Article ID: 1003-6326(2005)06-1388-06
Preparation of ZrO2-Al2O3 micro-laminated coatings on stainless steel and their high temperature oxidation resistance
YAO Ming-ming(姚明明)1, 2, HE Ye-dong(何业东)1,
GOU Ying-jun(缑英俊)2, GAO Wei(高 唯)3
(1. Beijing Key Laboratory for Corrosion, Erosion and Surface Technology,
University of Science and Technology Beijing, Beijing 100083, China;
2. School of Chemistry and Chemical Engineering, Ji′nan University,
Ji′nan 250022, China;
3. School of Engineering, The University of Auckland, Private Bag 92019,
Auckland, New Zealand)
Abstract: Micro-laminated ZrO2-Al2O3 coatings were prepared by electrochemical depositing ZrO2 film and Al2O3 film alternatively in ethanol solutions containing aluminum nitrate and zirconium nitrate, with small amounts of yttrium nitrate added respectively into both solutions. The micro-laminated ZrO2-Al2O3 coating is of nanostructure. FE-SEM analyses show that the cross section of the micro-laminated coatings has alternate six-layer films of ZrO2 and Al2O3, with the thickness of each layer in the range of nanometer or submicron. The surface of the micro-laminated coatings is composed of nano-particles. SEM, XRD and mass gain measurement were adopted to study the oxidation resistance of coatings on stainless steel. It has been found that all the coatings are effective in protecting the substrate from oxidation, and micro-laminated coatings exhibit more excellent protectiveness performance. Mechanisms accounting for such effects have been discussed.
Key words: ZrO2; Al2O3; micro-laminated coatings; electro-deposition; oxidation; stainless steel CLC
number: TG172.82 Document code: A
1 INTRODUCTION
In recent years, micro-laminated coatings with a great potential for applications have attracted considerable interest. The micro-laminated coatings can dramatically improve many properties including fracture toughness, fatigue behavior, impact behavior, oxidation resistance, hot corrosion resistance, etc[1]. It is evident that the formability, shape, and the fracture resistance of the coatings during forming and in service depend not only on the properties of the individual layers and their thickness ratio, but also on manufacturing techniques[2].
To date, many techniques have been developed to produce laminated composites, such as chemical vapor deposition(CVD), physical vapor deposition(PVD), electrophoresis, sol-gel and electro-deposition. Among these processes, the electro-deposition is an attractive method for the preparation of micro-laminated coatings since it is simple and inexpensive compared with CVD and PVD, etc. It is well known that electro-deposition methods can produce high quality metallic coatings, but it is rarely used to obtain oxide films. As early as 1993, HE et al[3-11] developed an electrochemical method to prepare oxide coatings or films. That is, the thin oxide films were prepared by an electrochemical deposition-pyrolysis in an organic solvent containing small amounts of yttrium, aluminum, zirconium or chromium salts. Recently, Marinova et al also used this electrochemical deposition method to prepare ZrO2[12, 13], La2O3[14], Al2O3[15] and CeO2[16], and successfully deposited these oxide films on metal substrate in a non-aqueous electrolyte.
To improve the resistance against high-temperature oxidation and electrochemical corrosion of metals, the surface on which the corrosive agents act may be modified by ceramic films. Among the oxides, ZrO2 and Al2O3 coatings have been extensively studied as barriers against oxidation on metal substrates. This is due to the fact that ZrO2 has the advantage of a higher mechanical toughness and a thermal expansion coefficient close to those of many metals, and can act as reactive element. While Al2O3 has a high melting point, stability at high temperature and a very low solubility particularly in molten salt, and it is expected to show an excellent corrosion resistance. Some reports have been dedicated to the study of corrosion protection of metals by ZrO2 or Al2O3 coatings respectively, but few of them involved the use of micro-laminated coatings of ZrO2 and Al2O3 on metals.
In the present study, an electrochemical method using a non-aqueous electrolyte was utilized for deposition of ZrO2 and Al2O3 micro-laminated coatings on stainless steel. The oxidation and spallation resistance of the micro-laminated ceramic coatings were investigated, and mechanisms accounting for such effects were also discussed.
