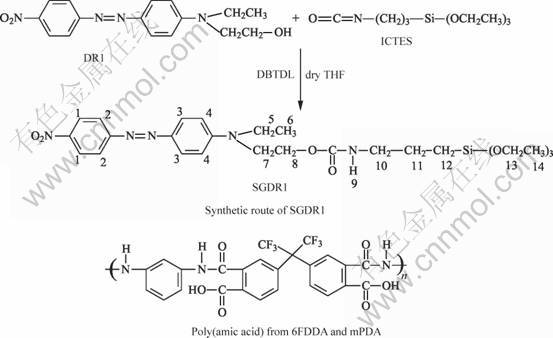
Fig.1 Synthetic route of SGDR1 and chemical structure of fluorinated poly(amic acid)
site were determined through DSC analysis that was performed by a TA Instruments 2010 at a heating rate of 10 ℃/min in nitrogen atmosphere (50 mL/min). The UV-Vis spectra were measured with a Shimadzu model UV-2501PC spectrophotometer. The contact angle was measured using First Ten Angstroms system with automatic gain control camera with deionized water as reference liquid. The surface energy was automatically computed through Girifalco-Good-Fowkes-Young Model. In water absorption test, the polyimide films on glass slide were cut into 20 mm×20 mm samples. The samples were then dried at 105 ℃ for 1 h, weighed, and kept in deionized water at 25 ℃ for 24 h and weighted again. SHG measurements were carried out with a p-polarized Q-switched Nd:YAG pulse laser operating with 10 Hz repetition rate and a 4-5 ns pulse width at 1 064 nm. A Y-cut quartz crystal was used as the reference.
3 Results and discussions
The 1H NMR spectrum and the spectrum peak assignment of SGDR1 were shown in Fig.2. Referred to Ref.[11], it can be found that the peaks at 8.37×10-6, 7.94×10-6, 7.84×10-6 and 6.92×10-6 are attributed to H atoms of phenylene H1, H2, H3 and H4, respectively. The peak at 7.20×10-6 was assigned to proton in carbamate, H9. The peaks at 4.15×10-6, 3.73×10-6, 3.67×10-6, 3.53×10-6, 2.96×10-6, 1.45×10-6 and 0.53×10-6 are assigned to H atom in methylene H8, H13, H7, H5, H10, H11 and H12, respectively. H atoms in methyl H6 and H14 were overlapped at the peak 1.15×10-6. FTIR spectra of DR1, ICTES and SGDR1 were shown in Fig.3. The stretching vibration of the hydroxyl group in DR1 is observed at around 3 273 cm-1. For ICTES, the strong absorption peak corresponding to N==C==O stretching was observed at 2 272 cm-1. In spectrum of SGDR1, the absorption peaks of N==C==O and OH vanished and the new absorption peaks at 3 327, 1 690 and 1 542 cm-1 emerged, which contributed by the NH stretching, carbonyl (C==O) stretching and NH bending, respectively. In addition, asymmetric stretching and symmetric stretching of NO2 were observed at 1 509 and 1 343 cm-1, respectively. The experimental element analysis results are in agreement with the theoretical ones. Based on 1H NMR, FTIR and element analysis results, structure of SGDR1 was confirmed as expected.
The thermal properties of SGDR1 were characterized through DSC and TGA. The results show that the melting point of the SGDR1 is 97 ℃ and the onset decomposition temperature is 225 ℃, which ensures that the chromophore could not decompose during curing at 200 ℃. The UV-Vis results show that the maximum absorption wavelength lmax of SGDR1 in NMP and THF are 498 and 474 nm, respectively.
