钒渣提钒和铬循环冶金工艺:(I)含铬钒酸盐溶液分离回收钒
来源期刊:中国有色金属学报(英文版)2021年第3期
论文作者:王学文 杨明鄂 孟钰麒 高大雄 王明玉 付自碧
文章页码:807 - 816
Key words:chromium-containing vanadate solution; calcium salt precipitating vanadium; sodium bicarbonate leaching; ammonium salt precipitating vanadate; cyclic metallurgical process
摘 要:研究含铬钒酸盐溶液分离回收钒的循环冶金工艺,工艺过程包括溶液中钒的选择性沉淀、沉淀物中钒的浸出及五氧化二钒的制备。在含铬钒酸盐溶液中加入Ca(OH)2 进行球磨,不仅可以使其中的钒选择性沉淀析出, 而且沉淀物的浸出动力学性能得到显著改善。在含铬钒酸盐溶液中按 Ca/V摩尔比1.75:1加入Ca(OH)2,室温球磨60 min,钒的沉淀率达99.59%, 而铬的沉淀率只有0.04%。过滤得到的沉钒渣按液固比4:1 mL/g加水,并按NaHCO3/V摩尔比2.74:1加入NaHCO3,室温搅拌60 min,过滤得浸出液,钒的浸出率为99.35%。所得浸出液先通入CO2调pH至 8.0,再按NH4HCO3/NaVO3摩尔比1:1加入NH4HCO3,室温搅拌8 h结晶析出NH4VO3,过滤后,含氨的结晶液重新用于钒酸钙沉淀的浸出,温室搅拌1 h后钒的浸出率>99%,最后,将所得NH4VO3晶体在560 °C煅烧2 h得到纯度为99.6%的V2O5。
Abstract: The separation and recovery of V from chromium-containing vanadate solution were investigated by a cyclic metallurgical process including selective precipitation of vanadium, vanadium leaching and preparation of vanadium pentoxide. By adding Ca(OH)2 and ball milling, not only the V in the solution can be selectively precipitated, but also the leaching kinetics of the precipitate is significantly improved. The precipitation efficiency of V is 99.59% by adding Ca(OH)2 according to Ca/V molar ratio of 1.75:1 into chromium-containing vanadate solution and ball milling for 60 min at room temperature, while the content of Cr in the precipitate is 0.04%. The leaching rate of V reaches 99.35% by adding NaHCO3 into water according to NaHCO3/V molar ratio of 2.74:1 to leach V from the precipitate with L/S ratio of 4:1 mL/g and stirring for 60 min at room temperature. The crystals of NH4VO3 are obtained by adjusting the leaching solution pH to be 8.0 with CO2 and then adding NH4HCO3 according to NH4HCO3/NaVO3 molar ratio of 1:1 and stirring for 8 h at room temperature. After filtration, the crystallized solution containing ammonia is reused to leach the precipitate of calcium vanadates, and the leaching efficiency of V is >99% after stirring for 1 h at room temperature. Finally, the product of V2O5 with purity of 99.6% is obtained by calcining the crystals at 560 °C for 2 h.
Trans. Nonferrous Met. Soc. China 31(2021) 807-816
Xue-wen WANG1, Ming-e YANG1,2, Yu-qi MENG1, Da-xiong GAO1, Ming-yu WANG1, Zi-bi FU3
1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. Hunan Automotive Engineering Vocational College, Zhuzhou 412001, China;
3. State Key Laboratory for Comprehensive Utilization of Vanadium and Titanium Resources, Pangang Group Research Institute Co., Ltd., Panzhihua 617000, China
Received 16 April 2020; accepted 28 September 2020
Abstract: The separation and recovery of V from chromium-containing vanadate solution were investigated by a cyclic metallurgical process including selective precipitation of vanadium, vanadium leaching and preparation of vanadium pentoxide. By adding Ca(OH)2 and ball milling, not only the V in the solution can be selectively precipitated, but also the leaching kinetics of the precipitate is significantly improved. The precipitation efficiency of V is 99.59% by adding Ca(OH)2 according to Ca/V molar ratio of 1.75:1 into chromium-containing vanadate solution and ball milling for 60 min at room temperature, while the content of Cr in the precipitate is 0.04%. The leaching rate of V reaches 99.35% by adding NaHCO3 into water according to NaHCO3/V molar ratio of 2.74:1 to leach V from the precipitate with L/S ratio of 4:1 mL/g and stirring for 60 min at room temperature. The crystals of NH4VO3 are obtained by adjusting the leaching solution pH to be 8.0 with CO2 and then adding NH4HCO3 according to NH4HCO3/NaVO3 molar ratio of 1:1 and stirring for 8 h at room temperature. After filtration, the crystallized solution containing ammonia is reused to leach the precipitate of calcium vanadates, and the leaching efficiency of V is >99% after stirring for 1 h at room temperature. Finally, the product of V2O5 with purity of 99.6% is obtained by calcining the crystals at 560 °C for 2 h.
Key words: chromium-containing vanadate solution; calcium salt precipitating vanadium; sodium bicarbonate leaching; ammonium salt precipitating vanadate; cyclic metallurgical process
1 Introduction
Vanadium is an important nonferrous metal element which is widely applied in metallurgical, material and chemical industries [1,2]. Vanadium and chromium are the accompanying elements in the ore of vanadium titano-magnetite [3,4]. During the smelting of the ore, vanadium and chromium are mainly enriched in converter slag. The converter slag is usually called vanadium slag [5], which is the main material of V2O5 production [6,7].