2 EXPERIMENTAL
An electrochemical deposition method was used to prepare micro-laminated oxide coatings on the sample surface. The concentrations of Zr(NO3)4・5H2O+8%(mass fraction, the same below if not mentioned) Y(NO3)3・6H2O and Al(NO3)3・9H2O+1%Y(NO3)3・6H2O were both 0.1mol/L respectively in ethanol solutions. The specimen acted as the cathode, and two platinum sheets(25mm×15mm) were used as the anodes. The distance between the anodes and cathode was 15 mm. The applied current density was about 0.005A/cm2 via adjusting voltage, the deposition time was 20 to 90s, and a post-heat treatment for each run was performed in air at 300℃ for 30min. This process was alternately repeated up to a desired times. In order to compare the oxidation resistance of laminated coatings with that of non-laminated coatings, the deposition conditions of each layer should be the same.
The stainless steel has a composition (mole fraction) of 19.8%Cr, 9.08%Ni, 0.37%Ti, and 70.75%Fe. The specimens were cut to a size of 15mm×10mm×2mm. All surfaces of the specimens were ground to 1500 grit SiC paper, followed by ultrasonic cleaning with alcohol and de-ionized water.
High temperature oxidation of the specimens was carried out in a horizontal furnace at 900℃ in air for 100h. The specimens were placed in quartz crucibles, which had been heated to a constant mass. After a certain time period of oxidation, mass gains were weighed using an electronic balance with an accuracy of 10-5g. Oxidation kinetic curves were plotted separately by using the mass gains and spallation mass of the specimens vs time.
The cross sections and surfaces of the micro-laminated oxide coatings were characterized with a high-resolution FE-SEM(Phillips XL-30).The composition and phases of the oxide scales formed by oxidation at 900℃ for 100h were analyzed by EDS and XRD. The surface morphology of the oxide scales after oxidation at 900℃ for 100h was observed using SEM.
3 RESULTS
Fig.1(a) shows the FE-SEM cross section micrograph of ZrO2 and Al2O3 micro-laminated coatings on substrates heated at 600℃ in air for 1h. The alternate six-layer coatings of ZrO2 and Al2O3 are observed clearly and the thickness of each layer is in the range of nanometer or submicron. Fig.1(b) shows the FE-SEM surface micrograph of ZrO2 and Al2O3 micro-laminated coatings on substrates heated at 600℃ in air for 1h. The coatings are composed of nano-particles, in which a few micro-holes are observed. The sizes of particles by electro-deposition method are almost the same as that of particles obtained by sol gel technique[17], but electro-deposition coatings are denser than sol gel coatings.

Fig.1 FE-SEM micrographs of ZrO2 and Al2O3 micro-laminated films on substrates heated at 600℃ in air for 1h
Fig.2 shows the morphologies of the outer surface of the stainless steels with coated two-layer and six-layer ZrO2 and Al2O3 micro-laminated coatings heated at 600℃ in air for 1h. The coatings of two layers present cracks on the surface, while the coatings by six depositions are homogeneous and continuous. Therefore, six- layer coatings are chosen as the criterion of oxidation resistance experiment.

Fig.2 SEM micrographs of ZrO2 and Al2O3 micro-laminated coatings on stainless steel heated at 600℃ in air for 1h
Oxidation kinetics of stainless steel substrates with and without coatings at 900℃ in air for 100h are illustrated in Fig.3. It can be seen from Fig.3 that all surface-applied ZrO2, Al2O3, or ZrO2 and Al2O3 micro-laminated coatings on the alloy can significantly improve the oxidation resistance at high temperature. The specimen without coatings show a mass gain of about 1.2mg/cm2 after oxidation at 900℃ in air for 100h, compared with 0.6mg/cm2 for the specimen coated with ZZZZZZ(Z: 1 layer of ZrO2 ), 0.5mg/cm2 for the specimen coated with AAAAAA(A: 1 layer of Al2O3), 0.4mg/cm2 for the specimen coated with ZAZAZA. By comparing Fig.3(a) with Fig.3(b), it can be seen that the oxide scale spallation shows a similar trend as the mass gain, that is, the large mass gains correspond to the large-scale spallation.
