J. Cent. South Univ. (2014) 21: 4409-4414
DOI: 10.1007/s11771-014-2442-1
Effects of Al and Sn on electrochemical properties of Mg-6%Al-1%Sn (mass fraction) magnesium alloy as anode in 3.5%NaCl solution
HUANG Qiao(黄俏), YU Kun(余琨), YANG Shi-hai(杨士海),WEN Li(文利), DAI Yi-long(戴翼龙), QIAO Xue-yan(乔雪岩)
School of Materials Science and Engineering, Central South University, Changsha 411083, China
 Central South University Press and Springer-Verlag Berlin Heidelberg 2014
Central South University Press and Springer-Verlag Berlin Heidelberg 2014
Abstract: Mg-6%Al-1%Sn (mass fraction) alloy is a newly developed anode material for seawater activated batteries. The electrochemical properties of Mg-1%Sn, Mg-6%Al and Mg-6%Al-1%Sn alloys are measured by galvanostatic and potentiodynamic tests. Scanning electron microscopy (SEM) with energy dispersive spectrometry (EDS) is used to characterize the microstructures of the experimental alloys. The results show that the Mg-6%Al-1%Sn alloy obtains more negative discharge potential (-1.38 V (vs SCE)) in hot-rolled condition. This is attributed to the fine dynamically recrystallized grains during the hot rolling process. After the experimental alloys are annealed at 473 K for 1 h, the discharge potentials of Mg-6%Al-1%Sn alloy are more negative than those of Mg-6%Al alloy under different current densities. After annealing at 673 K, the discharge potentials of Mg-6%Al-1%Sn alloy become more positive than those of Mg-6%Al alloy. Such phenomenon is due to the coarse grains and the second phase Mg2Sn. The discharge potentials of Mg-1%Sn shift positively obviously in the discharge process compared with Mg-6%Al-1%Sn alloy. This is due to the corrosion products pasting on the discharge surface, which leads to anode polarization.
Key words: magnesium alloy; electrochemical properties; polarization; anodic dissolution
1 Introduction
Magnesium alloys are excellent materials used as anodes for seawater-activated battery due to their properties, such as rapid activation process, low electrode potential, high specific strength and high current capacity [1-2]. As a result, they can be used in many under-water instruments such as sonobuoy, air-sea rescue equipment and military devices [3]. Different magnesium alloys were produced to meet different applications, working from a few minutes to several days, and discharging in low and high current density, even in pulsed and constant operation [4]. The principle of such battery can be expressed as the following reaction: Mg+2H2O→Mg(OH)2+H2 [5-7]. However, since magnesium alloys are prone to form a hydroxide film on the surface in seawater, the electrochemical reaction process is impeded by Mg(OH)2 film, leading to low current utilization efficiency [8-10]. The previous studies with Mg-Ga-Hg alloy and AP65 alloy have demonstrated that good electrochemical properties can be obtained by adding alloying element into the magnesium matrix or using plastic deformation to change the microstructure of the magnesium anode [6, 11]. But those anode magnesium alloys contain toxic elements, mercury and lead, which are harmful to the environment. Tin is a nontoxic element which is also beneficial to improving the discharged potential of magnesium. So, in this work, a Mg-Al-Sn anode alloy is developed to research and evaluate the effects of tin and aluminium on the electrochemical properties of magnesium matrix in NaCl solution as an anode.
2 Experimental
A Mg-6%Al-1%Sn (mass fraction) experimental alloy was prepared through ingot metallurgy method. Mg-1%Sn and Mg-6%Al alloys were also prepared for comparison. These three kinds of experimental alloys were melt by using high purity magnesium (99.9%, mass fraction), aluminum (99.99%, mass fraction), tin (99.99%, mass fraction) in a furnace at 1023 K with the protection of argon. The molten alloys were cast into a steel mold. Then, all the as-cast magnesium alloy ingots were homogenized at 673 K for 16 h and quenched in the air. The homogenized alloys were hot rolled for several passes to obtain the sheets with thickness of 2 mm. During every rolling pass, the deformed alloys were reheated to 673 K for 30 min to obtain a good deformability. In the end, the rolled sheets were annealed at 473 K or 673 K for 1 h.
