Process of electroless plating Cu-Sn-Zn ternary alloy
来源期刊:中国有色金属学报(英文版)2006年第1期
论文作者:何新快 陈白珍 胡更生 邓凌峰 周宁波 田文增
文章页码:223 - 228
Key words:electroless plating; Cu-Sn-Zn ternary alloy; color-change resistance; corrosion resistance
Abstract: Cu-Sn-Zn ternary alloy layer with 10 μm thickness was prepared through electroless plating method. The influences of process conditions including the concentration of metallic salts, reductant and complex agent on Cu-Sn-Zn alloy were studied in details. The stability to bear color changes and corrosion resistance of Cu-Sn-Zn film layer were studied through air-exposure experiment and electrochemical analyses test respectively. The results show that the performances of Cu-Sn-Zn film layer are obviously superior to brass matrix. By use of SEM,EDS and XRD, the morphology, microstructure and chemical composition were investigated. The results show that complex agent can increase the content of Sn and Zn, improve the evenness and compactness and decrease needle holes, therefore the properties of electroless plating layer such as the stability to bear color changes and corrosion resistance are improved remarkably. The probable mechanism of complex agent was discussed.
基金信息:the Natural Science Foundation of Hunan Province, China

HE Xin-kuai(何新快)1, 2,CHEN Bai-zhen(陈白珍)2, HU Geng-sheng(胡更生)1
DENG Ling-feng(邓凌峰)1, 2, ZHOU Ning-bo(周宁波)2, TIAN Wen-zeng(田文增)2
1. School of Packaging and Printing, Zhuzhou Institute of Technology, Zhuzhou 412008, China;
2. School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China
Received 15 January 2005; accepted 4 October 2005
Abstract: Cu-Sn-Zn ternary alloy layer with 10 μm thickness was prepared through electroless plating method. The influences of process conditions including the concentration of metallic salts, reductant and complex agent on Cu-Sn-Zn alloy were studied in details. The stability to bear color changes and corrosion resistance of Cu-Sn-Zn film layer were studied through air-exposure experiment and electrochemical analyses test respectively. The results show that the performances of Cu-Sn-Zn film layer are obviously superior to brass matrix. By use of SEM,EDS and XRD, the morphology, microstructure and chemical composition were investigated. The results show that complex agent can increase the content of Sn and Zn, improve the evenness and compactness and decrease needle holes, therefore the properties of electroless plating layer such as the stability to bear color changes and corrosion resistance are improved remarkably. The probable mechanism of complex agent was discussed.
Key words: electroless plating; Cu-Sn-Zn ternary alloy; color-change resistance; corrosion resistance
1 Introduction
Electroless plating tin and tin alloy is an attractive technique to produce function plating layer with prior performances of corrosion resistance and decoration[1, 2], due to the possibility of uniform deposition on complex shapes, conductive and non-conductive substrates, hardness and relatively simple equipment[3-7]. At present, these plating layers are applied in many fields, such as chemical industry, machinery, electron and spaceflight. A lot of domestic and overseas scientific workers are interested in electroless plating tin, especially in electroless plating tin binary alloy[8-12]. But the study of electroless plating tin ternary alloy is relatively little, and the study of Cu-Sn-Zn ternary alloy electroless plating is still not reported. The electroless plating tin binary alloy layers, for example Cu-Sn plating layer, have bad stability to bear color changes, from gold to gray, appearing dark spots,and so on[13-16]. To solve these problem, it is necessary to develop a new process to obtain Cu-Sn-Zn ternary alloy electroless plating layer, which does not fade and has better corrosion resistance than binary alloy. In this study, Cu-Sn-Zn ternary alloy electroless plating layer with 10 μm thickness was successfully prepared on brass substrate, which did not fade when exposed in air after a year, and showed better corrosion resistance than brass substrate.
2 Experimental
2.1 Materials and electroless plating
The matrix used was brass(H65), whose size was 40 mm×20 mm×2 mm. Electroless baths were prepared using analytic grade reagents and distilled water. Brass substrates were polished mechanically to mirror bright finish and degreased in acetone solution. Before electroless plating, the substrates were activated in a 10% H2SO4 solution. After each of these pretreatment steps the substrates were cleaned in distilled water. Electroless plating was performed for 10-15 min at room temperature in electroless bath containing: CuCl2 12 g/L, SnCl2 20 g/L, ZnCl2 40 g/L, NaH2PO2 40 g/L, HC l80 mL/L, H3COONa 25 g/L, complex agent A 25 g/L, complex agent B 0.15 g/L, pH 4-5. During electroless deposition the bath was agitated mechanically.
