Key words: surfactant; single bubble; shear thinning fluids; velocity; drag coefficient
Cite this article as: LI Shao-bai, FAN Jun-geng, LI Run-dong, WANG Lei, LUAN Jing-de. Effect of the surfactants on hydrodynamics characteristics of bubble in shear thinning fluids at low Reynolds number [J]. Journal of Central South University, 2018, 25(4): 805�C811. DOI: https://doi.org/10.1007/s11771-018-3785-9.
1 Introduction
The motion of bubbles in a gas�Cliquid two-phase system is frequently encountered in several industrial processes, such as in gas strippers, bubble columns, and biochemical reactors, as well as distillation [1]. In such equipment, the hydrodynamic characteristics of bubbles are closely related to the residence time and gas�Cliquid mass transfer; hence, for design and optimization of those processes, it is crucial to research the motion behavior of bubbles. Even for practical applications regarding bubble swarms motion, the hydrodynamic characteristic of single bubbles can provide better knowledge of the overall behavior. As a result, over the years, considerable research efforts have been focused on the hydrodynamic characteristic of single bubbles in clean gas�Cliquid two-phase systems [2�C6]. And others efforts can be achieved from the review of KULKARNI et al [7]. However, it is crucial to state that in practical industrial processes, it is difficult to maintain a pure liquid phase, and the presence of impurities is inevitable in the majority of the aforementioned processes. Hence, the motion behavior of bubbles is easily affected by those impurities in three respects: reduction of terminal velocity [8], prevention of coalescence [9], and hindrance of mass transfer [10]. The reduction of the terminal velocity of the bubble can be attributed to the bubble surface mobility, which is determined by the area covered with the surfactant molecules. As the liquid phase is contaminated, the surfactant molecules will accumulate on the bubble surface, and shear stress generates due to the bubble relative motion with the surrounding liquid phase. This results in the decreasing of the bubble terminal velocity [11]. If the bubble surface is sufficiently covered by surfactant molecules under the appropriate conditions, the motion behavior of the bubble is the same as that of solid spheres [12]. This is the famous stagnant cap hypothesis [13], which has been successfully confirmed by several experimental studies [14�C18] and numerical simulation [19].
Aforementioned studies mainly focus on Newtonian fluids. However, several fluids in practical processing industries exhibit shear thinning behavior. Compared to Newtonian fluids, much less is known about the effect of surfactant on bubble motion behavior in shear thinning fluids due to its complicated rheological characteristic. Despite several difficulties, a few efforts have still been devoted to the study of the motion behavior of the bubble rising in shear thinning fluids. RODRIGUE et al [20, 21] have experimentally investigated the effect of surfactants on the terminal velocity of a single bubble in shear thinning fluids and employed thermodynamics and physical approximation for theoretically examining the motion behavior of the single bubble at low Reynolds number. TZOUNAKOS et al [22] studied the influence of surfactants on the terminal velocity, shape, and drag coefficient of freely rising bubbles in shear thinning fluids. Recently, NALAJALA et al [23] have numerically simulated the flow and drag phenomena of contaminated bubbles in shear thinning liquids using ANSYS Fluent. All those papers have concluded that the presence of surfactant decreases the terminal velocity of the bubble, albeit under the primary condition of a large Reynolds number, on the other hand, few studies have been reported the effects of surfactants on a bubble at low Reynolds number in shear thinning fluids, which is becoming more important for food as well as daily chemical and anaerobic fermentation industries.
The present research work is to study the hydrodynamic characteristics, particularly, the terminal velocity and drag coefficient of single bubble in shear thinning fluids at low Reynolds number. The effect of surfactant concentration and liquid-phase rheological properties on the terminal velocity and drag coefficient of the single bubble was also experimentally investigated.
2 Experimental
2.1 Experimental apparatus
Figure 1 shows the experimental set up. The principal part of the bubble formation system comprised a rectangular bubble column with dimensions of 0.15 m��0.15 m��1.5 m. Nitrogen was injected via an orifice at the bottom of the bubble column from a gas cylinder using a valve and rotameter (within +0.01 cm3/s). The inner diameter of orifices used in the experiments was 1.0 mm. Bubble motion behavior was determined using a high-speed camera (Motion Pro Y5, REDLAKE Global USA) with a lens (Nikon, 24�C85mm/f2.8-4). In this study, the process of bubble motion was captured at a rate of 100 frames/s with a resolution of 500��1728 pixels. For ensuring that the bubble motion reached the steady state, an observing field with dimensions of 140 mm��140 mm was located at above 700 mm from the nozzle. The selected sequence of frames was analyzed using Matlab 10.0 with a self-written code, and the bubble shape and terminal velocity were obtained. And all the studied bubble shape is basically spherical at low Reynolds number (Re<50). All experiments were conducted at room temperature and pressure.
