(LiF-CaF2)eut-xLnF3 (Ln=Sm, Gd)���γ������������������ӫ�����
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2021���4��
�������ߣ�Miroslav BOCA Jarmila MLYNARIKOVA Iveta MACKOVA Dusan JANICKOVIC Adriana CZIMEROVA ʯ����
����ҳ�룺1151 - 1164
�ؼ��ʣ����Σ��������ϵͳ��SmF3��GdF3�����չ��ף�ӫ����ף���������
Key words��molten salts; rapidly cooled molten system; SmF3; GdF3; absorption spectra; fluorescence spectra; luminescence properties
ժ Ҫ��(LiF-CaF2)eut-xSmF3(-xGdF3) (x=0.03~0.50)����ͨ����Ȼ��ȴ�Ϳ�����ȴ������õ���״����״������ò��ͨ��X�������䡢���չ���ӫ����������֣�LiF�����ȶ������ṹ����LnF3����ֱ��Ӱ������ṹ�;�̬��(LiF-CaF2)eut-xSmF3(-xGdF3)��ϵ�Ĺ��·���������LnF3����Ӱ�죬��SmF3����Ϊ0.03ʱ��Sm3+�ķ����ǿ��������۲���������ȴ��ʽ����Ʒ��ò��Ӱ�����ѧ���ԣ���е��ĥ����Ʒ��ӫ�����ܺͷ���ǿ�������о�������ӫ�������һ�ֿ����ڼ�����֤�״��������Ѻ�ɽ��;ֲ��Գ��ԵĹ�������
Abstract: Samples of (LiF-CaF2)eut-xSmF3(-xGdF3) (x=0.03-0.50) were prepared by spontaneous cooling as well by very fast cooling (using the rapid solidification processing (RSP) method) which provide two types of sample morphology: riffle-like and sphere-like fragments. All types of samples were studied by XRD, optical absorption and fluorescence spectroscopy. The presence of LiF stabilises the cubic cell dimension, and the content of lanthanide fluorides presents influence on phases formed, as well as the crystallinity of the system, in both the spontaneously and rapidly-cooled systems. The photoluminescence properties of the investigated systems are dependent on the lanthanide fluoride content. The intensity of the emission bands, corresponding to the Sm3+ ion, reaches the maximum when the Sm content is x(SmF3)=0.03. An even stronger dependence is observed of optical properties on the morphology of the samples, i.e. whether the samples are spontaneously cooled or processed by the RSP method followed by mechanical grinding. Mechanical grinding enhances the luminescent properties and leads to higher emission intensities. It is also shown that the photoluminescent spectroscopy is suitable for detection/approval of lowered local symmetry via significant splitting of spectral bands.
Trans. Nonferrous Met. Soc. China 31(2021) 1151-1164
Miroslav BOCA1, Jarmila MLYNARIKOVA1, Iveta MACKOVA1, Dusan JANICKOVIC2, Adriana CZIMEROVA1, Zhong-ning SHI3
1. Institute of Inorganic Chemistry, Slovak Academy of Sciences, DubravskA cesta 9, SK-845 36 Bratislava, Slovakia;
2. Institute of Physics, Slovak Academy of Sciences, DubravskA cesta 9, SK-84 511 Bratislava, Slovak;
3. School of Metallurgy, Northeastern University, Shenyang 110819, China
Received 12 May 2020; accepted 30 November 2020
Abstract: Samples of (LiF-CaF2)eut-xSmF3(-xGdF3) (x=0.03-0.50) were prepared by spontaneous cooling as well by very fast cooling (using the rapid solidification processing (RSP) method) which provide two types of sample morphology: riffle-like and sphere-like fragments. All types of samples were studied by XRD, optical absorption and fluorescence spectroscopy. The presence of LiF stabilises the cubic cell dimension, and the content of lanthanide fluorides presents influence on phases formed, as well as the crystallinity of the system, in both the spontaneously and rapidly-cooled systems. The photoluminescence properties of the investigated systems are dependent on the lanthanide fluoride content. The intensity of the emission bands, corresponding to the Sm3+ ion, reaches the maximum when the Sm content is x(SmF3)=0.03. An even stronger dependence is observed of optical properties on the morphology of the samples, i.e. whether the samples are spontaneously cooled or processed by the RSP method followed by mechanical grinding. Mechanical grinding enhances the luminescent properties and leads to higher emission intensities. It is also shown that the photoluminescent spectroscopy is suitable for detection/approval of lowered local symmetry via significant splitting of spectral bands.
Key words: molten salts; rapidly cooled molten system; SmF3; GdF3; absorption spectra; fluorescence spectra; luminescence properties
1 Introduction
The embedding of rare-earth ions in various host materials has attracted a lot of attention in materials science due to the unique optical properties. The interest is not only theoretical, due to a very interesting spectroscopic property, but also practical, due to the potential applications of these materials. One promising area of applications for which they are suitable is the field of photonics, including solid-state lasers and optoelectronic devices [1,2]. The luminescence properties of many doped fluoride systems have been investigated. For example, the luminescence properties were studied for simple binary fluorides in host matrices of simple fluorides of Li-Cs or Mg-Ba [3-5], and for more complicated ternary fluorides with host matrices such as MNF4 (M=Li-Cs, N=Y, Sc or others), KMF3 (M=Zn, Cd, Mg, Mn) [6-8], or La3Zr4F25, LaZr3F15 [9] among others and even more complicated hosting systems, such as glasses or ceramics (SiO2-Al2O3-CaF2-SmF3) [10,11]. Various properties have been proven to be relevant to the optical properties: the effect of particle size/shape/position (nanoparticles) in e.g. rare earth elements-doped (LiF or Ca)/LaF3 [12,13], the dependence of energy gap on the host matrix [14], and frequently, the position of the dopant in the structure of hosting matrix, for example, RbLa0.95Eu0.05F4 [15]. The dopants are mostly lanthanides, but other elements have also been used, such as Mn in host matrices Na2ZrF6, K2ZrF6 [16] and K3AlF6 [17].
Compared to oxide-based materials, fluoride- containing compounds have a higher luminescence and higher quantum efficiency [18]. Sm3+ exhibits absorption over a wide range of the spectrum, from the near-infrared to the UV region, thus including significant emission in the visible spectral region [19]. The intensest transition occurs around 600 nm. Emissions in the red, deep red and green areas of the spectrum are also observed. These strong emissions originate from the 4G5/2 state, which is populated non-radiatively by the 6Pj excited state [20]. Relatively large energy gap between the 4G5/2 level and the closest lower lying energy levels makes doping with Sm3+ useful in a wide range of applications, such as hole burning, high-density optical storage, and colour displays [21]. As a result, materials doped with Sm3+ and the other trivalent rare-earth ions (i.e. Nd3+ and Gd3+) have attracted substantial attention due to their high intensity emissions [22].
Most studies of luminescent properties involve a host matrix which is a crystallographically well-defined system of single crystals and glasses. Even nonstoichiometric phases with a composition of (Ca or Ba)xLnyF2x+3y are no exceptions to this. As examples, we may mention the compounds Ca0.9Nd0.1F2.1 [23] and Ca0.88Gd0.12F2.12 [24], and solid solutions of Ca1-xNdxF2+x, Ba1-xNdxF2+x and Ba1-xUxF2+x (0
The new topic of rapid solidification process (RSP) in molten salt was initiated by our research group only recently. The reasons to apply rapidly cooling process in molten salts arise from the following expectations:
(1) To reach the state when the solidified system is comparable with the liquid system (completely amorphous state) in terms of potentially problematic structural features (i.e. crystallization speed is still as high as the cooling rate [26,29]);
(2) To introduce as much as possible defects in the structure as well as to introduce the highest possible deviation from local symmetry;
(3) To reach at least metastable state of the system (partial amorphous state, glassy phases or metastable crystalline phases).