The conventional process of V2O5 production with vanadium slag includes sodium roasting, leaching and purification, V2O5 production, and wastewater purification [8]. V2O5 is produced by adding (NH4)2SO4 into the purified solution to obtain the precipitate of ammonium polyvanadate under pH 1.8-2.1 at 85-95 °C, and then to calcine the precipitate at about 550 °C for 2-3 h [9]. After vanadium precipitation, a solution containing Cr(VI) and a small amount of V(V) is formed. The contents of Cr(VI) and V(V) are 0.5-5.0 and 0.1-1.0 g/L, respectively, in the solution. Cr(VI) and V(V) are all harmful to lives, and sometimes can be fatal [10]. To protect the environment, many methods were used to treat the solution, including solvent extraction [11,12], ion exchange [13], chemical and electrochemical precipitation, membrane filtration and sorption [14]. However, these methods are not suitable for industrial production because of high cost and secondary pollution. For example, the Cr(VI) and V(V) can be completely separated and recovered from the solution using ion exchange resin D314 [13], but the operating life of the resin is shortened as Cr(VI) and V(V) are all strong oxidants in acid solution. Therefore, the solution has no choice but to be reduced with Na2SO3 or iron scurf to form the precipitate containing Cr(III), V(IV) and V(III), which is usually called V-Cr-bearing reducing slag [15]. After filtration, the solution containing trace amounts of V and Cr is evaporated to form the mixture of Na2SO4 and (NH4)2SO4 [16]. The mixture of Na2SO4 and (NH4)2SO4 and V-Cr- bearing reducing slag are all dangerous solid waste, and the handling of them have become a thorny problem in many plants in China.
In order to avoid the generation of typical solid wastes in the production of V2O5 with vanadium slag, researches have been carried out, including adding calcium to precipitate vanadium [6,8]. Although the precipitation of V can reach >99% by adding excessive amount of lime into vanadate solution under stirring [17], the leaching performance of the obtained precipitate of calcium vanadates is not good. Using ammonium carbonate salt solution to leach the precipitate, the leaching efficiency of V was 93.5%-94.3% [17]. Even if the precipitate was leached twice with sodium carbonate solution, the leaching efficiency of V was only 95.0%-96.5% [18]. Moreover, when the leaching solution is used to crystallize NH4VO3 by adding ammonium carbonate, and then the crystallized solution is returned to leach V from the precipitate, the escape of ammonia is a difficult problem to overcome. Due to the escape of ammonia, the production site environment is harsh, and the operators cannot accept it. That is why Pangang Group Research Institute Co., Ltd. entrusted the authors to carry out technical research on this problem.
It has been found that adding calcium salt to precipitate V under ball milling not only significantly improved the leaching efficiency of V from calcium vanadate precipitates, but also successfully solved the technical problem of ammonia escape, and developed the cyclic metallurgical process to extract V and Cr from vanadium slag. The so-called cyclic metallurgical process refers to a process that can extract valuable metals from metallurgical materials economically, efficiently, cleanly and environment-friendly, and can also separate and recycle chemical raw materials and water used in the process.
Using the cyclic metallurgical process, can not only V and Cr be separated and recovered from vanadium slag, but also the production of V-Cr-containing reducing slag and ammonia nitrogen wastewater be avoided [19]. In the process, by ball milling, Ca(OH)2 is added into the leaching solution of the vanadium slag roasted with sodium to make V selectively precipitated. After filtering, PbCO3 is added into V precipitated solution to precipitate and enrich Cr. Finally, NaHCO3 and water are separated and recovered by electrodialysis to realize the recycling of NaHCO3 and water in the process. The present work focused on the separation and recovery of vanadium.
2 Experimental
2.1 Materials and analysis
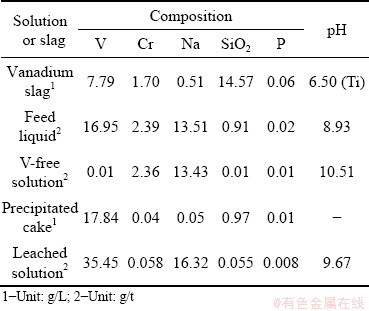
The chromium-containing vanadate solution was provided by Pangang Group Vanadium & Titanium Resources Co., Ltd., which was obtained by leaching vanadium slag roasted with Na2CO3. The solution was used as the feed liquid in the experiments. The compositions of the solution and the vanadium slag are both listed in Table 1. All the reagents used in tests are of analytical grade. The compositions of experimental samples were determined by chemical methods and inductively coupled plasma emission spectroscopy (ICP) with a PS-6 Plasma Spectrovac, Baird (USA). The X-ray diffraction (XRD) patterns were recorded on a Rigaku Miniflex diffractometer with Cu Kα X-ray radiation at 35 kV and 20 mA. The pH was determined with ORION-230A which was made in USA, and the degree of accuracy was 0.01. The internal structure of the precipitate particles was observed with 4XC metallographic microscope (Shanghai Optical Instrument No. 5 Factory Co., Ltd.).
Table 1 Compositions of solution and vanadium slag in cyclic metallurgical process to separate and recover V
2.2 Experimental procedure
The separation and recovery of vanadium from feed liquid were performed according to the flow sheet shown in Fig. 1, which includes the procedure of vanadium selective precipitation, vanadium leaching and vanadium pentoxide preparation.
