�ȴ���������̬Mg5Gd�Ͻ�ʴ��Ϊ��Ӱ��
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2021���4��
�������ߣ��ܸ��� ��� ��ɿ� �ι���
����ҳ�룺939 - 951
�ؼ��ʣ�þ�Ͻ�ϡ������ʴ�ԣ�����֯���ȴ���
Key words��magnesium alloy; rare earth; corrosion resistance; microstructure; heat treatment
ժ Ҫ��ͨ���������顢�绯ѧ���ԡ�ɨ��绯ѧ������ʴ��ò�������ֶ��о��ȴ������ն�����̬Mg5Gd�Ͻ���3.5 wt.% NaCl����Mg(OH)2��Һ�и�ʴ��Ϊ��Ӱ�켰���������ڴﵽ���þ�Ͻ���ʴ�Ե�Ŀ�ġ�������������ܴ�������������Mg5Gd�Ͻ�ĸ�ʴ���ʣ�����ʹ�丯ʴ����ȣ���ʴ�ӱ�dz������Ҫ�����ڹ��ܴ��������۽�þ�����д����ĸ�Gd��������������ܴ�������˻����ܽ�һ������Mg5Gd�Ͻ�ĸ�ʴ�ٶȡ����˻�����У���þ�����г�������������δ������Ե���ż��ʴ������Ҫ����Ϊ������������������ɵı����Բ���Ĥ��ȫ�����˿������Ӷ���ֹ��ż��ʴ�IJ�����
Abstract: The influence of heat treatment on the corrosion behavior of rolled Mg5Gd alloys in 3.5 wt.% NaCl solution saturated with Mg(OH)2 was characterized by immersion test, electrochemical test, scanning electrochemical microscopy (SECM) and corrosion morphology analysis in order to improve the corrosion resistance of Mg alloys. The results showed that solution treatment reduced the corrosion rate of the Mg5Gd significantly, resulting in relatively uniform corrosion and shallow corrosion cavities due to the dissolution of Cd-containing particles. The following aging process could further decrease the corrosion rate. Precipitation of nano-sized Cd-containing particles did not cause apparent micro-galvanic corrosion, which could be attributed to the formation of a protective corrosion product film fully covering the particles.
Trans. Nonferrous Met. Soc. China 31(2021) 939-951
Fu-yong Cao1, Jing Zhang1, Ke-Ke Li1, Guang-Ling Song1,2,3
1. Center for Marine Materials Corrosion and Protection, College of Materials, Xiamen University, Xiamen 361005, China;
2. State Key Laboratory of Physical Chemistry of Solid Surfaces, College of Chemistry and Chemical Engineering, Xiamen University, Xiamen 361005, China;
3. School of Engineering, The University of Queensland, St. Lucia, Qld 4072, Australia
Received 12 April 2020; accepted 28 December 2020
Abstract: The influence of heat treatment on the corrosion behavior of rolled Mg5Gd alloys in 3.5 wt.% NaCl solution saturated with Mg(OH)2 was characterized by immersion test, electrochemical test, scanning electrochemical microscopy (SECM) and corrosion morphology analysis in order to improve the corrosion resistance of Mg alloys. The results showed that solution treatment reduced the corrosion rate of the Mg5Gd significantly, resulting in relatively uniform corrosion and shallow corrosion cavities due to the dissolution of Cd-containing particles. The following aging process could further decrease the corrosion rate. Precipitation of nano-sized Cd-containing particles did not cause apparent micro-galvanic corrosion, which could be attributed to the formation of a protective corrosion product film fully covering the particles.
Key words: magnesium alloy; rare earth; corrosion resistance; microstructure; heat treatment
1 Introduction
Mg alloys have been widely applied in various fields such as automobile, aerospace and electronics for their high specific strength, favorable casting capability and ease of recycling [1-5]. However, the bad mechanical properties and poor corrosion resistance are still the main drawbacks that limit the widespread applications of Mg alloys [6,7].
Alloying is a useful technology to improve both the mechanical properties and corrosion resistance of Mg alloy [8-10]. In recent years, Mg-Gd based alloys have attracted much attention due to their excellent tensile strength [11], superior creep resistance [12], and relatively good corrosion resistance [13]. There are substantial reports that the mechanical properties could be improved significantly by the addition of Gd with the formation of a long period stacking ordered (LPSO) second phase(s) [14-16]. Generally, the LPSO structure is mainly obtained through heat treatment. YAMASAKI et al [17] indicated that heat treatment produced a 14H-type LPSO structure that was absent in the as-cast Mg-Zn-Gd alloy. This is consistent with the (Mg, Zn)3Gd eutectic phase in the as-cast Mg-Gd-Zn-Zr alloys transformed into an X phase with a 14H-LPSO structure after T4 heat treatment [18]. However, the modification of the microstructure during heat treatment is really challenging for the corrosion resistance of Mg alloys as the precipitation of second phases or impurities is a vital factor in the formation of micro-galvanic corrosion [19,20]. Therefore, some researchers [2,3,18] have investigated the influence of heat treatment on the corrosion behavior of Mg-Gd based alloys to clarify the mechanism of the corrosion of Mg-Gd alloy.
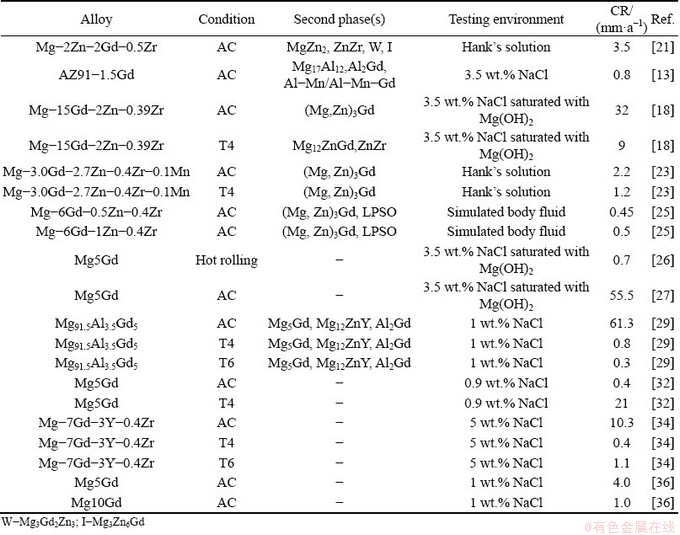
Table 1 summarizes some typical studies on the corrosion behavior of Mg-Gd based alloys in recent years [13,18,21-37]. The following key points could be concluded from the previous research.
