Influence of substitution of Nd3+ for Bi3+ on structure and piezoelectric properties of SrBi2-χNdχNb2O9 (χ=0, 0.1, 0.2 and 0.4)
来源期刊:中国有色金属学报(英文版)2009年第6期
论文作者:SUN Lin 褚君浩 杨平雄 越方禹 李亚巍 冯楚德 毛朝梁
文章页码:1454 - 1458
Key words:bismuth layer-structured ferroelectrics; X-ray Rietveld method; synchrotron radiation XAFS; piezoelectric properties
Abstract: SrBi2-χNdχNb2O9 (χ=0, 0.1, 0.2 and 0.4) bismuth layer-structured ferroelectric ceramics were prepared by the solid-state reaction sintering method. The accurate position of Nd element in SrBi2-χNdχNb2O9 ceramics was determined by the X-ray Rietveld method and Synchrotron radiation X-ray absorption fine structure (XAFS) technology. The partial substitution of Nd3+ for Bi3+ leads to the decrease in the distortion of NbO6 octahedron for SrBi2-χNdχNb2O9 ceramics and also lowers the piezoelectric properties of SrBi2-χNdχNb2O9 ceramics. Meanwhile, the temperature coefficient of resonant frequency (TCF) decreases when Nd element partially replaces Bi element in SrBi2-χNdχNb2O9 ceramics.
基金信息:supported by China Postdoctoral Science Foundation
supported by the National Basic Research Program of China
supported by the National Natural Science Foundation of China
supported by the Natural Science Foundation of Shanghai, China

SUN Lin(孙 琳)1, CHU Jun-hao(褚君浩)1, YANG Ping-xiong(杨平雄)1, YUE Fang-yu(越方禹)1,
LI Ya-wei(李亚巍)1, FENG Chu-de(冯楚德)2, MAO Cao-liang(毛朝梁)2
1. Key Laboratory of Polar Materials and Devices of Ministry of Education,
East China Normal University, Shanghai 200062, China;
2. Shanghai Institute of Ceramics, Chinese Academy of Sciences, Shanghai 200050, China
Received 10 August 2009; accepted 15 September 2009
Abstract: SrBi2-χNdχNb2O9 (χ=0, 0.1, 0.2 and 0.4) bismuth layer-structured ferroelectric ceramics were prepared by the solid-state reaction sintering method. The accurate position of Nd element in SrBi2-χNdχNb2O9 ceramics was determined by the X-ray Rietveld method and Synchrotron radiation X-ray absorption fine structure (XAFS) technology. The partial substitution of Nd3+ for Bi3+ leads to the decrease in the distortion of NbO6 octahedron for SrBi2-χNdχNb2O9 ceramics and also lowers the piezoelectric properties of SrBi2-χNdχNb2O9 ceramics. Meanwhile, the temperature coefficient of resonant frequency (TCF) decreases when Nd element partially replaces Bi element in SrBi2-χNdχNb2O9 ceramics.
Key words: bismuth layer-structured ferroelectrics; X-ray Rietveld method; synchrotron radiation XAFS; piezoelectric properties
Bismuth layer-structured ferroelectrics (BLSFs) have attracted much interest for their fatigue-free properties in nonvolatile ferroelectric thin film random access memory and high temperature piezoelectric devices. BLSFs can be described by the general formula (Bi2O2)2+(Am-1BmO3m-1)2- (m=1, 2, 3, 4, 5, 6). The crystal structure of these compounds consists of fluorite-like (Bi2O2)2+ layers interleaved with (m-1) perovskite-like blocks (Am-1BmO3m-1)2-[1]. SrBi2Nb2O9 (SBN) is a member of BLSFs family (i.e. m=2, A=Sr2+, B=Nb5+), which is composed of Bi2O2 layers and perovskite-like SrNb2O7 units with double NbO6 octahedral layers.