The procedure of vanadium selective precipitation is performed by adding Ca(OH)2 into feed liquid according to Ca/V molar ratio of 1.35-1.75 and ball milling for 20-70 min under different speeds at room temperature. After filtration, the V free solution is used to recover chromium.
The vanadium in the precipitate was leached with NaHCO3 solution. Meanwhile, the calcium was changed into calcium carbonate. The leaching was carried out by adding NaHCO3 into water according to NaHCO3/V molar ratio of 1.96-2.74 with L/S ratio of 4:1 mL/g and leaching for 10-120 min at different temperatures. Then, the crystals of NH4VO3 were obtained by adding NH4HCO3 into the leaching solution according to NH4HCO3/V molar ratio of 1:1 under pH 8. After NH4VO3 crystal was separated, the solution was returned to leach V so as to avoid the discharge of ammonia-nitrogen wastewater. Finally, the product of V2O5 with purity 99.6% was obtained by calcining the NH4VO3 crystals at 560 °C for 2 h.
3 Results and discussion
3.1 Selective precipitation of vanadium
3.1.1 Vanadium precipitation method
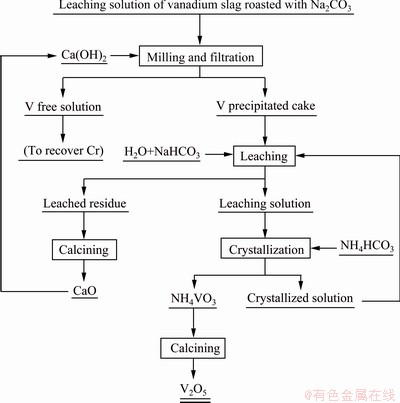
Fig. 1 Schematic flow-sheet of modified production process of V2O5 with vanadium slag
It was known that calcium vanadate is insoluble, while calcium chromate is soluble and its solubility is >20 g/L at room temperature [20]. Therefore, Ca(OH)2 can be used as the precipitator to selectively precipitate vanadium from chromium-containing vanadate solution. According to Ref. [8], the forms of calcium vanadate precipitates are different under different pH. The precipitate is Ca(VO3)2 under pH 5-8; when the pH is 9-10.6, the precipitate is Ca2V2O7; if the pH is 10.6-12, the precipitate is Ca3(VO4)2 [18]. It is found that by adding Ca(OH)2 into feed liquid according to Ca/V molar ratio of 1.5:1 and stirring for 1 h at room temperature, the precipitation efficiency of V is only 42.31%, the pH of precipitated solution is 9.3, the surface of the precipitate particles is Ca(VO3)2, and there is a lot of unreacted Ca(OH)2 inside the particles; by stirring for 1 h at 80 °C, the precipitation efficiency of V is 88.24%, the pH is 9.7, the surface of the precipitate particles is mainly Ca2V2O7, and unreacted Ca(OH)2 inside the particles is reduced; by ball milling for 1 h at room temperature, the precipitation efficiency of V is 97.31%, the pH is 10.2, the precipitate particles are all aggregates of Ca3(VO4)2, and unreacted Ca(OH)2 in the particles is difficult to find. As we all know, Ca(OH)2 is slightly soluble in water. When Ca(OH)2 is added into feed liquid under stirring, the precipitates of calcium vanadate formed by the reaction immediately adhere to the surface of Ca(OH)2. As the thickness of the calcium vanadate coating increases, the reaction rate decreases and eventually the reaction stops. However, under ball milling, the calcium vanadate attached to the surface of Ca(OH)2 can be stripped off in time, and the reaction interface is constantly updated to ensure that the added Ca(OH)2 can be fully utilized. Therefore, ball milling to precipitate vanadium was used in the subsequent experiments.
3.1.2 Effect of Cr concentration
Figure 2 shows the experimental results obtained by adding Ca(OH)2 into feed liquid with different Cr concentrations according to Ca/V molar ratio of 2:1 and ball milling for 60 min under 120 r/min at room temperature. The concentration of Cr in feed liquid is adjusted by adding Na2CrO4. Figure 2 shows that Cr concentration has little effect on the precipitation efficiency of V. With the increase in Cr concentration from 1.5 to 15.5 g/L, the concentration of V in the precipitated solution hardly changes, which is maintained at ~0.05 g/L, that is, the precipitation efficiency of V is about 99.7%, and the content of Cr in the precipitates is <0.05%. This indicates that by adding Ca(OH)2, V can be selectively precipitated from the leaching solutions of low-chromium or high-chromium vanadium slag.
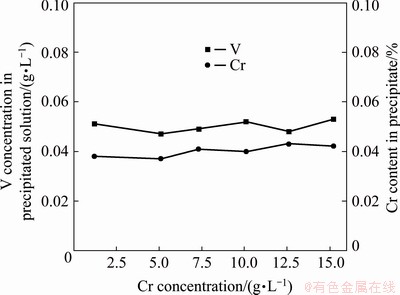
Fig. 2 Effect of Cr concentration on vanadium precipitation (Ca/V molar ratio of 2:1)
3.1.3 Effect of Ca(OH)2 addition
Figure 3 shows the experimental results obtained by adding Ca(OH)2 into feed liquid according to different Ca/V molar ratios and ball milling for 60 min under 120 r/min at room temperature. As can be seen, the precipitation efficiency of V increases from 84.23% to 99.59% with the increase in Ca/V molar ratio from 1.35:1 to 1.75:1, and then there is no significant change with the further increase in Ca/V molar ratio. However, the precipitation efficiency of Cr is always kept at ~1.5%, which is caused by mechanical entrainment. Therefore, the Ca/V molar ratio was maintained at 1.75:1 in the subsequent experiments.