(1) The added Gd into Mg alloys could react with the alloying elements to form LPSO second phases, such as Al-Mn-Gd, Al2Gd and (Mg2Zn)3Gd, which could modify the microstructure significantly.
(2) The addition of Gd could indeed improve the corrosion resistance of Mg alloy. However, this benefit is severely related to the amount of Gd and microstructure of Mg alloy [13,21,24,32,36]. Excessive addition of Gd may result in a corrosion rate even higher than that of the parent Mg alloy [21,24,36].
(3) The heat treatment or hot rolling has a substantial influence on the corrosion behavior of Mg-Gd based alloys, which is attributed to the dissolution or transformation of the second phases [18,22,23,25,29]. However, there is a discrepancy of different results. Many researchers [18,22,23,29] found that T4 treatment can reduce the corrosion rate significantly, while ZHANG et al [25] indicated that T4 treatment increased the corrosion rate of the Mg-Gd-Zn-Zr alloys. Furthermore, WANG et al [22] and LU et al [29] found that T6 treatment could further decrease the corrosion rate, whereas LIANG et al [34] indicated that the corrosion rate of Mg-7Gd-3Y-0.4Zr increased from 0.4 mm/a (T4) to 1.1 mm/a (T6).
In summary, the earlier studies on the corrosion behavior of Mg-Gd based alloys have made great contribution to the understanding of Gd effect on Mg corrosion. However, its mechanism still remains equivocal. In addition, many researchers added Gd into the Mg alloys containing the alloying elements such as Al or Zn which complicated the understanding of Gd effect [13,21,23,25,29].
Table 1 Corrosion rates (CR) for Gd-containing Mg alloys (AC: as-cast; T4: solution treated; T6: solution treatment + aging)
Therefore, in this study, we try to understand the influence of Gd addition on the corrosion of pure Mg. And then, the analysis of the mechanism of heat treatment on the corrosion of Mg-Gd was carried out systematically and extensively. Finally, it is expected to explore some insights into the improvement of the corrosion resistance of Mg-Gd alloy which may facilitate the application of Mg-Gd alloy in bio-degradation materials and automobile industry.
2 Experimental
2.1 Materials and specimens
The hot-rolled Mg5Gd alloy was prepared as described previously [26,38]. High-purity Mg (99.9 wt.%) was melted in an aluminum titanate crucible with addition of some commercially pure Gd from Alfa Aesar Company Ltd. A small amount of Zr was added to purify the melt. Then, the ingots were hot-rolled to 3 mm-thick belts. The chemical composition of the Mg5Gd was determined by iCAP 7000 Series optical emission spectroscopy (ICP-OES) from Thermo Scientific Company as follows (wt.%): 4.9 Gd, 0.07 Si, 0.04 Mn, 0.04 Zr, 0.03 Al, 0.02 Fe, 0.02 Ca, and Mg balance.
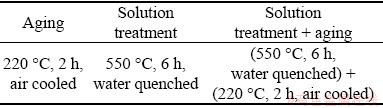
Table 2 presents the details of the heat treatment for the Mg5Gd specimens after hot rolling. The heat treatment was carried out in an electromagnetic induction furnace from Ningbo Shenguang Electric Furnace Company Limited with the flowing argon. The microstructure of the rolled Mg5Gd before and after heat treatment was characterized using Leica DVM6 optical microscope (OM), Hitachi TM3000 scanning electron microscope (SEM) equipped with energy dispersive spectroscopy (EDS) and X-ray diffraction (XRD) with Cu K�� radiation on a Rigaku D/max 2550 X-ray diffractometer. The angle range was 5�� to 90��. Each specimen for microstructure analysis was ground with a 5 vol.% HNO3 solution.
2.2 Immersion test and electrochemical measurement
Each Mg5Gd specimen was immersed in a 3.5 wt.% NaCl solution saturated with Mg(OH)2. Reagent grade chemicals and distilled water were used. The temperature was (24��1) ��C and the immersion test duration was 3 d. The setup of the immersion test was the same as described in our previous research [20]. The hydrogen evolved during the test was collected into a burette from Tianbo Company Limited by a funnel over the specimen. After the immersion test, the specimen was immersed in a 200 g/L CrO3 + 2 g/L AgNO3 solution to remove the corrosion products.
Table 2 Heat treatment process for hot-rolled Mg5Gd specimens
The average corrosion rate, PAH (mm/a), was calculated from the hydrogen evolution volume, VH (mL/cm2), by [26]
PAH=2.088VH/t (1)
where t is the immersion time. After immersion test, each specimen was characterized using OM and SEM equipped with EDS.
A Gamry Reference 600+ electrochemical workstation and a typical three-electrode electrolytic cell were used to carry out the electrochemical measurements. The counter electrode was a Pt foil of 1.5 cm �� 1.5 cm, and the reference electrode was Ag/AgCl/Sat. KCl. The electrolyte was the 3.5 wt.% NaCl solution saturated with Mg(OH)2. Electrochemical impedance spectroscopy (EIS) was measured daily in the frequency range from 100 kHz to 10 mHz with an AC amplitude of 5 mV. The polarization resistance (Rp) was obtained as the resistance at the lowest frequency, which did not include the solution resistance (Rs). The polarization curves were measured from -300 mV to 500 mV at a scan rate of 1 mV/s after 3 d immersion in the solution. The Tafel extrapolation method was used to evaluate the corrosion current density, Jcorr (mA/cm2), using the software of CorrView-3.0 (by Scribe Associates, Inc., USA).
2.3 Scanning electrochemical microscopy (SECM) test
SECM test was conducted using a M470 from Bio Logic Science Instruments in a three-electrode system with a Pt probe of 10 ��m in diameter as the working electrode, a Ag/AgCl/3 mol/L KCl as the reference electrode and a Pt foil as the counter electrode. Some ferrocene-methanol (FcMeOH) was added into the test solution as redox mediator in the feedback mode [39]. The tip of the Pt probe was biased at 0.5 V (vs Ag/AgCl/3 mol/L KCl) to ensure the complete diffusion-limited oxidation of ferrocene to ferrocinium [40]. The distance between the probe and the Mg surface was set at ~30 ��m and the scanning area was set as 1000 ��m �� 1000 ��m with a scan rate of 25 ��m/s. The normalized current ratio of IT/IT,�� (tip current/steady state current) was adopted in the current study when IT,�� was obtained from the corresponding approaching curve.