BLSFs have been widely studied because of their excellent fatigue-free properties[2-4]. In previous studies, we have found that Pr3+, La3+, Nd3+and Sm3+ elements can be introduced into Bi site for SBN ceramics and a relaxor behavior is reduced by the substitution of these lanthanide (Ln) elements for Bi ions[5-8]. However, there are few detailed studies about the accurate position of the dopant element like Nd3+ for these Ln-doped SBN ceramics[9]. On the other hand, SBN is suitable for fine tolerance resonator applications due to its low temperature coefficient of resonant frequency (TCF) value, low electromechanical coupling coefficient (Kp), and high mechanical quality factor (Qm). In addition, the substitution of Nd3+ for Bi3+ is most likely to further lower TCF value like the case of Sm-doped SBN[8]. Therefore, in the present work, the accurate position of Nd dopant was investigated by means of the X-ray Rietveld refinement method and XAFS technique and the effects of the Nd dopant on the structure and piezoelectric properties of SrBi2-χNdχNb2O9 (χ=0, 0.1, 0.2 and 0.4) were also studied in detail.
2 ExperimentalThe conventional solid-state reaction sintering method was used to prepare SrBi2-χNdχNb2O9 (χ=0, 0.1, 0.2 and 0.4) ceramics. Details about the preparation procedure were described elsewhere[7]. The ceramic samples for piezoelectric measurement were poled in a silicon oil bath at 180 ℃ under the electric field of about 8-10 kV/mm for 5-15 min.
Phase identification of samples was carried out by X-ray diffractometer (model: D?max2550 V, Rigaku, Tokyo, Japan) with monochromic Cu Kα radiation. Intensities were measured in steps of 0.02? for 1 s in the 2θ range of 10?-120? at room temperature. Crystallographic and structural parameters were refined by the Rietveld method using the TOPAS software. XAFS measurements at Nd-L3 absorption edge were performed using synchrotron radiation at the National Synchrotron Radiation Laboratory of China. The P―E hysteresis loops were measured by a TF analyzer (Model 2000, aixACCT Systems, Germany) at the temperature of 150 ℃ with a frequency of 1 Hz. The piezoelectric measurements were performed for 48 h after the poling. The piezoelectric constants, d33, were measured using a quasi-static d33 meter (Model ZJ-2, Institute of Acoustics, Chinese Academy of Science). The planar coupling factor Kp and the mechanical quality factor Qm were determined using the resonance-antiresonance method. Resonance and anti-resonance frequencies in the temperature range from -20 to 140 ℃ were measured using precision impedance analyzer (Agilent 4294A, USA) combined with a temperature-controlling oven (VT7004, Germany). TCF was calculated using
TCF=Δfr/(fr, 20 ℃×100) (1)
where Δfr is the variation of the resonance frequency within the temperature range from -20 to 80 ℃, and fr, 20 ℃ is the resonance frequency at 20 ℃.
3 Results and discussion
3.1 Analysis of X-ray Rietveld refinement and XAFS measurement for SrBi2-χNdχNb2O9
SrBi2-χNdχNb2O9 (χ=0, 0.2 and 0.4) ceramics powders were carried out by X-ray Rietveld refinement. Fig.1 shows the fitting result of X-ray Rietveld refinement of SrBi1.8Nd0.2Nb2O9 (i.e. χ=0.2) ceramics powder and Table 1 gives the Rietveld refined structural parameters for this sample. Some previous studies about SBN ceramics powders have been reported that the exchange of Sr2+ and Bi3+ occurs in SBN[10-12]. So, in our refinement for SrBi2-χNdχNb2O9, we assumed that some exchange of Sr2+ and Bi3+ existed for these three samples. The refinement was performed in the orthorhombic space group A21am. The results of refinement for these samples are presented in Table 1 and Table 2. It can be seen from Table 2 that the refinement parameters for these samples including three reliability factors (Rwp, Rexp and Rp) are reasonable values, which represent the reliability of the refined structural parameters. Table 1 shows that the exchange amount of Sr2+ and Bi3+ for χ=0.2 sample is about 16.2%. Similarly, this exchange of Sr2+ and Bi3+ also happens for χ=0 and 0.4 samples and the exchange amount is 16.7% and 15.1%, respectively, according to their refinement. On the other hand, considering that the radius of Nd3+(0.111 nm, coordinate number 8) is closer to Bi3+ (0.117 nm, coordinate number 8) and larger than Sr2+ (0.126 nm, coordinate number 8) and both Nd and Bi ions are trivalent, the refinement was performed based on the assumption that Nd3+ occupies the Bi-site in Bi2O2 layers for the Nd-doped SBN samples (χ=0.2 and 0.4). The reasonable structure parameters obtained by Rietveld refinement reflect that Nd3+ replaces Bi3+ for SrBi2-χNdχNb2O9 samples.
