
Barium phosphate conversion coating on die-cast AZ91D magnesium alloy
LIU Feng(刘 锋)1, 2, SHAN Da-yong(单大勇)1, HAN En-hou(韩恩厚)1, LIU Chang-sheng(刘常升)2
1. State Key Laboratory for Corrosion and Protection, Institute of Metal Research,
Chinese Academy of Sciences, Shenyang 110016, China;
2. School of Materials and Metallurgy, Northeastern University, Shenyang 110016, China
Received 12 June 2008; accepted 5 September 2008
Abstract: Poor corrosion resistance limits the application of magnesium alloys. Conversion coating is widely used to protect magnesium alloys because of easy operation and low cost. A novel conversion coating on die-cast AZ91D magnesium alloy containing barium salts was studied. The optimum concentrations of Ba(NO3)2, Mn(NO3)2 and NH4H2PO4 are 25 g/L, 15 mL/L and 20 g/L, respectively, based on orthogonal test results. The treating time, solution temperature and pH value are settled to be 5-30 min, 50-70 ℃ and 2.35-3.0, respectively. The corrosion resistance of barium conversion coating is better than that of manganese-based phosphate conversion coating by immersion test. The coating is composed of Ba, P, O, Mg, Zn, Mn and Al by EDX analysis.
Key words: barium phosphate conversion coating; die-cast AZ91D magnesium alloy; micro-cracks; corrosion resistance
1 Introduction
Low density, high strength to mass ratio, good electromagnetic features and easy recycle make magnesium alloy widely used in the fields of aircraft[1], automobile and 3C products[2]. But poor corrosion resistance limits the application of magnesium alloy[3]. Surface treatment[4] including anodic oxidation[5], metal coating[6], conversion coating and organic coating is an effective way to improve the corrosion resistance of magnesium alloy. Conversion coating is widely used due to easier operation and lower cost. Phosphate conversion coating[7-9], stannate conversion coating[10], organic conversion coating[11-12] and rare-earth conversion coating[13] are studied recently, and some metal-salts are added into the phosphate conversion coating solution as film forming matters, such as zinc salts and manganese salts.
But conversion coating which has already been reported can not provide enough protection for magnesium alloy in severe rugged environment. Through experiment, a kind of barium salt phosphate conversion coating on AZ91D was got and the concentration of the ingredients, pH value, temperature and treating time were settled. The corrosion resistance of barium conversion coating is better than that of manganese- based phosphate conversion coating.
2 Experimental
Die-cast AZ91D magnesium alloy was cut into plates with the size of 30 mm×20 mm×2 mm.
The treatment processes were as follows: mechanical polishing (800# silicon paper)→alkali washing (25 g/L NaOH solution, 70 ℃)→pickling (20 g/L NH4H2PO4 solution, room temperature)→conversion coating treatment→drying, and washing was necessary after each step.
Ba(NO3)2, Mn(NO3)2 (50% in mass fraction) solution and NH4H2PO4 were selected as the ingredients of treating solution, of which the concentrations were settled through orthogonal test. The test project contained three factors and three levels: the concentra- tions of these three ingredients were considered as three influence factors, and each factor consisted of three levels (see Table 1). Polarization curves were used to evaluate the corrosion resistance of the coatings.
The pH value, temperature and the treating time were settled through electrochemical test. All the electrochemical tests were conducted in 3.5% NaCl solution using a conventional three electrode cells at room pH value 2.75; solution temperature 50 ℃; treating time 20 min temperature (reference electrode: SCE). The specimens were sealed by silicon rubber with an exposure area of 1 cm2. In the polarization curves test, the initial scanning potential was -0.1 V vs open circuit potential and the scanning rate was 0.5 mV/s. In the impedance test, a frequency range from 100 kHz to10 mHz was swept with a sinusoidal potential signal with amplitude of 5 mV.
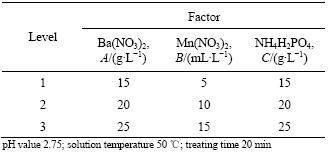
Table 1 Orthogonal test factors and levels
The corrosion resistance of this kind of conversion coating was compared with manganese phosphate conversion coating[14] through immersion test.
SEM and EDX were used to investigate the morphology and composition of the coating.
3 Results and discussion
3.1 Orthogonal test results
Effects of the three elements on corrosion resistance are reduced in turn according to the quantities of Ba(NO3)2, Mn(NO3)2 and NH4H2PO4 by comparing R values in Table 2. With increasing quantities of Ba(NO3)2 and Mn(NO3)2, the corrosion resistance becomes better in the range selected, the quantity of NH4H2PO4 has no significant influence on the corrosion resistance. The optimum quantities are 25 g/L, 15 mL/L and 20 g/L, which is just No.9 experiment in Table 2.
3.2 Influence of pH, temperature and treating time
3.2.1 Influence of pH
Fig.1 shows the polarization curves of the coatings formed at different pH values (solution temperature is 50 ℃, treating time is 20 min). The anode curves have a slight shift towards left with increasing the pH value, which means that the corrosion resistance of coating is better when the pH value is higher.