The specimens in hot-rolled, 473 K and 673 K annealed states were prepared for the electrochemical test. An IM6ex electrochemical test system with a three- electrode configuration was used in electrochemical test. The reference electrode was a saturated calomel electrode (SCE) and the counter electrode was a platinum plate. The electrolyte of the electrochemical test was 3.5% (mass fraction) NaCl solution, and the test temperature was (283±2) K. A FEI Quanta-200 SEM with EDS and a Polyvar-MET metallographic microscope were used to observe the microstructure and analyze the composition of the experimental alloys.
3 Results and discussion
3.1 Microstructure evolution of experimental alloys
Since the electrochemical properties of an alloy are mainly determined by its microstructure, it is important to observe and analyze the microstructure of the experimental magnesium alloys before analyzing their electrochemical behaviors. The morphologies of the prepared alloys under homogenization condition are shown in Fig. 1.
With reference to the binary phase diagram, Mg2Sn is the only phase existing between magnesium and tin and Mg17Al12 is the phase existing between magnesium and aluminum. The Mg2Sn phase with a sphere shape distributes discontinuously along the grain boundaries of the homogenized Mg-Sn alloy (Fig. 1(a)). The discontinuous precipitates can be observed in Mg-Al alloy in Fig. 1(b)). The size of Mg17Al12 precipitate on the grain boundaries is larger than that in the matrix. And in Fig. 1(d), it can be seen that Mg-6%Al-1%Sn alloy also gets a discontinuous precipitate morphology. The precipitates have a lamellar morphology with marked anisotropy of growth. This is almost the same as the Mg-6%Al alloy. According to the ternary equilibrium phase diagram, no ternary phase exists among magnesium, aluminum and tin. With the EDS result obtained from Fig. 1(e) and Fig. 1(f), in Mg-6%Al-1%Sn alloy, Mg17Al12 phases precipitate both at the grain boundaries and in the matrix, but Mg2Sn phases only precipitate in the matrix. These Mg2Sn phases have a lamellar shape, which is different from that of sphere shape in Mg-1%Sn alloy. The differences of morphology, size and distribution of phases among these three experimental alloys influence the electrochemical properties, due to their cathodic characterization towards the Mg matrix.
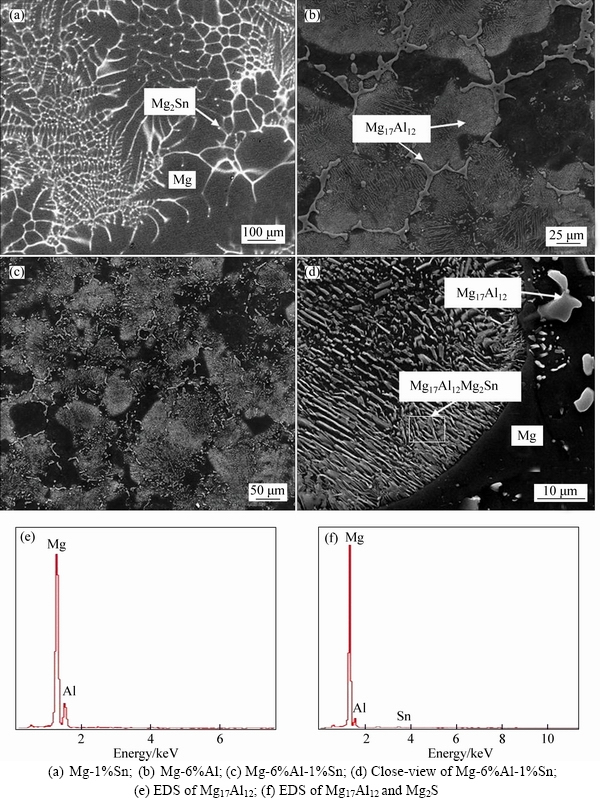
Fig. 1 Microstructure and phase identification of experimental alloys in homogenization condition:
After hot rolling process, the grain size of Mg-6%Al-1%Sn alloy is refined. These small-size grains distribute along the rolling direction. The equiaxed grains show that dynamic recrystallization occurs in discontinues areas (Fig. 2(a)). When being annealed at 473 K (Fig. 2(b)), many small recrystallized grains are distributed in the vicinity of the elongated ones. The fine grains become coarse when being annealed at 673 K for 1 h (Fig. 2(c)). Such coarse grains would shift the discharge potential positively, which needs to be avoided as an anode material [6].