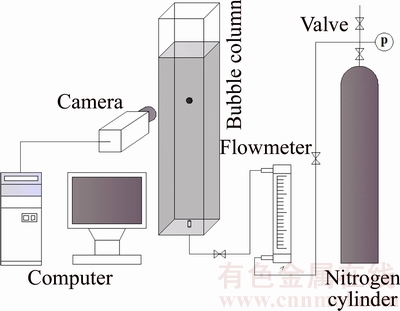
Figure 1 Schematic representation of experimental apparatus and process
2.2 Materials
In this study, different concentrations(0.25wt% and 0.50wt%) of aqueous CMC (guaranteed reagent, Sigma) solutions were used as experimental liquids, double-distilled water was used, and high purity nitrogen with 99.999% purity was used as the gas phase. For every test fluid, three concentrations (0 mg/L, 5 mg/L, and 10 mg/L) of sodium dodecyl sulfate (SDS) were added to observe the effect of surfactant concentration on the bubble hydrodynamic characteristics.
The surface tension of the liquid was measured using a tensiometer (DCAT21, data physics, Austria) with an accuracy of ��0.1 kg/m3. The rheological property was determined using a programmatic rheometer (Brookfield, DV-III, USA) with a shear rate ranging from 0.1 to 100 1/s. As can be observed in Figure 2, CMC solutions exhibit shear thinning behavior. The variation of the apparent viscosity with the shear rate can be described by the power-law model:
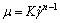 (1)
(1)
where K is the consistency index, and n is the flow index.
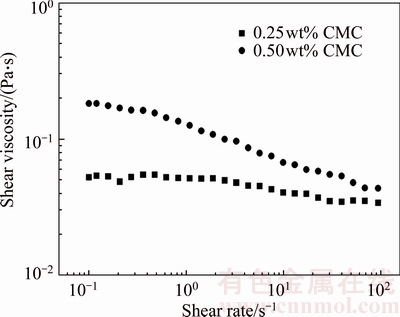
Figure 2 Rheological characteristics of test liquids
Table 1 summarizes the physical properties of experimental fluids. As can be observed in Table 1, SDS concentration does not exhibit any effect on the rheological property of CMC solutions.
3 Mathematical description
When bubbles freely rise in stagnant CMC aqueous solutions, the predominant forces acting on the bubble are buoyancy and drag force. When the bubble motion attains the steady state, buoyancy force and drag force balance each other, based on Newton��s second law:
Table 1 Rheological and physical properties of experimental fluids
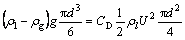 (2)
(2)
where ��l and ��g are the densities of liquid and gas, respectively; g is the gravitational acceleration; U is the terminal velocity of the bubble; CD is the drag coefficient; and d is the diameter of the bubble.
By neglecting the density of gas as compared with that of the liquid, CD can be can be expressed as follows:
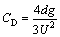 (3)
(3)
The drag coefficient of the bubble can be calculated using Eq. (3) when the bubble attains steady motion.
From Figure 2, the viscosity of experimental liquids varies with shear rate; thus, it is helpful to define the Reynolds number (Re). Here, the viscosity of experimental liquids follows the power- law model,  , and
, and  is calculated by
is calculated by 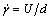 . Re is defined as follows:
. Re is defined as follows:
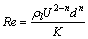 (4)
(4)
LI et al [24] has experimentally investigated the motion of a single bubble rising in a CMC solution and proposed a new empirical correlation of the bubble drag coefficient by introducing the effect of the bubble shape and rheological property of the liquid phase. However, for a spherical bubble, the drag coefficient can be expressed as follows:
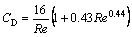 (5)
(5)
For Re approach to zero, Eq.(5) could regress to CD=16/Re, namely, the H�CR model.
DEWSBURY et al [25] have discussed the difference of light solid sphere and bubble rising in fluids. They found that the drag coefficient of a light solid sphere is greater than that of the bubble at the same Re, with a scaling factor of 3/2. The drag coefficient for a light solid sphere can be calculated as follows:
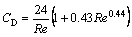 (6)
(6)
It converged with the Stokes equation CD=24/Re at low Reynolds numbers.