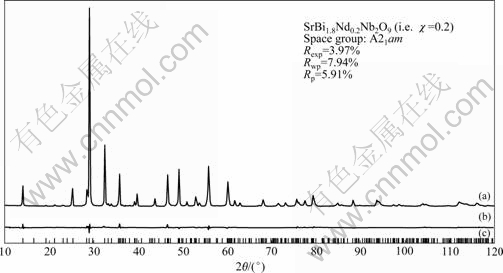
Fig.1 Fitting result of X-ray Rietveld refinement of SrBi1.8Nd0.2Nb2O9 (i.e. χ=0.2) ceramics powder: (a) Experimental data spectrum; (b) Difference between data points and fitted line; (c) Position of diffraction peaks
Table 1 Rietveld refined structural parameters for SrBi1.8Nd0.2Nb2O9 (i.e. χ=0.2)
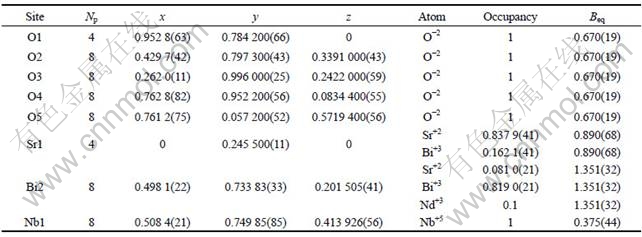
Table 2 Rietveld refined lattice constants for SrBi2-χNdχNb2O9 (χ=0, 0.2 and 0.4)
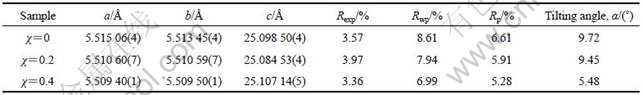
Table 2 lists the lattice constant of SrBi2-χNdχNb2O9 (χ=0, 0.2 and 0.4) samples and their titling angle for NbO6 octahedron, which was obtained by the Rietveld refinement. Compared with those of SBN (i.e, χ=0), the lattice constants a and b decrease by 0.1% and 0.07%, respectively, and the lattice constant c increases by 0.03%. Thus, the structural change mainly occurs in a-b plane when Nd3+ partially substitutes for Bi3+ in the Bi2O2 layer for Nd-doped SBN samples. It should be noted that the titling angle for NbO6 octahedron decreases from 9.72? for χ=0 to 9.45? for χ=0.2 and 5.48? for χ=0.4 sample with the increase in the Nd content for Nd-doped SBN samples. The decrease in the titling angle is related with the 6s2 lone-pair electrons of Bi ions in Bi2O2 layers[8].
