J. Cent. South Univ. Technol. (2008) 15: 34-38
DOI: 10.1007/s11771-008-0008-9
Coating of LiNi1/3Mn1/3Co1/3O2 cathode materials with alumina by solid state reaction at room temperature
PENG Zhong-dong(彭忠东), DENG Xin-rong(邓新荣), DU Ke(杜 柯), HU Guo-rong(胡国荣),
GAO Xu-guang(高旭光), LIU Ye-xiang(刘业翔)
(School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China)
Abstract: Alumina coated LiNi1/3Mn1/3Co1/3O2 particles were obtained by a simple method of solid state reaction at room temperature. The reaction mechanism of solid state reaction at room temperature was investigated. The structure and morphology of the coating materials were investigated by XRD, SEM and TEM. The electrochemical performances of uncoated and Al2O3-coated LiNi1/3Co1/3Mn1/3O2 cathode materials were studied within a voltage window of 3.00-4.35 V at current density of 30 mA/g. SEM, TEM and EDS analytical results indicate that the surface of LiNi1/3Mn1/3Co1/3O2 particles is coated with very fine Al2O3 composite, which leads to the improved cycle ability though a slight decrease in the first discharge capacity is observed. It is proposed that surface treatment by solid state reaction at room temperature is a simple and effective method to improve the cycle performance of LiNi1/3Co1/3Mn1/3O2 particles.
Key words: solid state reaction; preparation; electrochemical property; LiNi1/3Mn1/3Co1/3O2; alumina; coating
1 Introduction
The synthesis of composite particles, consisting of core particle covered by a coating of different materials, has opened new promising directions for materials research. It can be expected that the coating materials may stabilize the core materials to prevent possible undesirable interactions with the environment or may improve electrochemical, optical, magnetic, conductive, adsorptive, and surface reactive properties of the dispersed matter to meet certain requirements[1-2].
The cathode material LiNi1/3Co1/3Mn1/3O2 is one of the most promising cathode for rechargeable lithium ion batteries owing to its high reversible capacity at high potential. In order to develop high cyclability of layered cathode material for an advanced lithium-ion batteries, one of the approaches is to coat it with a thin layer of electrochemically inactive metal oxide materials[3-5]. The improved cycling performance and capacity retention of the coated cathode materials is thought to be successful in minimizing the side reactions within the batteries by placing a protective barrier layer between cathode material and the liquid electrolyte during (de) intercalation process[6].
The coating can be obtained by a variety of methods including impregnation, chemical vapor deposition method[7], precipitation and sol-gel techniques[8-9]. These methods all require the core material to be suspended in solvent(e.g. water). Coating by solid state reaction at room temperature is a new method that is simple and environmentally friendly. The advantages such as high yield, strong selectivity, low cost, no need of solvent and eliminating reunite problem[10-13] make it easy to be industrialized, and it can also reduce by-products to the greastest extent. Solid state reaction at room temperature is an efficient method for synthesizing nano-particles[14], atom cluster compounds[15], coordination compounds[16]. However, no reports on coating lithium-ion battery cathode material by this method have been found until now.
In this work, the method of solid state reaction at room temperature was introduced to synthesize Al2O3-coated LiNi1/3Co1/3Mn1/3O2 lithium ion cathode materials. The possible reaction mechanism of preparing Al2O3-coated LiNi1/3Co1/3Mn1/3O2 lithium ion cathode materials through solid state reaction at room temperature was studied by thermodynamics and kinetics.
2 Experimental
LiNi1/3Co1/3Mn1/3O2 particles in micrometer were prepared in our laboratory[17]. Al2(SO4)3?18H2O and LiOH?H2O were reagent grade. LiNi1/3Co1/3Mn1/3O2 powder was mixed and ground softly with Al2(SO4)3?18H2O (total metal ion molar ratio, n(Ni+Mn+Co)/n(Al) =10?1). Stoichiometric ground LiOH?H2O was then added to the mixture and ground softly for 40 min. During the process of grinding, the reactants became wet. Then the mixture was aged for 2 h and washed with distilled water. At last, the product (Al(OH)3-coated LiNi1/3Co1/3Mn1/3O2) was dried at 60 ℃ and then heated at 700 ℃ for 30 min to obtain the Al2O3 coated LiNi1/3Co1/3Mn1/3O2 particles.