3.2 Discharge behaviors of experimental alloys
To make a thorough understanding of the effects of Al and Sn on the discharge behavior of Mg-6%Al-1%Sn alloy, the potentiodynamic polarization, galvanostatic discharge and electrochemical impedance spectroscopy measurement were carried out, and Mg-1%Sn and Mg-6%Al alloys were used for comparison.
The potentiodynamic polarization curves of magnesium alloy in the rolled condition are shown in Fig. 3. The polarization parameters obtained from the polarization curves are given in Table 1.
The results show that different compositions of magnesium alloy would lead to different polarization parameters. The Mg-1%Sn alloy has the most negative corrosion potential. The corrosion potential of Mg-6%Al alloy is slightly more positive than that of Mg-1%Sn alloy. While Mg-6%Al-1%Sn alloy gets the most positive discharge potential during discharge process. The corrosion current density is an important parameter to evaluate the corrosion rate, and high corrosion current density leads to high corrosion rate. Hence, the potentiodynamic polarization results indicate that Mg-1%Sn alloy is easy to be activated. This is because Mg-1%Sn alloy gets the largest corrosion current density as shown in Table 1. By comparing Mg-6%Al-1%Sn, Mg-1%Sn and Mg-6%Al alloys, the additive of Al(Mg17Al12) phase would make the corrosion potential shift positively. The addition of Sn(Mg2Sn) phase can shift the corrosion potential of Mg-6%Al positively and enhance the discharge current density as well.

Fig. 2 Morphologies of rolled and annealed Mg-6%Al-1%Sn alloy:
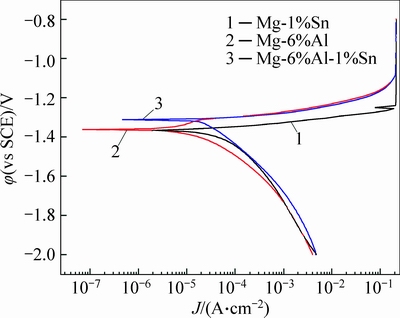
Fig. 3 Potentiodynamic polarization behavior of magnesium alloys in rolled condition
Table 1 Parameters from polarization curves
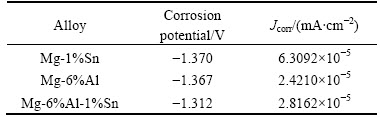
The galvanostatic discharge curves of three prepared magnesium alloys in rolled condition with different current densities of 10, 100 and 200 mA/cm2 are shown in Fig. 4.
Apparently, with the current density increasing, all the discharge potentials shift positively. Mg-6%Al-1%Sn alloy has the most negative potential when it is discharged at the current density of 10 mA/cm2 (Fig. 4(a)) and 100 mA/cm2 (Fig. 4(b)). Mg-1%Sn alloy can be discharged steadily with current density of 10 mA/cm2 (Fig. 4(a)), but it is polarized at the current density of 100 mA/cm2 (Fig. 4(b)) and 200 mA/cm2 (Fig. 4(c)). This is because the corrosion products past the discharge surface and cannot be removed easily. Tin has a good effect on reducing the polarization potential at the discharge current density of 10 mA/cm2 and 100 mA/cm2 by comparing Mg-6%Al alloy with Mg-6%Al-1%Sn alloy, because the potential of Mg-6%Al-1%Sn alloy is more negative than that of Mg-6%Al alloy. This effect is attributed to the intermetallic Mg2Sn. Mg2Sn can break the Mg(OH)2 film formed on the surface of anode so that the electrolyte is easy to contact with the magnesium matrix. Such process enables the anode to dissolve continuously. The Mg17Al12 phase is beneficial to the stripping of corrosion products, hence avoiding the polarization of the electrode. It is also good for the improvement of the utilization efficiency of anode according to the Refs. [11-12]. But when the experimental metals are discharged under high current density like 200 mA/cm2, the effect of decreasing discharge electrode potential of Sn(Mg2Sn) phase is not apparent.
