用甲酸铜预制块在空气中模压烧结形成全致密黏结层
来源期刊:中国有色金属学报(英文版)2021年第6期
论文作者:Yun-Ju LEE Jong-Hyun LEE
文章页码:1717 - 1728
关键词:结构材料;显微组织;力学性能;甲酸铜预制块;烧结连接; 热解
Key words:structural materials; microstructure; mechanical properties; copper formate preform; sinter bonding; pyrolysis
摘 要:使用由甲酸铜颗粒组成的预制块,在250~350 °C于空气中用压力辅助烧结法制备耐高温的黏结层。烧结过程中,甲酸铜在210 °C发生热解,同时生成H2 和CO,且去除铜块体表面的天然氧化物和其他生成的氧化物,形成界面结合。而且,原位还原Cu(II)生成的Cu加速烧结连接。在300~350 °C和5 MPa压力下烧结180~300 s后得到的黏结层具有足够的剪切强度:20.0~31.5 MPa。此外,在250 °C条件下,当压力增加到10 MPa时,在极短的时间(30 s)内,剪切强度达到21.9 MPa, 300 s后形成接近全致密的黏结层。因此,甲酸铜颗粒预制块在高速烧结连接中具有广阔的应用前景。
Abstract: Pressure-assisted sinter bonding was performed in air at 250-350 °C using a preform comprising copper formate particles to form a bondline that is sustainable at high temperatures. H2 and CO generated concurrently by the pyrolysis of copper formate at 210 °C during the sinter bonding removed the native oxide and other oxides grown on bulk Cu finishes, enabling interface bonding. Moreover, Cu produced in situ by the reduction of Cu(II) accelerated the sinter bonding. Consequently, the bonding achieved at 300-350 °C under 5 MPa exhibited sufficient shear strength of 20.0-31.5 MPa after 180-300 min of sinter bonding. In addition, an increase in pressure to 10 MPa resulted in shear strength of 21.9 MPa after an extremely short time of 30 s at 250 °C, and a near-full-density bondline was achieved after 300 s. The obtained results indicate the promising potential of the preform comprising copper formate particles for high-speed sinter bonding.
Trans. Nonferrous Met. Soc. China 31(2021) 1717-1728
Yun-Ju LEE, Jong-Hyun LEE
Department of Materials Science and Engineering, Seoul National University of Science and Technology, 232 Gongneung-ro, Nowon-gu, Seoul 139-743, Korea
Received 10 June 2020; accepted 4 February 2021
Abstract: Pressure-assisted sinter bonding was performed in air at 250-350 °C using a preform comprising copper formate particles to form a bondline that is sustainable at high temperatures. H2 and CO generated concurrently by the pyrolysis of copper formate at 210 °C during the sinter bonding removed the native oxide and other oxides grown on bulk Cu finishes, enabling interface bonding. Moreover, Cu produced in situ by the reduction of Cu(II) accelerated the sinter bonding. Consequently, the bonding achieved at 300-350 °C under 5 MPa exhibited sufficient shear strength of 20.0-31.5 MPa after 180-300 min of sinter bonding. In addition, an increase in pressure to 10 MPa resulted in shear strength of 21.9 MPa after an extremely short time of 30 s at 250 °C, and a near-full-density bondline was achieved after 300 s. The obtained results indicate the promising potential of the preform comprising copper formate particles for high-speed sinter bonding.
Key words: structural materials; microstructure; mechanical properties; copper formate preform; sinter bonding; pyrolysis
1 Introduction
Traditional bonding techniques that use solder alloys are expected to be partially replaced by sinter bonding to form high-temperature-sustainable bondlines in various devices and components in automotive vehicles, as well as electronic and electrical products [1-8]. For example, the peak junction temperature of high-end power devices based on SiC materials that provide high power as well as low switching loss is approaching 300 °C [3-7]. From the viewpoint of thermo-mechanical reliability, even solder alloys containing a large amount of Pb cannot withstand the harsh operating conditions of such systems because of the low remelting temperature of the bondline, which is similar to the bonding temperature. In contrast, the thermo-mechanical issues can be overcome using Ag or Cu particles for the sinter bonding, because the remelting temperature of the formed bondline is significantly enhanced in comparison with the low sinter bonding temperature attributable to the large initial surface area of metal particles [1-9]. In other words, while the sinter bonding using particles can be achieved at temperatures similar to the soldering temperatures, the formed bondline is sustainable at high temperatures, unless the temperature approaches the melting temperature of bulk Ag or Cu. Moreover, Ag and Cu have very high thermal conductivities [8,10], which are favorable for the aforementioned applications.