Composite electrodes were prepared by mixing 80% LiNi1/3Co1/3Mn1/3O2/Al2O3-coated LiNi1/3Co1/3Mn1/3O2 particles, 10% acetylene black(AB) and 10% polyvinylidene fluoride(PVDF). The mixture was spread on an aluminum foil and dried at 120 ℃ for 24 h. The charge-discharge cycling tests were performed using the CR2025 coin-type cell. The separator was a Celgard 2400 microporous polylene membrane. Lithium metal was used as anode in this study. The electrolyte was 1.0 mol/L LiPF6/EC+DEC (1?1, volume ratio). The cells were assembled in a glove box filled with ultra-pure argon gas. The charge-discharge capacity and cycling performance of the cells were galvanostatically performed at constant current density of 30 mA/g with a voltage window of 3.00-4.35 V(vs Li/Li+) using a LAND CT2001A computer-controlled battery testing system at room temperature. The crystal structure of cathode materials was identified from an X-ray diffraction (XRD) pattern obtained by diffractometry (Rigaku D/max 2550 VB+). TEM analysis was performed on Tecnai G220 operated at 200 kV. The morphology and EDS were observed by scanning electron microscopy (SEM, JEOL JSM-6360LV).
3 Results and discussion
3.1 Thermodynamic and kinetic analysis of alumina coating
The solid state reactions at room temperature are always ignored. Similar to the chemical reaction in solution, the chemical reaction in solid must satisfy the thermodynamics law that the change of the whole reaction Gibbs energy should be less than zero. When the reaction thermodynamics condition is satisfied, reactant structure will be the key condition that determines the solid state reactions at room temperature will happen or not. The effect of the solid structure on chemical reactions can also be known from the reaction temperature. If the solid state reactions happen, the reactant molecule must long-range move and collide each other. However, the force of the long-range motion and colliding each other can be known from the melting point of the solid reactant.
Usually, the temperature under which the solid state reaction can happen is decided by the lower TAMMANN temperature(Tm) of reactant[18]. Here, the TAMMANN temperature is the temperature when the solid interior diffusion becomes prominent. TAMMANN and MANSURI[18] pointed out that the temperature has something to do with solid melting point(tA) of the denotation of absolute temperature standard: Tm=k(tA+273), k was a coefficient, 0.3 to metal, 0.5 to inorganic compound and 0.9 to organic compound. Actually, in order to get a high reaction speed, usually a higher reaction temperature will be used. For example, to inorganic compound, it is 2/3, as shown in Fig.1. Therefore, the reaction of molecule solid state will happen at room temperature when the TAMMANN temperature is less than 300 K.
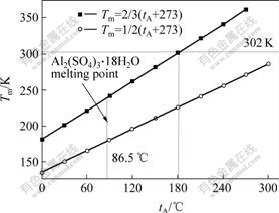
Fig.1 Correlation of melting point and TAMMANN temperature
To sum up, the thermodynamic and kinetic conditions that the solid state reaction can happen are as follows:
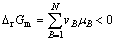 (1)
(1)
Tm≤300 K (2)
Eqn.(1) denotes the thermodynamic condition, and Eqn.(2) denotes the kinetic condition. Only meeting the two conditions together can the solid state reaction at room temperature happen.
Al2(SO4)3?18H2O reacts with LiOH?H2O at room temperature as follows:
Al2(SO4)3?18H2O+6LiOH?H2O→
2Al(OH)3+3Li2SO4+24H2O (3)
The ΔrGm of Eqn.(3) is less than zero and the melting point of Al2(SO4)3?18H2O is 86.5 ℃, as shown in Fig.1. Therefore, viewing from both the thermodynamic and kinetic analysis point, the reaction can happen. The above theoretical analysis will be proved in the following EDS and TEM experiment.
3.2 Analysis of experimental results
Fig.2 shows the X-ray diffraction pattern of the uncoated and coated LiNi1/3Co1/3Mn1/3O2 powders. The spectrum of uncoated LiNi1/3Co1/3Mn1/3O2 powders indicates that the material is similar to that of LiCoO2 (α-NaFeO2 type, space group R3m) and no impure peaks appear. It can be observed that the spectrum of the coated LiNi1/3Co1/3Mn1/3O2 powders is almost the same as that of the uncoated one. The absence of any other signals in the spectrum indicates the coated Al2O3 is probably not crystal, but amorphous in nature.