After being annealed at 473K, as shown in Fig. 5, the effect of reducing polarization of Sn(Mg2Sn) is easy to be obtained as the potential of Mg-6%Al-1%Sn alloy is more negative than that of Mg-6%Al alloy at current density of 10 mA/cm2 (Fig. 5(a)), 100 mA/cm2 (Fig. 5(b)) and 200 mA/cm2 (Fig. 5(c)). Mg-1%Sn alloy is discharged steadily with low current density, but polarized at current density of 100 mA/cm2 and 200 mA/cm2. The discharge curve of Mg-1%Sn alloy fluctuates severely when being discharged at 200 mA/cm2, and the potential at some point even reaches to positive range. The reason is also attribute to the hard-stripping of corrosion products [13-14].
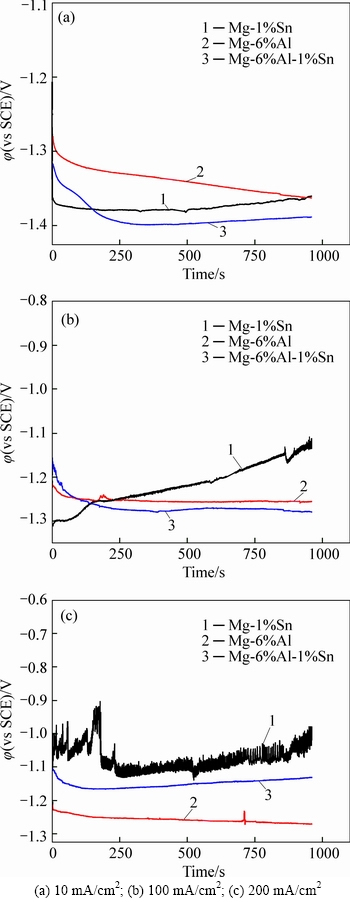
Fig. 4 Galvanostatic discharge behavior of magnesium alloys under rolled condition with different current densities:
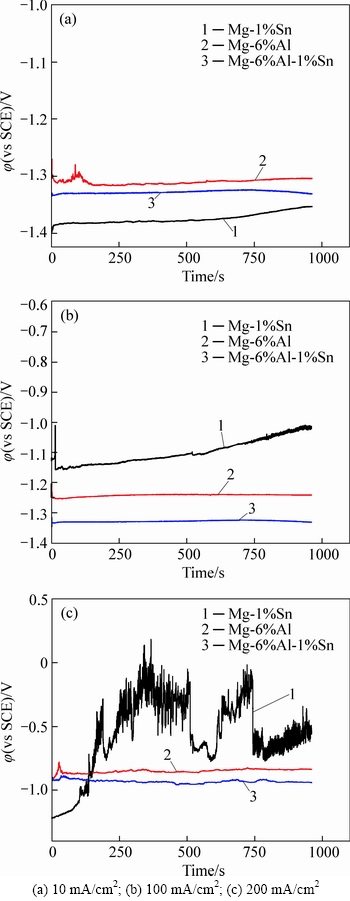
Fig. 5 Galvanostatic discharge behavior of magnesium alloys under 473 K annealed condition with different current densities:
Figure 6 shows the results of galvanostatic discharge curves after being annealed at 673 K. In this condition, the polarization potentials of Mg-6%Al-1%Sn alloy is more positive than that of Mg-6%Al alloy, which is different from the results of the previous studies. This is because of the coarse grains after being annealed at 673 K. The curves of Mg-1%Sn alloy begin to fluctuate under low current density like 10 mA/cm2, indicating that high annealing temperature is not available to obtain steady discharge potential for a metal material like magnesium used for seawater-activated battery anode.
