J. Cent. South Univ. Technol. (2010) 17: 202-210
DOI: 10.1007/s11771-010-0031-5 
Preparation of new tissue engineering bone-CPC/PLGA composite and its application in animal bone defects
ZHENG Zhi(郑治)1,2, WANG Jian-long(王剑龙)1, MI Lei(米雷)1,
LIU Yong(刘咏)2, HOU Guang-hui(侯光辉)1, CHEN Dan(陈旦)3, HUANG Bai-yun(黄伯云)2
1. The Third Xiangya Hospital, Central South University, Changsha 410011, China;
2. State Key Laboratory of Powder Metallurgy, Central South University, Changsha 410083, China;
3. Xiangya School of Medicine, Central South University, Changsha 410013, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2010
Abstract: To investigate the feasibility of implanting the biocomposite of calcium phosphate cement (CPC)/polylactic acid-polyglycolic acid (PLGA) into animals for bone defects repairing, the biocomposite of CPC/PLGA was prepared and its setting time, compressive strength, elastic modulus, pH values, phase composition of the samples, degradability and biocompatibility in vitro were tested. The above-mentioned composite implanted with bone marrow stromal cells was used to repair defects of the radius in rabbits. Osteogenesis was histomorphologically observed by using an electron-microscope. The results show that compared with the CPC, the physical and chemical properties of CPC/PLGA composite have some differences in which CPC/PLGA composite has better biological properties. The CPC/PLGA composite combined with seed cells is superior to the control in terms of the amount of new bones formed after CPC/PLGA composite is implanted into the rabbits, as well as the speed of repairing bone defects. The results suggest that the constructed CPC/PLGA composite basically meets the requirements of tissue engineering scaffold materials and that the CPC/PLGA composite implanted with bone marrow stromal cells may be a new artificial bone material for repairing bone defects because it can promote the growth of bone tissues.
Key words: biomaterials; mesenchymal stem cells (MSCs); calcium phosphate cement (CPC); polylactic acid-polyglycolic acid (PLGA); bone defects
1 Introduction
Repairing of bone defects, especially large segmental bone defects caused by tumors, fracture or trauma, has been a huge difficulty harassing orthopedic and cranio-facial surgery. Clinically, there are three common treatments: autogenous bone graft, allogeneic bone transplantation and synthetic bio-substitute transplantation. However, they all have their own disadvantages or shortcomings, such as increased trauma and limited supply for autogenous bone graft, problems of immunogenic response for allogeneic bone transplantation, and poor biocompatibility for synthetic bio-substitute transplantation. In 1995, CRANE et al [1] proposed a principle of a method based on engineering and life science, i.e., the concept of bone tissue engineering. This concept represents a completely new treatment mode in which seed cells are combined and cultured with a biological carrier material (i.e., scaffold) to form bone tissues with physiological function in the body so as to replace and repair the function of diseased bone tissues. This method is promising in solving inadequacies of various methods of repairing bone defects, and thus may bring hope for repairing large segmental bone defects. For the reasons above, it has become a focus in study of bone materials.
Requirements of ideal bone tissue engineering scaffold material are as follows [2].
(1) A good biocompatibility. Besides the general requirements of biomedical materials, such as non-toxic and non-teratogenic, the scaffold materials will also be conducive to seed cell adhesion and proliferation. Moreover, their degradation products not only have no poisoning effect on cells but also do not cause any inflammatory reaction, and are even beneficial to cell growth and differentiation.
(2) A good biodegradability. Matrix materials should be degradable after it finishes its role of scaffold, with degradation rate corresponding to cell growth rate and degradation time artificially regulated in accordance with tissue growth characteristics.
(3) Three-dimensional porous structure. Matrix materials can be processed into three-dimensional structure, with the best porosity over 90% and a high ratio of area to volume. This structure can provide large surface area and space, which is conducive to cell growth and adhesion, deposition of extracellular matrix, entry of nutrients and oxygen, excretion of metabolites, as well as the growth of blood vessels and nerves.
(4) A good plasticity and a certain degree of mechanical strength. Matrix materials should have a good plasticity, being made into a certain shape in advances, and have a certain degree of mechanical strength, providing support for regenerated tissues until regenerated tissues have their own biomechanical properties.