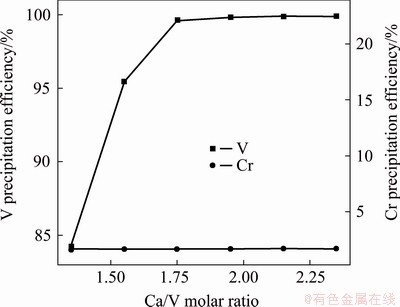
Fig. 3 Effect of Ca(OH)2 addition on vanadium precipitation (Ball milling for 60 min)
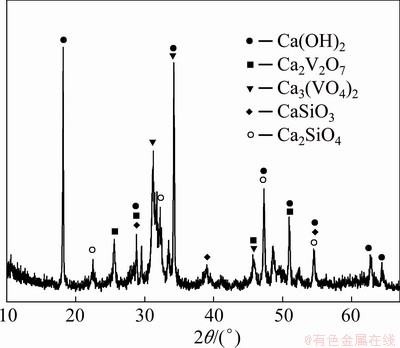
Fig. 4 XRD pattern of V precipitated cake
Figure 4 shows the XRD pattern of the precipitate obtained by adding Ca(OH)2 into feed liquid according to Ca/V molar ratio 1.75:1 and ball milling for 60 min under 120 r/min at room temperature. The compositions of V free solution and precipitated cake are listed in Table 1. As seen, there are Ca3(VO4)2, Ca2V2O7, CaSiO3, Ca2SiO4 and Ca(OH)2 in the cake. This indicates that the form of V in the cake is mainly Ca3(VO4)2, and the effective utilization rate of added Ca(OH)2 reaches ~85.7%. Adding Ca(OH)2 to precipitate V can cause the reaction of solid-solid transformation to occur in solution, so the added Ca(OH)2 is difficult to be fully utilized, especially by stirring. During the experiment, it is found that with the increase in Ca(OH)2 addition, the existing form of calcium vanadate is different. It turns from Ca(VO3)2, Ca2V2O7 to Ca3(VO4)2, and the transformation is faster under ball milling. The precipitation can be expressed by the following equations:
2VO3- +Ca2+=Ca(VO3)2↓ (1)
Ca(VO3)2+Ca(OH)2=Ca2V2O7↓+H2O (2)
Ca2V2O7+Ca(OH)2=Ca3(VO4)2↓+H2O (3)
SiO44-+ 2Ca2+=Ca2SiO4↓ (4)
SiO32-+Ca2+=CaSiO3↓ (5)
3.1.4 Effect of ball milling time
Figure 5 shows the experimental results obtained by adding Ca(OH)2 into feed liquid according to Ca/V molar ratio of 1.75:1 and ball milling for different time under 120 r/min at room temperature. It is shown that the initial speed of V precipitation in the solution is very fast, and then it becomes slow. As the reaction progresses, the probability of contact between Ca(OH)2 and V in the solution becomes low. The precipitation efficiency of V reaches 77.43% after ball milling for 20 min, and then it increases from 77.43% to 99.15% with the increase in ball milling time from 20 to 40 min. Then, the precipitation efficiency of V remains almost the same with the further increase in ball milling time. Therefore, the ball milling time should be ≥40 min.
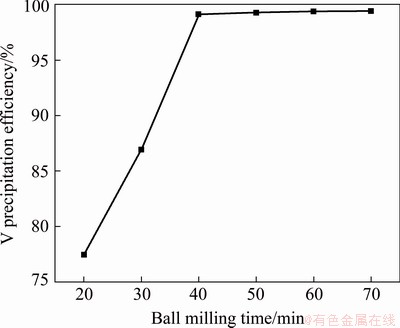
Fig. 5 Effect of ball milling time on vanadium precipitation (Rotating speed of 120 r/min)
3.1.5 Effect of rotating speed
Figure 6 shows the experimental results obtained by adding Ca(OH)2 into feed liquid according to Ca/V molar ratio of 1.75:1 and ball milling for 60 min under different speeds at room temperature. As seen, the precipitation efficiency of V increases from 86.13% to 99.25% with the increase in ball milling rotating speed from 40 to 100 r/min. Then, it increases slowly with the further increase in rotating speed. This indicates that the calcium vanadate precipitates are easy to break, and maintaining ball milling rotating speed at 100-120 r/min can meet the process requirements.