FTIR spectra of the fluorinated poly(amic acid)/SGDR1composites cured at 65 and 200 ℃ are shown in Fig.4. After curing of poly(amic acid)/SGDR1 at 200 ℃ for 1 h, the characteristic absorption peak of imide group at 1 780 cm-1 emerged. It is noted that the characteristic absorption peak of Si―O―C2H5 around
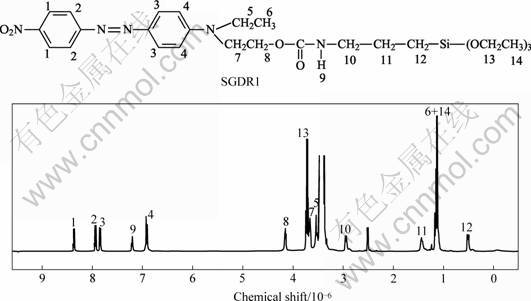
Fig.2 1H NMR spectrum of SGDR1
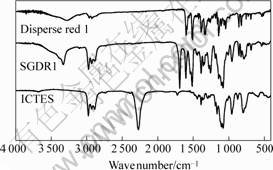
Fig.3 FTIR spectra of disperse red 1, ICTES and SGDR1
1 070 cm-1 disappeared and the absorption peak around
1 100 cm-1 becomes broader after curing. This elucidated that Si―O―Si bond has formed. The negligible change of the symmetric nitro absorption at 1 340 cm-1 and the C―N stretching of the aromatic nitro compound at 860 cm-1 also indicate that the NLO moieties did not decompose during the curing process. The FTIR results suggest that fluorinated polyimide/SGDR1composites were well prepared through curing at 200 ℃.
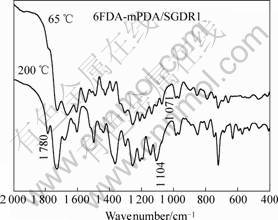
Fig.4 FTIR spectra of fluorinated poly (amic acid)/ SGDR 1
composites cured at 65 ℃ and 200 ℃
Thermal properties of fluorinated polyimide/SGDR1 composite were characterized by DSC and TGA and the results are shown in Fig.5. It can be found that the composite film have high tg (266 ℃) and td5 (433 ℃). tg of the fluorinated polyimide was measured to be 233 ℃. The results clearly show that tg of composite is higher than that of fluorinated polyimide, which is attributed to the formation of the silicon crosslink network.
UV-Vis absorption was used to characterize the transparency of NLO composite film. Fig.6 shows the UV-Vis spectrum of fluorinated polyimide/SGDR1 composite film. It can be found that λmax is 485 nm. According to the UV results of SGDR1, the absorption

Fig.5 DSC and TGA of fluorinated polyimide/SGDR1 composite film
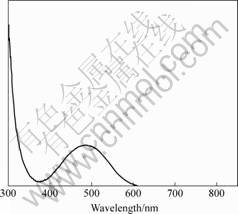
Fig.6 UV-Vis spectrum of fluorinated polyimide/SGDR1 composite film
peak is attributed to SGDR1.
As well known, water absorption and moisture content are very important to the electro-optical devices. It was reported that incorporation of fluorine or silicon into materials is an effective method to decrease the water absorption [12,13]. The relationship between contact angle and time of the composite film is shown in Fig.7. The average contact angle, surface energy and water absorption of the composite film are measured to be 90.58? , 18.17 mN/m, and 0.08%. The results show that the composite film exhibits excellent hydrophobic properties, which can significantly improve the stability and reliability of the optoelectronic materials and devices.
SHG results show that the NLO composite film possess d33 value of 16.77 pm/V. And its temporal stabilities of d33 value were studied at both room temperature and 100 ℃, respectively. Results show that there is no decay of d33 value at room temperature after 720 h. Fig.8 shows its temporal stability of d33 value at 100 ℃. It can be found that the composite film remained
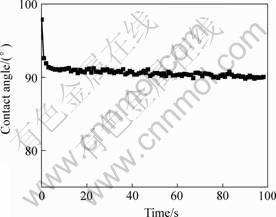
Fig.7 Contact angles of fluorinated polyimide/SGDR1 composite film
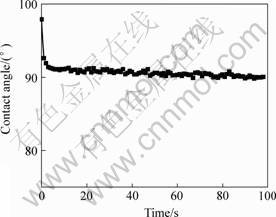
Fig.8 Temporal stability of d33 of NLO fluorinated polyimide /SGDR1 composites at 100 ℃
85% of the original d33 value after 720 h at 100 ℃. Compared with the reported sol-gel films [6], the results clearly reveal that fluorinated polyimide/SGDR1 composite exhibited higher temporal stability of d33 value. It is suggested that the incorporation of fluorinated polyimide into the inorganic materials could lead to better comprehensive properties especially higher stability. The better properties might be attributed to the existence of the fluorine in the side-chain of the polyimide and the formation of the silicon crosslinked network in the composite. The mechanism is still on investigation.