(5) Regulation of surface micro-environment. Interaction between cells and materials should be conducive to cell adhesion and proliferation, and more importantly, able to activate cell-specific gene expression and maintain normal cell phenotype expression. Among them, the cell adhesion to materials, which is basic for cell proliferation and differentiation is mainly regulated by receptor ligand on the cell surface.
At present, scaffold materials applied in bone tissue engineering are natural biomaterials, bioceramic materials, biopolymer materials and biocomposite materials. However, each kind of materials has its own merits and shortcomings and cannot fully meet the above requirements. Thus, the shortcomings of current scaffold materials have become factors limiting the development of bone tissue engineering research [3-4].
In this work, calcium phosphate cement (CPC) was combined with polylactic acid-polyglycolic acid (PLGA) by an improved manufacturing process and selected bone marrow stromal cells that have many advantages (e.g., strong proliferation ability, strong ability to differentiate into osteoblasts, and easy preparation) as seed cells. The preparation method and physico-chemical properties of the CPC/PLGA composite were studied. In addition, the osteogenic activity of this new tissue engineering bone was also investigated by implanting the cultured composite of CPC/PLGA with the seed cells-MSCs into the bone defect areas in animals, in hope of developing a new superior scaffold material.
2 Experimental
2.1 Preparation of CPC/PLGA scaffold
PLGA was added with a small amount of liquid nitrogen and then ground adequately in a milling pot. PLGA particles screened by a 0.3 mm stainless steel sieve were collected. CPC (Shanghai Rebone Biomaterials Co., Ltd., China) and PLGA particles were weighted respectively and mixed evenly. The mixture was stirred into pastes by adding a setting fluid at a ratio of mass (g) to volume (mL) of 3:1. The pastes were then filled into a mould and compressed for shaping. After standing at room temperature for 15 min, the shaped pastes were immersed into Ringer’s solution for 3 d and then dried in air.
2.2 Determination of physico-chemical properties of materials
2.2.1 Setting time
The initial and final setting times were determined using Gilmore needles method [5], i.e., the initial setting time was measured via a needle that was light in mass (m=113.4 g) and large in terms of cross sectional area (d=2.13 mm), and the final setting time was determined by a needle that was heavy in mass (m=453.6 g) and small in terms of cross sectional area (d=1.06 mm).
2.2.2 Compressive strength and elastic modulus
Compressive strength was tested immediately after immersion without drying in order to obtain a result close to the in vivo actual strength. The compressive strength of the cylindrical sample (diameter of 6 mm and height of 9 mm) was tested on a CSS-44100 electronic universal testing machine at a compression rate of 1 mm/min. Two end surfaces of the samples were maximally rubbed down using a piece of sand paper before testing. The averages of six parallel samples were used to represent the results of the experiment.
The formula for calculating compressive strength is shown as follows:
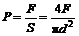 (1)
(1)
where P represents the compressive strength of the test sample, MPa; F refers to the tolerable maximal pressure load of the sample, N; and d signifies the diameter of the sample, mm.
The compressive elastic modulus of the cylindrical sample was treated as elastic modulus. The crushed sample was then immersed into acetone and vacuum- dried for 24 h to terminate the hydration reaction of CPC. XRD and SEM analyses were then performed.
2.2.3 Degradability in vitro
The prepared CPC/PLGA composite was immersed into the body-fluid-simulating Ringer’s solution and taken out a moment later. The degradation of the material was observed by SEM. The shape, size and connection of large pores left after degradation of PLGA were observed. Porosity was measured by using the mercury intrusion method.
2.2.4 Phase composition
The phase composition of the powder sample was analyzed by an X-ray diffractometer with a D/max- 2550VB (Rigaku, Japan) at a voltage of 40 kV and a current of 300 mA, using Cu target and Ka rays.
2.2.5 Microstructure
The microstructure of the sample was observed by a JSM-5600LV scanning electron microscope (SEM) (Japan).