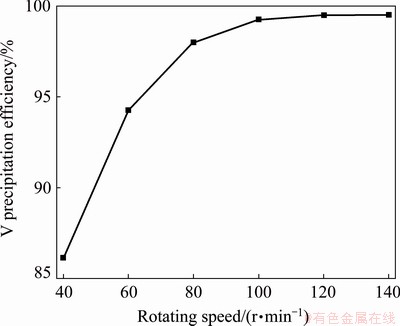
Fig. 6 Effect of ball milling rotating speed on vanadium precipitation (At room temperature)
3.2 Vanadium leaching
3.2.1 Vanadium leaching method
Figure 4 shows that the V existing in the precipitated cake is calcium vanadates, and the V can be leached with NaHCO3 or Na2CO3. Exploration experiments show that the leaching performances of the precipitate obtained by different methods are different. By adding Ca(OH)2 into feed liquid according to Ca/V molar ratio of 1.75:1 and stirring, and then adding NaHCO3 into water according to NaHCO3/V molar ratio of 3:1 to leach V from the precipitate with L/S ratio of 4:1 mL/g and stirring for 1 h at 80 °C, the leaching efficiency of V is 95.12%, and the pH of leaching solution is 10.58; while by ball milling for 1 h at room temperature, the leaching efficiency of V is 99.61%, and the pH is 11.13. The content of V in the ball milling leached residue is 0.15%, while that in the stirring leached residue is 0.95%, which is consistent with the results (0.91% V) obtained by other authors [18]. The V in the stirring leached residue is mainly wrapped by calcium carbonate. However, when V precipitated cake is obtained by ball milling under other conditions, and then NaHCO3 is added into water according to NaHCO3/V molar ratio of 3:1 to leach V from the precipitate with L/S ratio of 4:1 mL/g and stirring for 1 h at room temperature, the leaching efficiency of V is 99.27% and the pH is 9.65. This indicates that the mechanical activation by ball milling significantly increases the specific surface area, lattice distortion, and microscopic stress of the precipitate [21], and reduces unreacted Ca(OH)2 encapsulated therein, thus improving the kinetic conditions of the precipitate leaching. In addition, there is no V wrapped by calcium carbonate in the residue obtained by using NaHCO3 solution to leach the precipitate formed by ball milling. The leaching of V from the precipitate can be expressed by the following equations:
Ca2V2O7+2NaHCO3=2NaVO3+2CaCO3↓+H2O (6)
Ca3(VO4)2+3NaHCO3=2NaVO3+3CaCO3↓+H2O+NaOH (7)
NaOH+NaHCO3=Na2CO3+H2O (8)
Ca(OH)2+2NaHCO3=CaCO3↓+Na2CO3+2H2O (9)
Ca(OH)2+Na2CO3=CaCO3↓+2NaOH (10)
It can be seen that using NaHCO3 solution to leach the precipitate formed by ball milling, the main reactions are Eqs. (6), (7) and (8), and the main components of the leaching solution are NaVO3 and NaHCO3 because the pH of the leaching solution is 9.65. However, when the precipitate obtained by stirring is leached with NaHCO3 solution, in addition to the Reactions (6), (7) and (8), Reactions (9) and (10) also occur during the leaching, because the precipitate contains a large amount of unreacted Ca(OH)2, which causes the main components of the leaching solution to became NaVO3 and Na2CO3, and the pH of the leaching solution increases to 11.13. Because the precipitated cake is mechanically activated, it has good leaching performance. For easy operation, the stirring leaching is used in the subsequent experiments.
3.2.2 Effect of NaHCO3 addition
Figure 7 shows the experimental results obtained by adding NaHCO3 into water according to different NaHCO3/V molar ratio to leach V from the precipitate with L/S ratio of 4:1 mL/g and stirring for 60 min at 30 °C. As can be seen, the leaching efficiency of V increases from 84.13% to 99.38% with the increase in NaHCO3/V molar ratio from 1.96:1 to 2.74:1. Then, it does not change with the further increase in NaHCO3/V molar ratio. This indicates that the V in the precipitate can be completely leached as the addition of NaHCO3 is close to twice the theoretical amount because the V in the cake mainly exists in the form of Ca3(VO4)2. Therefore, the NaHCO3/V molar ratio is maintained at 2.74:1 in the subsequent experiments.
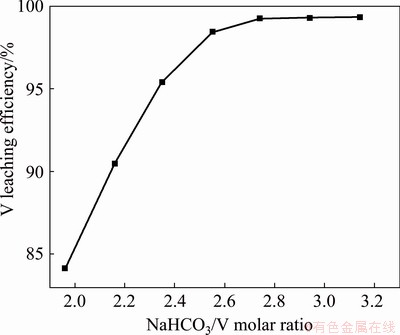
Fig. 7 Effect of NaHCO3 addition on V leaching efficiency (Stirring for 60 min at 30 °C)
The composition of the leaching solution is listed in Table 1 as well. It can be seen that V can be completely leached with NaHCO3 from the precipitate, while impurities Si and P are all remained in the leached residue. The pH of the solution is 9.67, which indicates that the main components of the leaching solution are NaVO3 and NaHCO3.
3.2.3 Effect of stirring time
Figure 8 shows the experimental results obtained by adding NaHCO3 into water according to NaHCO3/V molar ratio of 2.74:1 to leach V from the precipitate with L/S ratio of 4:1 mL/g and stirring for different time at 30 °C. Figure 8 shows that the V in the precipitate can be easily leached. The leaching efficiency of V increases rapidly from 88.35% to 99.28% with the increase in stirring time from 10 to 60 min, and then it maintains almost the same with the further increase in stirring time. Therefore, the stirring time should be maintained for ≥60 min.