4 Conclusions
A NLO alkoxysilane dye SGDR1 and a fluorinated polyimide/inorganic composite film were prepared. Incorporation of fluorinated polyimide into the inorganic material can significantly increase the thermal stability and long-term stability of d33. The composite film also exhibits better comprehensive properties than the reported inorganic sol-gel materials.
References
[1] DHANUSKODI S, MARY P A A. Growth and characterization of a new nonlinear optical crystal-ammonium borodilactate[J]. J Crystal Growth, 2003, 253: 424-428.
[2] MAREDR S R, KIPPELEN B, JEN A K Y, PEYHGAMBARIAN N. Design and synthesis of chromophores and polymersfor electro-optic and photorefractive applications[J]. Nature, 1997, 388: 845-851.
[3] Gunter P. Nonlinear Optical Effects and Materials[M]. New York Springer-Verlag, 2000. 163-168.
[4] IKUSHIMA J, FUJIWARA T, SAITO K. Silica glass: A material for photonics[J]. J Appl Phys, 2000, 88: 1201-1213.
[5] JENG R J, CHE YM, CHEN H I, KUMAR J, S. TRIPATHY K, Phenoxysilicon polymer with stablesecond-order optical nonlinearity[J]. Macromolecules, 1993, 26: 2530.-2534
[6] CHOI D H, PARK J H, LEE J H, LEE S D. Stability of second-order nonlinear optical properties in sol-gel matrix bearing silylated chalcone and disperse red 1[J]. Thin Solid Films, 2000, 360: 213-221.
[7] JENG R J, CHEN C C, CHANG C P, CHEN C T, SU W C, Thermally stable corosslinked NLO materialsbased on maleimides[J]. Polymer, 2003, 44: 143-155.
[8] HSUIE G H, LEE R H, JENG R J. All sol-gel organic-inorganicnonlinear optical materials based onmelanines and an alkoxysilane dye[J]. Polymer, 1999, 40: 6417-6428.
[9] YANG S Y, GE Z Y, YIN D X, LIU J G, LI Y F, FAN L. Synthesis and characterization of novel fluorinated polyimides derived from 4,4-[2,2,2-trifluoro-1-(3-trifluoromethylphenyl)ethylidene] di- phthalic anhydride and aromatic diamines[J]. J Polym Sci Part A: Polym. Chem, 2004, 42: 4143-4152.
[10] PALMLOF M, HJERTBERG T, SULTAN B A. Crosslinking reactions of ethylene vinyl silanecopolymers at prosessing temperature[J]. J Appl Polym Sci, 1991, 42: 1193-1203.
[11] CUI Y J, WANG M Q, CHEN L J, QIAN G D, Synthesis andspectroscopic characterization ofan alkoxysilane dye containing C. I. Disperse Red 1[J]. Dyes and Pigments, 2004, 62: 43-47.
[12] REN L, ZENG F S, NING P, CHEN Z Q, KO T M, Effect of addition orders on theproperties of fluorinecontaining copolyimides[J]. J Appl Polym Sci, 2000, 77: 3252-3258.
[13] LI Ren, LI G Y, SHEN J R, JIA D M. Effects of monomer addition sequences on the properties of silicon-containing copolyimides[J]. Polym Int, 2005, 54(7): 1097-1101.
(Edited by CHEN Ai-hua)
Corresponding author: LI Guo-yuan; Tel: +65-67904583; Fax: +65-67909081; E-mail: asgyli@ntu.edu.sg