Fig.2 shows the SEM images of the coatings formed at different pH values. It can be seen that this kind of conversion coating presents network feature. The micro-cracks become narrow with increasing the pH value. The conversion coating is a kind of colloid in solution, and the volume of the colloid will shrink when water escaped from the coating. If the frame of colloid is not strong enough and the speed of the water escaping
Table 2 Orthogonal test results
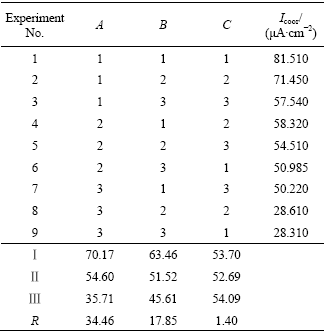
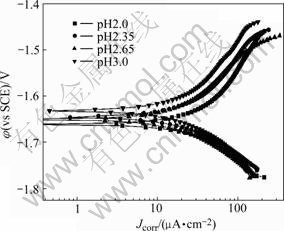
Fig.1 Polarization curves of coatings formed at different pH value
from the colloid is fast, the micro-crack will appear[15]. It has more possibility to induce micro-cracks when the frame of coating is non-uniform for the cracks will occur in the weaker region. When the pH value is lower, the deposition rate of indissoluble phosphate salts is higher and the coating is eroded by the solution more seriously which will induce non-uniform thickness. At the same time the coating is thicker[16], so there are more micro- cracks in the surface of coating formed at pH 2.0.
From Fig.1 and Fig.2, the relationship between micro-cracks and corrosion resistance of the coating can be found. The corrosion resistance of the coating is worse with the wider micro-cracks, because more entryway for the corrodibility ion can be provided.
So the pH value ranging from 2.35 to 3.0 is suitable for this kind solution.

Fig.2 Images of coatings formed at different pH values: (a) 2.0; (b) 2.35; (c) 2.65; (d) 3.0
3.2.2 Influence of temperature
Fig.3 shows the polarization curves of the coatings when the treating temperatures are different (pH value is 2.5). The anode and cathode curves both move towards left, which means that the anode and cathode reaction process are all restrained with the increasing of solution temperature.
The better corrosion resistance coating can be obtained at a temperature ranging from 50 ℃ to 70 ℃.

Fig.3 Polarization curves of coatings formed at different temperatures
3.2.3 Influence of treating time
Fig.4 shows the EIS results of the coatings when the treating time is different (solution temperature, pH value are 55 ℃ and 2.5). Fig.5 shows the equivalent circuit for the EIS of coatings, Rs represents solution electric resistance between working electrode and counter electrode, Rt is the electric resistance of the coating, CPE substitutes C to represent the capacity character of coating because of dispersion effects which may arise from surface defects and local charge inhomo- geneity. EIS simulation parameters for the conversion
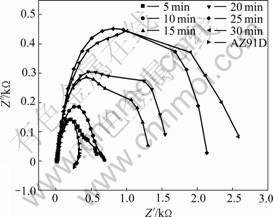
Fig.4 EIS results of coatings for different treating times
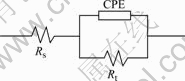
Fig.5 Equivalent circuit for EIS of conversion coating
coating are listed in Table 3. It is recognized that the coating with higher Rt value and lower CPE value has better corrosion resistance. So when the treating time ranges from 25 min to 30 min, the coatings have better corrosion resistance by analyzing the results listed in Table 3[17].
Table 3 EIS impedance parameters of conversion coating for different treating times
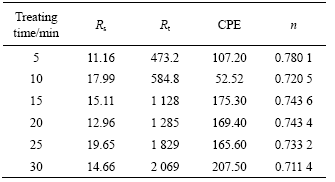
So the treating time ranging from 25 min to 30 min is suitable.
Treating time, solution temperature and pH values are settled through previous experiments, they are 25-30 min, 50-70 ℃ and 2.35-3.0, respectively.
3.3 Immersion test
Fig.6 shows the photos of two kinds of conversion coatings after immersion test. Some gas sends out from

Fig.6 Photos of conversion coatings by different processes after immerged in 3.5% NaCl for 48 h: (a) Barium phosphate conversion coating; (b) Manganese phosphate conversion coating
both of coatings after immerging for 5 min; the number of spots where the gas gets out decreases after immerging for 5 h for barium conversion coating, but it has not much decrease for another; when the immersion time is increased to 48 h, the color of the former does not change very much and keeps grey, but the color of the latter changes from grey to brown and the corrosion spots are bigger. So it can be concluded that the barium conversion coating has better corrosion resistance through immersion test.
3.4 EDX result
Fig.7 shows the EDX result of conversion coating. It can be seen that the coating is composed of Ba, P, O, Mg, Zn, Mn and Al.