2.2 Perfomances measurement and analyses of electroless plating layer
KYKY2800 SEM was used to observe the morphology of Cu-Sn-Zn electroless plating layer, the composition of layer was analyzed by EDS. The thickness was measured by the following formula: d=(m2-m1)/S×ρ. Where m2, m1 are respectively the mass of Cu-Sn-Zn ternary alloy electroless plating specimen and substrate; S is the area of the electroless plating specimen; ρ=ρ(Cu)×w(Cu)+ρ(Sn)×w(Sn)+ ρ(Zn)×w(Zn) (ρ(Cu)=8.9 g/cm3, ρ(Sn)=7.29 g/cm3, ρ(Zn)=7.09 g/cm3, w is the mass fraction of Cu, Sn, Zn in the alloy). The stability of the layer to bear color changes was studied through air-exposure experiment by observing its rate of color and luster alteration in room, and the corrosion resistance of the layer was investigated through electrochemical analyses test in 1 mol/L H2SO4, 3.5% NaCl and 10% NaOH solution respectively using electrochemical work station CHI600b. Cu-Sn-Zn plating layer was used as work electrode, the large area platinum slice as auxiliary electrode, and the reference electrode was a saturated calomel electrode (SCE).
3 Results and discussion
3.1 Influences of main salts
The important function of main salts(CuCl2, SnCl2 and ZnCl2) in bath is to provide Cu2+, Sn2+ and Zn2+ in the process of electroless deposition, and their concentrations have great influences on the composi- tions and performances of Cu-Sn-Zn ternary alloy. In order to obtain their influences simply and effectively, the method of keeping other compositions unchanging and only changing one of the concentrations of Cu2+, Sn2+, Zn2+, reductant and complex agent respectively was adopted, and the results are shown in Fig.1.The optimum content of salts, reductant and complex agent in bath was determined by air-exposure experiment, electrochemical analyses and deposition rate test.
As shown in Fig.1, the contents of Cu, Sn and Zn in the Cu-Sn-Zn alloy increase respectively with the increasing of main salts(CuCl2, SnCl2 and ZnCl2) concentration, but the increasing rate of Cu is faster than those of Sn and Zn, because Cu2+ is easier to obtain the electrons provided by reductant to deposit on brass substrate than Sn2+ and Zn2+. Figs.1(b) and (c) indicate that Cu content decreases when the main salts(SnCl2, ZnCl2) concentrations increase respec- tively, but its content almost does not change when the main salts(SnCl2, ZnCl2) concentrations are over 16 g/L and 10 g/L. However, Sn and Zn contents relatively increase or decrease with the main salt (SnCl2, ZnCl2) concentration increasing respectively. At last Sn and Zn contents maintain stable. The experiments also show that when CuCl2 concentration is over 20 g/L, Sn and Zn contents, especially Sn (3%), are very little. The Cu-Sn-Zn alloy containing different contents of Sn and Zn was exposed in air to test the stability to bear color changes. The results show that when the Sn and Zn contents are over 43%(mass fraction), it is still bright after a year’s exposing. While the color of the alloy, of which Sn and Zn contents are less than 40%, changes from platinum to gray, and dark spots appear on its surface after half a year, and when Sn content is less than 6%, it is seriously faded in one or two months. So, the optimum concentrations of CuCl2 , SnCl2 and ZnCl2 in the bath are 12 g/L, 20 g/L and 40 g/L.