Such prepared samples can be subjected to analysis by any suitable method (e.g. IR, NMR, XPS, XRD and SEM/TEM). However, in the case of lanthanides NMR, one of the most powerful method, it cannot be used due to the presence of unpaired electrons (except La3+). Thus, the aim of this work is to find out whether luminescent spectroscopy can be used to retrieve some important information on properties of rapidly- cooled systems through the variation of available energy levels. More specifically, luminescent properties might detect differences on the structural properties of samples prepared in equilibrium conditions (e.g. spontaneous slow cooling) or under non-equilibrium conditions (e.g. RSP samples) or what role plays in the morphology or mechanical treatment on the luminescent properties. From the above mentioned variety of systems, the systems of (LiF-CaF2)eut-SmF3(-GdF3) are selected for investigation.
2 Experimental
The following chemicals were used to prepare the mixtures: LiF (Sigma-Aldrich, 99.0%), CaF2 (Merck, pure), SmF3 (ChemPur, 99.9%) and GdF3 (ChemPur, 99.9%). LiF and CaF2 were dried in vacuum at 723 K for 2 h. A glovebox (JACOMEX) with an argon atmosphere (Messer, Slovakia, purity 99.999%) was used for storage of the chemicals. The moisture and oxygen content in the dry-box were kept under 5��10-6 all time. Samples were homogenized before each experiment in a glovebox, with a LiF/CaF2 molar ratio of 0.79/0.21. For RSP experiments, the samples were pressed in the form of tablets.
2.1 Rapid solidification process
The apparatus for RSP in Fig. 1 (specifically, the melt spinning method) consisted of an induction heating coil and a rotating Cu-wheel for cooling the sample. The samples were melted in a graphite crucible in an inert Ar atmosphere. The distance between the orifice (for the outflow of the melt at the bottom of the graphite crucibles) and the rotating cylinder was approximately 0.2 mm. This setup of the apparatus permitted the melts to solidify at a cooling rate of about 106 K/s. A detailed description of the experimental technique may be found in Refs. [26,30]. The spontaneously- cooled samples were used from the thermal analysis experiments reported in Ref. [31]. The analysed samples are summarised in Table 1.

Fig. 1 Scheme of experimental setup for preparation of deeply undercooled samples by melt spinning
Table 1 List of spontaneously-cooled samples studied by luminescence spectroscopy
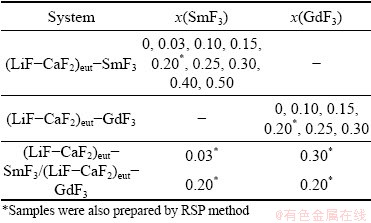
2.2 X-ray powder diffraction
X-ray powder diffraction (XRD) patterns were measured using an Empyrean PANalytical diffracto- meter with Cu K�� radiation in Bragg-Brentano geometry using a ��-filter (Ni). A solid-state PIXcel detector was used for recording XRD patterns, with an active length of 3.347��. Measurements were carried out at room temperature, with 2�� varied from 10�� to 80�� at a step of 0.026��. The time per step was 26 s. Phase analysis was performed with X��Pert HighScore Plus PANalytical software using the PDF2 2011 database.
2.3 SEM analysis
The surface morphology of the samples was analysed using a scanning electron microscope (SEM) JEOL EVO 40 (Carl-Zeiss, Germany). The surface of the samples was covered by carbon vapour.
2.4 Absorption spectroscopy
UV/Vis absorption spectra were recorded in the visible region using a Cary5000 spectro- photometer (Varian Inc.) equipped with an internal Praying Mantis Diffuse Reflectance Accessory and a Labsphere spectralon reflectance module as a standard. The absorption spectra were calculated using Kubelka-Munk algorithm (Varian CaryWinUV software package). The background spectrum was subtracted from the raw absorption spectra in all cases. For comparison, spectra of pure rare-earth fluorides were also recorded.
2.5 Fluorescence spectroscopy
Steady-state fluorescence spectra of the prepared samples were recorded using Fluorolog FL 3 (Horiba Jobin Yvon, Japan) fluorescence spectrophotometer equipped with a 450 W xenon short-arc lamp as an excitation source (wavelength range of 200-900 nm, excitation slit: 6 nm, emission slit: 6 nm, excitation and emission gratings: 1200 groove/nm blazed at 330 nm and with a standard photomultiplier tube (PMT) emission detector, voltage: 1000 V). The emission spectra were recorded for different excitation wavelengths according to which rare-earth dopants were present. Otherwise, the experimental setup and measurement parameters, i.e. excitation wavelength, slit width, bandpass, emission gratings, were identical for all samples.
3 Results and discussion
3.1 Diffraction analysis
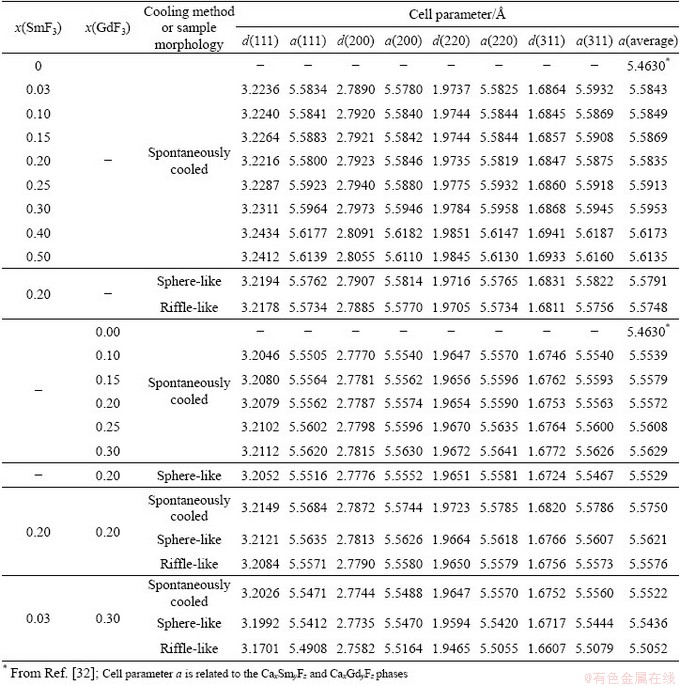
Spontaneous cooling of molten mixtures of (LiF-CaF2)eut-xSmF3 can theoretically result in one (or more) of three cases. The first is the crystallisation of the initially-present phases, the second is the crystallisation of new ternary fluorides, and the third is the crystallisation of solid solutions or some non-stoichiometric phases. In fact, crystalline LiF is observed at all studied compositions and its content decreases with increasing SmF3 content. Unreacted crystalline SmF3 is observed when contents of SmF3 with x(SmF3)>0.1 are present. On the other hand, crystalline CaF2 is only observed with contents of SmF3 up to x(SmF3)=0.1 (Fig. 2). There are two reasons why CaF2 is not observed at higher contents of SmF3. The first is the fact that, in the eutectic LiF-CaF2 system, only 21 at.% of CaF2 is present, and the relative amount of CaF2 decreases with increasing content of SmF3. Secondly, some part of the CaF2 might combine with SmF3 to form a new phase or solid solution. (Note that if we consider the formation of Ca0.65Sm0.35F2.35 (similar to Ca0.65La0.35F2.35), only about 11.3 at.% of SmF3 would be needed to consume all CaF2. On other hand if we consider the formation of Ca0.88Sm0.12F2.12 (similar to Ca0.88Gd0.12F2.12; also cubic phase), only about 2.8 at.% of SmF3 would be consumed. This means that probably both mentioned effects play role in the decrease of CaF2 diffractions the formation of new phase and the relative decrease of CaF2 content.) To investigate whether this happens, four diffraction peaks were analysed: (111), (200), (220) and (311). The diffraction peak (200) that has very low intensity for pure CaF2 is visible on new cubic phases formed in the system containing SmF3 (and also GdF3). This is caused by the change of electron density in these cubic phases due to the incorporation of SmF3 into the lattice. The appearance of (200) diffraction peak should be sufficient proof of the formation of the CaxSmyFz cubic phase related to CaF2. Table 2 summarises the cell parameter a, calculated on the assumption that the newly-formed phase is some modified form of CaF2. This assumption can be made on the basis of Ref. [33], where the binary system of CaF2-SmF3 was investigated regarding the formation of a Ca1-xSmxF2+x phase. Figure 3 shows the content dependence of the cell parameter a (cubic) for the system (LiF-CaF2)eut-SmF3. It may be seen that the presence of LiF stabilises the cell dimension, as in the (LiF-CaF2)eut-LaF3 system [26]. Contrarily, in binary systems CaF2-SmF3 and CaF2-LaF3 [33], a more pronounced dependence of the cell parameter a on the content of LnF3 is observed, which is explained by the formation of solid solutions characterized by unordered distribution of Ca2+ and Ln3+ cations retaining the cubic space group.
