J. Cent. South Univ. Technol. (2010) 17: 295-299
DOI: 10.1007/s11771-010-0045-z 
Ionization behavior of deep level donors in passive film formed on surface of stainless steel in 5% salt solution
WANG Chao(����), SHENG Min-qi(ʢ����), ZHONG Qing-dong(���춫),
CHOU Kuo-chih(�ܹ���), LU Xiong-gang(³�۸�)
Shanghai Key Laboratory of Modern Metallurgy and Material Processing, Shanghai University,
Shanghai 200072, China? Central South University Press and Springer-Verlag Berlin Heidelberg 2010
Abstract: Deep level donor��s ionization behavior of passive film formed on the surface of stainless steel was investigated by Mott-Schottky plots. It is indicated that transformation process of deep level donors�� ionization behavior of passive film on surface of stainless steel can be divided into 4 stages with rising immersion time. At the initial immersion stage (10 min), Fe(��) located in the octahedral sites of the unit cell is not ionized and the deep level does not appear in Mott-Schottky plots. At the second stage (9-38 h), Fe(��) located in the octahedral sites starts to be ionized, which results in deep level donors�� generation and density of deep level donors almost is constant with augmenting immersion time but the thickness of space charge layer is more and more thicker with rising immersion time. At the third stage (48 h-12 d), density of deep level donors rises with increasing immersion time and the thickness of passive films space charge layer decreases. At last stage (above 23 d), both the space charge layer��s thickness and density of deep level donors are no longer changed with increasing immersion time. In the overall immersion stage, the shallow level donors�� density is invariable all the time. The mechanism of deep level donor��s ionization can be the generation of metal vacancies, which results in crystal lattice��s aberration and the aberration energy urges the ionization of Fe(��) in octahedral sites.
Key words: stainless steel; deep level donor; Mott-Schottky plots; donor density; space charge layer; aberration energy
1 Introduction
As a hot topic, the relationship between character of passive film on the surface of steel and steel��s anti-corrosion performance was studied for many years. In recent years, it has been still an interesting subject to many electrochemists and material scientists. In general, passive film on surface of stainless steel could be interpreted in terms of the semi-conductive film. Capacitance measurements and photo electrochemistry are often utilized to study the conducting behavior of passive films and to obtain the donor density (ND) and the flat band potential (Ufb) by analysis of Mott-Schottky plots. The frequency dependency of passive film��s capacitance measurements indicated that the passive films were amorphous or strongly disordered semiconductors [1-4].
Previous researchers proved the passive film formed on the surface of stainless steel in electrolyte was made of an inner layer rich in chromium oxide and an outer layer rich in iron oxides, with a spinel structure [5-9]. The Mott-Schottky analysis can reveal the properties of different oxide layers. According to Ref.[7], below -0.5 V, the passive film displayed the chromium-rich layer��s semiconductor properties and behaved like a p-type semiconductor; between -0.5 and 0.4 V, the passive film exhibited iron-rich layer��s semiconductor characters and behaved like an n-type semiconductor in the Mott-Schottky plots. When the potential became higher than 0.5 V, i.e., in the stainless steel��s transpassive potential region, the Mott-Schottky curves reflected the chromium-rich layer��s semiconductor properties again with a negative slope.
It should be particularly pointed out that, in the potential region from -0.5 to 0.4 V, there was a break which separated the linear from 2 segments with different positive slopes. According to Ref.[4], this change can be related to the existence of a deep level formed by Fe(��) placed in the octahedral sites of the unit cell of the spinel structure. HAKIKI et al [7] maintained that all the donor species in the tetrahedral positions were ionized in a relative low potential region. When the applied potential reached a relatively high potential, Fe(��) ions located in the octahedral sites of spinel structure can also be ionized and make the passive film��s capacitance behavior, showing that there exist two donor levels in the band gap of the semiconductor film formed on the surface of the stainless steel electrode. CARMEZIM et al [3] found that the Mott-Schottky plots of the films formed on AISI 446 at pH 4.5 and 8.4 showed two different slopes. The existence of the second slope at relatively high potential may reveal a deep donor level that becomes oxidized at higher anodic polarizations [3]. The same feature was observed before in the 304 alloy and the shallow and deep levels were associated with the presence of Fe(��) ions in tetrahedral and octahedral positions, respectively [5]; the existence of two donor levels in oxide structure in stainless steels also was confirmed by photo electrochemical studies [10-11]. Similar feature of two donor levels observed in the passive film of iron in crystalline ��-Fe2O3 is ascribed to ions at octahedral and tetrahedral positions in the unit cell of a spinel structure [12-13]. FERREIRA et al [6-7] studied different passive films formed on surface of 304 stainless steels at different temperature by Mott-Schottky plots and photo electrochemical measurements. They indicated that the peak situated at 1.9 eV in the plots of (��h��)0.5 vs h�� can be associated with transitions from the valence band to the second donor level localized in the band gap and this level is situated at 0.4 eV below the conduction band [6-7].