Despite the lower material cost and the superior electrochemical migration properties of Cu compared to those of Ag, the main drawbacks of using Cu for sinter bonding are its susceptibility to oxidation and long sinter-bonding time [4,5,11]. However, oxidation-resistant Cu sinter bonding has been achieved using various methods, including the use of formic acid vapor or forming gases as a reducing atmosphere [4,12,13]. Therefore, the long bonding time, such as several minutes or an hour, required for achieving adequate bonding strength, is the most prominent issue that hinders the industrial application of Cu. The fastest pressure-assisted sinter bonding time reported to date is 5 min, although Cu nanoparticles and N2 atmosphere were used in the study [14]. The rapid formation of a near-full-density bondline is another main research goal because interparticulate voids accelerate the initiation and propagation of cracks, and the rates of these two processes depend on the void sizes [15]. A representative strategy to form a near-full- density bondline is to use metal nanoparticles, which are expensive. Moreover, the dispersion and mixing of pastes will be challenging when using nanoparticles.
To overcome the aforementioned problems simultaneously, we adopted a preform comprising micrometer-sized copper formate particles as an effective Cu-based bonding material. The strategy of exploiting the decomposition of copper formate particles during thermal bonding allowed us to achieve rapid sinter bonding. Both the initially compact microstructure of the preform and the lack of a vaporizing solvent are expected to contribute to enhanced bondability and rapid formation of a near-full-density bondline. Furthermore, to assess the possibility of industrial application, we also investigated the sinter bonding of this Cu-based preform in air. Figure 1 shows a schematic diagram of the suggested high-speed sinter bonding technique.
2 Experimental
Copper formate particles, which were used as the main material for sinter bonding, were synthesized via a wet precipitation method [16]. Firstly, 50 g of copper(II) powder (CuO, 95%, Daejung Chemical Co., Ltd.) and 246 mL of formic acid (HCOOH, 85%, Samchun Chemical Co., Ltd.) were taken in a beaker, and the mixture was stirred for 90 min at 250 r/min using a magnetic bar. The obtained slurry was filtered, and the collected copper formate particles were washed thrice with ethanol. Finally, the washed copper formate sludge was dried in a vacuum oven at room temperature for 1 h.
The dried copper formate particles were shredded using a mill. Then, preforms consisting of the shredded copper formate particles were fabricated by pelletization. The copper formate particles (0.1 g) were filled into a cavity of 15 mm in diameter and compressed for 2 min under a load of 2 t to form a pellet with a thickness of approximately 410 μm. Finally, the pellet was ground to an area of 3 mm × 3 mm to transform it into the preform material.
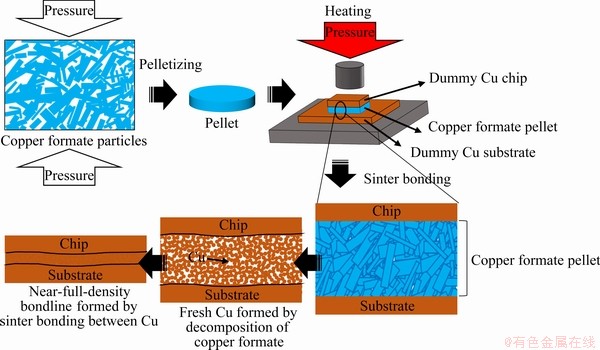
Fig. 1 Schematic representation of formation of preform comprising copper formate particles and achievement of high- speed sinter bonding
The sinter bonding tests were conducted using dummy Cu dies with dimensions of 3 mm × 3 mm × 1 mm and dummy Cu substrates with dimensions of 10 mm × 10 mm × 1 mm. The prepared preform was placed on the substrate, and then the die was placed and aligned on the preform. Thereafter, the resulting sandwich-structure was heated in air to the final temperature, which changed from 250 to 350 °C. The die bonding was carried out at a pressure of 5-10 MPa, which was maintained during the entire bonding process from the beginning of heating.
The morphologies of the fabricated and shredded copper formate particles, and the micro- structure of the preform, the formed bondlines, and fracture surfaces were observed by high-resolution scanning electron microscopy (HR-SEM, SU8010, Hitachi). Furthermore, the phase of the fabricated copper formate particles was determined by X-ray diffractometry (XRD, DE/D8 Advance, Bruker). The decomposition temperature and thermal behavior of the pellet were studied by thermo- gravimetry-differential thermal analysis (TG-DTA, DTG-60, Shimadzu). For these measurements, the samples were heated to 350 °C in air at a heating rate of 20 °C/min. The shear strength of the sinter-bonded die was determined from the average value of the maximum stress values measured during the shearing of five samples using a speed of 200 μm/s at a shear-tip height of 200 μm above the substrate surface using bond test equipment (Dage-4000, Nordson Dage).