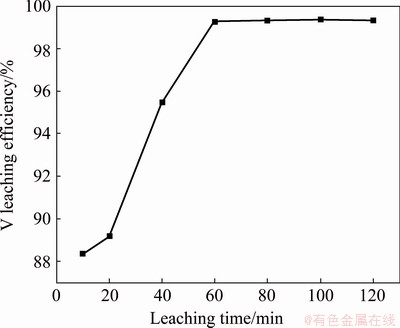
Fig. 8 Effect of stirring time on V leaching efficiency (NaHCO3/V molar ratio of 2.74:1)
3.2.4 Effect of leaching temperature
Figure 9 shows the experimental results obtained by adding NaHCO3 into water according to NaHCO3/V molar ratio of 2.74:1 to leach V from the precipitate with L/S ratio of 4:1 mL/g and stirring for 60 min at different temperatures. It can be seen that after mechanical activation, the precipitate has good leaching performance. Even if after stirring for 1 h at room temperature, the leaching efficiency of V can reach >99.2%, and the effect of leaching temperature on leaching efficiency of V is insignificant. Therefore, the leaching of V should be carried out at room temperature.
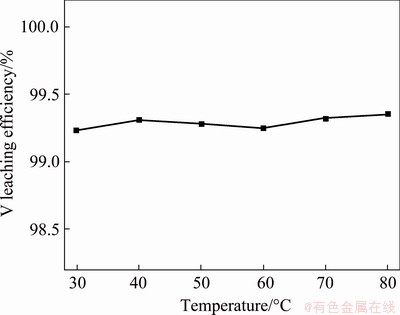
Fig. 9 Effect of temperature on V leaching efficiency (Stirring for 60 min)
3.3 Preparation of vanadium pentoxide
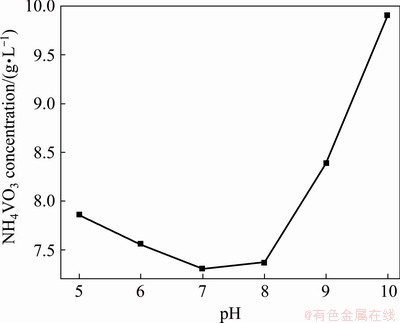
Fig. 10 Solubility of NH4VO3 in water under different pH values at 25 °C
The solubility of NH4VO3 in water under different pH values is shown in Fig. 10, which is obtained by adding excess NH4VO3 into water under different pH values, stirring at 50 °C and keeping for 24 h at 25 °C. The concentration of V in the solution is determined after filtration. Figure 10 indicates that the crystallization of NH4VO3 in the leaching solution should be carried out under pH 7-8, and the leaching of V from the precipitate should be performed under pH>9 with the NaHCO3 solution containing ammonia. In order to avoid the escape of ammonia, the pH of leaching solution is adjusted to be 8.0 with CO2, and NH4VO3 is crystallized by adding NH4HCO3 into leaching solution according to NH4HCO3/NaVO3 molar ratio of 1:1 and stirring for 8 h at room temperature. After NH4VO3 is crystallized, the solution is turned into the NaHCO3 solution containing ammonia. The crystallized solution can be returned to the leaching of the precipitate, and then the crystal of NH4VO3 can be obtained once again by adding an appropriate amount of NH4HCO3 into the leaching solution. V2O5 with purity of 99.6% is produced by calcining NH4VO3 crystals at 560 °C for 2 h. The production of V2O5 can be expressed by the following equations:
NaVO3+NH4HCO3=NaHCO3+NH4VO3↓ (11)
2NH4VO3=V2O5+2NH3↑+H2O↑ (12)
3.4 Cyclic leaching experiment
In order to verify that the crystallized solution can be reused in the leaching of the precipitated, the cyclic leaching experiment was performed. The experimental results are given in Table 2. As can be seen, the leaching efficiency of V is ≥99.17% under the conditions: L/S ratio of 4:1 mL/g, pH 9.6-9.7 and stirring for 1 h at room temperature. This indicates that when the molar ratio of NH4HCO3/NaVO3 is 1:1, the residual ammonium in the crystallized solution has no effect on the leaching of V from the precipitate, which is consistent with the experimental results shown in Fig. 10. However, it is found that when using the crystallized solution to leach the precipitate obtained by adding Ca(OH)2 into the feed liquid and stirring, the leaching effect of vanadium is not ideal: by stirring for 1 h at room temperature, the leaching efficiency of V is 76.3%; by stirring for 1 h at 80 °C, the leaching efficiency of V is 94.8%, which is consistent with the results obtained by other authors [17,18]. When the leaching temperature rises to close to 80 °C, the escape of ammonia is already very serious. This fully reflects the advantages of using ball milling to precipitate calcium vanadate.
It is found that by ball milling, can not only the unreacted Ca(OH)2 be significantly reduced in the precipitate, but also the particle size of the precipitate become finer and there is no calcium silicate shell on the particle surface, and these are all beneficial to the leaching of V. This is precisely because the precipitated cake formed by ball milling has good leaching performance. When the crystallized solution of NH4VO3 is returned for the leaching of the precipitated cake, the high leaching efficiency of V can be obtained by stirring leaching at room temperature. At the same time, because there are few unreacted Ca(OH)2 in the precipitated cake, the pH of the leaching solution is always maintained below 10, and the lower pH at room temperature effectively suppresses the escape of ammonia.
Table 2 shows that the leached residue can be reused in the V precipitation after it is roasted at 900 °C. Although the content of the free CaO in the roasted product gradually decreases with the increase in the number of recycling, the effect on the activity of the V precipitation reaction is not obvious. Even if the SiO2 in the leached residue is enriched to 15%, as the precipitant is added according to the molar ratio of the free CaO in the calcined product to the V in the solution (Ca/V) ≥1.75, by ball milling, the precipitation efficiency of V can be maintained ≥99.15%.