Mott-Schottky approach became very popular in studying donors�� properties of passive film on the surface of stainless steel, but it mainly involved in the properties of donors in different media or tested conditions. But transformation of donor level with rising the immersion time of passive film which will contribute to understanding the anticorrosion ability in passive film��s overall degradation process has not been researched nearly until now. Based on the above views, Mott-Schottky plots were utilized to study the changing characters of film��s deep donor level during immersion time in salt electrolyte in this work.
2 Experimental
One electrode sample was prepared in this work. This electrode was made of 304 stainless steel rod. It was embedded in epoxy resin, and resulted in exposed steel area being about 0.785 cm2. The working surface of the electrodes was polished with emery paper (800 grit), and then cleaned with deionized water, absolute alcohol and deionized water in turn, finally dried in the air. The sodium chloride (NaCl) solution was made from analytical grade reagent and deionized water.
Capacitance measurement was carried out in a conventional three-electrode system with a computer- controlled electrochemistry station Model CHI660C. The counter electrode was a platinum sheet and the reference electrode was a saturated calomel electrode (SCE). All potentials mentioned in this work were defined with respect to SCE reference electrode. All experiments were carried out at 30 ��.
3 Results and discussion
3.1 Mott-Schottky plots of stainless steel in 5%NaCl solution
It was proved by previous researchers [14-19] that the relationship between C-2 and applied potential was in good agreement with well-known Mott-Schottky equations when stainless steel electrode was immersed in electrolyte. The Mott-Schottky equations are as follows:
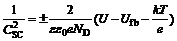 (1)
(1)
where ��+�� for the n-type semiconductor and ���C�� for p-type semiconductor; CSC is the capacitance of space charge layer; �� is the dielectric constant of the semiconductor (12 was applied in this work) [10]; ��0 is the permittivity of free space (8.854��10-12 F/m); e is the charge of electron; ND and NA are the densities of donors and acceptors, respectively; U is the sweeping potential and Ufb is the flat band potential; k is Boltzmann constant; and T is thermodynamic temperature. As a result, N is determined from the slope of the experimental CSC-2 vs U plot. In the present work, a frequency of 1 000 Hz was used for the capacitance measurement [6]. The validity of the Mott-Schottky analysis is based on the assumption that the capacitance of the space-charge layer is much less than that of the Helmholtz layer. Then, the data points in CSC-2 vs U plots can describe the semi- conductive behavior of the depletion region [15].
It is clearly shown in Fig.1 that the Mott-Schottky plot is nearly without evident break in the potential range from -0.5 to 0.2 V in the initial immersion stage (10 min). This phenomenon can be ascribed to an air-formed passive film on the surface of stainless steel. Air-formed passive film was almost not destroyed at the beginning of immersion. Because it was properties of homogeneity and waterproof resulted in Fe(��) ions located in the octahedral sites being more difficultly ionized in this stage. The space charges entirely came from shallow level, i.e., ionization of donors in the tetrahedral positions in this stage. The air-formed passive film was still working on alloy��s anti-corrosion performance in this stage.
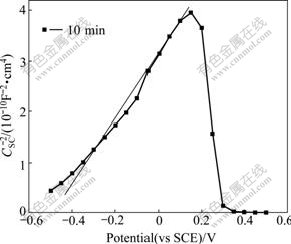
Fig.1 Mott-Schottky plot of 304 stainless steel in 5% NaCl solution at initial immersion stage
When the immersion time was above 9 h, a break generated in Mott-Schottky curves in the potential range of -0.3 V (9 h) to 0 V (38 h) in Fig.2. It explained that the ionization of Fe(��) ions was related to aggression of the water and ions to passive film. Otherwise, the capacitance decreased in this stage and these linear segments between break-emerging potential and approximate 0.2 V were almost parallel. The slope of linear segment before the break almost kept constant with rising immersion time. The flat band potential of the film in this potential region was -0.5 V, which was similar to others�� results [4]. Some results can be gained from the Mott-Schottky response in this immersion stage. Firstly, since between -0.5 and 0.2 V the slope of Mott-Schottky plots was positive in this potential region, the ferrite film��s properties were not significantly affected by electrolyte. Secondly, it can be speculated that the passive film��s space charge layer became thicker in this stage because space charge capacitance value became lower with increasing immersion time. At last, although the ionization of Fe(��) ions in octahedral sites initiated in this stage, the charge carriers�� concentration of deep donor level was not considerably increased.