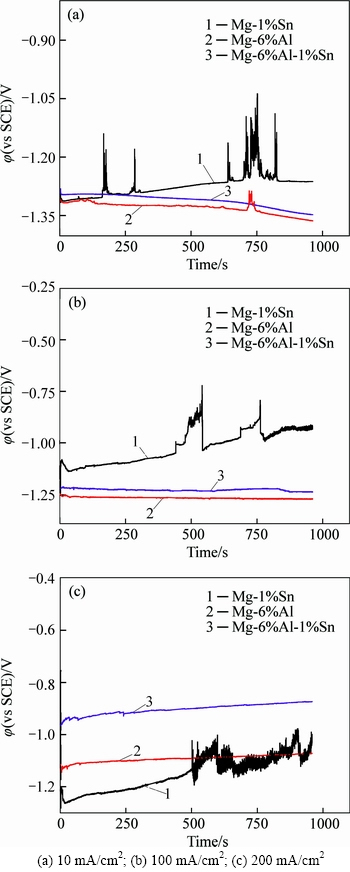
Fig. 6 Galvanostatic discharge behavior of magnesium alloys under 673 K annealed condition with different current densities:
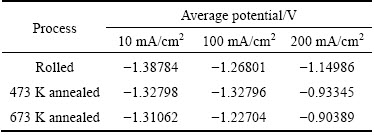
The parameters obtained from the galvanostatic discharge curves of Mg-6%Al-1%Sn alloy are given in Table 2.
The data show that as the current density increases, the electrode potential shifts toward positive direction. The potential is -1.387 V in rolled condition at current density of 10 mA/cm2. It shifts positively by about 0.24 V when being discharged at current density of 200 mA/cm2. This kind of trend can be got in 473 K and 673 K annealed alloy. This means that it is unavoidable that magnesium alloys are polarized with discharge current density increasing. As for an anode material, we expect relatively negative discharge potential in the discharge process so that the functions of outside devices can be met [15]. In this work, the discharge potentials of 473 K annealed and 673 K annealed alloys are -0.93345 V and -0.90389 V with the current density of 200 mA/cm2, and they are hard to meet the requirement of the devices that work with high current density. The rolled condition obtains the most negative discharge potential when the alloy is discharged under the same current density except for the 473 K annealed condition with current density of 100 mA/cm2.
Table 2 Parameters obtained from galvanostatic discharge curves of Mg-6%Al-1%Sn alloy
4 Conclusions
The Mg-1%Sn, Mg-6%Al, Mg-6%Al-1%Sn alloys in rolled, 473 K annealed, 673 K annealed conditions exhibit different electrochemical properties using potentiodynamic polarization, galvanostatic discharge and electrochemical impedance spectroscopy measures. Mg-1%Sn alloy obtains the most negative corrosion potential and the largest corrosion current density. Its corrosion products are easy to paste on the metal surface and hard to strip, which leads to apparent polarization when galvanostatic discharge is carried out. Compared with Mg-1%Sn alloy, Mg-6%Al-1%Sn alloy is discharged steadily in rolled and 473 K annealed conditions with current density of 10 mA/cm2 and 100 mA/cm2. This means that the additive of Al(Mg17Al12) is proper to resist polarization. The discharge potential of Mg-6%Al-1%Sn alloy becomes negative with the addition of tin. But when it is discharged with current density of 200 mA/cm2 in 673 K annealed condition, Mg-6%Al-1%Sn alloy fails to apply good electrode potential due to the coarse grains.