2.3 Isolation and culture of rabbit bone marrow stromal cells
The inner wall of a 5 mL syringe was moistened using a normal saline solution containing 2 000 U/mL heparin. Under a sterile condition, the needle of a 20 mL syringe was inserted into the iliac medullary cavity from the iliac crest of the rabbit that had been anesthetized by intraperitoneally injecting pentobarbital sodium. 1 mL of saline solution containing 1 000 U/mL heparin was sucked into 5 mL syringe. The heparin saline was mixed evenly with a total of 5 mL bone marrow immediately after the marrow was aspirated bilaterally. The mixture was laid in a Percoll separation medium of the same volume and then low-speed centrifuged at 1 000 r/min for 5 min. Then, the supernatant was removed and single-cell suspension was prepared. A blood cell counting chamber was used for counting. The cells were inoculated in a 25 cm2 culture flask at a density of 5×105 mL-1. A culture medium was then added to make the volume up to 5-8 mL. The cells were then incubated in a saturated humidity incubator under conditions of 37 ℃ and 5% CO2. The medium was semi-replaced after 24 h and totally replaced after 72 h. Afterwards, total replacement of the medium was done every 2-3 d. An inverted microscope was used for daily observation. The primary cells were digested by 0.25% pancreatin for 3 min and serially subcultured at a density of 1×105 mL-1 according to a ratio of 1:2.
2.4 Combined culture of cells and materials
After being digested by 0.25% pancreatin, passage 2 cells were inoculated onto the material at a density of 5×107 mL-1 and then incubated in a saturated humidity incubator under conditions of 37 ℃ and 5% CO2.
2.5 Animal grouping and observation indexes
Forty Japanese white rabbits (irrespective of sex) weighing 2.5-3.0 kg were randomized into four groups (10 rabbits per group). After the animal was anesthetized, the radius was exposed by layer-by-layer dissociation. A dental saw was used to cut a length of 15 mm radial and periosteal defects [6]. The bone-repair material was implanted into the bone defects after it was washed by a normal saline solution. A new tissue engineered bone was implanted in group A; the CPC/PLGA composite was implanted in group B; CPC combined with seed cells was used for implantation in group C; and simple CPC was implanted in group D. The surrounding soft tissues were sutured layer-wise via a 3/0 silk thread. The animals were kept in separate cages after operation. The general condition and incision of the animals were observed [7-8]. The growth of callus was analyzed through X-ray film observation taken by exposing animals in each group to the same irradiation condition. The formation of new bones in the bone defect areas was grossly observed. For electron microscopy, blunt cross sections near two ends of the bone material were collected in each group at week 2, 4 and 8, respectively. A JSM-5600LV SEM was used to obtain images of the samples that were made into a size of 5 mm×1 mm×1 mm. After the animals were executed, the samples were collected from the repairing area of bone defects for histological observation. The samples were sectioned after decalcification, fixation in formaldehyde for 24 h, and paraffin imbedding. Following hematoxylin-eosin staining, the histomorphology of the implanted material was observed under a light microscope.
3 Results
3.1 Physico-chemical properties
3.1.1 Setting time
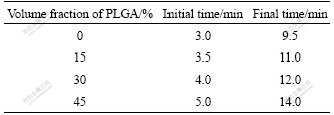
The CPC-PLGA composite powder was set by using ratios of solid (mass, g) to liquid (volume, mL) being 2:1, 3:1 and 4:1, respectively. Table 1 shows the setting time of CPC-PLGA composites containing different volume fractions of PLGA.
Table 1 Setting time of CPC-PLGA composites containing different volume fractions of PLGA
3.1.2 Compressive strength and elastic modulus
Changes in compressive strength of the CPC-PLGA composites containing different volume fractions of PLGA are shown in Fig.1. As shown in Fig.1, the maximal values (24.3 and 40.5 MPa, respectively) occurred when the volume fraction of PLGA reached 30%. Fig.2 displays changes in elastic modulus of the CPC-PLGA composites with different volume fractions of PLGA, and the elastic modulus of the CPC-PLGA composites ranges between 2.5 and 13.8 GPa.
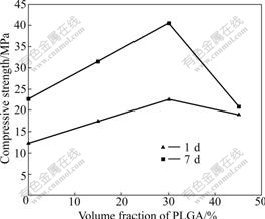
Fig.1 Compressive strength of CPC-PLGA composites varying with volume fraction of PLGA
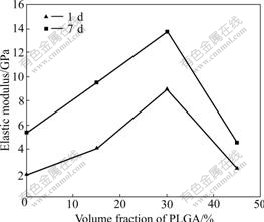
Fig.2 Elastic modulus of CPC-PLGA varying with volume fraction of PLGA contents
3.1.3 Degradability in vitro and analysis of physical phase
Fig.3 shows the XRD patterns of the CPC-PLGA composite samples containing different volume fractions of PLGA after being immersed in the simulated body fluid, and the position of each diffraction peak is basically the same except the intensity of each diffraction peak.