Our assumption is that some solid solution or non-stoichiometric compound has been formed; the formation of a ternary Li/Sm/F phase (e.g. LiSmF4) cannot be proven, as there exist no data on such a compound. Existence of a small number of ternary lithium fluorides (LiYF4 [34-39], LiCeF5 [40], Li2TbF6 [41,42], Li4TbF8 [43], LiYbF4 [44] and LiLuF4 [45]) has been proven by single crystal analysis. Other lithium lanthanide fluorides have been reported based only on XRD results (Li4CeF8 [46], LiCe4F17 [46], Li2PrF6 [47], LiGdF4 [48], LiTbF4 [48], LiHoF4 [49], LiErF4 [49] and LiTmF4 [48]). Additionally, there have been several non-stoichiometric compounds reported with a composition of Ca1-xLnxF2+x (Ln=Nd or Ga), although so far there is no similar compound with Sm [50] (Table 3).
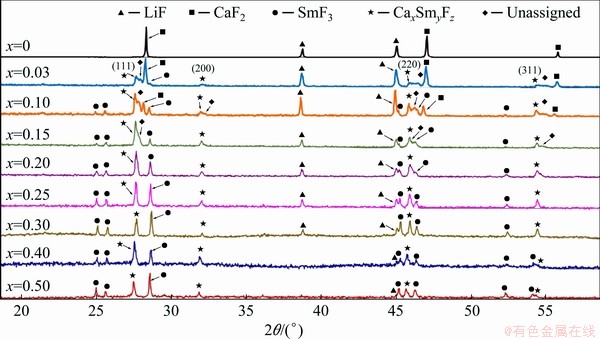
Fig. 2 XRD diffraction patterns of spontaneously-cooled melts of (LiF-CaF2)eut-xSmF3 system
Table 2 Calculated cell parameters based on (111), (200), (220) and (311) diffractions of (LiF-CaF2)eut-xLnF3 (Ln=Sm, Gd) system
The (LiF-CaF2)eut-0.20SmF3 system was then subjected to the RSP (for details see Ref. [26]). The rapidly-cooled sample was composed of fragments of both riffle-like and sphere-like morphology (Fig. 4); the two fragment types were separated for further analysis.
The cell parameters of these phases are similar to those of the spontaneously-cooled samples (Table 2 and Fig. 5). This means that rapid cooling does not result in such significant structural changes as identified in the (LiF-CaF2)eut-LaF3 system, where rapid solidification results in a significant increase of the cell parameter in comparison to spontaneously cooled samples [26]. This indicates that the results reported for phase diagrams should be considered in terms of cooling regimes (rates) to be sure of reaching as close as possible to equilibrium state. The cooling rate, moreover, might influence various systems in different extents.
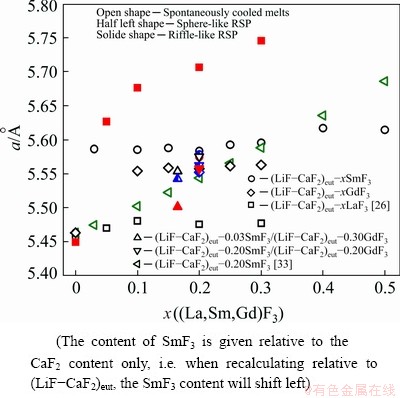
Fig. 3 Content dependence of cell parameter for different systems
Table 3 Some examples of cubic non-stoichiometric compounds

In the case of the spontaneously-cooled (LiF- CaF2)eut-GdF3 system, the situation is different to the previous system. Crystalline LiF is observed at all studied compositions, although its content decreases with increasing GdF3 content (Fig. 6). Unreacted crystalline GdF3 is not observed; LiGdF4 is detected instead, with its content increasing with increasing GdF3 content (it must be noted that the existence of this compound has been suggested in the past only on the basis of XRD patterns [48]). The presence of crystalline CaF2 is not detected in the mixtures; instead, a solid solution or non- stoichiometric phase Ca/Gd/F is identified. Figure 3 shows the content dependence of the cubic cell parameter a, estimated on the basis of the (111), (220), (220) and (311) diffractions (Table 2). The results show the presence of LiF stabilizes the cell dimension as shown in the (LiF-CaF2)eut-SmF3 and (LiF-CaF2)eut-LaF3 systems [26]. Alternatively, in CaF2-GdF3 and CaF2-LaF3 [32], a more pronounced dependence of the cell parameter a on the content of LnF3 is observed. Moreover, some weak diffractions remain unassigned.
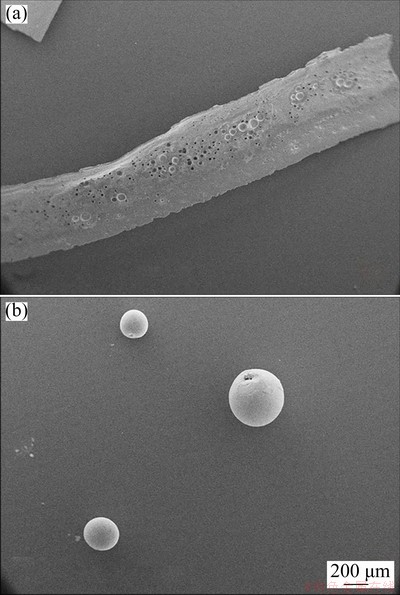
Fig. 4 SEM images of riffle-like (a) and sphere-like (b) fragments of rapidly-cooled (LiF-CaF2)eut-0.20SmF3 system
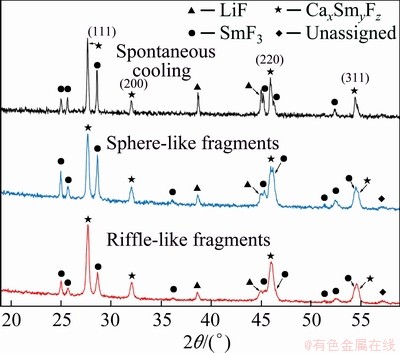
Fig. 5 XRD patterns of spontaneously-cooled melts, sphere-like fragments and riffle-like fragments of (LiF-CaF2)eut-0.20SmF3 system
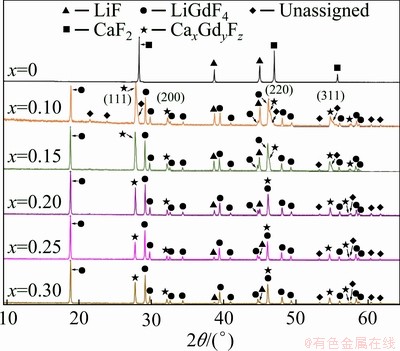
Fig. 6 XRD patterns of spontaneously-cooled melts of (LiF-CaF2)eut-xGdF3 system
RSP treatment of the (LiF-CaF2)eut-0.20GdF3 system also (as with the (LiF-CaF2)eut-0.20SmF3 system) does not result in any significant increase in the cell parameter (Fig. 7 and Table 2), unlike the previously-studied (LiF-CaF2)eut-LaF3 system. The XRD pattern of the riffle-like sample shows only the presence of a non-crystalline phase, or phases.