In order to further determine whether the doped Nd3+ indeed substitutes for Bi3+ in Bi2O2 layers or not, synchrotron radiation XAFS technique is used to probe the local environment of doped Nd3+ in SBN ceramics. Fig.2 shows the extended-XAFS spectra of Nd-L3 absorption edge in SrBi2-χNdχNb2O9 (χ=0.2 and 0.4) samples. Since Nd-L3 absorption edge lies in the 6 208 eV position and Nd-L2 absorption edge is close to Nd-L3, the range of selected energy varies from 6 100 to 6 700 eV. As illustrated in Fig.2, some vibration peaks appear near the high energy side of Nd-L3 edge, which just is attributed to the extended X-ray absorption fine structure. In addition, It can be seen from Fig.2 that the energy position of Nd-L3 absorption edge is the same and three little vibration peaks behind Nd-L3 edge have the similar shape for χ=0.2 and χ=0.4 samples. These observations reflect that the Nd atoms in χ=0.2 and χ=0.4 samples have the same electrovalent structure and the similar local coordination structure[13-14]. Fig.3 gives Fourier transform magnitudes of the Nd-L3 edge extended-XAFS spectrum of SrBi2-χNdχNb2O9 (χ=0.2 and 0.4) samples. The strongest peak in Fig.3 represents the distance between Nd atom and the nearest coordination atom O (i.e. the first coordination shell peak). It is obvious that the first coordination shell peak of χ=0.2 sample (near 2.2 ?) is the same as that of χ=0.4 sample and both the second coordination shell peaks of χ=0.2 and 0.4 samples appear at about 3.4 ?, which further indicates that the local structures around Nd atoms in χ=0.2 and χ=0.4 samples are very similar. Furthermore, it was ever reported by WACHSMUTH et al[15] that the first and second coordination shell peaks for the Bi atom of Ba0.6Sr0.4Bi2Nb2O9 were near 2.20 and 3.61 ? using synchrotron radiation extended-XAFS technology. More importantly, the configurations of the first and second coordination shell peaks for Nd-L3 edge of SrBi2-χNdχNb2O9 (χ=0.2 and 0.4) samples are very similar to those of Bi-L3 edge of Ba0.6Sr0.4Bi2Nb2O9. These results above further indicate that Nd3+ really occupies Bi-site in Bi2O2 layers for SrBi2-χNdχNb2O9 samples. Therefore, the combination of X-ray Rietveld refinement and synchrotron radiation extended-XAFS technology strongly confirm that the doped Nd3+ successfully substitutes for Bi3+ in SrBi2-χNdχNb2O9 samples.
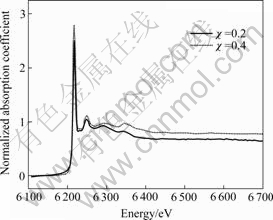
Fig.2 Extended-XAFS spectra of Nd-L3 edge in SrBi2-χNdχNb2O9 (χ=0.2 and 0.4) samples
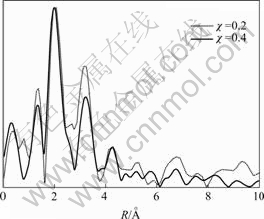
Fig.3 Fourier transform magnitudes of Nd-L3 edge extended-XAFS spectrum of SrBi2-χNdχNb2O9(χ=0.2 and 0.4) samples (R: interatomic distance between Nd atoms and other atoms)
3.2 Ferroelectric and piezoelectric properties
Fig.4 shows the P―E hysteresis loops of SrBi2-χNdχNb2O9 (χ=0, 0.1, 0.2 and 0.4) ceramics measured at 150 ℃. The P―E hysteresis loops of all samples approach saturation under the strong external electric field. With the increase in Nd content, both the coercive field (2Ec) and the remanent polarization (2Pr) gradually decrease. For instance, 2Ec decreases from 91.3l kV/cm for χ =0 to 70 kV/cm for χ=0.2 and 2Pr also decreases from 15.6 μC/cm2 for χ =0 to 11.90 μC/cm2 for χ=0.2. It may be related to the decrease in NbO6 structural distortion due to the increase in Nd content for SrBi2-χNdχNb2O9 (χ=0, 0.1, 0.2) ceramics. As for χ=0.4 sample, its P―E hysteresis loop is not characteristic of the ferroelectric hysteresis loop because of its low transition temperature (Tm=75 ℃) and it is observed that both 2Pr and 2Ec of χ=0.4 sample approach zero. These results indicate that the partial substitution of Nd3+ for Bi3+ leads to the deterioration of the ferroelectric properties of Nd-doped SBN ceramics.
