合金成分对铝导线力学性能与导电性能的影响
来源期刊:中国有色金属学报(英文版)2014年第10期
论文作者:钞润泽 管西花 管仁国 铁 镝 连 超 王 祥 张 俭
文章页码:3164 - 3169
Key words:alloy composition; Al wires; mechanical properties; electrical conductivity; continuous rheo-extrusion
摘 要:为了获得既有良好力学性能又有较高导电性能的铝导线,采用连续流变挤压技术制备出直径为9.5 mm的Al-0.16Zr、Al-0.16Sc、Al-0.12Sc-0.04Zr(质量分数,%)和高纯铝(99.996%)4种铝导线,随后对3种铝合金导线进行热处理,并进行分析测试。结果表明:向高纯铝中单独添加0.16% Sc和0.16% Zr可以提高铝导线的抗拉强度,降低铝导线的导电性能。在高纯铝中添加0.12%Sc和0.04%Zr的合金也具有相同的规律。经过热处理,Al-0.12Sc-0.04Zr铝合金导线可以达到抗拉强度和导电率的最优综合性能:抗拉强度为160 MPa,导电率为64.03% (IACS)。
Abstract: In order to obtain the Al wires with good mechanical properties and high electrical conductivities, conductive wires of Al-0.16Zr, Al-0.16Sc, Al-0.12Sc-0.04Zr (mass fraction, %) and pure Al (99.996%) were produced with the diameter of 9.5 mm by continuous rheo-extrusion technology, and the extruded materials were heat treated and analyzed. The results show that the separate additions of 0.16% Sc and 0.16% Zr to pure Al improve the ultimate tensile strength but reduce the electrical conductivity, and the similar trend is found in the Al-0.12Sc-0.04Zr alloy. After the subsequent heat treatment, the wire with the optimum comprehensive properties is Al-0.12Sc-0.04Zr alloy, of which the ultimate tensile strength and electrical conductivity reach 160 MPa and 64.03% (IACS), respectively.
Trans. Nonferrous Met. Soc. China 24(2014) 3164-3169
Run-ze CHAO1, Xi-hua GUAN 2, Ren-guo GUAN1, Di TIE1, Chao LIAN1, Xiang WANG1, Jian ZHANG1
1. School of Materials and Metallurgy, Northeastern University, Shenyang 110819, China;
2. School of Transportation and Mechanical Engineering, Shenyang Jianzhu University, Shenyang 110168, China
Received 28 October 2013; accepted 21 March 2014
Abstract: In order to obtain the Al wires with good mechanical properties and high electrical conductivities, conductive wires of Al-0.16Zr, Al-0.16Sc, Al-0.12Sc-0.04Zr (mass fraction, %) and pure Al (99.996%) were produced with the diameter of 9.5 mm by continuous rheo-extrusion technology, and the extruded materials were heat treated and analyzed. The results show that the separate additions of 0.16% Sc and 0.16% Zr to pure Al improve the ultimate tensile strength but reduce the electrical conductivity, and the similar trend is found in the Al-0.12Sc-0.04Zr alloy. After the subsequent heat treatment, the wire with the optimum comprehensive properties is Al-0.12Sc-0.04Zr alloy, of which the ultimate tensile strength and electrical conductivity reach 160 MPa and 64.03% (IACS), respectively.