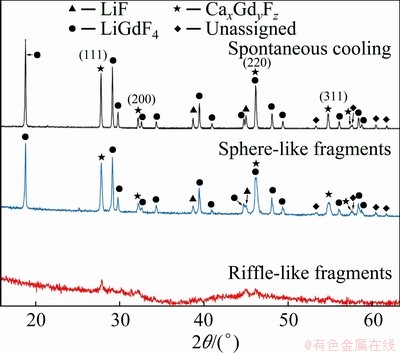
Fig. 7 XRD patterns of spontaneously-cooled melts, sphere-like fragments and riffle-like fragments of (LiF-CaF2)eut-0.20GdF3 system
For the purpose of investigating the luminescence properties of the systems containing both Sm3+ and Gd3+, two other systems were analyzed. The first one was a mixture of (LiF-CaF2)eut with x(SmF3)=0.2 and x(GdF3)=0.2, and the second was a mixture of (LiF-CaF2)eut with x(SmF3)=0.03 and x(GdF3)=0.3. Both systems were treated by the RSP method, and riffle-like and sphere-like fragments were separated and analysed by XRD and compared to spontaneously-cooled samples.
In spontaneously-cooled system (LiF-CaF2)eut- 0.20SmF3/(LiF-CaF2)eut-0.20GdF3 (labelled as the 20/20 system, according to the content of the lanthanide fluorides), the only initial phase still present is the LiF phase (Fig. 8 and Table 2). The initial GdF3 transforms to a LiGdF4 phase and the formation of a Ca/Sm+Gd/F phase is detected, together with some still undefined diffractions. Assuming a cubic phase-space, we find a=5.5750 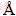 , which lies between those of the Ca/Sm/F and Ca/Gd/F systems (Fig. 3).
, which lies between those of the Ca/Sm/F and Ca/Gd/F systems (Fig. 3).
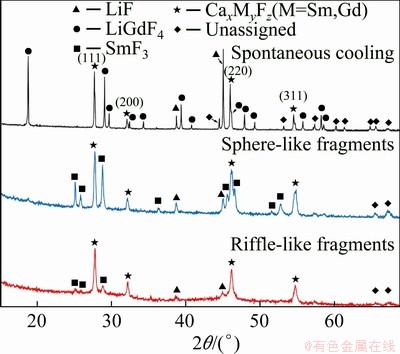
Fig. 8 XRD patterns of spontaneously-cooled melts, sphere-like and riffle-like fragments of (LiF-CaF2)eut- 0.20SmF3/(LiF-CaF2)eut-0.20GdF3 system
In the case of RSP-treated (LiF-CaF2)eut- 0.20SmF3/(LiF-CaF2)eut-0.20GdF3 samples (riffle- like and sphere-like), the cell parameter is slightly lower, but this decrease seems to be unimportant. Interestingly, RSP formation of a crystalline LiGdF4 phase is completely suppressed and the formation of a crystalline SmF3 phase is identified instead.
The reasons for the disappearance of LiGdF4 remain unclear. However, it should be mentioned that this compound was reported to melt incongruently [52]. It seems that during the RSP treatment, the speed of cooling is fast enough to suppress the crystallization of this phase in favour of concurrent processes of Ca/Gd/F phase crystallisation (or solid solution formation).
In the case of (LiF-CaF2)eut-0.03SmF3/(LiF- CaF2)eut-0.30GdF3 system (hereafter labelled as 3/30 due to the lanthanide fluoride content), the total lanthanide fluoride content in the mixture is 16.5 at.%. This differs from the 20/20 system, as well as the (LiF-CaF2)eut-0.30GdF3 (0/30) system. In the spontaneously-cooled 3/30 sample, the presence of LiF, LiGdF4 and Ca/Gd/F phases is identified, along with some unassigned diffractions (Fig. 9 and Table 2). However, the ratio of the diffraction intensities of these phases significantly differs from that of the spontaneously-cooled 0/30 system. Surprisingly, the presence of a crystalline CaF2 phase is detected in the 3/30 system, which is not observed in the 20/20 or 0/30 systems.
Fig. 9 XRD patterns of spontaneously cooled melts, sphere-like and riffle-like fragments of (LiF-CaF2)eut- 0.03SmF3/(LiF-CaF2)eut-0.30GdF3 system
In the RSP sphere-like fragments of the 3/30 system, the formation of a crystalline LiGdF4 phase is not suppressed as it is in the 20/20 system. Additionally, the presence of a CaF2 phase is observed, as found in the spontaneously cooled 3/30 sample. The general trend of lower values of the cell parameter a for RSP samples continues in the 3/30 system, although in this case the decrease is more significant (Fig. 3).
The differences in spontaneously cooled samples and RSP treated ones arise either from differences in phase diagrams reflecting the stability of the formed phases as well as in the kinetics of their formation or from energy and space relationships in the systems under special conditions. In the previous report [26], it was mentioned that the riffle-like fragments were subjected to a higher quenching rate while sphere- like samples represent the part of the melt at lower quenching rate. Indeed, the violation of the equilibrium conditions was the primary focus and obtained products provide the proof of such successful treatment.
3.2 Absorption and emission spectra of spontaneously-cooled systems
The absorption and fluorescence spectra provide information about the excited states of the ions in rapidly-cooled (LiF-CaF2)eut-(Gd or Sm)F3 systems. Moreover, these measurements give valuable spectroscopic information about the effect of the SmF3 and GdF3 content on the eutectic melt and information about how the luminescence properties change depending on the sample preparation. Complex spectroscopic measurements on the LiF-CaF2 molten system containing varying SmF3 and GdF3 content were performed.
The selected absorption spectra of the (LiF-CaF2)eut-xSmF3 systems are shown in Fig. 10, including the absorption spectrum of pure SmF3 for comparison. Sm3+ exhibits strong and sharp absorption bands over a wavelength range of 330-600 nm. These bands are assigned to f-f transitions, from the 6H5/2 ground state to various excited states of Sm3+ ions. The discrete absorption bands located at 344, 361, 375, 389, 400, 415, 441, 462 and 479 nm are assigned to the transitions from the 6H5/2 ground state to excited states of 4D7/2, 4D3/2, 6P7/2, 4L15/2, 6P3/2, 6P5/2, 4G9/2, 4I13/2 and 4M15/2, respectively [53-55], while the undoped (LiF-CaF2)eut sample does not show any absorption band as shown in Fig. 10. However, it should be noted that Sm3+ possess vast number of excited states, and thus the observed excitations might be the combination of more transitions, e.g. 6H5/2��4L13/2+6P3/2+4F7/2 [56]. The intensity of the absorption bands gradually increases with increasing content of Sm in the samples. Our measurements indicate that the intensity of the Sm3+ ion absorption bands reach a maximum value when the Sm dopant content is x(SmF3)=0.25. The shape of the absorption spectra and intensity of the bands do not change with increasing Sm content and the spectra at x=0.25, 0.30, 0.40 and 0.50 show almost identical shape and intensity. An excitation wavelength of 400 nm, corresponding to the most intense absorption band in Fig. 10 is selected for recording the emission spectra of the system (LiF-CaF2)eut-xSmF3 over a wavelength range of 500-700 nm (Fig. 11).
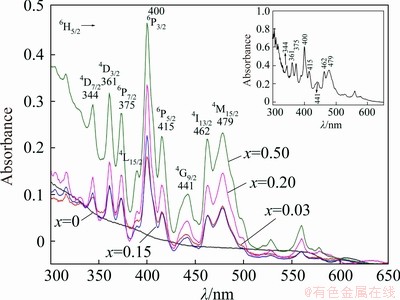
Fig. 10 Absorption spectra of spontaneously-cooled melts of (LiF-CaF2)eut-xSmF3 system (The inset shows the absorption spectrum of SmF3 powder)
Selected emission spectra of (LiF-CaF2)eut- xSmF3 systems are shown in Fig. 11 for the region of 500-740 nm under an excitation wavelength of 400 nm, with the emission spectrum of pure SmF3 included for comparison.