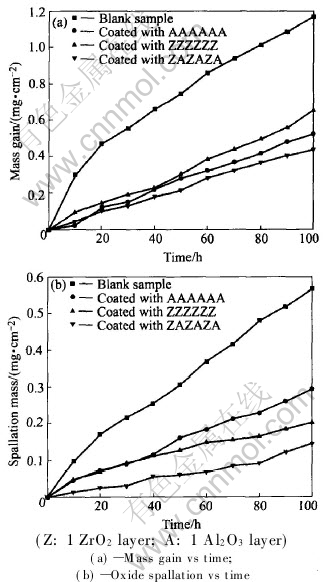
Fig.3 Oxidation kinetics of stainless steel substrates with and without coatings at 900℃ in air for 100h
SEM surface morphologies of the oxide scale after oxidation at 900℃ in air for 100h are shown in Fig.4. The alloy without coatings shows a rough surface with two different areas, the bright area and dark area as shown in Fig.4(a). In the bright area, the oxide scale is mainly composed of Cr and Fe oxides, the dark areas are holes and pores, indicating that the scale has broken away. The specimen with ZrO2 and Al2O3 micro-laminated coatings shows uniform scale morphology with no obvious spallation, as observed in Fig.4(b). The specimens with ZrO2 (Fig.4(c)) or Al2O3 (Fig.4(d)) coatings also show scales spallation to a certain degree. The leaf-like areas in Fig.4(c) are Zr rich oxide.

Fig.4 SEM surface morphologies after oxidation at 900℃ in air for 100h
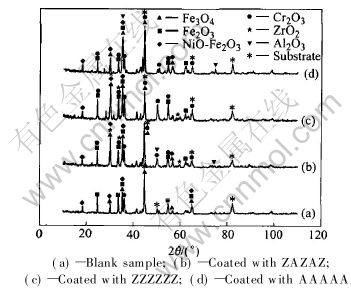
Fig.5 XRD spectra of specimens after oxidation at 900℃ in air for 100h
Fig.5 shows the XRD spectra of the four specimens with and without coatings after oxidation at 900℃ in air for 100h. It is shown that the oxide scales formed on the uncoated alloy are mixed Fe and Cr oxides. EDS analysis shows that the mass ratio of Cr to Fe for the specimens uncoated is 1.1, the mass ratio of Zr to Al and Fe for the specimens coated with ZrO2 and Al2O3 micro-laminated coatings is about 4∶2∶1, the mass ratio for the specimens coated with ZrO2 is about 3∶3∶1, and the mass ratio of Al to Cr and Fe for the specimens coated with Al2O3 is about 3∶2∶1, respectively. These results can be explained as that the sample coated with ZAZAZA six-layer coatings has no oxide spallation during oxidation at 900℃, so it still maintains the compositions of the original coatings. Since the diffusion velocity of Fe in alloy is faster than that of Cr in the alloy, there is a small quantity of Fe diffusing into surface. As to the samples coated with ZZZZZZ or AAAAAA, because they partially have oxide spallation, subjacent oxide films containing a large amount of Cr are exposed to surface; consequently, a lot of Cr can be detected by EDS spectrum. However, the Fe content of the surface of samples coated with ceramic coatings is very low, compared with the blank sample.
4 DISCUSSION
Micro-laminated coatings on stainless steel exhibit most excellent protection effect. Mechanisms accounting for such effects are discussed as follows.
4.1 Synergic effects of ZrO2 and Al2O3
The micro-laminated coatings can make use of the characteristics of ZrO2 and Al2O3, respectively. Al2O3 (containing trace yttrium) layer on the alloy surface have a positive influence on the oxidation and spallation resistance[6]. This layer can decrease the inward transport of oxygen to a certain degree, thereby reduce the oxidation rate. How-ever, Al2O3 layer may still spall at high temperature for a long time, because of thermal stress between the layer and alloy substrate, which is corresponding to the sample coated with AAAAAA as shown in Figs.4 and 5. As to ZrO2(containing trace yttrium) layer, owing to both Zr and Y are reactive elements, applied ZrO2 thin films on alloys containing Cr can promote the selective oxidation of Cr in alloys and enhance the adhesion between the oxide scale and the alloy. These properties are beneficial to enhancing the oxidation resistance of the alloy. But in another aspect, ZrO2 is a good oxygen ion conductor, which should restrict the ability for ZrO2 films to resist the oxidation of alloy imperfectly. This result is corresponding to the sample coated with ZZZZZZ as shown in Figs.4 and 5.