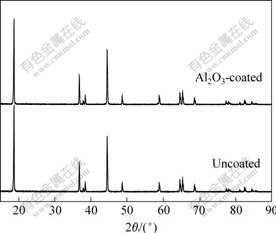
Fig.2 XRD patterns of uncoated and Al2O3-coated LiNi1/3Co1/3Mn1/3O2 powders
Fig.3 shows the surface morphologies of the uncoated and Al2O3-coated LiNi1/3Co1/3Mn1/3O2 composites. The surface morphologies of the uncoated LiNi1/3Co1/3Mn1/3O2 composites are very clean and smooth. By comparison, the surface morphology of Al2O3-coated LiNi1/3Co1/3Mn1/3O2 materials is blurry.
Fig.4 shows the TEM image of Al2O3-coated LiNi1/3Co1/3Mn1/3O2 materials. The crystalline grains are obviously coated with small particles and the thickness of the kernel is around 20 nm. The above layer in morphology should be the result of presence of the Al2O3 coating.
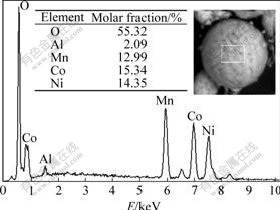
The surface composition of the coated LiNi1/3Co1/3Mn1/3O2 powders was analyzed by scanning electronic microscope energy spectrum. The relative surface compositions of the coated particles expressed quantitatively as molar fraction, derived from the EDS intensities, are shown in Fig.5. Al and O components appear on the surface of coated particles. It can be concluded that alumina is coated on the surface of the LiNi1/3Co1/3Mn1/3O2 particles. It can also prove that the reaction of Al2(SO4)3?18H2O with LiOH?H2O in solid state reaction route at room temperature is feasible.

Fig.3 SEM images of uncoated(a, b) and Al2O3-coated(c) LiNi1/3Co1/3Mn1/3O2 composite

Fig.4 TEM image of Al2O3-coated LiNi1/3Co1/3Mn1/3O2 particles
The electrochemical performances of uncoated and Al2O3-coated LiNi1/3Co1/3Mn1/3O2 materials were tested by Li half-cell. The typical discharge curves of different samples are presented in Fig.6. The initial discharge specific capacity of the Al2O3-coated LiNi1/3Co1/3Mn1/3O2 cathode material(147.6 mA?h/g) is lower than that of the
uncoated LiNi1/3Co1/3Mn1/3O2 cathode material(156.5 mA?h/g). However, the capacity retention of the LiNi1/3Mn1/3Co1/3O2 cathode material is significantly improved by coating a small amount of Al2O3. As shown in Figs.6 and 7, the uncoated LiNi1/3Co1/3Mn1/3O2 material loses about 10.29% of the initial discharge specific capacity (140.4 mA?h/g) after 20 cycles, while the Al2O3-coated LiNi1/3Co1/3Mn1/3O2 material behaves much better, losing about 2.44 % of its initial discharge specific capacity (144.0 mA?h/g) during the same number of cycles.
![文本框: keV]()
Fig.5 Scanning electronic microscope energy spectrum analysis of coated LiNi1/3Co1/3Mn1/3O2 particles
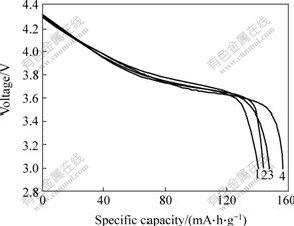
Fig.6 Discharge curves of uncoated and Al2O3-coated LiNi1/3Co1/3Mn1/3O2 electrode: 1―The 1st, uncoated LiNi1/3- Mn1/3Co1/3O2; 2―The 20th, uncoated LiNi1/3Mn1/3Co1/3O2; 3―The 1st, coated LiNi1/3Mn1/3Co1/3O2; 4―The 20th, coated LiNi1/3Mn1/3Co1/3O2
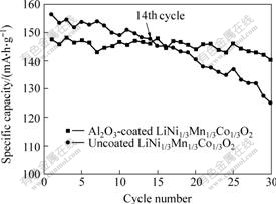
Fig.7 Cycling stability of uncoated and Al2O3-coated LiNi1/3Co1/3Mn1/3O2 electrode
The 3rd cyclic voltammetry records for the uncoated as well as the Al2O3-coated LiNi1/3Co1/3Mn1/3O2 electrodes are shown in Fig.8. Fig.8 shows that the similar specific cyclic voltammograms from the uncoated and Al2O3-coated LiNi1/3Co1/3Mn1/3O2 electrodes indicate that the improvement of capacity retention is not due to suppression of the phase transition. Another coating effect, which was reported in Ref.[19], is that the prevention of electrode reactions with electrolyte since the oxide-coated layer isolates them. Generally speaking, the reaction at the interface induces impedance increase and causes the capacity fading of the cathode[20].