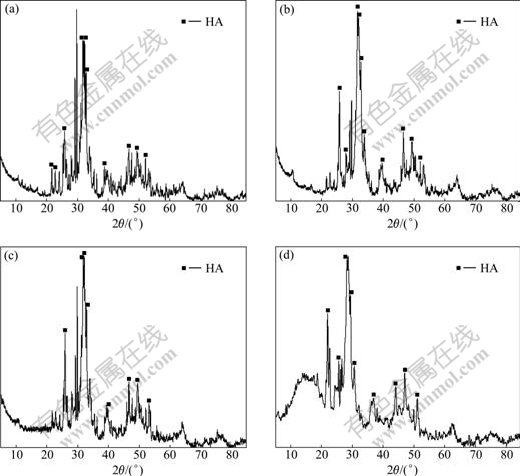
Fig.3 XRD patterns of different composites after being soaked for 7 d (HA: denotes hydroxyapatite): (a) CPC; (b) CPC-15%PLGA composite; (c) CPC-30%PLGA composite; (d) CPC-45%PLGA composite
3.1.4 Microstructures
Figs.4 and 5 show SEM images obtained under various magnifications for simple CPC material and CPC-30%PLGA composite material after being immersed in simulated body fluid for 7 d. As shown in Figs.4(a) and 5(a), micromorphologic differences between the hydration products of simple CPC and that of CPC- 30%PLGA are not significant, because the two hydration products exhibit a typical micromorphology of hydrated CPC and a loose porous structure. The changes in the aggregate morphology of HA crystals and the hydration product of CPC-30%PLGA composite are observed (see Fig.5(c)). As shown in SEM image in Fig.4(c), the aggregate morphology of the hydration products (HA) of simple CPC is rod-shaped (diameter of 70-100 nm and length of 0.2-0.5 mm). The hydration products (HA) of CPC-30%PLGA composite aggregate into different sizes (0.2-0.5 mm) of elliptical granules that are stacked tightly (see Fig.5(c)). In addition, as seen in Fig.5, numerous micropores (diameter of 1-5 mm) that are mutually connected appear in the microstructure of the CPC-30%PLGA composite.

Fig.4 SEM images of CPC soaked for 7 d: (a) Low magnifica- tion; (b) Middle magnification; (c) High magnification

Fig.5 SEM images of CPC-30%PLGA soaked for 7 d: (a) Low magnification; (b) Middle magnification; (c) High magnification
3.2 General condition of animals
In each group, post-operative animals were in a good general condition and wounds were healed completely 7 d after operation without occurrence of infection. No deaths occurred during feeding. No significant swelling, effusion and sinus formation could be seen in the affected limbs.
3.3 X-ray films and gross observation on part of samples
Group A: there is no definite borderline between the graft and normal bones on the 56th day after operation and the graft is morphologically irregular (Fig.6(a)). Group B: the boundary between the graft and normal bones is further blurred on the 56th day after operation and the graft is morphologically irregular (Fig.6(b)). Group C: bone density of the defect areas is increased on the 56th day after operation (Fig.6(c)). Group D: callus slowly grows. The graft is surrounded by a great amount of calluses on the 56th day after operation. Changes in the morphology of the graft are observed and shown in Fig.6(d).