3 Results
3.1 Microstructural characterization
Figure 1 presents the microstructure of the each Mg5Gd specimen after etching. Figures 1(a) and (b) show the OM and SEM images of the rolled Mg5Gd. They show that the grain size is about 20 ��m with some twin grains and Mg-Gd phase particles identified by EDS. Figures 1(c) and (d) show the microstructure of the aged Mg5Gd specimen which has more Mg-Gd phase particles. The grain size is about 20 ��m and the particles still distribute in the matrix. Figures 1(e) and (f) show the microstructure of the solution- treated Mg5Gd which has larger grains and less Mg-Gd phase particles than that of rolled and aged Mg5Gd. There are also no twin grains observed in the matrix. Figures 1(g) and (h) show the micro- structure of the solution-treated + aged Mg5Gd. It has large grains and some nano-sized Mg-Gd phase particles without twin grain in the matrix.
Table 3 presents the compositions of the matrix and the typical particles of each Mg5Gd specimen analyzed by EDS. It shows that the content of Gd dissolved in the Mg matrix increased after solution treatment (ST). The particles in each specimen contained large amount of Gd and Mg, and they were identified as Mg5Gd intermetallic. It was noted that the Gd content of the particles in solution-treated and solution-treated + aged (ST+A) specimen was lower than that in rolled (R) and aged (A) specimens, which was attributed to the small size of the particles after solution treatment and the Mg matrix contributed to the signals when the particles were analyzed by EDS.
Figure 2 shows the XRD patterns for the Mg5Gd specimen, indicating that only Mg peaks were detected. The Gd-containing particles were not detected, which was attributed to the low amount.

Fig. 1 Typical surface appearance of Mg5Gd specimens as revealed by OM (a, c, e, g) and SEM (b, d, f, h)
Table 3 Compositions of matrix and typical particles of each Mg5Gd specimen analyzed by EDS (wt.%)
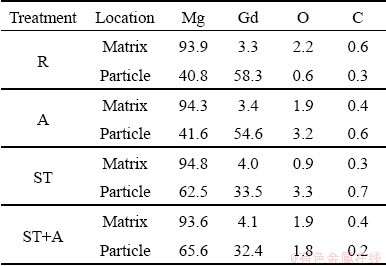
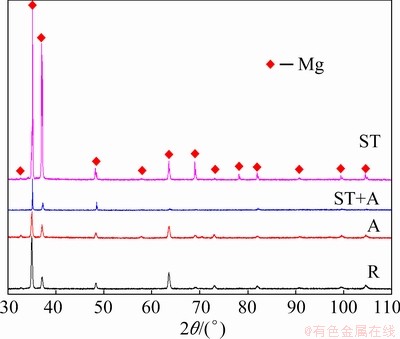
Fig. 2 XRD patterns for Mg5Gd specimens after rolling (R), aging (A), solution-treatment (ST) and solution-treatment+aging (ST+A)
3.2 Immersion test and electrochemical results
Figure 3 shows the hydrogen evolution volume for each Mg5Gd specimen immersed for 3 d at the open circuit potential (OCP) in the 3.5 wt.% NaCl solution saturated with Mg(OH)2. The aged Mg5Gd specimen evolved most hydrogen during the immersion test, followed by the rolled Mg5Gd specimen. The evolved hydrogen volumes of the solution-treated and (solution-treated + aged) Mg5Gd specimens were substantially lower than that of the aged Mg5Gd specimen. For aged and rolled Mg5Gd specimens, the hydrogen evolution rate increased apparently after the 1st day, while it kept almost constant during the whole immersion test for solution-treated and solution-treated + aged Mg5Gd specimens. Table 4 presents the average corrosion rate, PAH, from the evolved hydrogen volume for each Mg5Gd specimen, indicating the same sequence of the corrosion resistance for Mg5Gd specimen.

Fig. 3 Hydrogen evolution volume during immersion test at open circuit potential (OCP) in 3.5 wt.% NaCl solution saturated with Mg(OH)2 for 3 d at (24��1) ��C for different specimens
Table 4 Corrosion data evaluated for Mg5Gd specimens immersed at open circuit potential (OCP) in 3.5 wt.% NaCl solution saturated with Mg(OH)2 at (24��1) ��C for 3 d
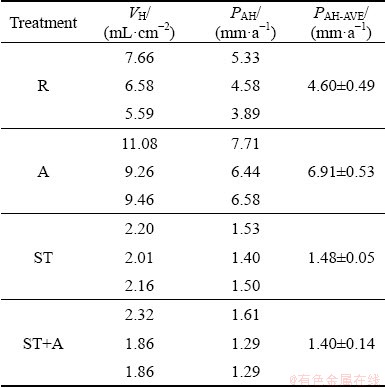
Figure 4(a) presents the Nyquist plots for rolled Mg5Gd specimen immersed for 3 d in 3.5 wt.% NaCl solution saturated with Mg(OH)2. It shows that the capacitance loop shrinks apparently with increase of the immersion time. There is an additional inductive loop observed at low frequencies on the Nyquist plots of 1, 2 and 3 d.
Figure 4(b) presents the Nyquist plots for aged Mg5Gd specimen, which are similar with those of rolled Mg5Gd specimen. The capacitance loop shrank apparently after 1 d immersion and an additional inductive loop at low frequencies was also observed.
Figure 4(c) presents the Nyquist plots for solution-treated Mg5Gd specimen immersed for 3 d in 3.5 wt.% NaCl solution saturated with Mg(OH)2. The capacitance loop shrank apparently with the immersion time. There was only one inductive loop at low frequencies and no additional one was observed for all the solution-treated Mg5Gd plots.
Figure 4(d) presents the Nyquist plots for solution-treated + aged Mg5Gd specimen. The capacitance loops of 1 d and 2 d were obviously smaller than those of 1 h and 6 h. There was an additional capacitance loop appeared on the 3 d Nyquist plot. However, there was no additional inductive loop observed at low frequencies which was similar with those of solution-treated Mg5Gd.
Table 5 presents the Rp values extracted from the fitting of the EIS data which statistically reveal the variation of EIS for each Mg5Gd specimen.