The Sm3+-doped (LiF-CaF2)eut systems show groups of typical emission lines at ~560, ~600, ~650 and ~710 nm assigned to 4G5/2��6H5/2, 4G5/2�� 6H7/2, 4G5/2��6H9/2 and 4G5/2��6H11/2 transitions, respectively, all corresponding also to pure SmF3, while the undoped (LiF-CaF2)eut sample does not exhibit any emission lines (Fig. 11). An increase in the content of Sm3+ leads to a decrease in the intensity of the fluorescence that can be ascribed to quenching effects promoted most probably through the cross-relaxation process.
The photoluminescence spectrum of the sample at x(SmF3)=0.03 is, however, completely different from the spectra with higher SmF3 content. The intensities of the bands are significantly stronger, and the shape and position of the spectral bands also differ from those of the other spectra. Significant splitting of the bands corresponding to the 4G5/2��6H5/2, 4G5/2��6H7/2 and 4G5/2��6H9/2 transitions is observed as well.
These splittings are on the level of 100- 300 cm-1 which might correspond to the ligand field effect that lifts the (2J+1)/2 degeneracy of the J levels in free ions [57,58]. Split bands are shown in Fig. 11 together with the schematic energy diagram of corresponding transitions including the number of Stark sublevels.
Another feature of the spectrum of the sample at x(SmF3)=0.03 is the occurrence of new bands (at 545, 585 and 620 nm). These can arise from emissions from higher excited states, e.g. 4F3/2. Similar situation was described also for LaMgB5O10: Sm3+ system [56] (4F3/2��6H5/2 at 549 nm, 4F3/2�� 6H7/2 at 587 nm and 4F3/2��6H9/2 at 629 nm).
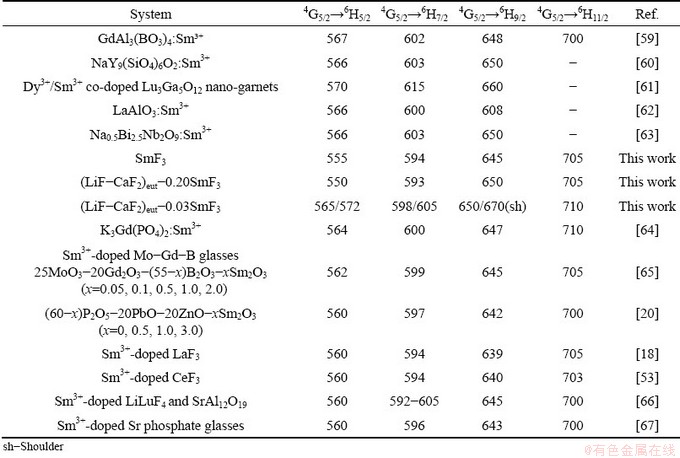
In Table 4, the aforementioned data are listed and compared with data for other systems. The 4G5/2��6Hj (j=5/2, 7/2, 9/2 and 11/2) transitions remain in similar positions for all cases, with only minor shifts.
The excitation spectra were also measured but no additional information is obtained. Moreover, less band resolution is observed in comparison to absorption spectra (thus excitation spectra are omitted).
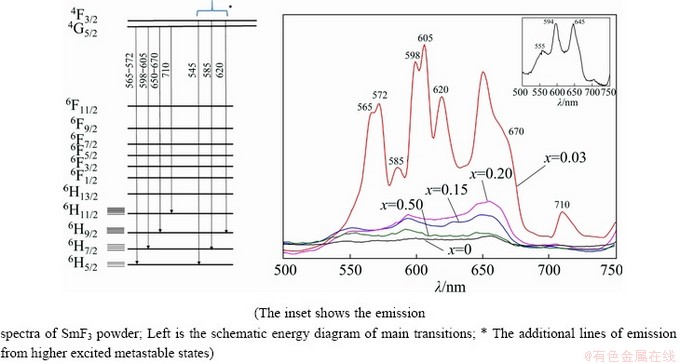
Fig. 11 Emission spectra of spontaneously-cooled melts of (LiF-CaF2)eut-xSmF3 system
Table 4 Emission wavelengths (nm) of transitions in Sm3+-containing systems
The system (LiF-CaF2)eut-xGdF3 is a less suitable case for measurements of absorption and emission spectra. The very large energy difference (~320 nm) between the ground state, 8S7/2, and the first excited state, 6Pj, and the absence of other states in between excludes the emission in the visible region. Thus, measurement of emission spectra for the system with GdF3 has been omitted.
3.3 Effect of morphology on spectral properties
In an earlier research work [26], it was mentioned that on application of the RSP method to molten systems (LiF-CaF2)eut-LaF3, the resulting samples exist in two distinct morphological forms: riffle-like and spherical fragments. For this reason, the next experiment was focused on the effect of the morphological forms on the optical properties of analysed samples. Based on the results of the emission spectra of one-component systems with various contents, two different systems were selected for further investigation. The first one is (LiF-CaF2)eut-0.03SmF3/(LiF-CaF2)eut- 0.30GdF3 (3/30) system, whose riffle-like fragments are labelled as 3/30r and spherical fragments are labelled as 3/30s. The second one is (LiF-CaF2)eut- 0.20SmF3/(LiF-CaF2)eut-0.20GdF3 (20/20) system, whose riffle-like fragments are labelled as 20/20r and spherical fragments are labelled as 20/20s. Due to the high sensitivity of the luminescence properties of the systems on the matrix used, some samples were also mechanically ground in an agate bowl. For these cases, the characters ��_g�� are appended to the label.
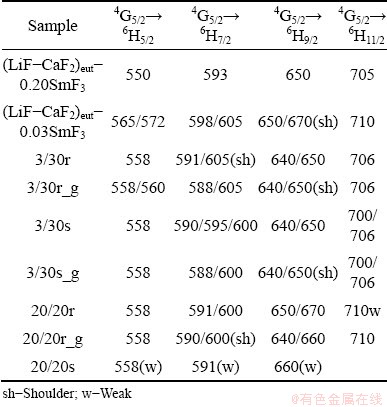
Figure 12 shows the emission spectra of all four forms of the 3/30 system, i.e. the two distinct morphological forms both as prepared by RSP and after mechanical grinding. It is evident that spectra of the sphere-like fragments of deeply undercooled systems show higher fluorescence than the riffle- like fragments. Next important aspect is the significant splitting of bands. This indicates that the host matrix of particular samples form Sm3+ should perform significant distortion from expected cubic symmetry (detected by XRD), affecting the energy levels degeneracy and energy level shifting. Table 5 summarises spectral bands of spontaneously cooled and RSP samples. As can be seen, it is problematic to identify any systematic shift (also shape, intensity or splitting) of the spectra as a whole. It is indicated that the samples perform some features of randomness, which is in accordance with the overall aim of the work to achieve randomness features of melt stacked in solid.
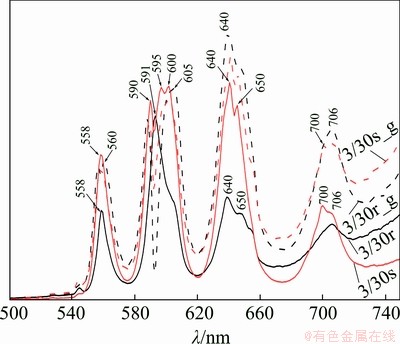
Fig. 12 Emission spectra of extremely rapidly-cooled 3/30 samples upon excitation at 400 nm
Table 5 Comparison of spectral bands (nm) of spontaneously-cooled and RSP samples
It should be pointed out that mechanical grinding leads to higher emission, an effect more pronounced for riffle-like fragments than for sphere-like fragments. It is not clear why this is the case. During RSP, rapid freezing of the structure of the molten salt occurs. It is found that the composition of rapidly-cooled samples includes a metastable FCC-type phase, in contrast to the spontaneously-cooled case, where only the initial components are present. It seems that the RSP method also gives a higher resolution of the emission spectrum and enhances the optical properties.