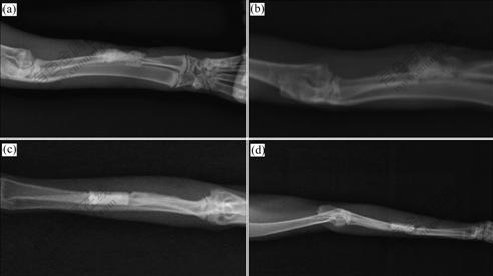
Fig.6 X-ray films of different groups on the 56th day after surgery: (a) Group A; (b) Group B; (c) Group C; (d) Group D
3.4 Histological examination
3.4.1 Light microscopy
Group A: a great number of mature bone trabeculaes can be seen on the 56th day after operation. In addition to relatively mature osteocytes and bone matrix, numerous mature osteoblasts and osteoclasts can also be observed in the bone trabeculaes (Fig.7(a)). Group B: calluses of bilateral broken ends are merged on the 56th day after operation, and few bone trabeculaes appear. Inside the bone trabeculaes, there are a large number of chondrocytes that tend to be mature and a small number of mature osteoblasts and osteoclasts (Fig.7(b)). Group C: the bone defect areas are filled with new bones on the 56th day after operation. The mature bone trabeculaes observed are less than those seen in group A. However, inside the bone trabeculaes, more mature chondrocytes can be seen with a few of osteoblasts and osteoclasts (Fig.7(c)). Group D: most of the defect areas are filled with fibrous tissues on the 56th day after operation. Proliferation of a small portion of chondrocytes as well as interspersed bone trabeculae and bone matrix formation can be seen (Fig.7(d)).
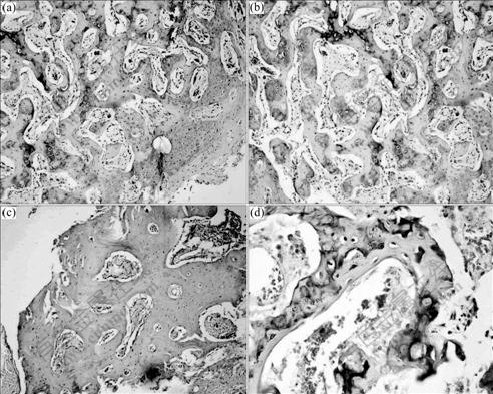
Fig.7 Light microscope pictures of different groups on the 56th day after surgery: (a) Group A; (b) Group B; (c) Group C; (d) Group D
3.4.2 Electron microscopy
On the 28th day after operation, in group A, most of the defect areas are filled with collagen fibrils that are interweaved into large collagen fiber bundles. The collagen fiber bundles, which are arranged in a loose and disordered manner, are embedded in bone cements and decalcified bone matrix particles. Mature bone trabeculaes are formed (Fig.8(a)). In group B, in the implantation area, there are interlaced collagen fibers, but no deposition of flaky or granular calcium salt crystals can be seen. The formation of bone trabeculaes, which is not significant, is superior to that in groups C and D, but inferior to that in group A (Fig.8(b)). In group C, in the implantation area, closely arranged and interlaced collagen fibers can be seen mainly among CPC particles. There are a small portion of precipitates of flaky and granular calcium salt crystals and a few of the formed bone trabeculaes (Fig.8(c)). In group D, most of them observed are closely arranged and interlaced collagen fibers.

Fig.8 SEM images of different groups on the 28th day after surgery: (a) Group A; (b) Group B; (c) Group C; (d) Group D
On the 56th day after operation, a large number of primary medullary cavities form in group A, and so do the mature bone trabeculaes (Fig.9(a)). In group B, most of bone trabeculaes form, and there are more bone trabeculaes than those formed in groups C and D, but less than those in group A. The formation of primary medullary cavity in group B is not comparable to that in group A (Fig.9(b)). In group C, no significant primary medullary cavity can be seen excerpt for a few of bone trabeculae formed in the implantation area (Fig.9(c)). In group D, no significant primary medullary cavity can be seen and only a very few of bone trabeculaes form in the implantation areas (Fig.9(d)).