Key words: alloy composition; Al wires; mechanical properties; electrical conductivity; continuous rheo-extrusion
1 Introduction
Since the electricity transmission by overhead wires has become the main way of the electricity transmission in electric power industry, long-span wires not only need good electrical conductivity to improve the transmission capacity and reduce line losses, but also should have enough mechanical strength to support their own mass and external natural load and improve their service life. The overhead conductive wires are mainly produced by Al alloy, while it is rather difficult to increase both the electrical conductivity and mechanical properties in the mean time. Therefore, much attention has been paid to the technology of producing Al alloy wires with both good electrical conductivity and mechanical properties [1]. There exist many factors that can influence the properties of Al alloy wires, such as chemical components, crystalline state, and processing technology. It was found that the Zr could improve the heat-resistant property of Al alloy and developed series of Al wires containing Zr, of which the electrical conductivity and the ultimate tensile strength could reach 60% (IACS) and 158.8-175 MPa [1]. Russian researchers found that the addition of Sc element could increase the recrystallization temperature and improve the thermal stability. Therefore, Sc was widely studied in the Al alloy [2,3]. Moreover, the physical and chemical properties of Zr and Sc are similar [4,5]. The joint addition of Zr and Sc to the Al alloy can form Al3Sc, Al3Zr and Al3(Sc,Zr) phases with good thermal stability. In particular, Al3(Sc,Zr) phase not only has all the advantages of Al3Sc phase, but also has better thermal stability [6-9]. Under the condition of Sc, the addition of Zr can enhance the beneficial effect of Sc in the Al alloy [7-9]. At present, the effect of Sc and Zr on the mechanical properties of Al alloy has been widely studied, however, the effect of Sc and Zr on the electrical conductivity is rarely reported. In addition, the continuous rheo-extrusion technology which can be used to produce conductive wires has amounts of advantages, such as energy conservation, near-net shape forming, good product quality and low cost. Therefore, it is considered as one of the most promising metal forming technologies [10,11]. Heat treatment plays an important role in the production of conductive wires with good properties. And it directly influences the final mechanical properties and electric conductivity [10]. In the present study, Zr and Sc were added into the pure Al. And the effect of trace amount of Zr and Sc on the mechanical properties and electrical conductivities of Al wires produced by continuous rheo-extrusion technology was studied.
2 Experimental
The raw materials in the experiment include pure Al (99.996%, mass fraction), Al-Sc master alloy (2.9% Sc), Al-Zr master alloy (4.6% Zr), which were produced by Yueyang Yuhua Metallurgical New Materials Co., Ltd. After the chemical analysis, the compositions of Al wires are listed in Table 1.
Table 1 Element content of Al wires (mass fraction, %)

The preheated and dry pure Al ingots were melted in the crucible resistance furnace. When the ingots melted and the temperature reached 720 °C, Al-Sc (2.9% Sc) and Al-Zr (4.6% Zr) master alloys were added into the melt after 10 min. Then the melt was stirred with the preheated ceramic rod. The melt was protected by argon gas all the time. Degassing and deslagging were adopted before pouring.

Fig. 1 Schematic diagram of continuous rheologic extrusion process
The Al wires were produced with DZJ350 continuous rheo-extrusion machine which was self- designed in Northeastern University, China. The schematic diagram of continuous rheo-extrusion process is shown in Fig. 1. The alloy melt was firstly poured into the roll groove of the continuous rheo-extrusion machine and solidified by chilling and shearing effect. Finally, the Al wires with the diameter of 9.5 mm were extruded through the extrusion die.
The TM-0910P Ceramic Fiber Muffle Furnace was used in the solution experiment of the Al wires, and JH-36A Well Resistance Furnace was used in the aging experiment of the Al wires. The parameters of heat treatment are listed in Table 2.
Table 2 Parameters of heat treatment

OLYMPUS DSX500 metallographic microscope and SSX-550 scanning electron microscope were used to observe and analyze the microstructure, and EDS analyses were performed. SB2230 Digital DC resistance tester was used to test the resistance value of Al wires with 120 mm in length. The electrical conductivity K of the materials could be obtained according to equations as follows:
 (1)
(1)
 (2)
(2)
where K is conductivity (IACS); ρ is electrical resistivity (Ω・mm2/m); R is conductor resistance (Ω); S is conductor cross-section (mm2); L is the length of the conductor (m).
3 Results and discussion
3.1 Effect of Zr and Sc on mechanical properties and electrical conductivities of as-extruded Al wires
Figure 2 shows the ultimate tensile strength and electrical conductivities of Al wires, including pure Al wire, Al-0.16Zr wire, Al-0.16Sc wire, Al-0.12Sc- 0.04Zr wire, which were produced by continuous rheo-extrusion technology. The ultimate tensile strength of Al-0.16Zr wire, Al-0.16Sc wire and Al-0.12Sc- 0.04Zr wire were 44.2, 51.76 and 62 MPa, which were increased by 5.2%, 23.2% and 47.6%, respectively, compared with the ultimate tensile strength of the pure Al wire (42 MPa). The separate addition of 0.16% Sc and 0.16% Zr to pure Al improved the ultimate tensile strength, and the similar trend was found in the Al-0.12Sc-0.04Zr alloy.