4 Results and discussion
Figures 3 and 4 plot the terminal velocity of the bubble at different CMC concentrations as a function of the bubble volume and SDS concentration. Figures 3 and 4 show the variation of the bubble velocity in the 0.25wt% and 0.50wt% CMC solutions, respectively. The bubble velocity increases with increasing bubble volume at any CMC solution concentration, and the effect of the surfactant results in the decrease of the terminal velocity of the bubbles for this range of volumes. However, no significant difference is observed on the effect of two surfactant concentrations (5 mg/L and 10 mg/L) on bubble velocity as the bubble volume is less than the critical volume (0.25wt% CMC: 100 mm3; 0.50wt% CMC: 110 mm3). Beyond the critical volume, the effect of surfactant concentration starts to emerge, and the bubble velocity decreases with increasing surfactant concentration. SHU et al [26] have found that surfactant molecules accumulate on the bubble surface and retard the internal circulation of the bubble surface. Thus, the drag curve for gas bubbles is similar to that of the rising solid particles. At a low bubble volume, the surfactant molecules cover the entire interface of the bubble at all experimental SDS concentrations; hence, the effect of SDS concentration on bubble velocity is insignificant. With increasing bubble volume, the bubble velocity increases, and the SDS molecules cannot completely cover the bubble surface; hence, SDS concentration exerts different effects on bubble velocity. Meanwhile, as can be observed in Figures 3 and 4, with further increase in the bubble volume, bubble velocity is not affected by the presence of contaminants. Besides, by the comparison of Figures 3 and 4, the bubble terminal velocity in 0.25wt% CMC solutions is greater than that in 0.50wt% CMC solutions, attributed to the apparent viscosity of CMC solutions, which could decrease the bubble terminal velocity. As shown in Figure 2, the shear viscosity of 0.25wt% CMC solution is less than that of 0.50wt% CMC solution at shear rates ranging from 0.1 to 1000 s�C1.
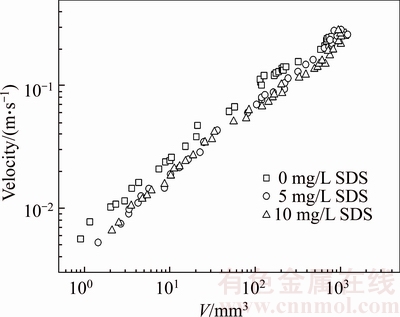
Figure 3 Plot of bubble velocity as function of bubble volume and SDS concentration in 0.25wt% CMC solutions
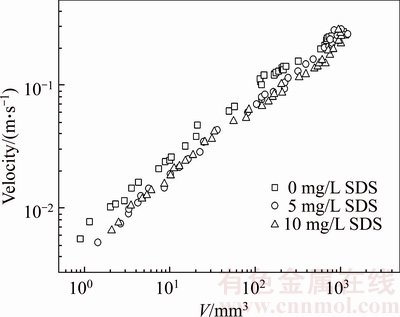
Figure 4 Plot of bubble velocity as function of bubble volume and SDS concentration in 0.50wt% CMC solutions
Figure 5 shows the plots of the drag coefficient of bubble rising in 0.25wt% CMC solutions as a function of SDS concentration. Drag coefficients were calculated using Eq. (3), while Reynolds numbers were calculated using Eq.(4). The surfactant significantly affects drag coefficients for a bubble with Re<10. For clean solutions, the drag coefficients exhibit good agreement with the data calculated by Eq.(5) and converge to the H�CR model, CD=16/Re, at low Reynolds numbers. For contaminated solutions, the drag coefficients exhibit good agreement with the data calculated by Eq.(6) and converge to the Stokes equation, CD=24/Re, at low Reynolds numbers.This confirms the previously reported results [27]. This was attributed to the effect of interface motion on bubble rise. If a bubble rises in liquid phase containing surfactant, the surfactant molecules will accumulate on the gas/liquid interface created by the bubble and liquid phase and decrease bubble surface mobility. Therefore, the bubble will has the drag coefficient as freely rising light solid sphere with similar size, namely, CD=24/Re. However, if a bubble rises in liquid phase without SDS, it have smaller drag coefficients than bubbles (and rigid particles) rising in liquid phase with SDS, namely, CD=16/Re.