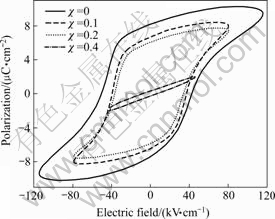
Fig.4 Hysteresis loops of SrBi2-χNdχNb2O9 (χ=0, 0.1, 0.2 and 0.4) samples
Table 3 shows the piezoelectric characteristics of SrBi2-χNdχNb2O9 (χ=0, 0.1 and 0.2) ceramics. As illustrated in Table 3, Nd-doped SBN ceramics have low dielectric constant εr, low piezoelectric constant d33, low electromechanical coupling coefficients Kp and high mechanical quality factor Qm compared with lead zirconate titanate (PZT) ceramics. Furthermore, it should be noted that d33 decreases from 11 pC/N for χ=0 to 6 pC/N for χ=0.2 when Nd content (χ) increases gradually in Nd-doped SBN ceramics. This result is consistent with the decrease in 2Pr due to the substitution of Nd3+ for Bi3+ in Bi2O2 layers. SBN is expected as the candidate for the fine-tolerance resonator application because it has low εr, low Kp, high Qm and low TCF value[16]. However, the TCF value of SBN is not low enough to satisfy the requirement of fine-tolerance resonator application. Thus, the doping-element method was used to decrease the TCF value. For example, there were some research papers which reported that the substitution of Ba2+ for Sr2+ induced the dielectric relaxor behavior and deceased TCF values for (Sr1-χBaχ)Bi2Nb2O9 ceramics [16-17]. Considering that the substitution of Nd3+ for Bi3+ also induces the dielectric relaxor behavior for SrBi2-χNdχNb2O9 ceramics[7], it is naturally expected this substitution can lead to lower TCF values. Fig.5 gives the temperature dependence of resonance frequency of SrBi2-χNdχNb2O9 (χ=0, 0.1 and 0.2) ceramics in the range from -20 to 140 ℃. It can be apparently seen from Fig.5 that TCF values of χ=0.1 and 0.2 samples are lower than that of χ=0 sample. In other words, the substitution of Nd3+ for Bi3+ decreases the TCF values of SBN ceramics. The reason for it is possibly related to the dielectric relaxor behavior of Nd-doped SBN ceramics[17].
Table 3 Piezoelectric characteristics of SrBi2-χNdχNb2O9 (χ=0, 0.1 and 0.2) ceramics
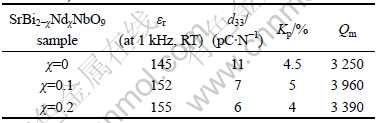
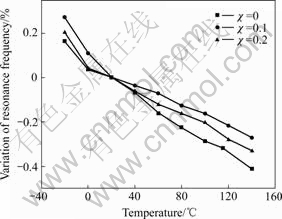
Fig.5 Temperature dependence of resonance frequency of SrBi2-χNdχNb2O9 (χ=0, 0.1 and 0.2) ceramics
4 Conclusions
1) SrBi2-χNdχNb2O9 (χ=0, 0.1, 0.2 and 0.4) ceramics were synthesized by the conventional solid-state reaction sintering method. The substitution of Nd3+ for Bi3+ in the Bi2O2 layers for SrBi2-χNdχNb2O9 ceramics were determined by the combination of X-ray Rietveld refinement and synchrotron radiation XAFS technique. X-ray Rietveld refinement also indicates that the substitution of Nd3+ for Bi3+ leads to the decrease in the structural distortion of NbO6 octahedron, which is described by title angle.
2) The substitution of Nd3+ for Bi3+ decreases the remnant polarization and piezoelectric constant, whereas it decreases the temperature coefficient of resonance frequency values of SBN ceramics. SrBi2-χNdχNb2O9 ceramics can be regarded as one of candidates for fine-tolerance resonator applications.
Acknowledgements
The X-ray absorption spectroscopy of XAFS measurements were performed on synchrotron radiation from beamline U7c at the National Sychrotron Radiation Laboratory in University of Science and Technology of China (USTC). The authors are grateful to PAN Zhi-yun in USTC for discussing the XAFS data and to LIU Fu-sheng in Shenzhen University for his help and advice on Rietveld refinement.
References
[1] AURIVILLIUS B, FANG P H. Ferroelectricity in the compound Ba2Bi4Ti5O18 [J]. Physics Review, 1962, 126: 893-896.
[2] ARAUJO C A, CUCHIARO J D, MCMILLAN L D, SCOTT M C, SCOTT J F. Fatigue-free ferroelectric capacitors with platinum electrodes [J]. Nature, 1995, 374: 627-629.
[3] PARK B H, KANG B S, BU S D, NOH T W, LEE J, JO W. Lanthanum-substituted bismuth titanate for use in non-volatile memories [J]. Nature, 1999, 401: 682-684.