3 Results and discussion
3.1 Copper formate particles and preforms
Figure 2 shows the XRD pattern of the copper formate particles synthesized from CuO particles. It has been reported that CuO was transformed into copper formate, as shown in Reaction (1) [17]:
CuO+2HCOOH→Cu(COOH)2+H2O (1)
XRD results confirmed the formation of two types of copper formate (PDF Nos. 32-0331 and 32-0332). The main peak of CuO almost disappeared after 90 min of reaction. Thus, copper formate could be easily synthesized by the simple stirring of CuO particles suspended in formic acid.
Figure 3 shows the morphologies of the synthesized copper formate particles and particles shredded using a mill. The synthesized copper formate particles have an irregular plate-like shape (Fig. 3(a)). Because the coarse plate-like particles were difficult to pelletize, they were shredded into tape-like particles with relatively similar lengths (Fig. 3(b)). This was possible due to the brittle nature of the material.
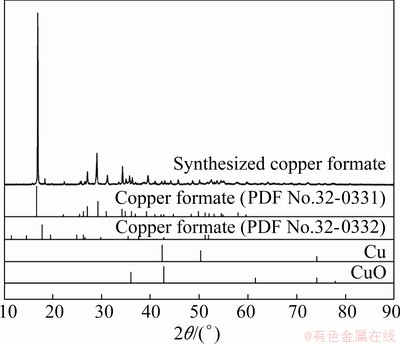
Fig. 2 XRD patterns of synthesized copper formate particles

Fig. 3 SEM images of synthesized (a) and shredded (b) copper formate particles
The cross-sectional microstructure of a pellet composed of the shredded copper formate particles is shown in Fig. 4. Interparticulate voids are not observable in the low-magnification image, and both the surfaces of the pellet show uneven contours owing to the loss of the particles during polishing with water. The thickness of the pellet used as a preform material was 410 μm.

Fig. 4 Cross-sectional optical microscopy image of copper formate pellet
The TG-DTA results for the copper formate pellet are shown in Fig. 5. The mass of the pellet decreased drastically from 210 °C, and a sharp exothermic peak appeared at the same temperature (peak temperature: 219.5 °C). This mass loss is due to the pyrolytic decomposition of copper formate, as shown in Reactions (2) and (3) [18-22]:
Cu(COOH)2→Cu+2CO2↑+H2↑ (2)
Cu(COOH)2→Cu+CO2↑+CO↑+ H2O↑ (3)
Gases such as carbon dioxide, hydrogen, and carbon monoxide are generated by the pyrolysis of copper formate (as shown in Reactions (2) and (3)), resulting in the mass loss of the sample due to out-gassing. The mass of the pellet reached 56.9% of its initial mass after this pyrolytic process. Subsequently, the mass of the sample increased gradually owing to the oxidation of the freshly reduced Cu. The exothermic heat generated during the decomposition of copper formate is anticipated to be advantageous for the sintering of the reduced Cu.
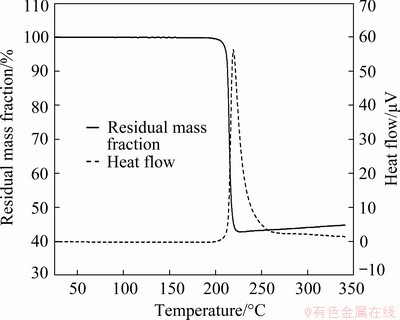
Fig. 5 TG-DTA curves of copper formate pellet
3.2 Shear strength
The temperature profiles of the bondlines during their sinter bonding at different temperatures of 250, 300, and 350 °C are displayed in Fig. 6. For the sample heated at 250 °C, the final temperature of 250 °C was attained rapidly in ~10 s. However, direct heating of the sample to 300 or 350 °C resulted in the bursting of the preform. Therefore, the final temperature of 300 or 350 °C was achieved by maintaining the sample at 250 °C for an additional 90 s after it reached that temperature. Thus, the final temperatures of 300 and 350 °C were achieved after a total heating time of 180 and 270 s, respectively. Hence, the bonding condition of 300 °C and 180 s implies that an air-cooling process was conducted immediately after the temperature of 300 °C was reached, and the bonding condition of 350 °C and 300 s implies an additional 30 s of maintenance at the final temperature of 350 °C followed by the air-cooling process.