After ball milling, calcium silicates are dispersed in the calcium vanadate precipitates. Figure 11 shows the XRD pattern of the calcined product. As can be seen, CaO and CaSiO3 exist as two separate phases in the calcined product, which is the key to lime recycling and its V precipitation activity does not decrease. Moreover, it is found that after the feed liquid is purified to remove Si, lime is added to precipitate V, and the content of Si in the resulting precipitate is very small, so the enrichment of Si can be avoided.
The above experimental results show that compared with the conventional process of V2O5 production with vanadium slag, although the procedures of using calcium to precipitate V and using NaHCO3 to leach V are used in the modified process (see Fig. 1), the purification of the leaching solution, the production of V-Cr-bearing reducing slag, and the treatment of the reduced and precipitation solution are reduced. In the modified process, by adding calcium to precipitate V, not only V and Cr can be completely separated, but also no V-Cr-bearing reducing slag is produced, and the crystallized solution of NH4VO3 is used to leach V from the precipitate without the discharging of ammonia nitrogen wastewater. Therefore, on the whole, the advantages of the modified process are very obvious.
Table 2 Experimental results of cyclic leaching of precipitated cake with NH4VO3 crystallized solution
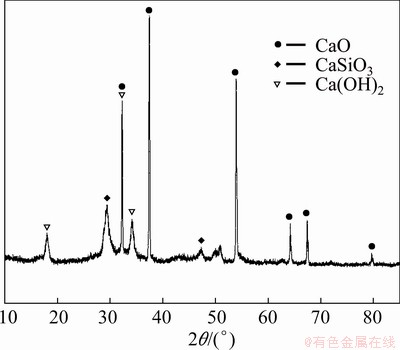
Fig. 11 XRD pattern of roasted product (Leached residue containing 12.75% SiO2)
4 Conclusions
(1) Vanadium can be effectively separated and recovered from chromium-containing vanadate solution by the cyclic metallurgical process including selective precipitation of vanadium, vanadium leaching and preparation of vanadium pentoxide.
(2) By adding Ca(OH)2 into the chromium- containing vanadate solution and ball milling, can not only V be selectively precipitated, but also the effective utilization of Ca(OH)2 be increased, and the leaching kinetics of the precipitate is significantly improved.
(3) Due to the precipitate of calcium vanadates formed by ball milling has good leaching performance, the V in it can be effectively leached with the crystallized solution of NH4VO3 at room temperature, so the escape of ammonia is greatly reduced. The water and NaHCO3 used in the process are both recycled. Therefore, there is no ammonia nitrogen wastewater discharged in V2O5 production.
Acknowledgments
The authors are grateful for the financial supports from the National Natural Science Foundation of China (51974369) and the Postgraduate Research Innovation Project of Central South University, China (2019zzts244).
References
[1] MASE A, SUGITA T, MORI M, IWAMOTO S, TOKUTOME T, KATAYAMA K, ITABASHI H. Study of vanadium-modified N/Si co-doped TiO2 in aqueous solution and its photocatalytic activity [J]. Chemical Engineering Journal, 2013, 225: 440-446.
[2] LIU Zuo-hua, LI Yan, CHEN Man-li, NUERAIHEMAITI A, DU Jun, FAN Xing, TAO Chang-yuan. Enhanced leaching of vanadium slag in acidic solution by electro-oxidation [J]. Hydrometallurgy, 2016, 159: 1-5.
[3] LI Hong-yi, LI Cui, ZHANG Meng, WANG Kang, XIE Bing. Removal of V(V) from aqueous Cr(VI)-bearing solution using anion exchange resin: Equilibrium and kinetics in batch studies [J]. Hydrometallurgy, 2016, 165: 381-389.
[4] QIU Hui-dong, ZHANG Hong, ZHAO Bo, ZHU Jian-fang, LIU De-rong. Dynamics study on vanadium extraction technology from chloride leaching steel slag [J]. Rare Metal Materials and Engineering, 2013, 42: 696-699.
[5] WANG Shuai, GUO Yu-feng, ZHENG Fu-qiang, CHEN Feng, YANG Ling-zhi, JIANG Tao, QIU Guan-zhou. Behavior of vanadium during reduction and smelting of vanadium titanomagnetite metallized pellets [J]. Transactions of Nonferrous Metals Society of China, 2020, 30: 1687-1696.
[6] CHEN Huo-sheng. Process study of V2O5 extraction from vanadium slag by lime roasting [J]. Steel Iron Vanadium Titanium, 1992, 13(6): 1-9. (in Chinese)
[7] WANG Ming-yu, CHEN Bian-fang, HUANG Sheng, WANG Xue-wen, LIU Biao, GE Qi. A novel technology for vanadium and chromium recovery from V-Cr-bearing reducing slag [J]. Hydrometallurgy, 2017, 171: 116-122.
[8] LIAO Shi-ming, BO Tan-lun. Foreign vanadium metallurgy [M]. Beijing: Metallurgical Industry Press, 1985. (in Chinese)
[9] WANG Xue-wen, GAO Da-xiong, CHEN Bian-fang, MENG Yu-qi, FU Zi-bi, WANG Ming-yu. A clean metallurgical process for separation and recovery of vanadium and chromium from V-Cr-bearing reducing slag [J]. Hydrometallurgy, 2018, 181: 1-6.