[4] WATANABE H, MIHARA T, YOSHIMORI H, ARAUJO A P. Preparation of ferroelectric thin films of bismuth layer structured compounds [J]. Japanese Journal of Applied Physics, 1995, 34: 5240-5244.
[5] HUANG Shi-ming, FENG Chu-de, CHEN Li-dong, WEI Xiao-wei. Dielectric properties of SrBi2-χPrχNb2O9 ceramics (χ=0, 0.04 and 0.2) [J]. Solid State Communication, 2005, 133: 375-379.
[6] HUANG Shi-ming, SUN Lin, FENG Chu-de, CHEN Li-dong. Relaxor behavior of layer structured SrBi1.65La0.35Nb2O9 [J]. Journal of Physics, 2006, 99: 076104.
[7] SUN Lin, FENG Chu-de, CHEN Li-dong, HUANG Shi-ming. Dielectric relaxation in layer-structured SrBi2-χNdχNb2O9 ceramics (χ=0, 0.05, 0.2, 0.35) [J]. Journal of American Ceramic Society, 2007, 90(1): 322-326.
[8] SUN Lin, FENG Chu-de, CHEN Li-dong, HUANG Shi-ming. Dielectric and piezoelectric properties of SrBi2-χSmχNb2O9 (χ=0, 0.05, 0.1, 0.2, 0.3, and 0.4) ceramics [J]. Journal of American Ceramic Society, 2007, 90(12): 3875-3881.
[9] SUN Lin, CHU Jun-hao, FENG Chu-de, CHEN Li-dong. Analysis of relaxor mechanism and structural distortion for SrBi1.6Nd0.4Nb2O9 bismuth-layer-structured ceramics [J]. Applied Physics Letters, 2007, 91: 242902.
[10] BLAKE S M, FALCONER M J, MCCREEDY M, LIGHTFOOT P. Cation disorder in ferroelectric Aurivillius phases of the type Bi2ANb2O9 ( A=Ba, Sr, Ca) [J]. Journal of Material Chemistry, 1997, 7: 1609-1613.
[11] ISMUNANDAR, KENNEDY B J. Effect of temperature on cation disorder in ABi2Nb2O9 (A=Sr, Ba) [J]. Journal of Material Chemistry, 1999, 9: 541-544.
[12] KENNEDY B J, HUNTER B A. Cation disorder in Pb-doped SrBi2Nb2O9 [J]. Chemistry Materials, 2001, 13: 4612-4617.
[13] SUNSTROM J E, KAUZLARICH S M, ANTONIO M R. Synthesis, structure and characterization of Ce1-χAχTiO3 (0.0≤χ≤≤0.8; A=Sr, Ba) [J]. Chemistry Materials, 1993, 5: 182-191.
[14] NEVES P P, DORIGUETTO A C, MASTERLARO V R, LOPES L P, MASCARENHAS Y P, MICHALOWICZ A, EIRAS J A. XAS and XRD structural characterization of lanthanum-modified PbTiO3 ceramic materials [J]. Journal of Physical Chemistry B, 2004, 108: 14840-14849.
[15] WACHSMUTH B, ZSCHECH E, THOMAS N W, BRODIE S G, GURMAN S J, BAKER S, BAYLISS S C. Structure model of Aurivillius compounds: An EXAFS study [J]. Physical State Solid A, 1993, 135: 59-79.
[16] ANDO A, KIMURA M, SAWADA T, HAYASHI K, SAKABE Y. Piezoelectric and ferroelectric properties of the modified SrBi2Nb2O9 ceramics [J]. Ferroelectrics, 2002, 268: 65-70.
[17] ANDO A, KIMURA M, SAKABE Y. Piezoelectric resonance characteristics of SrBi2Nb2O9-based ceramics [J]. Japanese Journal of Applied Physics, 2003, 42: 150-156.
Foundation item: Project(20080440083) supported by China Postdoctoral Science Foundation; Project(2007CB924901) supported by the National Basic Research Program of China; Project(60677022) supported by the National Natural Science Foundation of China; Project(09ZR1409200) supported by the Natural Science Foundation of Shanghai, China
Corresponding author: SUN Lin; Tel: +86-21-54345123; E-mail: lsun@ee.ecnu.edu.cn
DOI: 10.1016/S1003-6326(09)60051-0