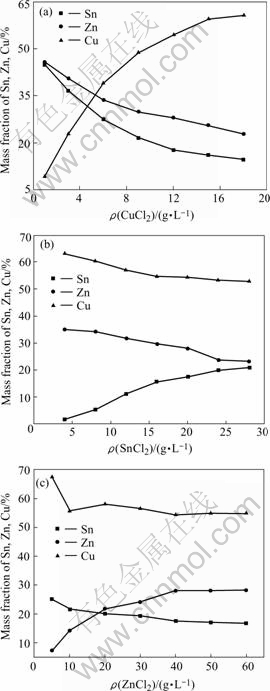
Fig.1 Influences of concentration of salt on composition of Cu-Sn-Zn alloy: (a) ρ(SnCl2)=20 g/L, ρ(ZnCl2)=40 g/L; (b) ρ(CuCl2)=12 g/L, ρ(ZnCl2)=40 g/L; (c) ρ(CuCl2)=12 g/L, ρ(SnCl2)=20 g/L
3.2 Influences of reductant
Experiments show that the reductant (NaH2PO2) concentration, which increases from 10 g/L to 50 g/L, has little influence on the deposition rate, which maintains 0.4 μm/min, when the buffer agent (CH3COONa) concentration is less than 15 g/L. The Cu-Sn-Zn alloy layer is not smooth and compact, and easily shells out, because the pH value of the bath, especially the pH value of interface between deposition substrate and solution, is not stable, due to the reaction: ![]() +H2O→
+H2O→![]() +OH-+2e-. But when the CH3COONa concentration is up to 25 g/L, NaH2PO2 concentration increases from 10 g/L to 40 g/L, the deposition rate increases obviously. However, when the NaH2PO2 concentration is over 40 g/L, the rate increases slowly. This is consistent with the result of Ref. [9]. The effect of NaH2PO2 on the rate and composition of Cu-Sn-Zn alloy is illustrated in Fig.2. The content of Sn increases from 14.26% to 18.15%, that of Zn from 23.18% to 28.12%, and that of Cu decreases from 62.56 % to 53.73%. The reason may be that Cu2+, Sn2+ and Zn2+ compete with each other to obtain electrons provided by reductant to deposit. When the NaH2PO2 concentration is low, the number of electrons is fewer, but Cu2+ is easier to obtain electrons to deposit, so Cu content is high. When NaH2PO2 concentration is high, the electron number is increased, the probability of Sn2+ and Zn2+ to obtain the electrons is increased. This leads to the increasing content of Sn and Zn. Considering the composition and the deposition rate, the optimum concentration of NaH2PO2 is 40 g/L.
+OH-+2e-. But when the CH3COONa concentration is up to 25 g/L, NaH2PO2 concentration increases from 10 g/L to 40 g/L, the deposition rate increases obviously. However, when the NaH2PO2 concentration is over 40 g/L, the rate increases slowly. This is consistent with the result of Ref. [9]. The effect of NaH2PO2 on the rate and composition of Cu-Sn-Zn alloy is illustrated in Fig.2. The content of Sn increases from 14.26% to 18.15%, that of Zn from 23.18% to 28.12%, and that of Cu decreases from 62.56 % to 53.73%. The reason may be that Cu2+, Sn2+ and Zn2+ compete with each other to obtain electrons provided by reductant to deposit. When the NaH2PO2 concentration is low, the number of electrons is fewer, but Cu2+ is easier to obtain electrons to deposit, so Cu content is high. When NaH2PO2 concentration is high, the electron number is increased, the probability of Sn2+ and Zn2+ to obtain the electrons is increased. This leads to the increasing content of Sn and Zn. Considering the composition and the deposition rate, the optimum concentration of NaH2PO2 is 40 g/L.
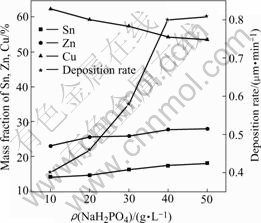
Fig.2 Influences of concentration of NaH2PO2 on deposition rate and composition of Cu-Sn-Zn alloy
3.3 Influences of complex agent
Complex agent A is an alcohol acid containing less than 8 carbon atoms, it acts as the complex function. Complex agent B is an organic compound containing coordinating N and S atoms, it acts as the stable function. The effect of complex agent A and B on electroless plating rate and composition of Cu-Sn-Zn alloy is shown in Fig.3.