Figure 5 presents the polarization curves measured at the 3rd day during immersion test in 3.5 wt.% NaCl solution saturated with Mg(OH)2 at (24��1) ��C for Mg5Gd specimens. It shows that there was no significant difference of the OCP and anodic branch among the rolled, aged and solution- treated + aged Mg5Gd specimens. However, the OCP of solution-treated Mg5Gd was 300 mV and the anodic polarization curve was apparently blocked. There was significant difference of the cathodic polarization curves for each Mg5Gd specimen that the cathodic curve of rolled Mg5Gd was activated while the solution-treated + aged one was blocked. Table 5 shows that the corrosion current density extracted from the polarization curve reveals the same corrosion resistance sequence with that from evolved hydrogen.
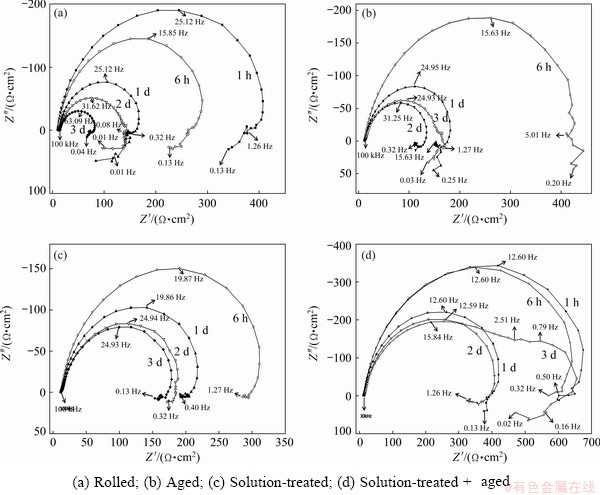
Fig. 4 Typical EIS data measured during immersion tested at OCP in 3.5 wt.% NaCl solution saturated with Mg(OH)2 for 3 d at (24��1) ��C for Mg5Gd specimens
Table 5 Electrochemical data evaluated by EIS fitting and Tafel extrapolation for Mg5Gd specimens immersed at OCP in 3.5 wt.% NaCl solution saturated with Mg(OH)2 at (24��1) ��C for 3 d
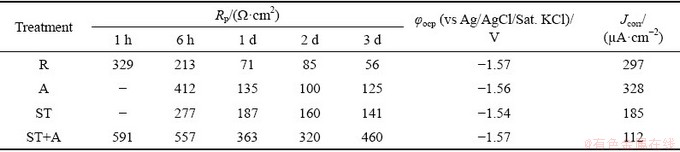
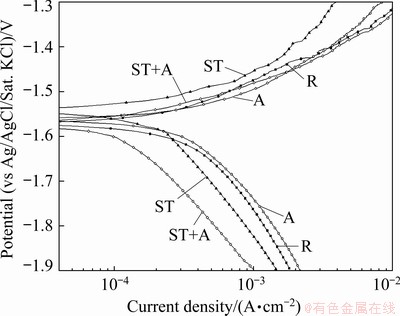
Fig. 5 Polarization curves measured at the 3rd day of immersion test in 3.5 wt.% NaCl solution saturated with Mg(OH)2 at (24��1) ��C for Mg5Gd specimens
3.3 Corrosion morphology
Figure 6 presents the typical corroded surface appearance for each Mg5Gd specimen after the removal of corrosion products including an optical overview image, a 3D optical image of the typical corrosion cavity and the SEM images for typical deeply corroded areas and typical shallow corroded areas. The scales of the figures are different to best present the most important corroded surface features.
Figures 6(a)-(d) present the surface appearance after immersion test at OCP in 3.5 wt.% NaCl solution saturated with Mg(OH)2 at (24��1) ��C for 3 d after removal of corrosion products for rolled Mg5Gd. Figure 6(a) shows that most area of the specimen suffered serious corrosion. Of the whole surface there were several larger cavities penetrated more than 300 ��m into the matrix as shown in Fig. 6(b). The rest area suffered slighter corrosion with shallower pits as shown in Figs. 6(c) and (d) compared with the larger cavities.
Figures 6(e)-(h) present the surface appearance of aged Mg5Gd specimen after immersion test, which is similar with that of rolled Mg5Gd specimen. Most parts suffered serious corrosion (Fig. 6(e)) with several large cavities (Fig. 6(f)) in the matrix. The rest parts still suffered apparent corrosion as shown in Figs. 6(g) and (h).
Figures 6(i)-(l) present the surface appearance of solution-treated Mg5Gd after immersion test. Figure 6(i) shows the whole specimen suffered slight corrosion with some small cavities in the matrix as shown in Fig. 6(j). The depth was less than 100 ��m which was significantly lower than those of rolled and aged Mg5Gd. Figures 6(k) and (l) show the localized corroded area and the slight corroded area of the solution-treated Mg5Gd.
Figures 6(m)-(p) show the surface appearance of solution treated + aged Mg5Gd specimen after the immersion test with the corrosion products removed. Figure 6(m) shows that it suffered the slightest corrosion compared with other Mg5Gd specimens. The typical corrosion cavity is small as shown in Fig. 6(n). Figures 6(o) and (p) show the relatively serious corroded area and slight corroded area.
The corrosion products of each Mg5Gd specimen were analyzed by EDS, and the main constituent is Mg(OH)2 containing Gd element. There is no significant difference of the constituents for each Mg5Gd specimen.
3.4 SECM results
Figure 7 shows the SECM images of each Mg5Gd specimen of a scanning tip at 25 ��m/s in 1.0 mmol/L FcMeOH + 0.1 mol/L NaCl. Figures 7(a) and (b) show that the normalized current ratio was more than 1, corresponding to active points for the rolled and aged specimen immersed in 0.1 mol/L NaCl. However, for the solution-treated and solution-treated + aged Mg5Gd specimens, no active points (corresponding to high normalized current ratio more than 1) were detected in the scanned area as shown in Figs. 7(c) and (d).