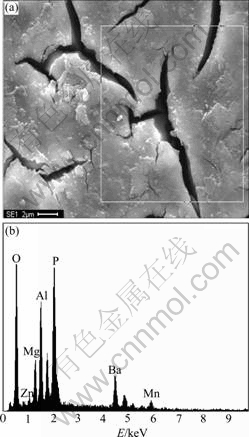
Fig.7 EDX result of conversion coating: (a) SEM image; (b) EDX pattern
4 Conclusions
1) The concentrations of Ba(NO3)2, Mn(NO3)2 and NH4H2PO4 are 25 g/L, 15 mL/L and 20 g/L, respectively, based on orthogonal test. The effects of three elements on corrosion resistance reduced in turn according to the quantity of Ba(NO3)2, Mn(NO3)2 and NH4H2PO4.
2) With increasing the treating time, solution temperature and pH value, the corrosion resistance of the coating gets better. The micro-cracks in the coating becomes narrower with increasing the pH value.
3) The treating time, solution temperature and pH value are settled to be 5-30 min, 50-70 ℃ and 2.35-3.0.
4) The corrosion resistance of the conversion coating is better than that of manganese phosphate conversion coating through immersion test.
5) The coating is composed of Ba, P, O, Mg, Zn, Mn and Al.
Acknowledgement
The authors thank the financial support from National Project of Scientific and Technical Supporting Programs Funded by Ministry of Science & Technology of China During the 11th Five-year Plan 2006BAE04B05-2 and the Chinese MOST Project 2007CB613705.
References
[1] COLE G S, SHERMA A M. Lightweight materials for automotive applications [J]. Material Characterization, 1995, 35(1): 3-9.
[2] IMMARIGEON J P, HOLT R T, KOUL A K, ZHAO L, WALLACE W, BEDDOES J C. Lightweight materials for aircraft applications [J]. Material Characterization, 1995, 35(1): 41-67.
[3] BALLERIMI G, BARDI U, Bignucolo R, Ceraolo G. About some corrosion mechanisms of AZ91D magnesium alloy [J]. Corrosion Science, 2005, 47(9): 2173-2184.
[4] Gray J E, Luan B. Protective coatings on magnesium and its alloys [J]. Journal of Alloys and Compounds, 2002, 336(1): 88-113.
[5] Shi Z M, Song G L, Atrens A. The corrosion performance of anodized magnesium alloys [J]. Corrosion Science, 2006, 48: 3531-3546
[6] Song Y W, Shan D Y, Han E H. High corrosion resistance multilayer nickel coatings on AZ91D magnesium alloys [J]. Surface Engineering, 2007, 23(5): 329-333.
[7] Zhao M, WU S, LUO J R, Fukuda Y, NAKAE H. A chromium- free conversion coating of magnesium alloy by a phosphate- permanganate solution [J]. Surface & Coatings Technology, 2006, 200(18): 5407-5412.
[8] Chong K Z, SHIH T. Conversion-coating treatment for magnesium alloys by permanganate-phosphate solution [J]. Materials Chemistry and Physics, 2003, 80: 191-200.
[9] NIU LY, JIANG Z H, LI G Y, GU C D, LIAN J S. A study and application of zinc phosphate coating on AZ91D magnesium alloy [J]. Surface & Coatings Technology, 2006, 200(9): 3021-3026.
[10] LIN C S, LIN H C, LIN K M, Lai W C. Formation and properties of stannate conversion coatings on AZ61 magnesium alloys [J]. Corrosion Science, 2006, 48(1): 93-109.
[11] Liu J R, GUO Y N, Huang W D. Study on the corrosion resistance of phytic acid conversion coating for magnesium alloys [J]. Surface & Coatings Technology, 2006, 201(3): 1536-1541.
[12] Yfantis A, Paloumpa I, Schmeier D, Yfantis D. Novel corrosion-resistant films for Mg alloys [J]. Surface and Coatings Technology, 2002, 151(1): 400-404.
[13] Rudd A L, Breslin C, Mansfeld F. The corrosion protection afforded by rare earth conversion coatings applied to magnesium [J]. Corrosion Science, 2000, 42(2): 275-288.
[14] ZHOU W Q, SHAN D Y, HAN E H. Preparation method of non-chromate conversion coating for Magnesium alloys and the solution: CN ZL02132772.6 [P]. 2002.
[15] Nicholas M. Martyak. Internal stresses in zinc-chromate coatings [J]. Surface and Coatings Technology, 1996, 88: 139-146.
[16] ZHOU W Q, SHAN D Y, HAN E H, KE W. Structure and formation mechanism of phosphate conversion coating on die-cast AZ91D magnesium alloy [J]. Corrosion Science, 2007, 50(2): 329-337.
[17] CAO C N, ZHANG J Q. An introduction to electrochemical impedance spectroscopy [M]. Beijing: Science Press, 2002. (in Chinese)
(Edited by HE Xue-feng)
Foundation item: Project(2006BAE04B05-2) supported by the National Project of Scientific and Technology of China during the 11th Five-year Plan; Project(2007CB613705) supported by the Chinese MOST
Corresponding author: HAN En-hou; Tel: +86-24-836885772; E-mail: ehhan@imr.ac.cn