Fig. 2 Ultimate tensile strength and electrical conductivities of as-extruded Al wires
However, the separate addition of 0.16% Sc and 0.16% Zr to pure Al reduced the electrical conductivity, and there existed the similar trend in the Al-0.12Sc- 0.04Zr alloy. As shown in Fig. 2, the electrical conductivities of as-extruded pure Al wires could reach 64.6% (IACS), while the electrical conductivities of as-extruded Al-0.16Zr wire, Al-0.16Sc wire and Al-0.12Sc-0.04Zr wire only reached 57.6% (IACS), 58.23% (IACS) and 56.7% (IACS), which were decreased by 10.8%, 9.86% and 12.3%, respectively, compared with the electrical conductivity of pure Al wire.
When the content of Al is close to 99% in Al-Zr alloy, a peritectic reaction of the alloy melt and Al3Zr to form Al solid solution occurs at 660.5 °C. The melting point of Al is only 0.5 °C lower than the peritectic reaction temperature. Under the condition of equilibrium solidification, the maximum solid solubility of Zr in Al is 0.28% and declines with decreasing the temperature. At 427 °C, the maximum solid solubility of Zr in Al drops to 0.05%-0.06%. However, the solidification process cannot be in the equilibrium state in reality. Under the condition of rapid cooling, the solid solution content of Zr in Al matrix is more than 0.2% and even reaches about 2.0%. Zirconium atoms in the matrix replace the Al part of the atomic lattice, forming the substitutional solid solution which is the main form of Zr in the alloy without heat treatment [12].
In the process of continuous rheo-extrusion, the alloy melt was poured into the roll groove of the rheo-extrusion machine. Due to the shearing and chilling effect of the extrusion roll, the melt solidified rapidly. At last, the Al wires were produced through the extrusion die. In the process, the temperature decreased dramatically. Most of the element Zr remained in the matrix in the form of solid solution. Therefore, solid solution strengthening was the main reinforcement form.
The solid solubility of Sc in Al is 0.27% at 655 °C and 0.07% at 527 °C, which decreases to 0 at room temperature [13]. The Al3Sc phases precipitate with decreasing the temperature to room temperature. However, for the sake of non-equilibrium solidification in the process of continuous rheo-extrusion, it is impossible that all the Sc elements precipitate from the Al matrix. A part of Sc elements still remains in the matrix in the form of solid solution. The main reinforcement form contains solid solution strengthening and precipitation strengthening.