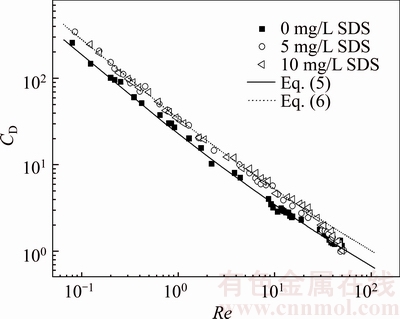
Figure 5 Drag coefficients of bubbles versus Reynolds number in 0.25wt% CMC solution as a function of SDS concentration
The difference in the drag coefficient of spherical bubbles with the Reynolds number between clean and contaminated liquids can be explained by the effect of internal bubble circulation. The internal circulation of a bubble appeared to decrease the friction at the gas/liquid interface, resulting in the decrease in the drag acting on the bubble. As a bubble rises in a contaminated solution, surfactant molecules cover the bubble surface and suppress the circulation of internal bubbles; hence, the bubble surface is similar to that of a light solid sphere with a quasi-fixed surface. This in turn results in the increase of drag, resulting in the reduction of increasing velocity [28]. Thus, for Re<10, the drag coefficient of bubbles in contaminated CMC solutions typically follows the Stokes model; hence, the effect of bubble internal circulation is insignificant. For 10 Figure 6 shows the effect of surfactant concentration on the relationship between the drag coefficients and Re of bubbles rising in the 0.50wt% CMC solution, which is qualitatively similar to effect in the 0.25wt% CMC solution shown in Figure 5. For Re < 10, the curve of the drag coefficient of the bubble in contaminated CMC solutions typically follows the Stokes model, while that in clean CMC solutions follows the H�CR model. For 10 more SDS molecules accumulate on the bubble surface, and the bubble surface mobility becomes worse. From Figure 7, high CMC concentration is conducive to the transition, which can be explained as follows: 1) CMC molecules prevent the surfactant molecules from reaching the bubble surface; 2) high CMC concentration indicates high viscosity, which leads to high bubble velocity at the same Re and promotes the internal bubble circulation [29]. 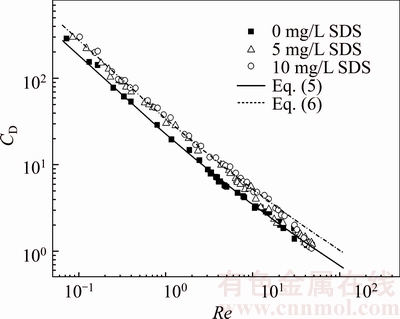
Figure 6 Drag coefficients of bubbles versus Reynolds number in 0.50wt% CMC solution as function of SDS concentration
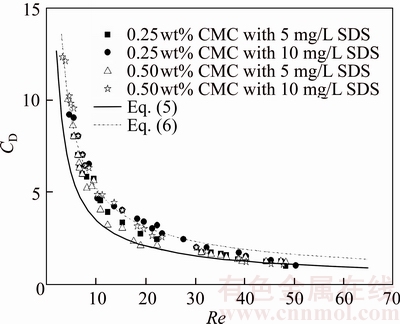
Figure 7 Effects of CMC and SDS concentrations on transition Reynolds numbers
5 Conclusions
In this study, the effects of surfactants on the hydrodynamic characteristics of a spherical bubble in non-Newtonian fluids at low Reynolds number (Re<50) were experimentally investigated. The terminal velocity of the bubble in clean and contaminated carboxymethylcellulose (CMC) solutions was obtained using a high-speed camera, and the drag coefficient was evaluated by the rheological model parameters, physical properties of the fluids, bubble velocity, and diameter. As a result, the following conclusions were obtained:
1) The presence of surfactant could decrease the spherical bubble terminal velocity at small volume (0.25wt% CMC: <100 mm3; 0.50wt% CMC: <110 mm3) due to the internal circulation flow of bubble interface, but this negative effect will recede and disappear with the increasing of bubble volume.
2) The curves of the bubble drag coefficient in contaminated CMC solutions exhibit behavior similar to that of solid spheres for Re<10, indicating no internal circulation flow at the bubble interface as compared to that in clean CMC solutions. However, for Re>50, the hydrodynamic characteristics of spherical bubbles in contaminated CMC solutions are similar to that in clean CMC solutions, implying no significant effect of the surfactant on the bubble motion in non-Newtonian fluids.
3) For 10 References
[1] CLIFT R, GRACE J R, WEBER M E. Bubbles, drops, and particle [M]. New York: Academic Press, 1978.
[2] LI Shao-bai, YAN Zheng, LI Run-dong, WANG Lei, LUAN Jing-de. Numerical simulation of single bubble rising in shear-thinning fluids by level set method [J]. Journal of Central South University, 2016, 23(4): 1000�C1006. DOI: 10.1007/s11771-016-3148-3.
