Trans. Nonferrous Met. Soc. China 20(2010) s1033-s1036

Characteristics of 100Cr6 bearing steel after thixoforming process performed with prototype device
?. ROGAL1, J. DUTKIEWICZ1, T. CZEPPE1 , J. BONARSKI1, B. OLSZOWSKA-SOBIERAJ2
1. Institute of Metallurgy and Materials Science of the Polish Academy of Sciences,
30-059 Krak��w, 25 Reymonta St. Poland;
2. Faculty of Foundry Engineering, University of Science and Technology,
30-059 Krak��w, 23 Reymonta St. Poland
Received 13 May 2010; Accepted 12 July 2010
Abstract: A device based on a high pressure die-casting(HPDC) machine was constructed, with a capacity to produce thixo-casts from steel. After inductive heating to the required semi-solid temperature range, the samples were transported in a protective argon atmosphere to a cylinder of modified HPDC and injected into a pre-heated die. Bearing steel 100Cr6 (after forging) was used as the feedstock material. The metallographic analysis of the steel showed a homogenous structure within a whole volume characterized by a grain size between 2 ?m and 8 ?m. Differential scanning calorimetry (DSC) analysis allowed to estimate the process temperature for thixoforming at 1 390 ?C which was attributed to about 45% of a liquid phase. With such technological parameters after the semi-solid processing the microstructure consisted of globular grains of size from 15 ?m to 40 ?m. The microstructure of globules indicated predominant presence of martensite (about 76% volume fraction) with residual austenite between plates. X-ray analysis confirmed that the thixo-cast samples contain mainly martensite and residual austenite. The hardness of the initial forged sample was HRC 27 and after the thixoforming process it was HRC 60, which confirms the presence of martensite and carbides. The thixo-casts were subjected to low tempering, at temperature of 150 ?C for a period of 2 h, in order to remove stresses created during the process of thixoforming and to increase plasticity. The hardness after tempering was HRC 62.3. Additionally, in order to identify phases present in the thixo-casts, transmission electron microscopy (TEM) was carried out.
Key words: SSM; semi-solid metal; thixoforming; 100Cr6 steel
1 Introduction
Thixoforming technology or semi-solid processing (SSP) is a method of forming parts from the solidus-liquidus range. In this process the thixotropic flow behaviour of metal in the solidus-liquidus range is used. The behaviour of Pb-Sn alloys was discovered by Spencer and all in 1970[1-3].
In order to obtain appropriate flow behaviour of the semi-solid slurry, a microstructure of an alloy prior to forming should consist preferably of solid metal spheroids in a liquid matrix[4]. The liquid fraction in the thixoforming processes should be within the range of 30%-60%. The essential factor influencing the final structure and properties of an alloy to be shaped in the semi-solid state is a method of the globular structure preparation. Many processes have been developed to produce the non-dendritic spherical microstructure, e.g. mechanical stirring, magnetohydrodynamic stirring (MHD), strain induced melt activated (SIMA) recrystallization and partial melting (RAP) and spray forming [5-6]. The thixoforming technology enables the production of near-net shape products for various industrial applications. For Al and Mg alloys, this technology has been used for a serial production of responsible parts in the automobile industry and aerospace engineering[7-10]. Currently, several research centres all over the world are conducting experiments on the flow behaviour of thixotropic iron alloys in the semi-solid state[11-13]. Until now, several steels have been qualified as suitable for the thixoforming process.
Research has centered on alloys with a wide enough semi-solid range. It has been estimated that the steels X210CrW12, HS6-5-2, 100Cr6 and C38 are the best for the thixoforming process[14-16]. The research has been conducted on the modification of existing steels to make them more suitable for the thixoforming process and the right heat treatment of thixo-casts. Concurrently, the problems of thixo-casts manufacturing technology have been dealt with, i.e. constructing and selecting material for the die, the precise control of the temperature in the semi-solid state[17-19]. In this study, the bearing steel 100Cr6 after forging was chosen as a feedstock for the thixoforming process as containing relatively high carbon content and a low content of other additions, which reflects a moderate cost. In the previous works on thixoforming of 100Cr6 steel, the carbon content was near 0.9%C, and the applied thixoforming temperature was in the range of 1 400-1 425 ?C. The parts after the process were of high hardness, suggesting dominant presence of martensite[20]. Therefore, tempering treatment was proposed[21]. However, no detailed phase analysis after thixoforming process and tempering treatment was performed. Thus, in the present research X-ray diffraction and transmission electron microscopy (TEM) studies of phase composition after thixo-process and tempering were performed.
2 Experimental
The feedstock used was the commercially available 100Cr6 steel bar after forging and softening. The average chemical composition, determined using emission optical spectrometer JY 10 000 RF JOBIN YVON, is shown in Table 1.
Table 1 Chemical composition of 100Cr6 steel (mass fraction, %)