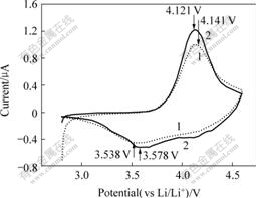
Fig.8 The 3rd cyclic voltammetry curve of uncoated and Al2O3-coated LiNi1/3Co1/3Mn1/3O2 electrodes at scan rate of 0.1 mV/s: 1―Uncoated LiNi1/3Mn1/3Co1/3O2; 2―Al2O3-coated LiNi1/3Mn1/3Co1/3O2
Fig.9 shows the comparison of the electrochemical impedance speceroscopy(EIS) profiles of uncoated and Al2O3-coated LiNi1/3Co1/3Mn1/3O2 samples at a charge potential of 4.35 V, respectively, as a function of cycle number. A high-frequency semicircle represents the impedance due to a solid-state interface layer formed on the surface of the electrodes, and a low-frequency semicircle is related to a slow charge transfer process at the interface and its relative double-layer capacitance at the film bulk oxide. The cell impedance is mainly determined by the cathode-side impedance[21], especially by the charge-transfer impedance. Thus, we focused on comparison of the a low-frequency semicircle that represents the charge-transfer impedance. The cell impedance of the uncoated cathode drastically increases during cycling, whereas the cell impedance of the coated cathode does not increase much. From these results, the smaller capacity loss of the coated LiNi1/3Co1/3Mn1/3O2 cathode to the uncoated LiNi1/3Co1/3Mn1/3O2 cathode appears due to the inactive Al2O3 coating layer on the cathode surface, which considerably decreases the impedance growth.
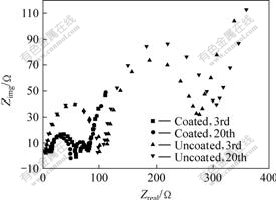
Fig.9 Nyquist plots of uncoated and Al2O3-coated LiNi1/3- Mn1/3Co1/3O2 samples
4 Conclusions
1) Alumina coating on LiNi1/3Mn1/3Co1/3O2 particles is obtained by solid state reaction at room temperature. The possibility of preparing Al2O3-coated LiNi1/3Co1/3Mn1/3O2 lithium ion battery cathode materials through solid state reaction at room temperature is studied by thermodynamics and kinetics.
2) The TEM, SEM, XRD and EDS experimental results show that Al2O3 is coated on the surface of LiNi1/3Mn1/3Co1/3O2 particles. The surface of LiNi1/3Mn1/3Co1/3O2 particles is coated with fine Al2O3 particles as a coating material for improving cyclic property at room temperature, though its first discharge specific capacity is slightly lower than that of the uncoated one. The uncoated LiNi1/3Mn1/3Co1/3O2 particles lose about 10.29% of its initial discharge capacity after 20 cycles, whereas the Al2O3-coated LiNi1/3Mn1/3Co1/3O2 behaves much better, only losing about 2.44 % of its initial discharge capacity during the same cycles.
3) The surface treatment of the solid state reaction at room temperature is a simple and effective way to improve the electrochemical performance of LiNi1/3Mn1/3Co1/3O2 material for lithium ion batteries.
References
[1] LIU Gui-xia, HONG Guang-yan. Synthesis of SiO2/Y2O3: Eu core-shell materials and hollow spheres[J]. Journal of Solid State Chemistry, 2005, 178(5): 1647-1651.
[2] GUO Hua-jun, LI Xin-hai, ZHANG Xin-ming, ZENG Su-ming, WANG Zhi-xing. Characteristics of LiCoO2, LiMn2O4 and LiNi0.45Co0.1Mn0.45O2 as cathodes of lithium ion batteries[J]. Journal of Central South University of Technology, 2005, 12(1): 44-49.
[3] KIM Y, KIM H S, MARTIN S W. Synthesis and electrochemical characteristics of Al2O3-coated LiNi1/3Co1/3Mn1/3O2 cathode materials for lithium ion batteries[J]. Electrochimica Acta, 2006, 52(3): 1316-1322.
[4] TONG Dong-ge, LAI Qiong-yu, WEI Ni-ni, TANG Ai-dong, TANG Lian-xing, HUANG Ke-long, JI Xiao-yang. Synthesis of LiCo1/3Ni1/3Mn1/3O2 as a cathode material for lithium ion battery by water-in-oil emulsion method[J]. Materials Chemistry and Physics, 2005, 94(2/3): 423-428.