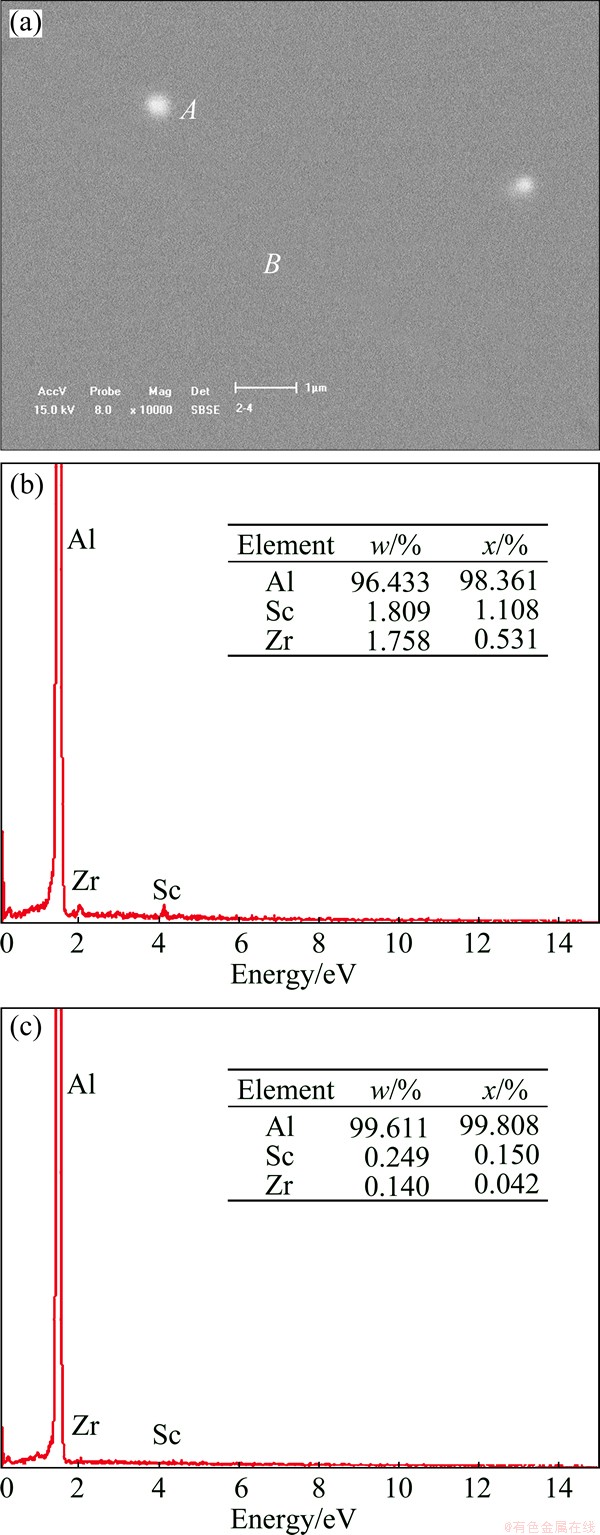
Fig. 3 SEM image of Al-0.12Sc-0.04Zr wire by continuous rheo-extrusion (a), and EDS results of point A (b) and point B (c)
Figure 3 shows the SEM image and EDS analysis result of the Al-0.12Sc-0.04Zr wire produced by continuous rheo-extrusion technology. As shown in Fig. 3(a), a small amount of precipitated phases with the diameter of 400 nm distributed dispersively in the microstructure of the Al-0.12Sc-0.04Zr wire. Figures 3(b) and (c) show the energy spectrum analysis result of the points A and B in Fig. 3(a), respectively. The contents of Sc and Zr in the precipitated phase are higher than those in the matrix. According to the reported studies, under the condition of casting, the precipitated phase is Al3(Sc,Zr) [14]. The stability of Al3(Sc,Zr) phase is better than that of both Al3Sc and Al3Zr phases [15]. The reason that the joint addition of 0.12% Sc and 0.04% Zr to pure Al improves the ultimate tensile strength of the Al wires with the largest extent is the generation of the Al3(Sc,Zr) phase.
As the temperature of the alloy decreased, the solid solubility of Zr and Sc decreased, leading to the precipitation of trace amount of Zr and Sc from the Al matrix in the continuous rheo-extrusion process. As shown in Fig. 3(a), the precipitated phase plays the role of precipitation strengthening, while the solute atoms plays the role of solid solution strengthening. Therefore, the ultimate tensile strength of Al-0.125Sc-0.04Zr is higher than that of other Al wires. Since the radius of the Zr and Sc atoms is bigger than that of Al atom, lattice distortion is produced in the alloy [16]. At room temperature, the crystal lattice is highly distorted due to supersaturated solid solution of Sc and Zr in Al matrix. The lattice distortion and precipitated phases can cause the conductive electron scattering, resulting in the decrease of the electrical conductivity. In addition, when Al atoms unite with Zr and Sc atoms, parts of free electrons in the alloy move into the electron shell of Zr and Sc atoms, causing the decrease of the amount of free electrons in the alloy, which also makes the electrical conductivity of the alloy decline [17].