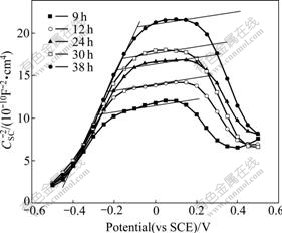
Fig.2 Mott-Schottky plots of 304 stainless steel in 5% NaCl solution at the second stage
However, after being immersed for 48 h, the semiconductor behavior of passive film exhibited another manner in the Mott-Schottky plots of Fig.3. The linear segment��s slope between -0.5 and -0.2 V was still constant with augmenting immersion time in this stage, which accounted for constant shallow donor density and the flat band in this segment was still -0.5 V, but the semiconductor behavior of passive film in the potential range between -0.2 and 0.3 V changed. In the region, the slope gradually became gentle and capacitance value also rised with increasing immersion time. The ionization of Fe(��) ions in octahedral sites resulted in deep donor density increasing and the thickness of passive film��s space charge layer thinning in this stage.
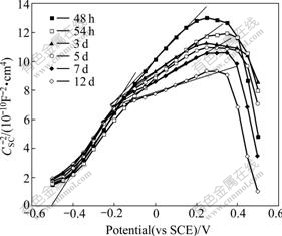
Fig.3 Mott-Schottky plots of 304 stainless steel in 5% NaCl solution at the third stage
In Fig.4, slopes of linear segments in all regions no longer considerably changed with increasing the immersion time. It is concluded from the phenomena that the passive film generated on the surface of stainless steel is in a relatively steady situation in this stage. The ionization of Fe(��) ions placed in the octahedral sites is completed in this stage.
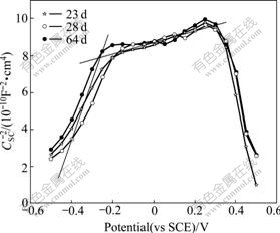
Fig.4 Mott-Schottky plots of 304 stainless steel in 5% NaCl solution at last stage
3.2 Transformation of deep level donor��s ionization behavior in degradation process by crystallology
As shown in Fig.5, Fe(��) ions in tetrahedral sites of the passive film are gradually ionized in the overall immersion time, but Fe(��) ions in octahedral sites are not ionized at the immersion time of 10 min, so the deep level donor was not generated in this moment. Because of the aggressive action of electrolyte, Fe(��) ions in tetrahedral sites were gradually ionized, which resulted in the generation of metal vacancies. The crystal lattice aberration also became more and more serious due to the vacancies, so Fe(��) ions in octahedral sites began to be ionized, which made the deep level appear after the stainless steel was immersed for more than 9 h, but between 9 and 38 h Fe(��) ions in the tetrahedral sites were not ionized entirely, the aberration energy mostly promoted the ionization of them since they are ionized more easily than Fe(��) ions in octahedral sites of lattice, which cause the deep level donors concentration to keep constant approximately at this stage.
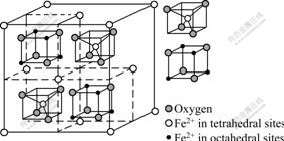
Fig.5 Sketch map of spinel ferrite crystal structure
Between 48 h and 12 d, Fe(��) ions in the tetrahedral sites were ionized entirely, so the aberration energy of lattice started to accelerate the ionization of Fe(��) in octahedral sites. As a result, the deep level donor concentration increased with rising the immersion time between 48 h and 12 d. Fe(��) in octahedral sites of lattice became ionized completely when the electrode was immersed for more than 23 d, and the deep level donor density no longer changed with rising the immersion time. In this stage, the passive film on the surface of stainless steel in NaCl solution reached a steady corrosion condition.
4 Conclusions
(1) There are different deep level donors�� ionization behaviors at different degradation stages in the overall corrosion process.
(2) At the beginning of immersion, deep donor level is not ionized.
(3) When the deep donor level��s ionization initiates, firstly, the deep donor density does not change and thickness of space charge layer increases with rising immersion time; and secondly, the deep donor density increases and thickness of space charge layer decreases; at last, passive film of stainless steel becomes steady and both thickness of space charge layer and density of carriers are no longer changed.
References
[1] WANG Qiao-ling, HUANG Gui-fang. Effect of sodium nitrite on corrosion of mild steel by using wire beam electrode [J]. Journal of Central South University: Science and Technology, 2008, 39(2): 317-321. (in Chinese)
[2] VIGNAL V, VALOT C, OLTRA R, VERNEAU M, COUDREUSE L. Analogy between the effects of a mechanical and chemical perturbation on the conductivity of passive films [J]. Corrosion Science, 2002, 44(7): 1477-1496.