Fig. 6 Surface appearance after immersion test at open circuit potential (OCP) in 3.5 wt.% NaCl solution saturated with Mg(OH)2 at (24��1) ��C for 3 d and after removal of corrosion products for rolled (a-d), aged (e-h), solution-treated (i-l), and solution-treated + aged (m-p) Mg5Gd specimens
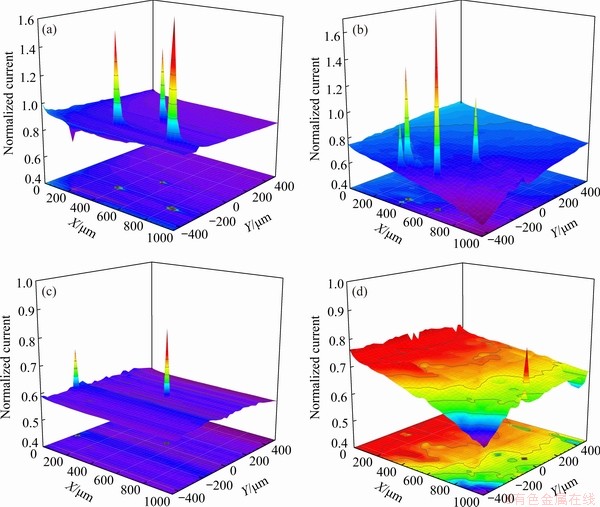
Fig. 7 SECM images of specimens immersed in 1.0 mmol/L FcMeOH + 0.1 mol/L NaCl for rolled (a), aged (b), solution-treated (c) and solution-treated + aged (d) Mg5Gd specimens (Tip-substrate distance: 30 ��m; Scan rate: 25 ��m/s; Tip potential: 0.5 V (vs Ag/AgCl/3 mol/L KCl))
4 Discussion
4.1 Influence of heat treatment
Figure 3 and Table 4 show that the corrosion resistances of the Mg5Gd alloys were ordered as follows: ST+A>ST >>R>A. This trend was totally consistent with that indicated by the polarization curves (Fig. 5 and Table 5) and the corroded morphologies (Fig. 6). This indicates that the aging treatment for the rolled Mg5Gd specimen does not reduce but increases the corrosion rate. This is mainly attributed to the precipitation of the Gd-containing particles or impurities in Mg matrix. The relatively short aging time and low temperature led to the precipitation of a small amount of Gd-containing particles or impurities as shown in Fig. 1. Such a small amount precipitation of the second phases could increase the corrosion rate significantly due to the galvanic corrosion [19].
The evolved hydrogen volume and the current density from the polarization curves strongly indicate that the solution treatment improves the corrosion resistance of the Mg5Gd specimen substantially. The grain size of the rolled Mg5Gd was obviously increased and the twin grains totally disappeared in the matrix. Some Gd-containing second phase was dissolved into the matrix during the matrix, as shown in Fig. 1. However, there was no phase transformation occurred during the solution treatment, as shown in Fig. 2. Figures 6(i)- (p) show that the low corrosion rate of Mg5Gd after solution treatment resulted from uniform corrosion and shallow cavities compared with that of Mg5Gd before solution treatment as shown in Figs. 6(a)-(h). Our previous research [41] indicated that specimens with casting porosity had high corrosion rates because of the corrosion associated with pores activating significant corrosion over the whole specimen surface. This feature can be identified from: (1) the corroded morphology with specific deep cavities, and (2) two inductive loops at low frequencies (e.g. a semi-circle inductive loop plus a quarter-circle inductive loop) [41]. In the current research, the rolled and aged Mg5Gd specimens had deep cavities. Meanwhile, double inductive loops were observed at low frequencies of EIS as shown in Figs. 4(a) and (b). Therefore, it is reasonable to propose that the deep cavities activated the corrosion of the whole Mg5Gd specimen during the immersion test for the rolled and aged Mg5Gd. The reduction or transformation of the deep corrosion cavities should be the main reason for the low corrosion rate of solution-treated and solution-treated + aged Mg5Gd, which is evidenced by the corroded morphology in Figs. 6(a)-(h) and the absence of double inductive loops in Figs. 4(c, d). It is noted in Ref. [41] that the activation corrosion of casting porosity is attributed to the damage of the somewhat protective corrosion product film formed on the specimen surface as: (1) the size and surface area of the porosity were increased by the corrosion; (2) the corrosion broke the integrity and compactness of the film over all of the surface, which led to significant corrosion adjacent to the porosity and also to significant corrosion over the whole specimen area. In the current research, the rolled and aged Mg5Gd specimens were as the same case as that of Mg0.1Si with casting porosity. The deep corrosion cavities on the Mg5Gd specimen increased the corrosion of the whole surface. However, the rolled and aged Mg5Gd had no initial casting porosity. Therefore, it is important to find out what caused the deep corrosion cavity in rolled and aged Mg5Gd.
Figure 1 shows that after solution treatment the amount of Gd-containing second phase decreased significantly, which dissolved into the Mg matrix. As a result, the content of Gd in the matrix increased a bit as shown in Table 3. The Gd-containing second phase has a higher potential than the matrix, which could induce serious micro-galvanic corrosion. This is considered as the main cause of the deep corrosion cavity in rolled and aged Mg5Gd specimens. After solution treatment, the deep corrosion cavities were absent as the large cathodic particles dissolved into the matrix, only left few and fine particles in the Mg matrix. As a result, the strong micro-galvanic corrosion was minimized and the corrosion resistance of Mg5Gd was increased substantially. This is evidenced by the corrosion morphology in Fig. 6. Furthermore, Gd in the Mg matrix could incorporate into the surface hydroxide layer to stabilize the degradation products and inhibit the penetration of harmful Cl- [42]. This is also consistent with the SECM results that the rolled and aged Mg5Gd specimens have active corrosion points which probably grow into the corroded cavities. However, there is no active point observed in the solution-treated and solution-treated + aged Mg5Gd specimens.
It is noted that the solution treatment increased the grain size of Mg5Gd, and may also cause the precipitation of impurities. They are detrimental to the corrosion resistance of Mg alloy [19,26,43]. However, the negative effects have been overwhelmed by the benefits of solution treatment. As a result, the overall influence of solution treatment is the improvement of the corrosion resistance of Mg5Gd. The aging treatment after solution treatment led to precipitation of some nano-sized Mg-Gd phase particles in the matrix as shown in Fig. 1. The immersion test results in Fig. 3 and corroded morphologies in Fig. 6 indicated that these particles did not cause apparent micro- galvanic corrosion. This was probably attributed to the fact that a thin but protective Gd-containing product film was formed on the nano-sized particles soon after the specimen was exposed in air. This film prevented the initiation and propagation of the micro-galvanic corrosion between the nano-sized particles and the Mg matrix. However, in the aged Mg5Gd specimens, due to the large size of the Mg-Gd phase particles, it was difficult to form such a protective product film on the whole particle. As a result, serious micro-galvanic corrosion between the Mg-Gd phase particles and the Mg matrix occurred. Furthermore, the aging process after solution treatment was beneficial to the further homogenization of the microstructure and the formation of the corrosion product film, which could be the reason that the corrosion rate of the solution-treated + aged Mg5Gd was slightly lower than that of the solution-treated Mg5Gd. This is also consistent with that the solution-treated + aged Mg5Gd shows the lowest activity in the polarization curves in Fig. 5. The above analysis might also interpret the results in Ref. [31] that the presence of nano-sized phase particles in Mg-10Gd did not lead to an increased corrosion rate.