3.2 Effect of Zr and Sc on electrical conductivities and mechanical properties of heat treated Al wires
In order to improve the mechanical properties and electrical conductivities of the Al-0.16Zr wire, Al-0.16Sc wire and Al-0.12Sc-0.04Zr wire produced by continuous rheo-extrusion process, heat treatment experiment was conducted subsequently. Figure 4 shows the changing curve of electrical conductivities and the ultimate tensile strength of Al alloy wires with the aging time after solid solution. It can be seen that both the electrical conductivities and ultimate tensile strength increase with the aging time, and tend to be stable at last.
As shown in Fig. 4(a), the optimal performance of Al-0.16Zr wire was obtained after solid solution at 520 °C for 1.5 h and aging treatment at 250 °C for 48 h. The ultimate tensile strength and electrical conductivity reach 49 MPa and 62.72% (IACS), respectively. In addition, after solid solution at 630 °C for 1.5 h and aging treatment at 320 °C for 6 h, both the Al-0.16Sc wire and Al-0.12Sc-0.04Zr wire reach the optimal performance. The ultimate tensile strength and electrical conductivities of Al-0.16Sc wire reach 110 MPa and 63.6% (IACS), while those of Al-0.12Sc-0.04Zr wire reach 160 MPa and 64.03% (IACS), respectively.

Fig. 4 Electrical conductivities and ultimate tensile strength of Al alloy wires at different aging time
Figure 5 shows the optimal comprehensive performance of Al wires. Compared with pure Al wire, the ultimate tensile strengths of Al-0.16Zr wire, Al-0.16Sc wire and Al-0.12Sc-0.04Zr wire are increased by 16.7%, 161.9% and 281%, respectively. The electrical conductivities of Al-0.16Sc wire, Al-0.16Sc wire and Al-0.12Sc-0.04Zr wire are decreased by 2.91%, 1.55% and 0.88%, respectively. In addition, compared with the as-extrusion wires, the ultimate tensile strengths of heat treated Al-0.16Zr wire, Al-0.16Sc wire and Al-0.12Sc-0.04Zr wire have been improved by 10.9%, 112.5% and 158.1%, while the electrical conductivities are increased by 8.9%, 9.22% and 12.85%, respectively. It can be concluded that the joint addition of 0.12% Sc and 0.04% Zr to the pure Al can more significantly increase the ultimate tensile strength and electrical conductivity of the heat treated Al alloy wires compared with the separate addition of 0.16% Sc and 0.16% Zr to the pure Al.

Fig. 5 Optimal comprehensive performance of four kinds of Al wires
Figure 6 shows the SEM images of heat treated Al-0.16Sc wire and Al-0.12Sc-0.04Zr wire. As shown in Fig. 6, the fine precipitated phases distribute dispersively in the matrix of heat treated Al-0.16Sc wire and Al-0.12Sc-0.04Zr wire. The amount of precipitated phases in the Al-0.12Sc-0.04Zr wire is less than that in the Al-0.16Sc wire, and the volume fraction of precipitated phase in the Al-0.12Sc-0.04Zr wire is larger than that in the Al-0.16Sc wire. The existing phases includes A13Sc, A13Zr and A13(Sc,Zr) in the Al- 0.12Sc-0.04Zr wire [18]. Al3Sc Ll2-type precipitated phase retains a coherent lattice relationship with Al matrix, while there exists a great coherency mismatch, which can introduce strong strain in the crystal lattice and inhibit the movement of the dislocations, and therefore prevent the grain growth. The coherency mismatch of Al3Sc and Al3Zr phases with the parent phase is about 1.2% and 0.8%, respectively [18]. Since the Sc can promote the recrystallization temperature to 600 °C, Al3Sc phase can not only prevent the grain growth but also completely inhibit the recrystallization compared with the Al3Zr phase [19].

Fig. 6 SEM images of heat treated Al-0.16Sc wire (a) and Al-0.12Sc-0.04Zr wire (b)
Similar to Al3Sc phase, A13(Sc,Zr) phase distributes dispersively in the matrix and even has more strong effect on strengthening the matrix and inhibiting the recrystallization. In addition, A13(Sc,Zr) phase is also more thermo-stable than the Al3Sc phase in the Al- matrix [20]. Therefore, the Al-0.12Sc-0.04Zr wire has better mechanical property.