[5] PENG Zhong-dong, HU Guo-rong, LIU Ye-xiang. Influence on performance and structure of spinel LiMn2O4 for lithium-ion batteries by doping rare-earch Sm[J]. Journal of Central South University of Technology, 2005, 12(1): 28-32.
[6] LI C, ZHANG H P, FU L J, LIU H, WU Y P, RAHM E, HOLZE R, WU H Q. Cathode materials modified by surface coating for lithium ion batteries[J]. Electrochimica Acta, 2006, 51(19): 3872-3883.
[7] ELKASABI Y, CHEN H Y, LAHANN J. Multipotent polymer coatings based on chemical vapor deposition copolymerization[J]. Advanced Materials, 2006, 18(12): 1521-1526.
[8] WANG F H, GUO R S, WEI Q T, ZHOU Y, LI H L, LI S L. Preparation and properties of Ni/YSZ anode by coating precipitation method[J]. Materials Letters, 2004, 58(24): 3079-3083.
[9] PHANI A R, GAMMEL F J, HACK T. Structural, mechanical and corrosion resistance properties of Al2O3-CeO2 nanocomposites in silica matrix on Mg alloys by a sol-gel dip coating technique[J]. Surface and Coatings Technology, 2006, 201(6): 3299-3306.
[10] ZHOU Tao-yu, YUAN Xin, HONG Jian-ming, XIN Xin-quan. Room-temperature solid-state reaction to nanowires of zinc sulfide[J]. Materials Letters, 2006, 60(2): 168-172.
[11] LI Fa-shen, WANG Hai-bo, WANG Li, WANG Jian-bo. Magnetic properties of ZnFe2O4 nanoparticles produced by a low-temperature solid-state reaction method[J]. Journal of Magnetism and Magnetic Materials, 2007, 309(2): 295-299.
[12] LIU Gui-xia, HONG Guang-yan, SUN Duo-xian. Coating Gd2O3: Eu phosphors with silica by solid-state reaction at room temperature[J]. Powder Technology, 2004, 145(2): 149-153.
[13] CUI Hong-tao, HONG Guang-yan, WU Xue-yan, HONG Yuan-jia. Silicon dioxide coating of CeO2 nanoparticles by solid state reaction at room temperature[J]. Materials Research Bulletin, 2002, 37(13): 2155-2163.
[14] CUI Hong-tao, HONG Guang-yan, YOU Hong-peng, WU Xue-yan. Coating of Y2O3: Eu3+ particles with alumina by a humid solid state reaction at room temperature[J]. Journal of Colloid and Interface Science, 2002, 252(1):184-187.
[15] SHAH J G, PATKI V A, SHIVAKAMY K, WANI B N, PATWE S J, RAO U R. On the material transport during solid state reactions at room temperature[J]. Applied Physics Communications, 1993, 12(1): 141-152.
[16] YUAN A Q, LIAO S, TONG Zh F,WU J, HUANG Z Y. Synthesis of nanoparticle zinc phosphate dihydrate by solid state reaction at room temperature and its thermochemical study[J]. Materials Letters, 2006, 60(17/18): 2110-2114.
[17] YU Xiao-yuan. Study on layered LiNi1/3Co1/3Mn1/3O2 and modified spinel LiMn2O4[D]. Changsha: Central South University, 2006. (in Chinese)
[18] TAMMANN G, MANSURI Q A. Hardness of amalgams Ag-Sn[J]. Anorg Chem, 1923, 132: 66-67. (in German)
[19] ZHANG Z R, LIU H S, GONG, Z L YANG Y. Electrochemical performance and spectroscopic characterization of TiO2-coated LiNi0.8Co0.2O2 cathode materials[J]. Journal of Power Sources, 2004, 129(1): 101-106
[20] CHEN Z H, DAHN J R. Methods to obtain excellent capacity retention in LiCoO2 cycled to 4.5 V[J]. Electrochimica Acta, 2004, 49(7): 1079-1090.
[21] CHEN C H, LIU J, AMINE K. Symmetric cell approach and impedance spectroscopy of high power lithium-ion batteries[J]. Journal of Power Sources, 2001, 96(2): 321-328.
(Edited by CHEN Wei-ping)
Foundation item: Project(50604018) supported by the National Natural Science Foundation of China
Received date: 2007-07-23; Accepted date: 2007-09-08
Corresponding author: DENG Xin-rong, Doctoral candidate; Tel: +86-731-8830474; E-mail: csudxr@mail.csu.edu.cn