The emission spectra of the 20/20 system (Fig. 13) show somewhat different emission lines and shapes, in contrast to the emission spectra discussed previously for the 3/30 system; especially extensive splitting is partially removed (see Table 5 also). Moreover, the sphere-like components emit only minimum light, and the positions of the peak maxima indicate only emissions comparable to pure SmF3. Mechanical grinding leads to the significant change of luminescent properties. The band at 670 nm present in the sample 20/20r (which is a shoulder in the spectra of the sample 0/20) disappears on grinding (20/20r_g) and intensity of emission increases on grinding for 20/20r_g. This would mean that the effect of symmetry lowering in the system 20/20 is suppressed. The emission spectra of the spontaneously-cooled 20/20 sample in powdered form (figure not shown) does not show any emission corresponding to the observation in the (LiF�CCaF2)eut�CSmF3 system with higher SmF3 content leading to luminescence quenching.
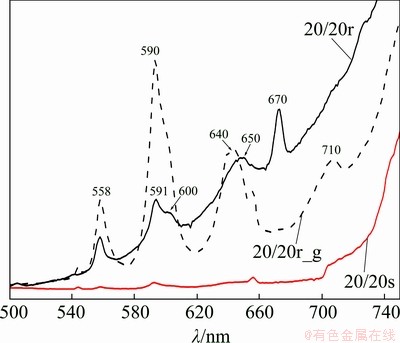
Fig. 13 Emission spectra of extremely rapidly-cooled 20/20 samples upon excitation at 400 nm
4 Conclusions
(1) The presence of LiF stabilises the cubic cell dimension, and the content dependence of the cell parameter changes only minimally with increasing the amount of SmF3 or GdF3 in spontaneously-cooled systems.
(2) Applying RSP shows that the ratio of lanthanum fluorides used influences the phases formed, as well as the crystallinity of the system in both spontaneously- and rapidly-cooled systems. The cell parameters of the discussed cubic phase(s) are generally the smallest for riffle-like fragments from RSP samples.
(3) The photoluminescent properties of the investigated systems are dependent on the lanthanide fluoride content in spontaneously-cooled samples. In the case of the x(SmF3)=0.03, the splitting of photoluminescent bands is observed due to the separation of energetically degenerated levels and new bands are observed due to the transitions from higher excited states as well.
(4) An additional dependence of photo- luminescent properties is observed on the morphological properties of the samples, i.e. whether the samples were processed by the RSP method followed by mechanical grinding.
Acknowledgments
This work was supported by the Slovak Research and Development Agency under contract No. APVV-15-0479, APVV-15-0738 and by the Slovak Grant Agency Vega Grant No. 2/0024/20. This work is the result of the project ��Effective Control of Production and Consumption of Energy�� from renewable sources ITMS 26240220028, supported by the Research and Development Operational Program supported by the European Regional Development Fund.
References
[1] ZHARIKOV E V, SMIRNOV V A. Luminescent dopants [C]//ROTMANS S R. Wide-Gap Luminescent Materials: Theory and Applications 1997. Boston, MA, US: Springer, 1997: 13-137.
[2] DIGONNET M J F. Rare-earth-doped fiber lasers and amplifiers [M]. 2nd ed. Boca Raton: CRC Press, 2001.
[3] PIKUZ T, FAENOV A Y, FUKUDA Y, KANDO M, BOLTON P, MITROFANOV A, VINOGRADOV A, NAGASONO M, OHASHI H, YABASHI M, TONO K, SENBA Y, TOGASHI T, ISHIKAWA T. Characterizing the luminescence properties of LiF crystal imaging detectors using femtosecond soft X-ray monochromatic free electron laser radiation [J]. Physica Status Solidi C, 2012, 9: 2239-2242.
[4] NAKAMURA F, KATO T, OKADA G, KAWAGUCHI N, FUKUDA K, YANAGIDA T. Scintillation and storage luminescence properties of MgF2 transparent ceramics doped with Ce3+ [J]. Optical Materials, 2017, 72: 470-475.
[5] RODNYI P A, KHANIN V M, VOLOSHINOVSKII A S, STRIGANYUK G B, GARIBIN E A, GUSEV P E, KRUTOV M A, DEMIDENKO A A. Optical and luminescence characteristics of BaF2 and BaF2:Tm in vacuum UV and UV spectral regions [J]. Optics and Spectroscopy, 2014, 117: 392-395.
[6] JANANI K, RAMASUBRAMANIAN S, SONI A K, RAI V K, THIYAGARAJAN P. Luminescence properties of LiYF4:Yb3+, Er3+ phosphors: A study on influence of synthesis temperature and dopant concentration [J]. Optik, 2018, 169: 147-155.
[7] RODRIGUEZ F, RIESEN H, G��DEL H U. Luminescence properties of Mn2+ in KMgF3 and KZnF3 perovskite crystals: Evidence of a dynamical Jahn-Teller effect [J]. Journal of Luminescence, 1991, 50: 101-110.
[8] GOLDBERG V, PACHECO D, MONCORGE R, di BARTOLO B. Luminescence characteristics of BaMnF4 and KMnF3 [J]. Journal of Luminescence, 1979, 18-19: 143-146.
[9] DEXPERT-GHYS J, RIBEIRO S J L, DUGAT P, AVIGNANT D. Crystal structures and luminescence properties of La3Zr4F25 and ��-LaZr3F15 [J]. Journal of Materials Chemistry, 1998, 8: 1043-1050.
[10] FEDOROV P P, LUGININA A A, KUZNETSOV S V, OSIKO V V. Nanofluorides [J]. Journal of Fluorine Chemistry, 2011, 132: 1012-1039.
[11] SECU M. Nanoparticles size effects in thermoluminescence of oxyfluoride glass-ceramics containing Sm3+-doped CaF2 nanocrystals [J]. Journal of Nanoparticle Research, 2011, 13: 2727-2732.
[12] KUROSAWA S, YAMAJI A, YOKOTA Y, FUTAMI Y, NISHIMOTO K, KAWAGUCHI N, FUKUDA K, YOSHIKAWA A. Growth and optical properties of LiF/LaF3 eutectic crystals [J]. Journal of the European Ceramic Society, 2014, 34: 2111-2115.
[13] JACOBSOHN L G, KUCERA C J, JAMES T L, SPRINKLE K B, di MAIO J R, KOKUOZ B, YAZGAN-KUKOUZ B, de VOL T A, BALLATO J. Preparation and characterization of rare earth doped fluoride nanoparticles [J]. Materials, 2010, 3: 2065-2068.
[14] BASIEV T T, DERGACHEV A Y, ORLOVSKII Y V, PROKHOROV A M. Multiphonon nonradiative relaxation from high-lying levels of Nd3+ ions in flouride and oxide laser materials [J]. Journal of Luminescence, 1992, 53: 19-23.
[15] ROLLET A L, ALLIX M, VERON E, DESCHAMPS M, MONTOUILLOUT V, SUCHOMEL M R, SUARD E, BARRE M, OCANA M, SADOC A, BOUCHER F, BESSADA C, MASSIOT D, FAYON F. Synthesis and structure resolution of RbLaF4 [J]. Inorganic Chemistry, 2012, 51: 2272-2282.
[16] KASA R, ADACHIA S. Mn-activated K2ZrF6 and Na2ZrF6 phosphors: Sharp red and oscillatory blue-green emissions [J]. Journal of Applid Physics, 2012, 112: 013506.