When a combination of Al2O3 layers with ZrO2 layers is in micro-laminated coatings on specimens, the oxidation resistance of the specimens can be improved greatly. From Figs.4 and 5, compared with samples coated with AAAAAA or ZZZZZZ, the mass gain and oxide spallation mass of the sample coated with ZAZAZA are less, and no evidence of spallation is observed. This is probably due to the fact that the thermal expansion coefficient of ZrO2 is very close to those of many metals or alloys, and suitable to act as intermediate layer, while Al2O3 is a very good diffusion barrier material, and suitable to act as top layer in micro-laminates. When micro-laminates are designed as a form of ZAZAZA…, advantages of ZrO2 and Al2O3 can complement, that is, ZrO2 can make the best of enhancing the adherence between coatings and substrates and preventing crack initiation, while Al2O3 can make the best of preventing diffusion of substrate species into coatings or vice versa during annealing and enhancing corrosion protection of coating and substrate, so the micro-laminated coatings of ZrO2 and Al2O3 can significantly improve the oxidation resistance of the alloys.
4.2 Effects of micro-laminated coatings
Micro-laminated ceramic coatings with a great potential for applications in the near future may be one of the most effective ways to toughen ceramics. From Fig.2, we can see that the thickness of each layer is in the range of nanometer or submicron in the laminated ceramic coatings. The layer spacing is so small that the defects size and dislocation movement can be effectively suppressed by a large number of interfaces in micro-laminated ceramic coatings. Consequently, the properties of the micro-laminated ceramic material are superior to those of appropriate single material. In deed, the micro-laminated ceramic coatings are more effective in resisting oxidation of stainless steel substrates at high temperature.
Two dominant ceramic toughening mechanisms, crack deflection and crack blunting, are shown schematically in Fig.6[18]. In many laminated systems, layer de-lamination can occur ahead of an advancing crack or as the result of a crack encountering an interface. These local de-laminations can result in crack deflection, which can significantly reduce the local stress intensity because of the large deviation in crack path. Crack blunting can be a result of propagating crack encountering a ruptured region or a large and hard particle. Further crack growth requires re-nucleation of the crack in the micro-laminated ceramic layer. This arresting and re-nucleation process results in a significant increase in the amount of energy required for crack growth. Consequently, micro-laminated ceramic materials can dramatically improve many properties including fracture toughness, corrosion resistance, etc.
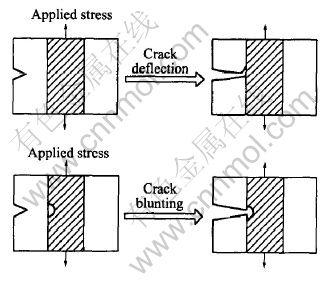
Fig.6 Schematic diagram of toughening mechanisms for micro-laminated composites
4.3 Effects of nano-composites
From Fig.2, the micro-laminated coating surfaces are composed of nano-particles, so the effects of nano-composites cannot be ignored, either. According to Refs.[19, 20], nano-phase can enhance the adherence between ceramic coatings and alloy substrates, more effectively peg cracks and prevent crack initiation, resist high flux of heat into substrates, promote the outwards diffusion of the solute element in the alloy and reduce the critical content required to form a selective oxide scale. Therefore, nano- composite coatings are effective in resisting the oxidation of the alloys at high temperature.
5 CONCLUSIONS
1) The micro-laminated ceramic coatings are prepared using electrochemical deposition. FE-SEM analyses show that the cross section of the micro-laminated coatings is composed of alternate six-layer coatings of ZrO2 or Al2O3, and the thickness of each layer is in the range of nanometer or submicron. The surface of the micro-laminated coatings is composed of nano-particles.
2) The micro-laminated coatings of ZrO2 and Al2O3 show excellent protectiveness to stainless steel at high temperature, which is superior to that of coatings of only single ZrO2 or Al2O3.
3) The relevant mechanisms of the excellent performance of the micro-laminated ceramic coating may be that the layer spacing is so small that the defects size and dislocation movement can be effectively suppressed by a large number of interfaces in micro-laminated ceramic coatings. Consequently, the properties of the micro-laminated ceramic material are superior to those of single material.
REFERENCES
[1]Lesuer D R, Syn C K, Sherby O D, et al. Mechanical behavior of laminated metal composite [J]. Int Mater Rev, 1996, 41(5): 169-197.
[2]Wadsworth J, Lesuer D R. Ancient and modern laminated composites [J]. Mater Charact, 2000, 45: 289-313.