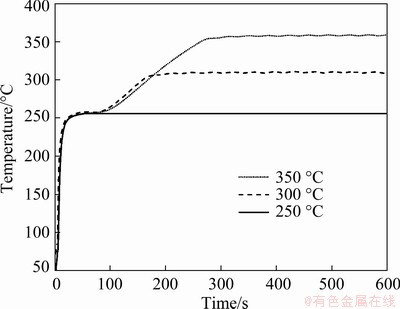
Fig. 6 Temperature profiles recorded during sinter bonding at 250, 300, and 350 °C
Figure 7 shows the average shear strength of the dummy Cu dies sinter-bonded using the copper formate preform as a function of the bonding pressure, temperature, and time. The strength obtained with each combination of bonding pressure and temperature generally increased with increasing bonding time, except for the samples under the condition of 5 MPa and 350 °C. For the samples under the condition of 5 MPa and 250 °C,which were formed using the gentlest bonding conditions used in this study, an insufficient strength of 11.8 MPa was achieved after bonding for 60 s. However, upon bonding for 300 s, the strength of the bondline approached that obtained by soldering a Pb-5Sn alloy [23]. Upon increasing the bonding temperature to 300 °C under the same pressure, the strength reached 20 MPa after only 180 s. Moreover, with an additional increase in time to 600 s, the strength reached an outstanding value of 36.9 MPa. At the higher temperature of 350 °C under the same pressure, the highest strength of 31.5 MPa was obtained after 300 s of bonding. However, the strength decreased slightly with a further increase in time to 600 s. When the pressure was increased to 10 MPa at 250 °C, a sufficient strength of 21.91 MPa was attained in 30 s, and a considerable strength of 31.2 MPa was achieved after 300 s. Therefore, high temperature and high pressure were found to be considerably effective for enhancing the bonding strength, while a longer bonding time could also generally lead to the increased strength. Furthermore, the observed speed of sinter bonding was found to be significantly faster than that reported previously [4,5,12,13].
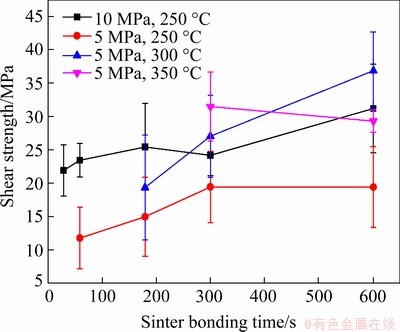
Fig. 7 Average shear strength of dummy Cu dies sinter-bonded using copper formate preform under different bonding conditions
3.3 Microstructures of bondlines and fracture surfaces
All the fracture surfaces indicate cohesive fracture, and the fracture occurred within the bondline rather than at the interface, although the amount of bondline adhering to the substrate decreased with decreasing shear strength. These results imply good sinter bonding of both the Cu chip/bondline and bondline/Cu substrate interfaces, even in the presence of the native oxide on the Cu surfaces and the growth of other oxides during heating in air. Therefore, the hydrogen and carbon monoxide gases emitted during the reactions (Eqs. (2) and (3)) are speculated to reduce the oxide layers on the Cu surfaces at the bonding temperature, eventually facilitating interfacial sinter bonding of the Cu interfaces under the assistance of external pressure. Furthermore, the emitted gas, which contains carbon dioxide, is expected to prevent the oxidation of the reduced copper. In other words, the emitted gases may act as a reducing and inert envelope against oxygen in air. The reduced copper fills up the concave surface regions even on the uneven upper and lower surface finishes, thus contributing to the strong interfacial sinter bonding of pure Cu species under the compressive pressure. The heat generated by pyrolysis also accelerates the sinter bonding between the formed pure Cu and interfaces.
The microstructures of the bondlines formed using unimodal Cu particles were dissimilar, according to the porous particulate structure observed in similar sintering processes. The representative microstructures obtained during sinter bonding using the copper formate preform at 250 °C under a pressure of 10 MPa are presented in Fig. 8. The active Cu species generated by the reduction of Cu ions at 210 °C coalesced to form Cu nanoparticles, and the Cu nanoparticles formed thus sintered rapidly under the assistance of the applied pressure, forming coarser Cu particles and thus eliminating interparticulate voids. Therefore, the remarkable sinterability of the Cu formed in situ is the main reason for the rapid sinter bonding.