In order to determine the temperature ensuring the appropriate amount of liquid fraction in the thixoforming process, a series of studies were carried out with SDT Q600 TA Instruments, using the heating rate of 20 ?C / min. The analysis of DSC-TGA (Fig.1) helped to establish the process temperature and showed that about 45% of liquid fraction exists at temperature of 1 390 ?C.
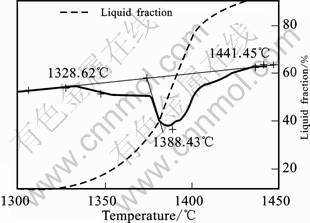
Fig.1 DSC-TGA curves for 100Cr6 steel after forging
Thixo-cast gear wheels were made using a specially built prototype device. The piston velocity of 1 m/s was applied. The locking force of the machine was 800 kN. A billet (diameter 30 mm, height 30 mm) of 100Cr6 steel was placed in the coil of an inductive heating machine. According to the heating curve in Fig.2, the temperature of 1 390 ?C was reached, which was equivalent to around 45% of the liquid fraction (heating time 260 s).
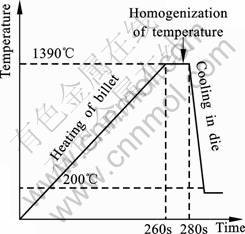
Fig. 2 Heating curve of billets in thixo-process
The billets were held at temperature of 1 390 ?C for around 20 s in order to homogenize the temperature within the whole volume. During the heating process a protective argon atmosphere was used and the heating time was limited. Next, the billet was moved to a cylinder of a high-pressure die-casting machine and pressed out by a piston into the die, pre-heated to temperature of 200 ?C, and covered with BN. The above procedure enabled the production of a series of gear wheel thixo-casts. Next, the thixo-casts were subjected to heat treatment: heating to temperature of 150 ?C for 2 h and then cooling down to room temperature. In order to estimate the functional quality of gear wheels, they were fitted into the machine and used in industrial conditions, where they show good functional properties.
The hardness measurements were carried out, using a Rockwell hardness tester fitted with a diamond cone penetrator. Scanning electron microscope, FEI E-SEM XL30, equipped with energy dispersive X-ray spectrometer EDAX GEMINI 4000, was used for the observation of the sample surface (previously ground and polished) in the controlled vacuum. X-ray diffractometry investigations of the alloy phase composition were performed using Co K�� (1.788 96 ?) filtered radiation on Philips PW 1710 diffractometer. The phase identification was performed using EVA 11.0 (Bruker) program. Samples for TEM observation were electropolished using jet method in electrolyte consisting of 20% HClO4 and 80% CH3OH at subzero temperatures.
3 Results and discussion
The bearing steel 100Cr6 (after forging) provided by the manufacturer in a softened state had a hardness of HRC 26. Fig.3 shows TEM micrograph and a selected area diffraction pattern as an insert taken from the area of the micrograph. It shows a clear perlitic structure consisting of alternating ferrite and secondary cementite plates. Electron diffraction pattern from the area shows strong reflections from ��-Fe and the less visible Fe3C of lattice described in Ref.[22]. The measurements of angles and distances between the reflections enabled a determination of the crystallographic relationship: [314] Fe3C// [110] ��-Fe and (2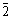
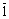 ) Fe3C//(1
) Fe3C//(1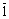 0) ��-Fe.
0) ��-Fe.

Fig.3 TEM micrograph of 100Cr6 steel after forging and SAEDP as insert with indexing of reflections
Gear wheels made of 100Cr6 steel are presented in Fig.4, and their optical microstructures are shown in Fig.5. Globular grains of supersaturated solid solution of size from 15 ?m to 40 ?m are visible. Inside the globular grains we can observe a structure consisting of martensite needles with residual austenite between them, which is formed due to cooling conditions exceeding critical cooling rate. The average hardness of the thixo-cast obtained in the above process was HRC 60.