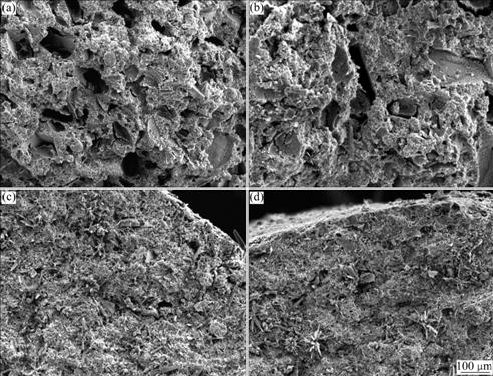
Fig.9 SEM images of different groups on the 56th day after surgery: (a) Group A ; (b) Group B; (c) Group C; (d) Group D
4 Discussion
Scaffold material plays an important role in bone tissue engineering and is a key influential factor for clinical application of tissue engineering bone. Currently, there are two types of cell matrix materials used in bone tissue engineering: natural cell matrix material and synthetic cell matrix material. However, these two matrix materials have different shortcomings. For instance, the disadvantages of CPC include great fragility and slow degradability in vivo. PLGA has such shortcomings as inadequate hydrophilicity, poor ability to adhere cells, and decreased pH values during degradation, which may cause aseptic inflammation. The preparation process for CPC endows its natural microporous structure, and bone conductivity affects its mechanical properties. The mechanical strength of the material decreases as the pore size or porosity increases. When the porosity reaches 63%, the compressive strength of CPC will be nearly 0 [9]. On the other hand, a smaller pore size or lower porosity may increase the mechanical strength of the material, which is not helpful for the inward growth of new bones. Therefore, good bone conductivity of the material should be maintained while the porosity and pore size should not be blindly reduced. Based on the description above, it is speculated that the shortcomings of simple CPC and PLGA matrix materials may be overcome and scaffold materials may be optimized by mixing CPC and PLGA according to a certain ratio. This study induced setting of the CPC/PLGA composite by using a ratio of solid (mass, g) to liquid (volume, mL) of 3:1. By gross observation, imaging analysis, histological examination and light microscopy observation, it is found that as the volume fraction of PLGA in the composite increases, the setting time is slightly prolonged but the setting speed of the composite is not adversely affected significantly. As the volume fraction of PLGA increases, the compressive strength of the CPC/PLGA composite is slightly increased compared with that of simple CPC. This may be attributed to calcium lactate (chemical reaction: Ca2++ 2CH3CHOHCOO-→Ca(CH3CHOHCOO)2), which is produced in chemical reactions between lactic acid groups in polylactic acid released after hydrolysis of a portion of PLGA and calcium ions contained in the CPC powder, resulting in accelerated setting reaction of CPC and increased compressive strength of the composite material. However, when the volume fraction of PLGA is further increased up to 40%, the connecting structure among CPC particles in the composite material becomes loose and the stable structure is destructed due to the presence of a large amount of PLGA; meanwhile, the strength of the CPC/PLGA composite is contrarily decreased slightly due to reduced strength of PLGA itself caused by hydrolysis and softening of PLGA. As the volume fraction of PLGA in the composite material increases, the compressive elastic modulus of the material is increased first to a range, which is basically consistent with the range (1.85-13.0 GPa) required for human bone elastic modulus [10], and then decreased when the volume fraction of PLGA reaches 40%. Based on these findings, it is assumed that the CPC/PLGA composite material may have good properties if the solid-liquid ratio is set to be 3:1. Further detections show as follows. (1) The initial and final setting time of this material are 3.5-5.0 min and 11-14 min, respectively, which basically meet the requirements for the setting time of clinically used CPC proposed by GINEBRA et al [11]. (2) The setting product of the CPC/PLGA composite is basically the same as that of simple CPC because both products have hydroxyapatite (Ca5(PO4)3OH, HA) as the main crystal phase and contain trace amount of incompletely reacted tetracalcium phosphate (Ca4(PO4)2O, TTCP). (3) X-ray observation shows that bone remodeling function is progressively increased as time is prolonged in the experimental groups, confirming the superior function of scaffold materials in remodeling and repairing large segmental bone defects [12-14]. (4) In 56 d of implantation, there are no acute or chronic toxic reactions observed, nor significant inflammatory reactions of the operative incision. All wounds are healed by first intention, which suggests favorable biocompatibility of the implanted material [15]. (5) histological observation finds earlier occurrence of both osteoblast and bone trabeculaes in the experimental group as compared with other groups. (6) By 56 d, the repairing effect on bone defects in the experimental group is superior to that in other groups; formation of primary medullary cavity can be seen in this group. All of above-mentioned results suggest that the CPC/PLGA composite is a good scaffold material in bone tissue engineering.
5 Conclusions
(1) The CPC/PLGA organic-inorganic composite materials may be used as a kind of novel tissue engineering scaffold material in clinical because they have good mechanical properties in line with the tissue engineering scaffold material conditions. PLGA has no obvious adverse effects on the setting time and final phase composition of CPC/PLGA composite. The setting time also meets the requirements in clinical application, and the setting product is mainly hydroxyapatite (HA).