Sinter bonding for 30 s at 5 MPa and 250 °C resulted in a bondline with a low shear strength of <10 MPa for the interface failure mode. Thus, such immature sintering did not yield a strong bondline at the Cu interface, which directly influenced the shear strength.
Figure 9 shows the backscattered electron (BSE) images of the overall bondline and the upper and lower interfaces of the bondline sintered for 60 s at 250 °C under 5 MPa pressure, along with the representative fracture surface. The copper formate particles were nearly reduced to pure Cu at the bondline, and Cu-to-Cu sintering at the Cu die/Cu particles and Cu particles/Cu substrate interfaces was partially observed. However, large voids were observed in the bondline, indicating a low-density structure. In the fracture surface of a dummy Cu chip, the contours of the reduced Cu particles were clearly observed. However, sintering between particles was only partial, and a small amount (a thin layer) of the sintered Cu was left on the substrate.

Fig. 8 Sequential microstructure images of bondline formed before (a) and after (b) sinter bonding at 250 °C and 10 MPa

Fig. 9 Backscattered electron (BSE) micrographs showing overall structure (a), upper interface (b) and lower interface (c) of bondline sinter-bonded for 60 s at 250 °C and 5 MPa, and SEM image of fracture surface (d)
When the bonding time was increased to 300 s at the identical temperature and pressure (Fig. 10), the number of voids decreased significantly, particularly at the interfaces, indicating the enhancement of the shear strength. However, a large amount of small-sized copper oxide (mainly Cu2O) phases were observed between the pure Cu particles owing to the exposure of the porous Cu microstructure to air at high temperatures. On the fracture surface on the Cu chip, shear bands in the partially dense regions as well as particles that were not densely sintered were observed simultaneously. The presence of shear bands normally indicates well-established metallic bonding.
The microstructures of the bondline sinter- bonded for 180 s at 300 °C under 5 MPa are presented in Fig. 11. A large void fraction was observed near the Cu chip/bondline interface owing to the out-gassing of carbon dioxide, hydrogen, and carbon monoxide generated by pyrolysis, and fracture occurred in this region. Moreover, the distribution of the copper oxide phase was intensified near the interface (although it was also observed within the bondline) as well as in some regions in the bondline. A large amount of small- sized copper oxide phases were also observed between the sintered Cu particles. On the fracture surface on the Cu chip, a densely sintered microstructure and partial shear bands were observed.

Fig. 10 BSE micrographs showing overall structure (a), upper interface (b) and lower interface (c) of bondline sinter-bonded for 300 s at 250 °C and 5 MPa, and SEM image of fracture surface (d)

Fig. 11 BSE micrographs showing overall structure (a), upper interface (b) and lower interface (c) of bondline sinter-bonded for 180 s at 300 °C and 5 MPa, and SEM image of fracture surface (d)
Figure 12 shows the change in the micro- structures upon increasing the sintering time to 300 s under identical conditions. Although a non-uniform microstructure was observed in the bondline, the weak microstructure at the Cu die/bondline interface was reinforced and the void fraction decreased significantly. Considering the microstructure shown in Fig. 11, the decrease in the void fraction near the Cu chip/bondline interface is attributed to sintering involving the formed copper oxide phase under the compressive pressure. Although the melting temperatures of the copper oxides are lower than that of Cu [24], the oxides formed on the Cu particle surface significantly suppress the sintering because the oxide shells are detached from the core Cu particles [25]. However, in this study, the applied pressure transformed the formed grain-like oxide to coarse copper oxides that were sintered without difficulty. The voids mainly existed above the middle region of the bondline, and the sintered oxide and small-sized oxide phases were randomly distributed throughout the bondline. On the fracture surface, a ductile elongated micro- structure was clearly observed, indicating the formation of stronger metallic bonding.
The microstructures obtained after the increase in temperature to 350 °C at 300 s under 5 MPa are shown in Fig. 13. Compared to that obtained at 300 °C, the bondline microstructure was more uniform. Meanwhile, a continuous copper oxide layer containing large voids was observed at the Cu die/bondline interface, whereas a non-continuous copper oxide layer containing large voids was observed at the bondline/Cu substrate interface. A significant amount of the copper oxide phase mainly existed above the middle region of the bondline, and the small-sized copper oxide phase was distributed throughout the bondline. In addition, the fracture surface showed a ductile elongated microstructure. Regardless of the bonding temperature and time, the samples bonded at 5 MPa generally showed a porous microstructure near the Cu die/bondline interface above the middle region of the bondline, because of the out-gassing phenomenon. Therefore, the microstructure of the bondline was not symmetric across the horizontal centerline of the bondline.