[17] SONG E H, WANG J Q, SHI J H, DENG T T, YE S, PENG M Y, WANG J, WONDRACZEK L, ZHANG Q Y. Highly efficient and thermally stable K3AlF6:Mn4+ as a red phosphor for ultra-high-performance warm white light-emitting diodes [J]. ACS Applied Materials & Interfaces, 2017, 9: 8805-8812.
[18] HA H M, HOA T T Q, van VU L, LONG N N. Optical properties and Judd�COfelt analysis of Sm ions in lanthanum trifluoride nanocrystals [J]. Journal of Materials Science: Materials in Electronics, 2017, 28: 884-891.
[19] DIEKE G H, CROSSWHITE H M. The spectra of the doubly and triply ionized rare earths [J]. Applied Optics, 1963, 2: 675-686.
[20] DOUSTI M R, GHOSHAL S K, AMJAD R J, SAHAR M R, NAWAZ F, ARIFIN R. Structural and optical study of samarium doped lead zinc phosphate glasses [J]. Optics Communications, 2013, 300: 204-209.
[21] FARRIES M C, MORKEL P R, TOWNSEND J E. Spectroscopic and lasing characteristics of samarium-doped glass fibre [J]. IEE Proceedings J: Optoelectronics, 1990, 137: 318-322.
[22] PANG S Y, MA F K, YU H, QIAN X B, JIANG D P, WU Y J, ZHANG F, LIU J, XU J Y, SU L B. Highly efficient continuous-wave laser operation of LD-pumped Nd,Gd:CaF2 and Nd,Y:CaF2 crystals [J]. Laser Physics Letters, 2018, 15: 055802.
[23] CATLOW C R A, CHADWICK A V, GREAVES G N, MORONEY L M. Direct observations of the dopant environment in fluorites using EXAFS [J]. Nature, 1984, 312: 601-604.
[24] GRIGOR��EVA N B, MAKSIMOV B A, SOBOLEV B P. X-ray diffraction study of Ca0.88Gd0.12F2.12 single crystals with a modified fluorite structure [J]. Crystallography Reports, 2000, 45: 718-720.
[25] GRANDJEAN D, CHALLIER T, JONES D J, VITSE P. Anion-excess fluorite type solid solutions MF2-NdF3 and MF2-UF4 (M=Ca, Ba): Conductivity and EXAFS spectroscopic study [J]. Solid State Ionics, 1992, 51: 297-303.
[26] SIMURDA M, BOCA M, SVEC P, JANICKOVIC D, SHI Z, MLYNARIKOVA J. Analysis of the extremely rapidly cooled molten system (LiF-CaF2)eut-LaF3 [J]. New Journal of Chemistry, 2018, 42: 4612-4623.
[27] DIEUDONNE B, CHABLE J, MAUVY F, FOURCADE S, DURAND E, LEBRAUD E, LEBLANC M, LEGEIN C, BODY M, MAISONNEUVE V, DEMOURGUES A. exploring the Sm1�CxCaxF3�Cx tysonite solid solution as a solid-state electrolyte: Relationships between structural features and F�Cionic conductivity [J]. The Journal of Physical Chemistry C, 2015, 119: 25170-25179.
[28] CHABLE J, DIEUDONNE B, BODY M, LEGEIN C, CROSNIER-LOPEZ M P, GALVEN C, MAUVY F, DURAND E, FOURCADE S, SHEPTYAKOV D, LEBLANC M, MAISONNEUVE V, DEMOURGUES A. Fluoride solid electrolytes: Investigation of the tysonite-type solid solutions La1-xBaxF3-x (x<0.15) [J]. Dalton Transactions, 2015, 44: 19625-19635.
[29] KUCHARIK M, KORENKO M, JANICKOVIC D, KADLECIKOVA M, BOCA M, OBONA J V. Rapid solidification of cryolite and cryolite-alumina melts [J]. Monatsh Chem, 2010, 141: 7-13.
[30] JACOBSON L A, MCKITTRICK J. Rapid solidification processing [J]. Materials Science and Engineering R: Reports, 1994, 11: 355-408.
[31] MLYNARIKOVA J, BOCA M, GURISOVA V, MACKOVA I, NETRIOVA Z. Thermal analysis and volume properties of the systems (LiF-CaF2)eut.-LnF3 (Ln=Sm, Gd, and Nd) up to 1273 K [J]. Journal of Thermal Analysis and Calorimetry, 2016, 124.
[32] STREL'TSOV V A, TSIREL'SON V G, OZEROV R P, GOLOVANOV O A. Electronic and thermal parameters of ions in calcium fluoride: Results of application of the regularized least-squares method [J]. Kristallografiya, 1988, 33: 90-97.
[33] SOBOLEV B P, FEDOROV P P. Phase diagrams of the CaF2-(Y,Ln)F3 svstems: I. Experimental [J]. Journal of the Less Common Metals, 1978, 60: 33-46.
[34] THOMA R E, WEAVER C F, FRIEDMAN H A, INSLEY H, HARRIS L A, YAKEL H A Jr. Phase equilibriums in the system LiF-YF3 [J]. The Journal of Physical Chemistry A, 1961, 65: 1096-1099.
[35] GORYUNOV A V, POPOV A I. Crystal structure of LiYF4 [J]. Zhurnal Neorganicheskoi Khimii 1992, 37: 276-279.
[36] GORYUNOV A V, POPOV A I, KHAJDUKOV N M, FEDOROV P P. Crystal structure of lithium and yttrium complex fluorides [J]. Materials Research Bulletin, 1992, 27: 213-220.
[37] GARCIA E, R. RYAN R. Structure of the laser host material LiYF4 [J]. Acta Crystallographica Section C, 1993, 49: 2053-2054.
[38] GRZECHNIK A, SYASSEN K, LOA I, HANFLAND M, GESLAND J Y. Scheelite to fergusonite phase transition in YLiF4 at high pressures [J]. Physical Review B, 2002, 65: 104102.
[39] LI S, AHUJA R, JOHANSSON B. Wolframite: The post-fergusonite phase in YLiF4 [J]. Journal of Physics: Condensed Matter, 2004, 16: S983.
[40] GRZECHNIK A, UNDERWOOD C C, KOLIS J W, FRIESE K. Crystal structures and stability of LiCeF5 and LiThF5 at high pressures: A comparative study of the coordination around the Ce4+ and Th4+ ions [J]. Journal of Fluorine Chemistry, 2013, 156: 124-129.
[41] LALIGANT Y, le BAIL A, FEREY G, AVIGNANT D, COUSSEINS J C. Determination of the crystal structure of lithium hexafluoroterbate(2-) from X-ray and neutron powder diffraction: An example of lithium in five-fold coordination [J]. European Journal of Solid State and Inorganic Chemistry, 1989, 25: 551-563.
[42] JOSSE M, EL-GHOZZI M, AVIGNANT D, ANDRE G, BOUREE F. Magnetic structures of the M2TbF6 (M=Li, K, Rb) fluorides: A complex behavior resulting from frustration [J]. Journal of Solid State Chemistry, 2007, 180: 1623-1635.
[43] EL-GHOZZI M, AVIGNANT D, COUSSEINS J C. X-ray powder diffraction study of lithium terbium fluoride Li4TbF8 [J]. European Journal of Solid State and Inorganic Chemistry, 1992, 29: 981-992.
[44] THOMA R E, BRUNTON G D, PENNEMAN R A, KEENAN T K. Equilibrium relations and crystal structure of lithium fluorolanthanate phases [J]. Inorganic Chemistry, 1970, 9: 1096-1101.
[45] GRZECHNIK A, FRIESE K, DMITRIEV V, WEBER H P, GESLAND J Y, CRICHTON W A. Pressure-induced tricritical phase transition from the scheelite structure to the fergusonite structure in LiLuF4 [J]. Journal of Physics: Condensed Matter, 2005, 17: 763-770.