(2) The general observation on animals after operation confirms that three-dimensional porous scaffold CPC/PLGA composite has a good biocompatibility but no toxicity, no significant effect on MSCs adhesion, proliferation, differentiation and secretion, and accordingly can be applied as bone tissue engineering scaffold material.
(3) With ability to promote growth of bone tissue and accelerate repairing of bone defects, the CPC/PLGA composite combined with MSCs may be a new type of compound artificial bone promising in clinical repairing bone defects.
References
[1] CRANE G M, ISHAUG S L, MIKOS A G. Bone tissue engineering [J]. Nat Med, 1995, 1(12): 1322-1324.
[2] ZHENG Lei, WANG Qian, PEI Guo-xian. Selection on extracellular matrix materials in bone tissue engineering [J]. Chinese Journal of Surgery, 2000, 38(10): 745-748.
[3] BAI Bing, YE Mao-chang. The current situation and future of extracellular matrix materials in bone tissue engineering [J]. Progress in Modern Biomedicine, 2007, 7(1): 138-141. (in Chinese)
[4] ZHANG Xin-feng, ZHANG Zhi-hong. Research development of mineralized biomaterials with osteoinductivity [J]. Progress in Modern Biomedicine, 2007, 7(5): 777-779. (in Chinese)
[5] DRIESSENS F C M, BOLTONG M G, BERMUDEZ O, PLANELL J A. Formulation and setting times of some calcium orthophosphate cements: A pilot study [J]. J Mater Sci: Mater in Med, 1993, 4(5): 503-508.
[6] BAADSGAARD K. Defect pseudoarthroses: An experimental study in rabbits [J]. Acta Orthop Scand, 1969, 40(6): 689-695.
[7] ZELLIN G, LINDE A. Treatment of segmental defects in long bones using osteopromotive membranes and recombinant human bone morphogenetic protein-2: An experimental study in rabbits [J]. Scand J Plast Reconstr Surg Hand Surg, 1997, 31(2): 97-104.
[8] FEI Zheng-qi, HU Yun-yu, WU Dao-chen, WU Hong, LU Rong, BAI Jian-ping, SONG Hong-xun. Preparation and property of a novel bone graft composite consisting of rhBMP-2 loaded PLGA microspheres and calcium. Phosphate cement [J]. Journal of Materials Science: Material in Medicine, 2007, 19(3): 1109-1116.
[9] LI Qi-hong. Progress in research of repairing large segmental bone defects of long bones in China [J]. Chinese Journal of Orthopaedics, 1997, 17: 13-14. (in Chinese)
[10] LSHIKAWA K, ASAOKA K. Estimation of ideal mechanical strength and critical porosity of calcium phosphate cement [J]. J Biomed Mater Res, 1995, 29(12): 1537-1543.
[11] GINEBRA M P, FERNANDEZ E, BOLTONG M G, BERMUDEZ O, PLANELL J A, DRIESSENS F C M. Compliance of an apatitic calcium phosphate cement with the short-term clinical requirements in bone surgery, orthopaedics and dentistry [J]. Clin Mater, 1994, 17(2): 99-104.
[12] XU Jin, XIONG Gui-rong, JIANG Ping. Relationships between stress distribution of mandible with age and incidence of fracture [J]. Journal of Jiangsu University: Medicine Edition, 2006, 16(4): 291-293. (in Chinese)
[13] ZHANG Wei. The preparation of, and experimental study on compound bioactive artificial bone (CBAB) [D]. Shanghai: The Second Military Medical University, 2001. (in Chinese)
[14] MURUGAN R, RAMAKRISHNA S. Nano-featured scaffolds for tissue engineering: A review of spinning methodologles [J]. Tissue Engineering, 2006, 12(3): 435-447.
[15] XIANG Qi-jun, LIU Yong, ZHENG Zhi, SU Zhe-an, WANG Jian-long, XIAO Fei. Preparation and some property investigations on composite of calcium phosphate cements/polyglycolic-polylactic acid [J]. Materials Science and Engineering of Powder Metallurgy, 2005, 10(6): 361-364. (in Chinese)
Foundation item: Projects(30370412, 30670558) supported by the National Natural Science Foundation of China
Received date: 2009-05-08; Accepted date: 2009-10-14
Corresponding author: ZHENG Zhi, PhD, Professor; Tel: +86-731-88618663; E-mail: zheng0813@163.com
(Edited by CHEN Wei-ping)