Figure 14 shows images of the bondline microstructure after 30 s of sinter bonding at a higher pressure of 10 MPa and decreased temperature of 250 °C. Large voids were scarcely observed, whereas small voids were relatively uniformly distributed. Moreover, the amount of the oxide phase also decreased, which suggested that less copper oxide was formed during the short sinter-bonding time of 30 s. The thickness of the formed bondline was 34.3 μm, indicating a drastic decrease in thickness compared to that of the initial preform because of the significant volume shrinkage caused by the decomposition of the preform (as shown in Reactions (2) and (3)). The reduced bondline thickness may be favorable considering the consistently decreasing trend in the profile of the bonded die. Further, the fracture surface showed a dense microstructure as well as the contours of the particles.

Fig. 12 BSE micrographs showing overall structure (a), upper interface (b) and lower interface (c) of bondline sinter-bonded for 300 s at 300 °C and 5 MPa, and SEM image of fracture surface (d)
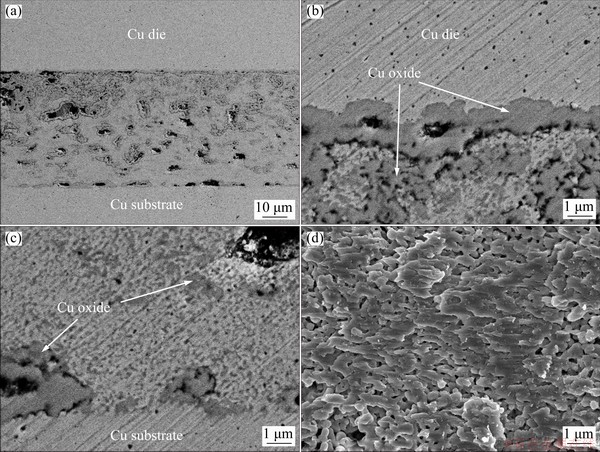
Fig. 13 BSE micrographs showing overall structure (a), upper interface (b) and lower interface (c) of bondline sinter-bonded for 300 s at 350 °C and 5 MPa, and SEM image of fracture surface (d)

Fig. 14 BSE images showing overall structure (a), upper interface (b) and lower interface (c) of bondline sinter-bonded for 30 s at 250 °C and 10 MPa, and SEM images of fracture surface (d)
When the bonding time was increased to 300 s at the same pressure and temperature, a denser bondline was achieved, as shown in Fig. 15(a). The magnified images confirm its close bonding with the Cu finishes at both the upper and lower interfaces (Figs. 15(b) and 15(c), respectively). The thickness of the bondline increased to 47.8 μm because the interfaces between the grain-like Cu particles were oxidized. Therefore, it is found that the oxidation of Cu contributes to the increase in volume of the bondline. Further, the fracture surface showed well-developed shear bands on the dense microstructures as well as particle contours.
Lastly, the microstructures of the bondline formed at 10 MPa with an increased bonding time of 600 s are displayed in Fig. 16. Although the dense microstructure of the bondline was still maintained, its thickness increased slightly to 51.1 μm due to further oxidation by the increased bonding time at 250 °C. Nevertheless, the uniformly elongated fracture mode was more developed on the fracture surface. Consequently, the 10 MPa samples showed almost symmetrical microstructures across the horizontal centerline of the bondline. Therefore, the strong compressive pressure of 10 MPa mitigates the effect of out-gassing, leading to a superior bondline with considerably reduced voids, especially near the Cu die/bondline interface.
Considering the aforementioned micro- structures, none of the bondline microstructures having a shear strength of more than 20 MPa showed a continuous void line near the Cu die/ bondline interface. Among the microstructures, the sample under the condition of 10 MPa, 250 °C and 30 s showed the absence of large voids near the Cu die/bondline interface and the most uniform distribution of voids. Furthermore, although the low pressure and high temperature conditions (5 MPa, 350 °C and 5 MPa, 300 °C) induced excessive interfacial oxidation or non-uniform oxidation when the bonding time approached 300 s, the high pressure and low temperature condition of 10 MPa and 250 °C yielded a uniformly oxidized microstructure, which is considered to be better.