4.2 Improvement of corrosion resistance
As Mg-Gd based alloy has superficial mechanical properties and acceptable corrosion resistance, it has attracted much attention and investigation in automobile industry and biodegradable materials area as summarized in Table 1. We have studied the Mg5Gd alloy for several years and try to improve the corrosion resistance through various attempts as shown in Fig. 8 [26-28]. The corrosion rate of the as-cast Mg5Gd was 55.5 mm/a in 3.5 wt.% NaCl solution saturated with Mg(OH)2, which was caused by the substantial Gd-containing second phases distributed along the grain boundaries [27]. It decreased to 30.0 mm/a after solution treatment as the heat treatment did not dissolve all the second phases in the matrix [27]. The corrosion resistance of solution- treated Mg5Gd was not satisfied. Then we tried hot rolling and sputter deposition methods to improve the corrosion resistance of as-cast Mg5Gd [26,28]. Both of the corrosion rates of Mg5Gd decreased dramatically to 0.66 mm/a. The superior corrosion resistance of sputtered Mg5Gd was mainly attributed to the single phase of the alloy and probably the low content of impurities [28]. While the improvement of the corrosion resistance of Mg5Gd by hot rolling was attributed to the fact that the alloy had a more homogeneous microstructure, and few, fine second-particles [26]. The corrosion resistance of Mg5Gd after hot rolling is significantly related to the temperature and processes during hot rolling, which is the main reason that the current hot rolling specimen has a different corrosion rate with that in Ref. [26]. In the current research, the rolled Mg5Gd had some Gd-containing particles in the matrix, which caused micro-galvanic corrosion, resulting in deep corroded cavities. These deep corroded cavities activated the corrosion over the whole specimen surface during the immersion test. The solution treatment dissolved most second phase particles into the Mg matrix and reduced the corrosion rate significantly. The aging process after solution treatment precipitated some nano-sized particles, which did not cause obvious micro-galvanic corrosion but blocked the corrosion instead. It is noted that all the above researches used the same Mg5Gd ingot produced in The University of Queensland [38]. Various corrosion rates of Mg5Gd were mainly attributed to the second phase particles, especially the size and amount of Gd-containing particles, and the microstructure. This strongly indicated that a homogenous microstructure with fine Gd-containing particles was beneficial to the improvement of corrosion resistance of Mg5Gd.
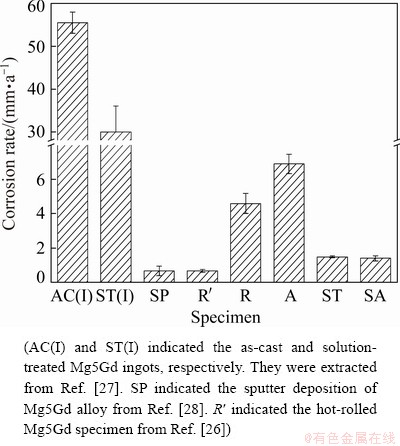
Fig. 8 Comparison of corrosion rate of Mg5Gd immersed in 3.5 wt.% NaCl solution saturated with Mg(OH)2
5 Conclusions
(1) The corrosion resistance of Mg5Gd decreases in the following order: solution treatment + aging > solution treatment >> rolling > aging.
(2) Solution treatment leads to significantly reduced corrosion rate and much more uniform corrosion damage of Mg5Gd, because the Gd-containing particles that can cause serious micro-galvanic corrosion are dissolved during the treatment.
(3) The rolled and aged Mg5Gd specimens have deep corrosion cavities and active surfaces due to the micro-galvanic effect induced by the Gd-containing particles.
(4) The aging process after solution treatment can further decrease the corrosion rate. The precipitation of nano-sized Mg5Gd particles did not cause apparent micro-galvanic corrosion, which was attributed to the formation of a protective product film fully covering the particles.
(5) The corrosion resistance of Mg5Gd can be related to the second phase particles, especially the size and amount of the Gd-containing particles, and the microstructure. A homogeneous microstructure with fine Gd-containing particles is beneficial to the improvement of corrosion resistance of Mg5Gd.
Acknowledgments
The authors are grateful for the financial supports from the National Natural Science Foundation of China (51801168, 51731008), Natural Science Foundation of Fujian Province (2018J05093), China, and National Environmental Corrosion Platform of China
References
[1] ABBOTT T B. Magnesium: Industrial and research developments over the last 15 years [J]. Corrosion, 2015, 71: 120-127.
[2] Atrens A, Song Guang-ling, CAO Fu-yong, SHI Zhi-ming, Bowen P K. Advances in Mg corrosion and research suggestions [J]. Journal of Magnesium and Alloys, 2013, 1: 177-200.
[3] Esmaily M, Svensson J E, Fajardo S, Birbilis N, Frankel G S, Virtanen S, Arrabal R, Thomas S, Johansson L G. Fundamentals and advances in magnesium alloy corrosion [J]. Progress in Materials Science, 2017, 89: 92-193.
[4] CHENG Yuan-fen, DU Wen-bo, LIU Ke, FU Jun-jian, WANG Zhao-hui, LI Shu-bo, FU Jin-long. Mechanical properties and corrosion behaviors of Mg-4Zn-0.2Mn- 0.2Ca alloy after long term in vitro degradation [J]. Transactions of Nonferrous Metals Society of China, 2020, 30: 363-372.
[5] FENG Yan, LEI Ge, HE Yu-qing, WANG Ri-chu, WANG Xiao-feng. Discharge performance of Mg-Al-Pb-La anode for Mg-air battery [J]. Transactions of Nonferrous Metals Society of China, 2018, 28: 2274-2286.