In the mean time, the precipitated phase can cause the electron scattering, leading to the decrease of electrical conductivity. However, the electron scattering effect caused by the precipitated phase is much smaller than that caused by solid solution atoms [16]. Therefore, the heat treated Al alloy wire has a better electrical conductivity.
4 Conclusions
1) The separate addition of 0.16% Sc and 0.16% Zr to pure Al improved the ultimate tensile strength but reduced the electrical conductivity, and the similar trend was found in the Al-0.12Sc-0.04Zr alloy.
2) The joint addition of 0.12% Sc and 0.04% Zr to the pure Al could increase the ultimate tensile strength and electrical conductivities of the heat treated Al alloy wires more obviously than the separate addition of 0.16% Sc and 0.16% Zr to the pure Al.
3) After the subsequent heat treatment, the wire with the optimum comprehensive properties was Al-0.12Sc- 0.04Zr, of which the ultimate tensile strength and electrical conductivity reached 160 MPa and 64.03% (IACS), respectively.
References
[1] HUANG Chong-qi, FENG Jin-quan, LI Wen-hao, DING Guan-sen, HUANG Guo-fei, QIN Yu-fang, FU Xu-ping, DENG Yong-hong. Large-capacity conductor for overhead transmission lines [J]. Electric Power Construction, 2005, 26: 2-5. (in Chinese)
[2] ZAKHAROV V V, ROSTOVA T D. Effect of scandium, transition metals, and admixtures on strengthening of aluminum alloys due to decomposition of the solid solution [J]. Metal Science and Heat Treatment, 2007, 49(9-10): 435-442.
[3] DAVYDOV V G, ROSTOVA T D, ZAKHAROV V V, FILATOV Y A, YELAGIN V I. Scientific principles of making an alloying addition of scandium to aluminum alloys [J]. Materials Science and Engineering A, 2000, 280(1): 30-36.
[4] WANG Ying, PAN Qing-lin, SONG Yan-fang, LI Chen, LI Zhi-feng, CHEN Qin, YIN Zhi-min. Recrystallization of Al-5.8Mg-Mn-Sc- Zr alloy [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(11): 3235-3241.
[5] MENG Yi, ZHAO Zhi-hao, CUI Jian-zhong. Effect of minor Zr and Sc on microstructures and mechanical properties of Al-Mg-Si-Cu- Cr-V alloys [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(7): 1882-1889.
[6] JIA Zhi-hong, ROYSET J, SOLBERG J K, LIU Qing. Formation of precipitates and recrystallization resistance in Al-Sc-Zr alloys [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(8): 1866-1871.
[7] LEE S, UTSUNOMIYA A, AKAMATSU H, NEISHI K, FURUKAWA M, HORITA Z, LANGDON T G. Influence of scandium and zirconium on grain stability and superplastic ductilities in ultrafine-grained Al-Mg alloys [J]. Acta Materialia, 2002, 50(3): 553-564.
[8] FULLER C B, SEIDMAN D N, DUNAND D C. Mechanical properties of Al(Sc,Zr) alloys at ambient and elevated temperatures [J]. Acta Materialia, 2003, 51(16): 4803-4814.
[9] MUSIN F, KAIBYSHEV R, MOTOHASHI Y, ITOH G. High strain rate superplasticity in a commercial Al-Mg-Sc alloy [J]. Scripta Materialia, 2004, 50(4): 511-516.
[10] GUAN Ren-guo, LI Xue, LI Jiang-ping, XIAO Zhao-hui, ZHU Li-jie, WEN Jing-lin. Continuous extending rheo-extrusion process for producing 6201 alloy tube [J]. The Chinese Journal of Nonferrous Metals, 2009, 19(8): 1398-1402. (in Chinese)
[11] GUAN Ren-guo, ZHAO Zhan-yong, CAO Fu-rong, HUANG Hong-qian, ZHANG Qiu-sheng. Effect of continuous rheo-extrusion and heat treatment on microstructure and mechanical properties of 6201 alloy [J]. Transactions of Materials and Heat Treatment, 2011, 32(9): 53-57. (in Chinese)
[12] XIE You-hua, YANG Shou-jie, DAI Sheng-long, LU Zheng. Application of element Zr in aluminum alloys [J]. Journal of Aeronautical Materials, 2002, 22 (4): 56-61. (in Chinese)
[13]  J, RYUM N. Scandium in aluminum alloys [J]. International Materials Reviews, 2005, 50(1): 19-44.