Fig. 15 BSE images showing overall structure (a), upper interface (b) and lower interface (c) of bondline sinter-bonded for 300 s at 250 °C and 10 MPa, and SEM image of fracture surface (d)

Fig. 16 BSE images showing overall structure (a), upper interface (b) and lower interface (c) of bondline sinter-bonded for 600 s at 250 °C and 10 MPa, and SEM image of fracture surface (d)
4 Conclusions
(1) Carbon dioxide, hydrogen, and carbon monoxide generated by the in situ reduction of the copper formate particles at 210 °C aided the removal of the native oxide and other oxides grown on bulk Cu finishes and suppressed the oxidation of the freshly formed Cu, leading to sintering of Cu nanoparticles at the Cu nanoparticle/bulk Cu interfaces, as well as the prompt growth of Cu nanoparticles.
(2) Sinter bonding at 250 and 300 °C under 5 MPa led to a shear strength approaching 20 MPa after 300 and 180 s, respectively. Moreover, when the temperature was increased to 350 °C, a high strength of 31.5 MPa was achieved after 5 min.
(3) When the pressure was increased to 10 MPa, a sufficient strength of 21.9 MPa was achieved after sinter bonding for only 30 s at 250 °C. Furthermore, sinter bonding for 5 min at the same pressure and temperature resulted in a near-full-density bondline.
(4) Although bonding at 5 MPa induced excessive interfacial oxidation or non-uniform oxidation upon increasing the bonding time to 300 s, bonding at 10 MPa led to a lower degree of oxidation owing to the prompt densification of the bondline structure.
(5) Although the increase in bonding temperature or time generally contributes to the increase in shear strength, the increase in bonding pressure effectively boosted both the shear strength and the bondline density. The rapid solid-state sinter bonding in air is attributed to the remarkable sinterability of in situ reduced Cu under the compressive pressure before its oxidation.
Acknowledgments
This work was supported by the Materials & Components Technology Development Program (10080187) funded by the Ministry of Trade, Industry & Energy (MI, Korea).
References
[1] FU S, MEI Y, LU G Q, LI X, CHEN G, CHEN X. Pressureless sintering of nanosilver paste at low temperature to join large area (≥100 mm2) power chips for electronic packaging [J]. Materials Letters, 2014, 128: 42-45.
[2] CABEZAS-VILLA J L, OLMOS L, VERGARA- HERNANDEZ H J, JIMENEZ O, GARNICA P, BOUVARD D, FLORES M. Constrained sintering and wear properties of Cu-WC composite coating [J]. Transactions of Nonferrous Metals Society of China, 2017, 27: 2214-2224.
[3] KISIEL R, SZCZEPANSKI Z. Die-attachment solutions for SiC power devices [J]. Microelectronics Reliability, 2009, 49: 627-629.
[4] LIU X, NISHIKAWA H. Low-pressure Cu-Cu bonding using in-situ surface-modified microscale Cu particles for power device packaging [J]. Scripta Materialia, 2016, 120: 80-84.
[5] LIU J D, CHEN H T, JI H J, LI M Y. Highly conductive Cu-Cu joint formation by low-temperature sintering of formic acid- treated Cu nanoparticles [J]. ACS Applied Materials & Interfaces, 2016, 8: 33289-33298.
[6] ZHANG H Q, WANG W G, BAI H L, ZOU G S, LIU L, PENG P, GUO W. Microstructural and mechanical evolution of silver sintering die attach for SiC power devices during high temperature applications [J]. Journal of Alloys and Compounds, 2019, 774: 487-494.
[7] CHIN H S, CHEONG K Y, ISMAIL A B. A review on die attach materials for SiC-based high temperature power devices [J]. Metallurgical and Materials Transactions B, 2010, 41: 824-832.
[8] SIOW K S. Mechanical properties of nano-silver joints as die attach materials [J]. Journal of Alloys and Compounds, 2012, 514: 6-19.
[9] WANG S, JI H J, LI M Y, WANG C Q. Fabrication of interconnects using pressureless low temperature sintered Ag nanoparticles [J]. Materials Letters, 2012, 85: 61-63.
[10] NATH P, CHOPRA K L. Thermal conductivity of copper films [J]. Thin Solid Films, 1974, 20: 53-62.
[11] LI W, CHEN M F, WEI J, LI W J, YOU C. Synthesis and characterization of air-stable Cu nanoparticles for conductive pattern drawing directly on paper substrates [J]. Journal of Nanoparticle Research, 2013, 15: 1949-1958.
[12] YANG W, AKAIKE M, SUGA T. Effect of formic acid vapor in situ treatment process on Cu low-temperature bonding [J]. IEEE Transactions on Components, Packaging and Manufacturing Technology, 2014, 4: 951-956.
[13] LI J J, LIANG Q, SHI T L, FAN J, GONG B, FENG C, FAN J H, LIAO G L, TANG Z R. Design of Cu nanoaggregates composed of ultra-small Cu nanoparticles for Cu-Cu thermo-compression bonding [J]. Journal of Alloys and Compounds, 2019, 772: 793-800.