[6] MA Li-na, YANG Yan, ZHOU Gang, REN Feng-juan, DENG Hong-ju, WEI Guo-bing, PENG Xiao-dong. Effect of rolling reduction and annealing process on microstructure and corrosion behavior of LZ91 alloy sheet [J]. Transactions of Nonferrous Metals Society of China, 2020, 30: 1816-1825.
[7] NEZAMDOUST S, SEIFZADEH D, HABIBI-YANGJEH A. Nanodiamond incorporated sol-gel coating for corrosion protection of magnesium alloy [J]. Transactions of Nonferrous Metals Society of China, 2020, 30: 1535-1549.
[8] AMIRENJAD M, RAJABI M, MOTAVALLI A. Effect of addition of Si on microstructure, mechanical properties, bio-corrosion and cytotoxicity of Mg-6Al-1Zn alloy [J]. Transactions of Nonferrous Metals Society of China, 2018: 28: 1755-1762.
[9] LI Chuan-qiang, TONG Zhi-pei, HE Yi-bin, HUANG Huai-pei, DONG Yong, ZHANG Peng. Comparison on corrosion resistance and surface film of pure Mg and Mg-14Li alloy [J]. Transactions of Nonferrous Metals Society of China, 2020, 30: 2413-2423.
[10] ZENGIN H, TUREN Y, AHLATCI H, SUN Y, KARAOGLANLI A C. Influence of Sn addition on microstructure and corrosion resistance of AS21 magnesium alloy [J]. Transactions of Nonferrous Metals Society of China, 2019, 29: 1413-1423.
[11] GAO Xiang, Nie Jian-feng. Enhanced precipitation- hardening in Mg�CGd alloys containing Ag and Zn [J]. Scripta Materialia, 2008, 58: 619-622.
[12] HE Shang-ming, ZENG Xiao-qing, PENG Li-ming, GAO Xiang, Nie Jian-feng, DING Weng-jiang. Precipitation in a Mg�C10Gd�C3Y�C0.4Zr (wt.%) alloy during isothermal ageing at 250 ��C [J]. Journal of Alloys and Compounds, 2006, 421: 309-313.
[13] YIN Zheng, CHEN Yang, YAN Hong, ZHOU Guo-hua, WU Xiao-quan, Hu Zhi. Effects of the second phases on corrosion resistance of AZ91-xGd alloys treated with ultrasonic vibration [J]. Journal of Alloys and Compounds, 2019, 783: 877-885.
[14] ZHANG Song, YUAN Guang-yin, LU Chen, DING Weng-jiang. The relationship between (Mg,Zn)3RE phase and 14H-LPSO phase in Mg�CGd�CY�CZn�CZr alloys solidified at different cooling rates [J]. Journal of Alloys and Compounds, 2011, 509: 3515-3521.
[15] PENG Qiu-ming, HOU Xiu-li, WANG Li-dong, WU Yao-ming, CAO Zhan-yi, WANG Li-ming. Microstructure and mechanical properties of high performance Mg�CGd based alloys [J]. Materials and Design, 2009, 30: 292-296.
[16] MORDIKE B L. Creep-resistant magnesium alloys [J]. Materials Science and Engineering A, 2002, 324: 103-112.
[17] YAMASAKI M, ANAN T, YOSHIMOTO S, KAWAMURA Y. Mechanical properties of warm-extruded Mg�CZn�CGd alloy with coherent 14H long periodic stacking ordered structure precipitate [J]. Scripta Materialia, 2005, 53: 799-803.
[18] LIU Jing, YANG Li-xin, ZHANG Chun-yan, ZHANG Bo, ZHANG Tao, LI Yang, WU Kai-ming, WANG Fu-hui. Role of the LPSO structure in the improvement of corrosion resistance of Mg-Gd-Zn-Zr alloys [J]. Journal of Alloys and Compounds, 2019, 782: 648-658.
[19] LIU Ming, Uggowitzer P J, Nagasekhar A V, Schmutz P, Easton M, Song Guang-ling, Atrens A. Calculated phase diagrams and the corrosion of die-cast Mg�CAl alloys [J]. Corrosion Science, 2009, 51: 602-619.
[20] Song Guang-ling, Atrens A. Understanding magnesium corrosion��A framework for improved alloy performance [J]. Advanced Engineering Materials, 2003, 5: 837-858.
[21] Chen Jun-xiu, Tan Li-li, Yu Xiao-ming, Yang Ke. Effect of minor content of Gd on the mechanical and degradable properties of as-cast Mg-2Zn-xGd-0.5Zr alloys [J]. Journal of Materials Science and Technology, 2019, 35: 503-511.
[22] Wang Jing-feng, Jiang Wei-yan, Ma Yao, Li Yang, Huang Song. Substantial corrosion resistance improvement in heat-treated Mg�CGd�CZn alloys with a long period stacking ordered structure [J]. Materials Chemistry and Physics, 2018, 203: 352-361.
[23] Gui Zhen-zhen, Kang Zhi-xin, Li Yuan-yuan. Mechanical and corrosion properties of Mg-Gd-Zn-Zr-Mn biodegradable alloy by hot extrusion [J]. Journal of Alloys and Compounds, 2016, 685: 222-230.
[24] Zhang Jin-yang, Xu Min, Teng Xin-ying, Zuo Min. Effect of Gd addition on microstructure and corrosion behaviors of Mg�CZn�CY alloy [J]. Journal of Magnesium and Alloys, 2016, 4: 319-325.
[25] Zhang Xiao-bo, Ba Zhi-xin, Wang Zhang-zhong, Xue Ya-jun. Microstructures and corrosion behavior of biodegradable Mg�C6Gd�CxZn�C0.4Zr alloys with and without long period stacking ordered structure [J]. Corrosion Science, 2016, 105: 68-77.
[26] Cao Fu-yong, Shi Zhi-ming, Song Guang-ling, Liu Ming, Dargusch M S, Atrens A. Influence of hot rolling on the corrosion behavior of several Mg�CX alloys [J]. Corrosion Science, 2015, 90: 176-191.
[27] Shi Zhi-ming, Cao Fu-yong, Song Guang-ling, Liu Ming, Atrens A. Corrosion behaviour in salt spray and in 3.5% NaCl solution saturated with Mg(OH)2 of as-cast and solution heat-treated binary Mg�CRE alloys: RE=Ce, La, Nd, Y, Gd [J]. Corrosion Science, 2013, 76: 98-118.