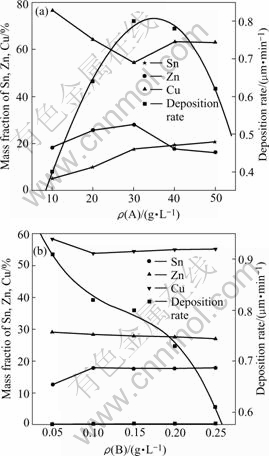
Fig.3 Influences of concentration of complex agent on deposition rate and composition of Cu-Sn-Zn alloy: (a) ρ(B)= 0.15 g/L; (b) ρ(A)= 30 g/L
When the complex agent A concentration is low, the deposition rate of Cu-Sn-Zn alloy increases rapidly with its concentration rising, while the concentration is over 30 g/L, the rate increases slowly, and when the concentration is above 40 g/L, the rate decreases. The reason may be that Cu2+, Sn2+ and Zn2+ deposit through coordination ions. When the complex agent A concentration is low, it is not enough to coordinate all Cu2+, Sn2+ and Zn2+, so the deposition rate is slow, and is increased with the increasing concentration of complex agent A. After the rate reaches the maximum, the deposition rate will drop down, due to the great residual adsorption of complex agent A on the reduction surface and preventing the coordination ions from depositing. Complex agent A also has great influence on the composition of Cu-Sn-Zn alloy (Fig.3(a)). When it is relatively low, the Sn and Zn contents increase with the increasing of complex agent A, while Cu content rapidly reduces. When the complex agent A is over 30 g/L, Sn content increases slowly, while Zn reduces. The reason may be that the amount of adsorption on the auto-catalytic reduction surface[16] is less when the complex agent A concentration is low, which accelerates the rate of deposition and crystallization. When the complex agent A concentration is high, the great amount of adsorption will poison the reduction reaction[15]. From the above discussion, the optimum concentration of complex agent A is determined to be 30 g/L.
As shown in Fig.3(b), the deposition rate drops rapidly with the increasing of complex agent B concentration, while the Zn content almost sustains unchangeable. When the complex agent B concentration is relatively low, the Sn content increases with the increasing of complex agent B concentration, while that of Cu reduces. When the complex agent B concentration is over 0.1 g/L, the composition of Cu-Sn-Zn alloy maintains stable. The complex agent B is probable to decrease the rate of anti-reduction reaction, and strengthen cathodic polarization, which leads to the formation of fine-grain, better evenness and compactness of the plating layer(Fig.4). Considering the quality of Cu-Sn-Zn alloy and the deposition rate, the optimum concentration of complex agent B is 0.15 g/L.
3.4 SEM morphology of different Cu-Sn-Zn plating alloy
Fig.4 shows the morphologies of different Cu-Sn- Zn alloy layers obtained from different electroless bath.
It can be found that the morphology changes remarkably. For the optimum samples, the best evenness and compactness can be observed and needle holes hardly appear on its surface from SEM images (Fig.4(a)). For the samples without complex agent A, although the compactness is good, but fine projection appears on its surface (Fig.4(b)). For the samples without complex agent B, not only a large number of needle holes appear on microstructure surface, but also the evenness and compactness get worse as shown in Fig.4(c). Fig.4 shows that both complex agents A and B can improve the evenness and compactness of plating layer. In the process of electroless plating, complex agent A can alter the crystal microstructure in deposition. The reason may be that complex agent A is adsorbed on the auto-catalytic reduction surface, which not only accelerates the deposition and crystallization rate to obtain fine-grain, but also postpones or prevents the bath decomposing and decreases hydrogen evolution to strengthen its stability, decrease needle hole and hydrogen brittleness. While complex agent B decrease the rate of anti-reduction reaction and strengthen cathodic polarization, which leads to the formation of fine-grain, better evenness and compactness of the plating layer. The air-exposure experiments shows that samples(a) do not fade after a year, samples(b) change from platinum to gray after 8 months, and samples(c) change from platinum to gray and dark spots appear on its surface in 30 d. This illustrates that complex agents A and B can strengthen the stability to bear color changes of Cu-Sn-Zn plating layer.

Fig.4 SEM morphologies of Cu-Sn-Zn alloys: (a) Optimum plating layer; (b) Plating without complex agent A; (c) Plating without complex agent B
3.5 EDS and XRD analyses of Cu-Sn-Zn plating alloy layer
Fig.5 and Fig.6 show the EDS and XRD spectra of Cu-Sn-Zn plating layer under optimum conditions. As shown in Fig.5, the main compositions are Cu, Sn, and Zn, and their atomic ratio is approximately 60∶30∶10. It shows that Cu2+, Sn2+ and Zn2+ obtain electrons and deposit on brass substrate with the help of reductant(NaH2PO2). The XRD pattern (Fig.6) shows that the layer is a Cu0.60Sn0.10Zn0.30 ternary alloy.