[3] BOZZANO G, DENTE M. Shape and terminal velocity of single bubble motion: A novel approach [J]. Computers & Chemical Engineering, 2001, 25(4�C6): 571�C576. DOI: 10.1016/S0098-1354(01)00636-6.
[4] LOTH E. Quasi-steady shape and drag of deformable bubbles and drops [J]. International Journal of Multiphase Flow, 2008, 34(6): 523�C546. DOI: 10.1016/ j.ijmultiphaseflow.2007.08.010.
[5] FAN Wen-yuan, MA You-guang, JIANG Shao-kun, LI Huai-zhi. An experimental investigation for bubble rising in non-Newtonian fluids and empirical correlation of drag coefficient [J]. Journal of Fluids Engineering, 2010, 132(2): 150�C153. DOI: 10.1115/1.4000739.
[6] RODRIGUE D. A general correlation for the rise velocity of single gas bubbles [J]. Canadian Journal of Chemical Engineering, 2004, 82(2): 382�C386. DOI: 10.1002/cjce. 5450820219.
[7] KULKARNI A A, JOSHI J B. Bubble formation and bubble rise velocity in gas-liquid system: A review [J]. Industrial & Engineering Chemistry Research, 2005, 44: 5873�C5931. DOI: 10.1021/ie049131p.
[8] KRZAN M, ZAWALA J, MALYSA K. Development of steady state adsorption distribution over interface of a bubble rising in solutions of n-alkanols (C5, C8) and n-alkyltrimethylammonium bromides (C8, C12, C16) [J]. Colloids & Surfaces a Physicochemical & Engineering Aspects, 2007, 298(1�C2): 42�C51. DOI: 10.1016/j.colsurfa. 2006.12.056.
[9] ZHANG Wei, NESSET J E, FINCH J A. A novel approach to prevent bubble coalescence during measurement of bubble size in flotation [J]. Journal of Central South University, 2014, 21(1): 338�C343. DOI: 10.1007/s11771-014-1945-0.
[10] MCCLURE D D, AI C L, KAVANAGH J M, FLETCHER D F, BARTON G W. Impact of surfactant addition on oxygen mass transfer in a bubble column [J]. Chemical Engineering & Technology, 2015, 38(1): 44�C52. DOI: 10.1002/ceat. 201400403.
[11] LEVEN M D, NEWMAN J. The effect of surfactant on the terminal and interfacial velocities of a bubble or drop [J]. AlChE J, 1976, 22: 695�C701. DOI:10.1002/aic.690220411.
[12] KARAMANEV D G. Rise of gas bubbles in quiescent liquids [J]. AlChE J, 1994, 40: 1418�C1421. DOI: 10.1002/aic.690400814.
[13] DAGAN Z, YAN Zong-yi, SHEN Hui-xian. The axisymmetric rise of a spherical bubble at the exit of an orifice in the presence of a stagnant cap of insoluble surfactants [J]. Journal of Fluid Mechanics, 1988, 190(5): 299�C319. DOI: 10.1017/S0022112088001326.
[14] ZHANG Yong-qin, FINCH J A. A note on single bubble motion in surfactant solutions [J]. Journal of Fluid Mechanics, 2001, 429: 63�C66. DOI: 10.1017/ S0022112000002755.
[15] ZHANG Yong-qin, MCLAUGHLIN J B, FINCH J A. Bubble velocity profile and model of surfactant mass transfer to bubble surface [J]. Chemical Engineering Science, 2001, 56(23): 6605�C6616. DOI: 10.1016/S0009-2509(01)00304-9.
[16] ALVES S S, ORVALHO S P, VASCONCELOS J M T. Effect of bubble contamination on rise velocity and mass transfer [J]. Chemical Engineering Science, 2005, 60: 1�C9. DOI: 10.1016/j.ces.2004.07.053.
[17] WANG Dan, ZHANG Xin-yi, WANG Jie, KOIDE T. Effect of small amount of water on CO2 bubble behavior in ionic liquid systems [J]. Industrial & Engineering Chemistry Research, 2014, 53(1): 428�C439. DOI: 10.1016/j.ssc.2008. 11.024.
[18] ZHANG Wen-hui, JIANG Xiao-ya, DU Wei-tao. Bubble rise velocity in a fiber suspension [J]. Industrial & Engineering Chemistry Research, 2013, 52(24): 8340�C8345. DOI: 10.1021/ie3034834.