[28] Schl��ter K, Shi Zhi-ming, Zamponi C, Cao Fu-yong, Quandt E, Atrens A. Corrosion performance and mechanical properties of sputter-deposited MgY and MgGd alloys [J]. Corrosion Science, 2014, 78: 43-54.
[29] Lu Fu-min, Ma Ai-bin, Jiang Jing-hua, Guo Yu, Yang Dong-hui, Song Dan, Chen Jian-qing. Significantly improved corrosion resistance of heat-treated Mg�CAl�CGd alloy containing profuse needle-like precipitates within grains [J]. Corrosion Science, 2015, 94: 171-178.
[30] Shi Ling-ling, Huang yuan-ding, Yang lei, Feyerabend F, Mendis C, Willumeit R, Kainer K U, Hort N. Mechanical properties and corrosion behavior of Mg�CGd�CCa�CZr alloys for medical applications [J]. Journal of the Mechanical Behavior of Biomedical Materials, 2015, 47: 38-48.
[31] Srinivasan A, Blawert C, Huang Yuan-ding, Mendis C L, Kainer K U, Hort N. Corrosion behavior of Mg�CGd�CZn based alloys in aqueous NaCl solution [J]. Journal of Magnesium and Alloys, 2014, 2: 245-256.
[32] KubASek J, VojtECh D. Structural and corrosion characterization of biodegradable Mg�CRE (RE=Gd, Y, Nd) alloys [J]. Transactions of Nonferrous Metals Society of China, 2013, 23: 1215-1225.
[33] Arrabal R, Matykina E, Pardo A, Merino M C, Paucar K, Mohedano M, Casaj��s P. Corrosion behaviour of AZ91D and AM50 magnesium alloys with Nd and Gd additions in humid environments [J]. Corrosion Science, 2012, 55: 351-362.
[34] Liang Shu-quan, Guan Di-kai, Tan Xiao-ping. The relation between heat treatment and corrosion behavior of Mg�CGd�CY�CZr alloy [J]. Materials and Design, 2011, 32: 1194-1199.
[35] Liu xian-bin, Shan da-yong, Song ying-wei, Chen rong-shi, Han En-hou. Influence of casting module on corrosion behavior of Mg-11Gd-3Y alloy [J]. Transactions of Nonferrous Metals Society of China, 2011, 21: 903-911.
[36] Hort N, Huang yuan-ding, Fechner D, StOrmer M, Blawert C, Witte F, Vogt C, Dr��cker H, Willumeit R, Kainer K U, Feyerabend F. Magnesium alloys as implant materials-Principles of property design for Mg�CRE alloys [J]. Acta Biomaterialia, 2010, 6: 1714-1725.
[37] Chang jian-wei, Guo xing-wu, He shang-ming, Fu peng-huai, Peng li-ming, Ding wen-jiang. Investigation of the corrosion for Mg�CxGd�C3Y�C0.4Zr (x=6, 8, 10, 12 wt%) alloys in a peak-aged condition [J]. Corrosion Science, 2008, 50: 166-177.
[38] Prasad A, Uggowitzer P J, Shi zhi-ming, Atrens A. Production of high purity magnesium alloys by melt purification with Zr [J]. Advanced Engineering Materials, 2012, 14: 477-490.
[39] Liu wen-juan, Cao fa-he, Xia yan, Chang lin-rong, Zhang jian-qing. Localized corrosion of magnesium alloys in NaCl solutions explored by scanning electrochemical microscopy in feedback mode [J]. Electrochimica Acta, 2014, 132: 377-388.
[40] Tan Cen, Rodriguez-Lopez J, Parks J J, Ritzert N L, Ralph D C, Abruna H D. Reactivity of monolayer chemical vapor deposited graphene imperfections studied using scanning electrochemical microscopy [J]. ACS Nano, 2012, 6: 3070-3079.
[41] Cao fu-yong, Shi zhi-ming, Song Guang-ling, Liu Ming, Dargusch M S, Atrens A. Influence of casting porosity on the corrosion behaviour of Mg0.1Si [J]. Corrosion Science, 2015, 94: 255-269.
[42] KubAsek J, VojtEch D, Lipov J, Ruml T. Structure, mechanical properties, corrosion behavior and cytotoxicity of biodegradable Mg�CX (X=Sn, Ga, In) alloys [J]. Materials Science and Engineering C, 2013, 33: 2421-2432.
[43] Liu Ming, SONG Guang-ling. Impurity control and corrosion resistance of magnesium�Caluminum alloy [J]. Corrosion Science, 2013, 77: 143-150.
�ܸ���1���� �1����ɿ�1���ι���1,2,3
1. ���Ŵ�ѧ ����ѧԺ ������ϸ�ʴ��������ģ����� 361005��
2. ���Ŵ�ѧ ��ѧ����ѧԺ �������������ѧ�����ص�ʵ���ң����� 361005��
3. School of Engineering, The University of Queensland, St. Lucia, Qld 4072, Australia
ժ Ҫ��ͨ���������顢�绯ѧ���ԡ�ɨ��绯ѧ������ʴ��ò�������ֶ��о��ȴ������ն�����̬Mg5Gd�Ͻ���3.5 wt.% NaCl����Mg(OH)2��Һ�и�ʴ��Ϊ��Ӱ�켰���������ڴﵽ���þ�Ͻ���ʴ�Ե�Ŀ�ġ�������������ܴ�������������Mg5Gd�Ͻ�ĸ�ʴ���ʣ�����ʹ�丯ʴ����ȣ���ʴ�ӱ�dz������Ҫ�����ڹ��ܴ��������۽�þ�����д����ĸ�Gd��������������ܴ�������˻����ܽ�һ������Mg5Gd�Ͻ�ĸ�ʴ�ٶȡ����˻�����У���þ�����г�������������δ������Ե���ż��ʴ������Ҫ����Ϊ������������������ɵı����Բ���Ĥ��ȫ�����˿������Ӷ���ֹ��ż��ʴ�IJ�����
�ؼ��ʣ�þ�Ͻ�ϡ������ʴ�ԣ�����֯���ȴ���
(Edited by Xiang-qun LI)
Corresponding author: Guang-ling Song; +86-18359252602; E-mail: glsong@xmu.edu.cn, guangling.song@hotmail.com
DOI: 10.1016/S1003-6326(21)65551-6
1003-6326/ 2021 The Nonferrous Metals Society of China. Published by Elsevier Ltd & Science Press
2021 The Nonferrous Metals Society of China. Published by Elsevier Ltd & Science Press