J, RYUM N. Scandium in aluminum alloys [J]. International Materials Reviews, 2005, 50(1): 19-44.
[14] KNIPLING K E, SEIDMAN D N, DUNAND D C. Ambient and high-temperature mechanical properties of isochronally aged Al-0.06Sc, Al-0.06Zr and Al-0.06Sc-0.06Zr (at.%) alloys [J]. Acta Materialia, 2011, 59(3): 943-954.
[15] TOLLEY A, RADMILOVIC V, DAHMEN U. Segregation in Al3(Sc,Zr) precipitates in Al-Sc-Zr alloys [J]. Scripta Materialia, 2005, 52(7): 621-625.
[16] ZHOU Wei-wei, CAI Bin, LI Jin-wen, LIU Zhi-xia, YANG Sheng. Heat-resistant Al-0.2Sc-0.04Zr electrical conductor [J]. Materials Science and Engineering A, 2012, 552: 353-358.
[17] YUAN Sheng-ping, PU Xiong, ZHANG Guo-jun, LIU Gang, WANG Rui-hong, SUN Jun, CHEN Kang-hua. Effects of multiple precipitates on electrical conductivity of aged Al-Mg-Si alloys [J]. The Chinese Journal of Nonferrous Metals, 2010, 20(11): 2070-2074. (in Chinese)
[18] FULLER C B, MURRAY J L, SEIDMAN D N. Temporal evolution of the nanostructure of Al (Sc,Zr) alloys: Part I―Chemical compositions of Al3(Sc1-xZrx) precipitates [J]. Acta materialia, 2005, 53(20): 5401-5413.
[19] PRUKKANON W, CHANPUM S, LIMMANEEVICHITI C. Effect of Sc on precipitation hardening of AlSi6Mg alloy [J]. Journal of Materials Science and Technology, 2008, 24(1): 17-20.
[20] LI Yi-yun, WANG Wen-Hsiung, Hsu Yung-Fu, Shan Trong. High-temperature tensile behavior and microstructural evolution of cold-rolled Al-6Mg-0.4Sc-0.13Zr alloy [J]. Materials Science and Engineering A, 2008, 497(1-2): 10-17.
钞润泽1,管西花2,管仁国1,铁 镝1,连 超1,王 祥1,张 俭1
1. 东北大学 材料与冶金学院,沈阳 110819;2. 沈阳建筑大学 交通与机械工程学院,沈阳 110168
摘 要:为了获得既有良好力学性能又有较高导电性能的铝导线,采用连续流变挤压技术制备出直径为9.5 mm的Al-0.16Zr、Al-0.16Sc、Al-0.12Sc-0.04Zr(质量分数,%)和高纯铝(99.996%)4种铝导线,随后对3种铝合金导线进行热处理,并进行分析测试。结果表明:向高纯铝中单独添加0.16% Sc和0.16% Zr可以提高铝导线的抗拉强度,降低铝导线的导电性能。在高纯铝中添加0.12%Sc和0.04%Zr的合金也具有相同的规律。经过热处理,Al-0.12Sc-0.04Zr铝合金导线可以达到抗拉强度和导电率的最优综合性能:抗拉强度为160 MPa,导电率为64.03% (IACS)。
关键词:合金成分;铝导线;力学性能;导电率;连续流变挤压
(Edited by Chao WANG)
Foundation item: Project (51222405) supported by the National Natural Science Foundation for Outstanding Young Scholars of China; Project (51034002) supported by the National Natural Science Foundation of China; Project (120502001) supported by the Fundamental Research Funds for the Central Universities of China
Corresponding author: Ren-guo GUAN; Tel: +86-24-83681463; E-mail: guanrg@smm.neu.edu.cn
DOI: 10.1016/S1003-6326(14)63456-7