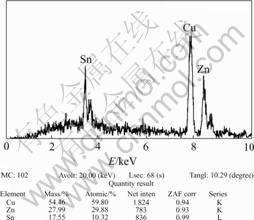
Fig.5 EDS spectra of Cu-Sn-Zn alloy layer
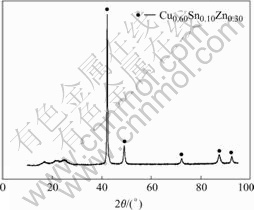
Fig.6 XRD spectra of Cu-Sn-Zn alloy layers
3.6 Electrochemical analyses test and thickness measurement of plating alloy layer
The thickness of Cu-Sn-Zn plating alloy layer obtained under optimum conditions was measured by the formula: d=(m2-m1)/S×ρ, its average thickness is approximately 10.06 μm. Fig.7 shows the corrosion curves of different Cu-Sn-Zn plating alloys.
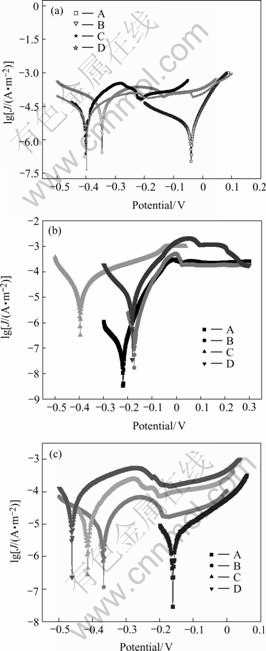
Fig.7 Corrosion curves of Cu-Sn-Zn alloys: (a) 1 mol/L H2SO4 solution; (b) 3.5% NaCl solution; (c) 10% NaOH solution; A, Optimum plating layer(Cu54.46Sn17.55Zn27.99); B, Plating without complex agent A(Cu63.16Sn19.09Zn17.74); C, Plating without complex agent B(Cu62.72Sn6.03Zn31.25); D, Brass substrate
As shown in Fig.7, the corrosion potential of plating alloy layer A is about 420 mV higher than that of brass substrate and its corrosion current density is the lowest in 1mol/L H2SO4 solution, which improves the corrosionresistance of the layer remarkably, while the corrosion potential of layer C is unchangeable and its corrosion current density is higher than that of brass substrate, so its corrosion-resistance is the worst (Fig.7(a)). Although the corrosion potential of layer A is a bit more negative than that of layer B and brass substrate, its corrosion current density is the lowest, layer B is the second, and layer C is the highest in 3.5% NaCl neutral solution, which proves that the corrosion-resistance and color-change resistance of layer A are the best in the neutral environment(in the air) and the layer C is easy to fade(Fig.7(b)). The corrosion potential of layers A, B and C are respectively about 300 mV, 100 mV and 50 mV higher than that of brass substrate in 10% NaOH solution, and the corrosion current density of layer A is the lowest, so the best anticorrosive layer in the alkali environment is layer A, the second is layer B, the worst is layer C(Fig.7(c)). Besides the evenness, compactness and lest needle holes on the surface, the main reason for the best corrosion-resistance of layer A is that the highest contents of Sn and Zn in the Cu-Sn-Zn alloy which reach 45.54%. Although the contents of Sn and Zn in layer B and C are approximately the same, the content of Sn in layer C is so low that its corrosion-resistance is the worst.
4 Conclusions
1) Cu-Sn-Zn ternary alloy layer with thickness up to 10 μm was prepared through electroless plating method. The optimum bath composition and plating conditions are as follows: CuCl2 12 g/L, SnCl2 20 g/L, ZnCl2 40 g/L, NaH2PO2 40 g/L; HC l80 mL/L, CH3COONa 25 g/L, complex agent A 25 g/L, complex agent B 0.15 g/L, time 10-15 min and room tempe- rature.
2) The electroless plating layer is Cu0.60Sn0.10Zn0.30 ternary alloy. The stability of Cu-Sn- Zn plating layer to bear color changes is excellent(no fading after a year) and the corrosion resistance of Cu-Sn-Zn plating layer is obviously superior to the brass matrix.
3) The performances of color-change resistance and corrosion resistance depend on the quality (evenness, compactness and needle holes) and compo- sition of Cu-Sn-Zn alloy plating layer. The better the evenness and compactness, the less the needle holes and the more the content of Sn and Zn, especially the more content of Sn in the Cu-Sn-Zn ternary alloy, the better the performances of it.
4) Complex agent A can increase the content of Sn and Zn through coordinating with Cu2+, Sn2+ and Zn2+. Although complex agent B can decrease deposition rate, it can improve the performances of evenness and compactness and decrease needle holes, therefore the properties of plating layer such as the stability to bear color changes and corrosion resistance are improved remarkably.