Fig. 4 Thixocast samples of gear wheels
The SEM microstructure with marked places of chemical analysis is shown in Fig.6. The chemical composition of globular grains (point 1 in Fig.6) is as follows: 0.9% C, 1.45% Cr, 0.49% Mn, 0.31% Si. The chemical composition analysis at grain boundaries (point 2 in Fig.6) is as follows: 2.5% C, 2.5% Cr, 0.16% Si, 0.6% Mn.

Fig.5 Optical microstructure of 100Cr6 steel after thixoforming

Fig. 6 SEM micrograph from Thixo-cast
In a bright field TEM micrograph (Fig. 7), bands of martensite needles and dispersed precipitates of carbides are visible. The electron diffraction pattern from the area shows reflections from ��-Fe (200) and (01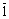 ) and weaker reflections from alloying carbides Fe3C (011) and (200) of an orthorombic lattice described in Ref.[22]. Additionally, in other diffractions, the carbide Cr7C3 of a lattice described in Ref.[22] was identified. The measurements of angles and distances between the reflections enabled the determination of the crystallographic relationship: [0
) and weaker reflections from alloying carbides Fe3C (011) and (200) of an orthorombic lattice described in Ref.[22]. Additionally, in other diffractions, the carbide Cr7C3 of a lattice described in Ref.[22] was identified. The measurements of angles and distances between the reflections enabled the determination of the crystallographic relationship: [0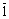 1] Fe3C// [011] ��-Fe.
1] Fe3C// [011] ��-Fe.
In order to identify and determine the percentage of individual structure components, an X-ray diffraction experiment was made. Fig.8 shows RD spectrum of three technological states. 1) 100Cr6 steel after forging and softening: a perlitic structure consisting of ferrite (��-Fe) and cementite (Fe3C). 2) After thixoforming: visible martensite peaks (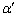 -Fe) estimated at 67% of the volume and residual austenite (��-Fe) at 33% of the volume. The spectrum does not show a volume contribution or presence of alloy carbides M3C and M7C3. The microstructure of thixo-casts consists of fine martensite needles, residual austenite and super dispersive carbides. They underwent
-Fe) estimated at 67% of the volume and residual austenite (��-Fe) at 33% of the volume. The spectrum does not show a volume contribution or presence of alloy carbides M3C and M7C3. The microstructure of thixo-casts consists of fine martensite needles, residual austenite and super dispersive carbides. They underwent  a low temperature tempering (150 ?C��2h��air cooling). This treatment resulted in an increase in the hardness up to HRC 62.3. 3) An increased volume of martensite (
a low temperature tempering (150 ?C��2h��air cooling). This treatment resulted in an increase in the hardness up to HRC 62.3. 3) An increased volume of martensite (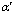 -Fe, 78.7%), residual austenite (��-Fe,15.3%) and �� carbide (6%) was observed. This indicated that a part of residual austenite transformed to martensite. In the same time, precipitation of �� carbide caused lower tetragonality of martensite.
-Fe, 78.7%), residual austenite (��-Fe,15.3%) and �� carbide (6%) was observed. This indicated that a part of residual austenite transformed to martensite. In the same time, precipitation of �� carbide caused lower tetragonality of martensite.