References
[1] FENG Yan, WANG Ri-chu, PENG Chao-qun, WANG Nai-guang. Influences of Mg21Ga5Hg3 compound on electrochemical properties of Mg-5%Hg-5%Ga alloy [J]. Transactions of Nonferrous Metals Society of China, 2009, 19: 154-159.
[2] DORON A, GURUKAR S S, ELENA L, ARIEL M, OREN M, ORITC, MICHELA B. Progress in rechargeable magnesium battery technology [J]. Advanced Materials, 2007, 19: 4260-4267.
[3] SHINOHARA M, ARAKIB E, MOCHIZUKIC M. Practical application of a sea-water battery in deep-sea basin and its performance [J]. Power Source, 2009, 187: 253-260.
[4] WILCOCK W S D, KAUFFMAN P C. Development of a seawater battery for deepwater applications [J]. Power Source, 1997, 66: 71-75.
[5] RENUKA R. AgCl and Ag2S as additive to CuI in Mg-CuI seawater activated batteries [J]. Corrosion Science, 2011, 53: 2035-2040.
[6] YU K, TAN X, HU Y, CHEN F, LI S. Microstructure effects on the electrochemical corrosion properties of Mg-4.1%Ga-2.2%Hg alloy as the anode for seawater-activated batteries [J]. Corrosion Science, 2011, 53: 2035-2040.
[7] WANG Nai-guang, WANG Ri-chu, PENG Chao-chun, FENG Yan, CHEN Bin. Effect of hot rolling and subsequent annealing on electrochemical discharge behavior of AP65 magnesium alloy as anode for seawater activated battery [J]. Corrosion Science, 2012, 64: 17-27.
[8] LIU L J, SCHLESINGER M. Corrosion of magnesium and its alloys [J]. Corrosion Science, 2009, 51: 1733-1737.
[9] VENKATESARA R K. Performance evaluation of Mg-AgCl batteries for under water propulsion [J]. Denfence Science, 2001, 5: 161-170.
[10] GUANG L S, ANDREJ A. Corrosion mechanisms of magnesium alloys [J]. Advance Engineering Materials, 1999, 1(1): 11-33.
[11] JONSSON M, THIERRY D, BOZEC N L. The influence of microstructure on the corrosion behavior of AZ91D studies by scanning Kelvin probe force microscopy and scanning Kelvin probe [J]. Corrosion Science, 2006, 48: 1193-1208.
[12] MERINO M C, PARDO A, ARRABAL R, MERINO S, CASAJUS P, MOHEDANO M. Influence of chloride ion concentration and temperature on the corrosion of Mg-Al alloys in salt fog [J]. Corrosion Science, 2010, 52: 589-594.
[13] ZHAO Jun, YU Kun, HU Yan-an, LI Shao-jun, TAN Xin, CHEN Fu-wen, YU Zhi-ming. Discharge behavior of Mg-4Ga-2Hg alloy as anode for seawater activated battery [J]. Electrochimica Acta, 2011, 56: 8224-8231.
[14] WANG Nai-guang, WANG Ri-chu, PENG Chao-qun, FENG Yan, ZHANG Xiang-yu. Corrosion behavior of Mg-Al-Pb and Mg-Al-Pb- Zn-Mn alloys in 3.5%NaCl solution [J]. Transactions of Nonferrous Metals Society of China, 2010, 20: 1936-1934.
[15] KONDOH K, ELSAYED A, IMAI H, UMEDA J, JONES T. Microstructures and mechanical responses of powder metallurgy non-combustive magnesium extruded alloy by rapid solidification process in mass production [J]. Material Design, 2010, 31: 1540- 1546.
(Edited by YANG Bing)
Foundation item: Project supported by the Open Fund of the Sate Key Laboratory of Powder Metallurgy (Central South University), China
Received date: 2013-07-25; Accepted date: 2013-10-21
Corresponding author: YU Kun, Professor, PhD; Tel: +86-731-88879341; Fax: +86-731-88876692; E-mail: yukunarticle@163.com