[19] LI Xiao-jin, MAO Zai-sha. The effect of surfactant on the motion of a buoyancy-driven drop at intermediate Reynolds numbers: a numerical approach [J]. Journal of Colloid & Interface Science, 2001, 240(1): 307�C322. DOI: 10.1006/jcis. 2001.7587.
[20] RODRIGUE D, de KEE D, FONG C F C M. An experimental study of the effect of surfactants on the free rise velocity of gas bubbles [J]. Journal of Non-Newtonian Fluid Mechanics, 1996, 66(2): 213�C232. DOI: 10.1016/0266- 3538(96)00044-9.
[21] RODRIGUE D, de KEE D, FONG C F C M. The slow motion of a single gas bubble in a non-Newtonian fluid containing surfactants [J]. Journal of Non-Newtonian Fluid Mechanics, 1999, 86(1): 211�C227. DOI: 10.1016/S0377- 0257(98)00209-2.
[22] TZOUNAKOS A, KARAMANEV D G, MARGARITIS A, BERGOUGNOU M A. Effect of the surfactant concentration on the rise of gas bubbles in power-law non-Newtonian liquids [J]. Industrial & Engineering Chemistry Research, 2004, 43(18): 5790�C5795. DOI: 10.1021/ie049649t.
[23] NALAJALA V S, KISHORE N. Drag of contaminated bubbles in power-law fluids [J]. Colloids & Surfaces, 2014, 443: 240�C248. DOI: 10.1016/j.colsurfa.2013.11.014.
[24] LI Shao-bai, MA You-guang, JIANG Shao-kun, FU Tao-tao, ZHU Chun-ying, LI Huai-zhi. The drag coefficient and the shape for a single bubble rising in non-Newtonian fluids [J]. Journal of Fluids Engineering, 2012, 134(8): 669�C679. DOI: 10.1115/1.4007073.
[25] DEWSBURY K, KARAMANEV D, MARGARITIS A. Hydrodynamic characteristics of free rise of light solid particles and gas bubbles in non-Newtonian liquids [J]. Chemical Engineering Science, 1999, 54(21): 4825�C4830. DOI: 10.1016/S0009-2509(99)00200-6.
[26] SHU T, MATSUMOTO Y. Surfactant effects on bubble motion and bubbly flows [J]. Annual Review of Fluid Mechanics, 2011, 43: 615�C636. DOI: 10.1146/annurev- fluid-122109-160756.
[27] HAYASHI K, TOMIYAMA A. Effects of surfactant on terminal velocity of a Taylor bubble in a vertical pipe [J]. International Journal of Multiphase Flow, 2012, 39: 78�C87. DOI: 10.1016/j.ijmultiphaseflow.2011.11.001.
[28] RODRIGUE D, BLANCHET J F. Surface remobilization of gas bubbles in polymer solutions containing surfactants [J]. Journal of Colloid & Interface Science, 2002, 256(2): 249�C255. DOI: 10.1006/jcis.2002.8578.
(Edited by HE Yun-bin)
���ĵ���
����ŵ���±�����Լ��Լ��б�ϡ�����������������Ե�Ӱ��
ժҪ���о��˵���ŵ����(Re<50)������Լ��Լ��б�ϡ�����������������Ե�Ӱ�졣Ϊ���ڱȽϣ����ø���������ɼ�ͼƬ�������õ���������δ�����Լ����ӱ�����Լ����ȼ���ά����(CMC)��Һ�е��ն��ٶȺ�ҷ��ϵ������������������������Сʱ(0.25wt% CMC: <100 mm3; 0.50wt% CMC: <110 mm3)�����ڱ�����Լ��������ݱ���ĸջ������ն��ٶ������Ľ��͡������������������������Լ���Ӱ����С��������ʧ����ҷ��ϵ�����߿�֪����Re<10ʱ�����б�����Լ���CMC��Һ�е�����ҷ��ϵ�����������С�����ƣ���10 �ؼ��ʣ�������Լ��������ݣ����б�ϡ���壻�ٶȣ�ҷ��ϵ��
Foundation item: Project(21406141) supported by the National Natural Science Foundation of China; Project(20141078) supported by the National Basic Research Program, China; Project(L201733) supported by the Research Foundation of Education Bureau of Liaoning Province, China
Received date: 2016-07-07; Accepted date: 2016-09-09
Corresponding author: LI Shao-bai, PhD, Associate Professor; Tel: +86-13674136938; E-mail: lishaobai1982@163.com; ORCID: 0000-0001-5121-1607