Fig. 7 TEM micrograph of 100Cr6 steel after thixoforming and SAEDP as insert with indexing of reflections
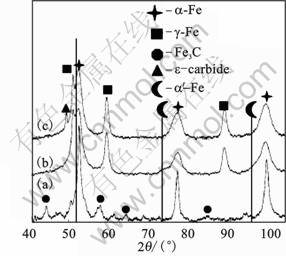
Fig.8 XRD spectra of 100Cr6 steel after forging (a), after thixoforming (b) and thixo-cast after low tempering (c)
4 Conclusions
1) A series of gear wheel thixo-casts with globular grain microstructure and hardness HRC 60 were produced from 100Cr6 steel, using a prototype device, at temperature of 1 390 ?C, corresponding to 45% of liquid phase, and piston velocity of 1 m/s. The microstructure of the obtained thixo-casts comprised fine needle like martensite inside spheroid grains and residual austenite. Dispersed carbides of Fe3C and M7C3 type were identified.
2) Low temperature tempering (at 150 ?C, 2 h, air cooling) reduced the volume fraction of the residual austenite by half (down to 15%) with a simultaneous rise of the martensite fraction up to 78.7% and with the precipitation of �� carbide (6%), which caused a decrease in the martensite tetragonality. The avarage thixo-cast hardness increased slightly after tempering up to HRC 62.3.
References
[1] MEHRABIAN R, FLEMINGS M C. Rheological behaviour of Sn-15 pct Pb in the crystallization range [J]. Met Trans, 1972, 3: 1925-1932.
[2] FLEMINGS M C. Behavior of metal alloys in the semisolid state[J]. Metall Trans A, 1991, 22: 957-981.
[3] FAN Z. Semisolid metal processing[J]. International Materials Reviews, 2002, 47(2): 49-85.
[4] ATKINSON H. Current status of semi-solid processing of metallic materials[M]//Advances in Material Forming. Paris, France: Springer-Verlag, 2007: 81-98.
[5] HIRT G, KOPP R. Thixoforming semi-solid metal processing[M]. Wiley-vch, 2009: 9-17.
[6] ATKINSON H. Modelling the semisolid processing of metallic alloys[J]. Progress in Materials Science, 2005, 50(3): 341-412.
[7] XINGGANG L, SHUISHENG X, YUNXI J, LEI L. Semi-solid processing technologies for magnesium alloys[J]. Materials Science Forum, 2005. 488/489: 307-312.
[8] GOVENDER G, IVANCHEV L, JAHAJEEAH N, BEAN R. Application of CSIR rheocasting technology for the production of an automotive component[J]. Solid State Phenomena, 2006, 116/117: 501-504.
[9] CZERWINSKI F. Magnesium injection molding[M]. Springer, 2008: 135-145.
[10] CHAYONG S, ATKINSON H V, KAPRANOS P. Thixoforming 7075 aluminium alloys[J]. Materials Science and Engineering A, 2005, 390: 3-12.
[11] PUETTGEN W, BLECK W, HIRT G. Thixoforming of steel�CA status report[J]. Adv Eng Materials, 2007, 9: 231.
[12] DUTKIEWICZ J, ROGAL ?, SO?K K, MITURA Z. Thixoforming Technology of high carbon X210CrW12 steel[J]. International Journal of Material Forming, 2009, 2(S1): 753-756.
[13] OMAR M Z, ATKINSON H V, HOWE A A, PALMIERE E J. Solid-liquid structure break-up in M2 tool steel for semi-solid metal processing[J]. J Mater Sci, 2009, 44(3): 869-874.
[14] KAPRANOS P, GHAZALI M J, MUENSTERMANN S, UIBEL K, TONNESENN T, TELLE R. Development of a self-heating ceramic tool for the thixoextrusion of high melting point alloys[J]. Solid State Phenomena, 2006, 116/117: 696-699.
[15] ROBELET M, RASSILI A, FISCHER D. Steel grades adapted to the thixoforming process: Metallurgical structure and mechanical properties[J]. Solid State Phenomena, 2006, 116/117: 712-716.
[16] LECOMTE-BECKERS J, RASSILI A, CARTON M, ROBELET M, KOEUNE R. Study of the liquid fraction and thermophysical properties of semi-solid steel and application to the simulation of inductive heating for thixoforming[M]. Berlin, Heidelberg: Springer, 2005: 321-347.
[17] KOPP R, SHIMAHARA H, SCHNEIDER J M, KURAPOV D, TELLE R, MUNSTERMANN S, LUGSCHEIDER E, BOBZIN K, MAES M. Characterization of steel thixoforming tool materials by high temperature compression tests[J]. Steel Research International, 2004, 75(8/9): 569-576.
[18] KOPP R, KALLWEIT J, MOLLER T, INGOD S. Forming and joining of commercial steel grades in the semi-solid state[J]. Journal of Materials Processing Technology, 2002, 130/131: 562-568.
[19] SCHONBOLHM A, GASPER R. Inductive reheating of steel billets into the semi-solid state based on pyrometer measurements[J]. Solid State Phenomena, 2006, 116/117: 734-737.
[20] PUTTGEN W, HALLSTEDT B, BLECK W, LOFFLER J F, UGGOWITZER P J. On the microstructure and properties of 100Cr6 steel processed in the semi solid state[J]. Acta Materialia, 2007, 55: 6553-6560.
[21] PUTTGEN W, HALLSTEDT B, BLECK W, UGGOWITZER P J. On the microstructure formation in chromium steels rapidly cooled from the semi-solid state[J]. Acta Materialia, 2007, 55: 1033-1042.
[22] VILLARS P, CALVERT L D. Pearson��s handbook of crystallographic data for intermetallic phases[M]. ASM International, 1991: 1894.
(Edited by YUAN Sai-qian)
Corresponding author: ?. ROGAL; Tel/fax: +48-12-2952861; E-mail: lukaszrogal@wp.pl