[3] CARMEZIM M J, SIM?ES A M, MONTEMOR M F, da CUNHA BELO M. Capacitance behaviour of passive films on ferritic and austenitic stainless steel [J]. Corrosion Science, 2005, 47(3): 581-591.
[4] MONTEMOR M F, FERREIRA M G S, HAKIKI N E, da CUNHA BELO M. Chemical composition and electronic structure of the oxide films formed on 316L stainless steel and nickel based alloys in high temperature aqueous environments [J]. Corrosion Science, 2000, 42(9): 1635-1650.
[5] SIM?ES A M P, FERREIRA M G S, RONDOT B, da CUNHA BELO M. Study of passive films formed on AISI 304 stainless steel by impedance measurements and photoelectrochemistry [J]. Journal of the Electrochemical Society, 1990, 137(1): 82-87.
[6] FERREIRA M G S, HAKIKI N E, GOODLET G, FATY S, SIM?ES A M P, da CUNHA BELO M. Influence of the temperature of film formation on the electronic structure of oxide films formed on 304 stainless steel [J]. Electrochimica Acta, 2001, 46(24/25): 3767-3776 .
[7] HAKIKI N E, MONTEMOR M F, FERREIRA M G S, da CUNHA BELO M. Semiconducting properties of thermally grown oxide films on AISI 304 stainless steel [J]. Corrosion Science, 2000, 42(4): 687-702.
[8] ABDULLAH S, SASHA O. Improvement of pitting corrosion resistance of a biomedical grade 316LVM stainless steel by electrochemical modification of the passive film semiconducting properties [J]. Electrochemistry Communications, 2007, 9(1): 76-82.
[9] MONTEMOR M F, SIM?ES A M P, FERREIRA M G S, da CUNHA BELO M. The role of Mo in the chemical composition and semiconductive behaviour of oxide films formed on stainless steels [J]. Corrosion Science, 1999, 41(1): 17-34.
[10] CARMEZIM M J, SIM?ES A M, FIGUEIREDO M O, da CUNHA BELO M. Electrochemical behaviour of thermally treated Cr-oxide films deposited on stainless steel [J]. Corrosion Science, 2002, 44(3): 451-465.
[11] GOODLET G, FATY S, CARDOSO S, FREITAS P P, SIM?ES A M P, FERREIRA M G S, da CUNHA BELO M. The electronic properties of sputtered chromium and iron oxide films [J]. Corrosion Science, 2004, 46(6): 1479-1499.
[12] LEITNER K, SCHLTZE J W, STIMMING U. Photoelectrochemical investigations of passive films on titanium electrodes [J]. Journal of the Electrochemical Society, 1986, 133(8): 1561-1568.
[13] STIMMING U, SCHULTZE J W. Semiconductor model of the passive layer on iron electrodes and its application to electrochemical reaction [J]. Electrochimica Acta, 1979, 24(8): 859-869.
[14] IVERSEN A K. Stainless steels in bipolar plates��Surface resistive properties of corrosion resistant steel grades during current loads [J]. Corrosion Science, 2006, 48(5): 1036-1058.
[15] MORRISON S R. Electrochemistry at semiconductor and oxidized metal electrodes [M]. New York: Plenum, 1980.
[16] S?NCHEZ M, GREGORI J, ALONSO M C, GARC?A-JARE?O J J, VICENTE F. Anodic growth of passive layers on steel rebars in an alkaline medium simulating the concrete pores [J]. Electrochimica Acta, 2006, 52(1): 47-53.
[17] HAMADOU L, KADRI A, BENBRAHIM N. Characterisation of passive films formed on low carbon steel in borate buffer solution (pH=9.2) by electrochemical impedance spectroscopy [J]. Applied Surface Science, 2005, 252(5): 1510-1519.
[18] ALVES V A, BRETT C H A. Characterisation of passive films formed on mild steels in bicarbonate solution by EIS [J]. Electrochimica Acta, 2002, 47(13/14): 2081-2091.
[19] HAKIKI N E, da CUNHA BELO M. Electronic structure of passive films formed on molybdenum-containing ferritic stainless steels [J]. Journal of the Electrochemical Society, 1996, 143(10): 3088-3096.
Foundation item: Projects(50571059, 50615024 ) supported by the National Natural Science Foundation of China; Project(NCET-07-0536) supported by Program for New Century Excellent Talents in University; Project(IRT0739) supported by Program for Innovative Research Team in University
Received date: 2009-04-21; Accepted date: 2009-07-09
Corresponding author: ZHONG Qing-dong, PhD, Professor; Tel: +86-13391312191; E-mail: qdzhong@shu.edu.cn
(Edited by YANG You-ping)