References
[1] ZHANG Jian. Electroless Cu-Sn-P plating and its deposit properties[J]. Materials Protection, 1995, 28(5): 7-9.(in Chinese)
[2] CAI Ji-Qing. Electroless Sn and Sn-Pb alloy plating[J]. Corrosion and Protection, 2001, 22(6): 270-271.(in chinese)
[3] LIANG Cheng-hao, CHEN Bang-yi, CHEN Wan, WANG Hua. Electrochemical behavior of Cu-Zn-Al shape memory alloy after surface modification by electroless plated Ni-P[J]. Rare Metals, 2004, 23(4): 317-321.(in Chinese)
[4] WANG Sen-lin, WU Hui-huang. Electroless plating of Ni-Zn(Fe)-P alloy on carbon steel sheets[J]. Trans Nonferrous Met Soc China, 2004, 14(S2): 157-161.
[5] DENG Fu-ming, CHEN Xiao-hua, CHEN Wei-xiang, LI Wen-zhu. Electroless plating Ni-P matrix composite coating reinforced by carbon nanotubes[J]. Trans Nonferrous Met Soc China, 2004, 14(4): 681-685.
[6] WANG Tian-xu, HU Yong-jun, MENG Ji-long. Novel technology of electroless Ni-W-P on plastics and its interface behavior[J]. Trans Nonferrous Met Soc China, 2004, 14(S2): 136-139.
[7] FU Yu-bin, ZHANG Li-de, ZHENG Ji-yong. Electroless deposition of Ni on template of halloysite and its magnetic property [J]. Trans Nonferrous Met Soc China, 2004, 14(S2): 152-156.
[8] LI Dao-hua. Structure and characterization of Cu-Sn-P/Ni-Sn-P alloy composite cladding materials on iron and steel surface[J]. Journal of China West Normal University, 2004, 25(2): 180-183. (in Chinese)
[9] ZHANG Bang-wei, XIE Hao-wen. Investigation of amorphous formation ability of Ni-Sn-Cu-P alloy deposit prepared by chemical reduction[J]. Rare Metal Materials and Engneering, 1998, 27(2): 79. (in Chinese)
[10] SI Xiu-li, WU Feng, CHU Song-zhu. Investugation of technology and performance of the electroless-deposited film of Ni-Sn-P alloy[J]. Function Materails, 1995, 26(2): 189-191. (in Chinese)
[11] XIE Hao-wen, ZHANG Bang-wei, XU Xie-wen, LIAO Shu-zhi. Investigation on preparation of quaternary amorphous Ni-Sn-Cu-P alloys by electroless plating[J]. Journal of Changsha Communications University, 1998, 14(3):24. (in Chinese)
[12] Chonan Y, Komiyama K, Onuki K, KIMURA T, NAGANO T. ![]() Influence of phosphorus concentration in electroless plated Ni-P alloy film on interfacial structures and strength between Sn-Ag-(-Cu) solder and plated Ni-P alloy film[J]. Materials Transactions, 2002, 43(8): 1840-1846.
Influence of phosphorus concentration in electroless plated Ni-P alloy film on interfacial structures and strength between Sn-Ag-(-Cu) solder and plated Ni-P alloy film[J]. Materials Transactions, 2002, 43(8): 1840-1846.
[13] Yoon J W, Jung S B. Growth kinetics of Ni3Sn4 and Ni3P layer between Sn-3.5Ag solder and electroless Ni-P substrate[J]. Journal of Alloys and Compounds, 2004, 376(1-2): 105.
[14] LI Ning. Practical Technology of Electroless Plating [M]. Beijing: Chemistry Industry Press, 2004. (in Chinese)
[15] XIA Chang-rong, GUO Xiao-xia, LI Fan-qing, CHEN Chu-sheng. Preparation of asymmetric Ni/ceramic composite membrane by electroless plating[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2001, 179(1-2): 229-235.
[16] Abd EL-Rehim S S, Shaffei M, EL-Ibiari N, ABDEL HALEM S. Effect of additives on plating rate and bath stability of electroless deposition of nickel-phosphorous-boron on aluminum[J]. Metal Finishing, 1996, 94(12): 29-33.
Foundation item: Project(04JJ40036)Supported by the Natural Science Foundation of Hunan Province, China
Corresponding author: HE Xin-kuai; Tel: +86-731-8876621; E-mail: h-xk@163.com
(Edited by YUAN Sai-qian)