[46] BELAIGUE, COUSSEINS. Systems MF-CeF4 (M=alkaline metal) [J]. Inorganic Chemistry Review, 1972, 9: 789. (in France)
[47] FELDNER K, HOPPE R. New fluorides of tetravalent cerium, praseodymium and terbium [J]. Inorganic Chemistry Review, 1983, 20: 351. (in France)
[48] BRUNTON G D, INSLEY H, MCVAY T N, THOMA R E, Crystallographic data for some metal fluorides, chlorides and oxides [M]. Washington DC, USA: Government Printing Office, 1965: 267.
[49] XUN X, FENG S, XU R. Hydrothermal synthesis of complex fluorides LiHoF4 and LiErF4 with scheelite structures under mild conditions [J]. Materials Research Bulletin, 1998, 33: 369-375.
[50] FEDOROV P. Systems of alkali and rare-earth metal fluorides [J]. Russian Journal of Inorganic Chemistry, 1999, 44: 1703-1727.
[51] TSYTSENKO A K, FRANK-KAMENETSKAYA O V, FUNDAMENSKII V S, FRANK-KAMENETSKII V A. Crystal structures of Ca(1-x)Nd(x)F(2+x) (x=0.1,0.2) from precisional X-ray diffraction data [J]. Kristallografiya, 1991, 36: 347-353.
[52] KLIMM D, DOS SANTOS I, RANIERI I, BALDOCHI S. Phase equilibria and crystal growth for LiREF4 scheelite laser crystals [C]//Materials Research Society Symposia Proceedings. Warrendale: Materials Research Society, 2009: 167-172.
[53] HUONG D T M, TIEN N T, VU L V, LONG N N. Synthesis and optical characterization of samarium doped cerium fluoride nanoparticles [J]. VNU Journal of Science: Mathematics �C Physics, 2015, 31: 23-31.
[54] OH S Y, KIM J Y, BAE S E, CHO Y H, YEON J W, SONG K. Spectroscopic analysis of trivalent cerium and holmium ions in LiCl-KCl eutectic melt at high temperature [J]. Journal of Luminescence, 2013, 134: 706-709.
[55] MEEJITPAISAN P, INSIRIPONG S, KEDKAEW C, KIM H J, KAEWKHAO J. Radioluminescence and optical studies of gadolinium calcium phosphate oxyfluoride glasses doped with Sm3+ [J]. Radiation Physics and Chemistry, 2017, 137: 62-67.
[56] SAKIRZANOVAS S, KATELNIKOVAS A, BETTENTRUP H, KAREIVA A, J��STEL T. Synthesis and photoluminescence properties of Sm3+-doped LaMgB5O10 and GdMgB5O10 [J]. Journal of Luminescence, 2011, 131: 1525-1529.
[57] B��NZLI J C G. On the design of highly luminescent lanthanide complexes [J]. Coordination Chemistry Reviews, 2015, 293-294: 19-47.
[58] KHAN L U, KHAN Z U, Bifunctional nanomaterials: Magnetism, luminescence and multimodal biomedical applications [C]//SHARMAS S K. Complex Magnetic Nanostructures: Synthesis, Assembly and Applications 2017. Cham, Springer International Publishing, 2017: 121-171.
[59] JAMALAIAH B C. Optimization of photoluminescence of GdAl3(BO3)4:Sm3+ phosphors for solid state lighting devices [J]. Journal of Molecular Structure, 2017, 1146: 546-553.
[60] MEI L F, LIU H K, LIAO L B, ZHANG Y Y, KUMAR R V. Structure and photoluminescence properties of red-emitting apatite-type phosphor NaY9(SiO4)6O2:Sm3+ with excellent quantum efficiency and thermal stability for solid-state lighting [J]. Scientific Reports, 2017, 7: 15171.
[61] PAMULURI H, RATHAIAH M, LINGANNA K, JAYASANKAR C K, LAVIN V, VENKATRAMU V. Role of Dy3+��Sm3+ energy transfer in the tuning of warm to cold white light emission in Dy3+/Sm3+ co-doped Lu3Ga5O12 nano-garnets [J]. New Journal of Chemistry, 2018, 42: 1260-1270.
[62] KAUR J, SINGH D, SURYANARAYANA N S, DUBEY V. UV induced thermoluminescence and photoluminescence studies of Sm3+ doped LaAlO3 phosphor [J]. Journal of Display Technology, 2016, 12: 928-932.
[63] ZHANG Q W, ZHANG Y, SUN H Q, SUN Q, WANG X S, HAO X H, AN S L. Photoluminescence, photochromism, and reversible luminescence modulation behavior of Sm-doped Na0.5Bi2.5Nb2O9 ferroelectrics [J]. Journal of the European Ceramic Society, 2017, 37: 955-966.
[64] GUPTA P, BEDYAL A K, KUMAR V, KHAJURIA Y, SHARMA V, NTWAEABORWA O M, SWART H C. Energy transfer mechanism from Gd3+ to Sm3+ in K3Gd(PO4)2:Sm3+ phosphor [J]. Materials Research Express, 2015, 2: 076202.
[65] RAJARAMAKRISHNA R, RUANGTAWEE Y, KAEWKHAO J. Sm3+-doped molybdenum gadolinium borate glasses for orange emission laser active medium [J]. Ukrainian Journal of Physics, 2018, 63: 721-732.
[66] MARZAHL D T, METZ P W, KRANKEL C, HUBER G. Spectroscopy and laser operation of Sm3+-doped lithium lutetium tetrafluoride (LiLuF4) and strontium hexaaluminate (SrAl12O19) [J]. Optics Express, 2015, 23: 21118-21127.
[67] OKASHA A, ABDELGHANY A M, MOHAMED S K, MARZOUK S Y, EL-BATAL H A, GAAFAR M S. Gamma ray interactions with samarium doped strontium phosphate glasses [J]. Journal of Materials Science: Materials in Electronics, 2018, 29: 20907-20913.
Miroslav BOCA1, Jarmila MLYNARIKOVA1, Iveta MACKOVA1, Dusan JANICKOVIC2, Adriana CZIMEROVA1, ʯ����3
1. Institute of Inorganic Chemistry, Slovak Academy of Sciences, DubravskA cesta 9, SK-845 36 Bratislava, Slovakia;
2. Institute of Physics, Slovak Academy of Sciences, DubravskA cesta 9, SK-84 511 Bratislava, Slovak;
3. ������ѧ ұ��ѧԺ������ 110819
ժ Ҫ��(LiF-CaF2)eut-xSmF3(-xGdF3) (x=0.03~0.50)����ͨ����Ȼ��ȴ�Ϳ�����ȴ������õ���״����״������ò��ͨ��X�������䡢���չ���ӫ����������֣�LiF�����ȶ������ṹ����LnF3����ֱ��Ӱ������ṹ�;�̬��(LiF-CaF2)eut-xSmF3(-xGdF3)��ϵ�Ĺ��·���������LnF3����Ӱ�죬��SmF3����Ϊ0.03ʱ��Sm3+�ķ����ǿ��������۲���������ȴ��ʽ����Ʒ��ò��Ӱ�����ѧ���ԣ���е��ĥ����Ʒ��ӫ�����ܺͷ���ǿ�������о�������ӫ�������һ�ֿ����ڼ�����֤�״��������Ѻ�ɽ��;ֲ��Գ��ԵĹ�������
�ؼ��ʣ����Σ��������ϵͳ��SmF3��GdF3�����չ��ף�ӫ����ף���������
(Edited by Bing YANG)
Corresponding author: Miroslav BOCA; Tel: +421-2-59410-400; Fax: +421-2-59410-444; E-mail: miroslav.boca@savba.sk
DOI: 10.1016/S1003-6326(21)65568-1
1003-6326/ 2021 The Nonferrous Metals Society of China. Published by Elsevier Ltd & Science Press
2021 The Nonferrous Metals Society of China. Published by Elsevier Ltd & Science Press