[10] TANDUKAR M, HUBER S J, ONODERA T, PAVLOSTATHIS S G. Biological chromium(VI) reduction in the cathode of a microbial fuel cell [J]. Environmental Science & Technology, 2009, 43: 8159-8165.
[11] NING Peng-ge, LIN Xiao, CAO Hong-bin, ZHANG Yi. Selective extraction and deep separation of V(V) and Cr(VI) in the leaching solution of chromium-bearing vanadium slag with primary amine LK-N21 [J]. Separation and Purification Technology, 2014, 137: 109-115.
[12] SUN Pan, HUANG Kun, WANG Xiao-qin, ZHENG Han, LIU Hui-zhou. Separation of V and Cr from alkaline aqueous solutions using acidified primary amine A-N1923-sodium tartrate system [J]. Separation and Purification Technology, 2017, 179: 504-512.
[13] FAN Ye-ye, WANG Xue-wen, WANG Ming-yu. Separation and recovery of chromium and vanadium from vanadium-containing chromate solution by ion exchange [J]. Hydrometallurgy, 2013, 13: 31-35.
[14] KACZALA F, MARQUES M, HOGLAND W. Lead and vanadium removal from a real industrial wastewater by gravitational settling/sedimentation and sorption onto Pinus sylvestris sawdust [J]. Bioresource Technology, 2009, 100: 235-243.
[15] WEN Shi-zu, YANG Ming-liang. Extraction of vanadium from reduced V-Cr slag [J]. Shanghai Metals (Nonferrous Fascicule), 1988, 9(2): 38-43. (in Chinese)
[16] WANG Xue-wen, WANG Hua-guang, GAO Da-xiong, CHEN Bian-fang, MENG Yu-qi, WANG Ming-yu. A clean technology to separate and recover vanadium and chromium from chromate solutions [J]. Hydrometallurgy, 2018, 177: 94-99.
[17] SHEN Xiao-qing, YANG Lin-sha, XU min, LI Zhong-jun. Study on the technology of recovering vanadium from vanadium-containing acid leaching solution [J]. Henan Chemical Industry, 1999(1): 16-18. (in Chinese)
[18] ZHUANG Li-jun, YANG Jin-ming. Study on the preparation of vanadium pentoxide from calcium vanadate residue [J]. Ferro-Alloys, 2017(10): 31-34. (in Chinese)
[19] WANG Xue-wen, WANG Ming-yu, YANG Ming-e, DU Yan-ping. Method for separating and recovering vanadium, chromium and sodium carbonate from the leaching solution of vanadium slag [P]. Chinese Patent 201711079106.5, 2017-11-09.
[20] DEAN J A. Langes’s handbook of chemistry [M]. 13th Ed. McGraw-Hill Inc, 1985.
[21] HUANG Qing-yun, XIANG Jun-yi, PEI Gui-shang, WANG Xin, Lü Xue-wei. Mechanical activation on extraction of vanadium from vanadium slag by calcification roasting-acid leaching process [J]. The Chinese Journal of Nonferrous Metals, 2020, 30(4): 858-865. (in Chinese).
王学文1,杨明鄂1,2,孟钰麒1,高大雄1,王明玉1,付自碧3
1. 中南大学 冶金与环境学院,长沙 410083;
2. 湖南汽车工程职业学院,株洲 412001;
3. 攀钢集团研究院有限公司 钒钛资源综合利用国家重点实验室,攀枝花 617000
摘 要:研究含铬钒酸盐溶液分离回收钒的循环冶金工艺,工艺过程包括溶液中钒的选择性沉淀、沉淀物中钒的浸出及五氧化二钒的制备。在含铬钒酸盐溶液中加入Ca(OH)2 进行球磨,不仅可以使其中的钒选择性沉淀析出, 而且沉淀物的浸出动力学性能得到显著改善。在含铬钒酸盐溶液中按 Ca/V摩尔比1.75:1加入Ca(OH)2,室温球磨60 min,钒的沉淀率达99.59%, 而铬的沉淀率只有0.04%。过滤得到的沉钒渣按液固比4:1 mL/g加水,并按NaHCO3/V摩尔比2.74:1加入NaHCO3,室温搅拌60 min,过滤得浸出液,钒的浸出率为99.35%。所得浸出液先通入CO2调pH至 8.0,再按NH4HCO3/NaVO3摩尔比1:1加入NH4HCO3,室温搅拌8 h结晶析出NH4VO3,过滤后,含氨的结晶液重新用于钒酸钙沉淀的浸出,温室搅拌1 h后钒的浸出率>99%,最后,将所得NH4VO3晶体在560 °C煅烧2 h得到纯度为99.6%的V2O5。
关键词:含铬钒酸盐溶液;钙盐沉钒;碳酸氢钠浸出;铵盐沉钒;循环冶金工艺
(Edited by Bing YANG)
Corresponding author: Ming-e YANG; Tel: +86-731-28431879; E-mail: yme8888@163.com
DOI: 10.1016/S1003-6326(21)65540-1
1003-6326/ 2021 The Nonferrous Metals Society of China. Published by Elsevier Ltd & Science Press
2021 The Nonferrous Metals Society of China. Published by Elsevier Ltd & Science Press