[3]LU Xin-ying, ZHU Ri-zhang, HE Ye-dong. An electrochemical method to prepare oxide films [J]. CN 93100181.1, 1993.
[4]HE Ye-dong, Stott F H. The selective oxidation of Ni-15%Cr and Ni-10%Cr alloy promoted by surface- applied thin oxide films [J]. Corrosion Science, 1994, 36(11): 1869-1884.
[5]HE Ye-dong, Stott F H. The effects of thin surface- applied oxide coatings on the selective oxidation of alloys [J]. Corrosion Science, 1996, 38: 1853-1868.
[6]LU Xin-ying, ZHU Ri-zhang, HE Ye-dong. Electrodeposited Thin oxide films [J]. Surface and Coatings Technology, 1996, 79: 19-24.
[7]LU Xin-ying, ZHU Ri-zhang, HE Ye-dong. Reactive-element effect of electrodeposited Y2O3 oxide films on the oxidation of Fe-25Cr and Fe-25Cr-10Al alloys [J]. Oxidation of Metals, 1995, 43(3/4): 217-236.
[8]LU Xin-ying, ZHU Ri-zhang, HE Ye-dong. Electrophoretic deposition of MCrAlY overlay-type coatings [J]. Oxidation of Metals, 1995, 43(3/4): 353-362.
[9]QI Hui-bin, Lees D G. The effects of surface-applied oxide films containing varing amounts of yttria, chromia, or alumina on the high-temperature oxidation behavior of chromia-forming and alumina-forming alloys [J]. Oxidation of Metals, 2000, 53: 507-527.
[10]LI Zhen-wei, GAO Wei, HE Ye-dong. Oxidation behaviour of a Ti3Al-Nb alloy with surface thin oxide films [J]. Oxidation of Metals, 2001, 56(5-6): 495-516.
[11]MA Jing, HE Ye-dong, WANG De-ren, et al.The effects of pre-oxidation and thin Y2O3 coating on the selective oxidation of Cr18-Ni9-Ti steel [J]. Materials Letters, 2004, 58: 807-812.
[12]Stefanov P, Stoychev D, Valov I, et al. Electrochemical deposition of thin zirconia films on stainless steel 316L [J]. Materials Chemistry and Physics, 2000, 65: 222-225.
[13]Espitia-Cabrera I, Orozco-Hernández H, Torres-Sánchez R. Synthesis of nanostructured zirconia electrodeposited films on AISI 316L stainless steel and its behaviour in corrosion resistance assessment [J]. Materials Letters, 2003, 58:191-195.
[14]Stoychev D, Valov P, Stefanov I, et al. Electrochemical growth of thin La2O3 films on oxide and metal surfaces [J]. Mater Sci Eng, 2003, C23:123-128.
[15]Stoychev D, Stefanov P, Nikolova D, et al. Preparation of Al2O3 thin films on stainless steel by electrochemical deposition [J]. Surface and Coatings Technology, 2004, 180-181: 441-445.
[16]Stefanov P, Atanasova G, Stoychev D, et al. Electrochemical deposition of CeO2 on ZrO2 and Al2O3 thin films formed on stainless steel [J]. Surface and Coatings Technology , 2004, 180-181: 446-449.
[17]LI Hai-bin, LANG Kai-ming, MEI Le-fu, et al. Oxidation resistance of mild steel by zirconia sol-gel coatings [J]. Mater Sci Eng A, 2003, A341: 87-90.
[18]Ritchie R O. Mechanisms of fatigue crack-propagation in metals, ceramics and composites [J] Mater Sci Eng A, 1988, A103(1): 15-28.
[19]Nihara K. New desigh concept of structural ceramics-ceramic nanocomposites [J]. J Ceram Soc Jpn, 1991, 99: 947-982.
[20]LIU Z, GAO W, Dahm D, et al. Oxidation behavior of micro-crystalline of 310 stainless steel [J]. J Univ Sci Technol Beijing, 1997, 5: 51-56.
(Edited by LONG Huai-zhong)
Foundation item: Project(50271010) supported by the National Natural Science Foundation of China
Received date: 2005-03-17; Accepted date: 2005-05-31
Correspondence: HE Ye-dong, Professor, PhD; Tel: +86-10-62332715; E-mail: htgroup@mater.ustb.edu.cn