[14] ISHIKAWA D, NAKAKO H, KAWANA Y, SUGAMA C, NEGISHI M, EJIRI Y, UEDA S, AN B N, WURST H, LEYRER B, BLANK T, WEBER M. Copper die-bonding sinter paste: Sintering and bonding properties [C]//Proc 7th Electronic System―Integration Technology Conference (ESTC). Dresden, Germany: IEEE, 2018: 1-10.
[15] CHAWLA N, DENG X. Microstructure and mechanical behavior of porous sintered steels [J]. Materials Science and Engineering A, 2005, 390: 98-112.
[16] LEE B Y. Synthesis and characterization of conductive paste using copper complex [D]. Seoul: Yonsei University, 2011.
[17] CHOI W L, KIM Y S, LEE K S, LEE J H. Characterization of the die-attach process via low-temperature reduction of Cu formate in air [J]. Journal of Materials Science: Materials in Electronics, 2019, 30: 9806-9813.
[18] GALWEY A K, JAMIESON D M, BROWN M E. Thermal decomposition of three crystalline modifications of anhydrous copper(II) formate [J]. The Journal of Physical Chemistry, 1974, 78: 2664-2670.
[19] MOUCHE M J, MERMET J L, ROMAND M, CHARBONNIER M. Metal-organic chemical vapor deposition of copper using hydrated copper formate as a new precursor [J]. Thin Solid Films, 1995, 262: 1-6.
[20] MOHAMED M A, GALWEY A K, HALAWY S A. Kinetic and thermodynamic studies of the nonisothermal decomposition of anhydrous copper (II) formate in different gas atmospheres [J]. Thermochimica Acta, 2004, 411: 13-20.
[21] YABUKI A, ARRIFFIN N, YANASE M. Low-temperature synthesis of copper conductive film by thermal decomposition of copper-amine complexes [J]. Thin Solid Films, 2011, 519: 6530-6533.
[22] YABUKI A, TANAKA S. Electrically conductive copper film prepared at low temperature by thermal decomposition of copper amine complexes with various amines [J]. Materials Research Bulletin, 2012, 47: 4107-4111.
[23] SUGANUMA K, KIM S J, KIM K S. High-temperature lead-free solders: properties and possibilities [J]. JOM, 2009, 61: 64-71.
[24] MORITA T, YASUDA Y. New bonding technique using copper oxide materials [J]. Materials Transactions, 2015, 56: 878-882.
[25] LI W L, LI L Y, GAO Y, HU D W, LI C F, ZHANG H, JIU J T, NAGAO S, SUGANUMA K. Highly conductive copper films based on submicron copper particles/copper complex inks for printed electronics: Microstructure, resistivity, oxidation resistance, and long-term stability [J]. Journal of Alloys and Compounds, 2018, 732: 240-247.
Yun-Ju LEE, Jong-Hyun LEE
Department of Materials Science and Engineering, Seoul National University of Science and Technology, 232 Gongneung-ro, Nowon-gu, Seoul 139-743, Korea
摘 要:使用由甲酸铜颗粒组成的预制块,在250~350 °C于空气中用压力辅助烧结法制备耐高温的黏结层。烧结过程中,甲酸铜在210 °C发生热解,同时生成H2 和CO,且去除铜块体表面的天然氧化物和其他生成的氧化物,形成界面结合。而且,原位还原Cu(II)生成的Cu加速烧结连接。在300~350 °C和5 MPa压力下烧结180~300 s后得到的黏结层具有足够的剪切强度:20.0~31.5 MPa。此外,在250 °C条件下,当压力增加到10 MPa时,在极短的时间(30 s)内,剪切强度达到21.9 MPa, 300 s后形成接近全致密的黏结层。因此,甲酸铜颗粒预制块在高速烧结连接中具有广阔的应用前景。
关键词:结构材料;显微组织;力学性能;甲酸铜预制块;烧结连接; 热解
(Edited by Wei-ping CHEN)
Corresponding author: Jong-Hyun LEE, Tel: +82-2-970-6612 (office), +82-10-2214-4974 (mobile), E-mail: pljh@snut.ac.kr
DOI: 10.1016/S1003-6326(21)65610-8
1003-6326/ 2021 The Nonferrous Metals Society of China. Published by Elsevier Ltd & Science Press
2021 The Nonferrous Metals Society of China. Published by Elsevier Ltd & Science Press

