Preparation of salt-based colloid palladium of high concentration
来源期刊:中国有色金属学报(英文版)2005年第1期
论文作者:郑雅杰 肖发新 易丹青 龚竹青 李新海
文章页码:190 - 194
Key words:salt-based colloid palladium; preparation; activation; stability
Abstract: A kind of salt-based colloid palladium of high concentration was prepared, with concentration up to 3.6g/L on amount of PdCl2. The optimal preparation conditions of the salt-based colloid palladium were that the PdCl2 and NaCl concentrations were respectively 3.6g/L and 175g/L, mole ratio of Sn to Pd was 50∶1, reaction temperature was 20-35℃, with urea, ascorbic acid and vanillin added in a proper amount. The test results of optimal condition show that the time of starting hydrogen-deposition is 9s, the time of completely coating copper on a test substrate is 2min, the stability time of colloid palladium is 98h after it is diluted into 0.1g/L(on amount of PdCl2) when the solution temperature is 20℃, the backlight lever of electroless copper plating layer is 10th grade of 10-grade system, the adhesion force of the copper layer is up to GB5270―85 of China, and the average particle size of the colloid palladium is 81 nm measured by Master Sizer.
ZHENG Ya-jie(郑雅杰)1, XIAO Fa-xin(肖发新)1, YI Dan-qing(易丹青)2,GONG Zhu-qing(龚竹青)1, LI Xin-hai(李新海)1
(1. School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China;
2. School of Materials Science and Engineering, Central South University, Changsha 410083, China)
Abstract: A kind of salt-based colloid palladium of high concentration was prepared, with concentration up to 3.6g/L on amount of PdCl2. The optimal preparation conditions of the salt-based colloid palladium were that the PdCl2 and NaCl concentrations were respectively 3.6g/L and 175g/L, mole ratio of Sn to Pd was 50∶1, reaction temperature was 20-35℃, with urea, ascorbic acid and vanillin added in a proper amount. The test results of optimal condition show that the time of starting hydrogen-deposition is 9s, the time of completely coating copper on a test substrate is 2min, the stability time of colloid palladium is 98h after it is diluted into 0.1g/L(on amount of PdCl2) when the solution temperature is 20℃, the backlight lever of electroless copper plating layer is 10th grade of 10-grade system, the adhesion force of the copper layer is up to GB5270―85 of China, and the average particle size of the colloid palladium is 81nm measured by Master Sizer.
Key words: salt-based colloid palladium; preparation; activation; stability CLC number: O614.82+3
Document code: A
1 INTRODUCTION
Electroless plating is one of the most important surface treatment technologies. An amount of activation particles must be adsorbed on substrate before electroless plating. The activator used in electroless plating influences directly on the uniformity, adhesion force of the layer and the time of starting hydrogen-deposition. Therefore, activator is most important to electroless plating[1, 2]. Two-step activation, colloid palladium, copper-tin colloid and ion palladium are applied in electroless plating[3]. The two-step activation is rarely applied in automatic production because of the complicated process. The copper-tin colloid is rarely applied because of little activation. Though the activation and stability is nice, the ion palladium has a high cost because unstable and expensive NaBH4 is used to reduce Pd2+. The colloid palladium is widely applied in electroless plating, especially the electroless copper plating in printed circuit board(PCB) due to the better activation, less dosage, simple technique and reliable quality.
There are two kinds of colloid palladium, which are an acid-based colloid palladium and a salt-based colloid palladium. The acidity of acid-based colloid palladium is so high that “pink ring” is produced on the hole wall after depositing copper[4-6]. In addition, the acid mist pollutes environment badly during production and application[7, 8]. In contrast to acid-based colloid palladium, salt-based colloid palladium can overcome the above disadvantages. As present, reports on salt-based colloid palladium are mostly the introductions of its components[2, 3, 9-13]. In these reports, the concentration of colloid palladium is in the range of 0.1 to 0.25g/L(on amount of PdCl2), while the reports on high concentration colloid palladium are few. In this paper, the palladium chloride concentration was up to 3.6g/L, while the concentration was diluted to 0.1g/L in application. Obviously, the salt-based colloid palladium has advantages such as low cost and convenient transport.
2 EXPERIMENTAL
2.1 Preparation of salt-based colloid palladium
An amount of stannous chloride was dissolved into some water. According to mole ratio of Sn to Pd(2∶1), the SnCl2 solution was gauged and added into PdCl2 solution with slow agitation and at constant temperature. After several minutes, the surplus SnCl2 solution was added into the above mixed solution of SnCl2 and PdCl2 when the color of blended solution turned brown dark. After agitating for 10min, the total mixed solution was dispersed into the stabilization solution which contained NaCl, SnCl2, urea, ascorbic acid and vanillin. After maturing 4h in a water bath heater, the salt-based colloid palladium was obtained.
2.2 Activation test of salt-based colloid palladium
In this paper, a PCB was used as the substrate of electroless plating. A piece of PCB was cut into many test pieces with size of 4cm×4cm. A test piece was first washed in alkali cleaner solution at 70-80℃ before etching the test piece in H2O2/H2SO4 solution. After etching, the test piece was dipped in the NaCl/(NH2)2CO solution, then the test piece was put in a activation solution of salt-based colloid palladium for about 5min at 20-30℃. The concentration of an activation solution was 0.1g/L(on amount of PdCl2).After activation, the test piece was dipped in HBF4 solution.
After the above pretreatment, copper was electrolessly plated on the test piece at 30℃. Cu2+ concentration was 2.3g/L in a bath solution. The time of starting hydrogen-deposition and the time of completely coating copper on a substrate are adopted to measure the activation of colloid palladium in the test. The time of starting hydrogen-deposition is the time when hydrogen bubbles occur in the bath since the test piece is dipped into a bath. The time of completely coating copper on a substrate is the time when the surface of test piece is coated with copper completely since the test piece is dipped into a bath. The activation solution was the colloid palladium diluted by the solution of NaCl and NH2CONH2, and the concentration of the activation solution was 0.1g/L (on amount of PdCl2). The test time was measured by a stopwatch.
2.3 Stability time of salt-based colloid palladium
50mL activation solution was filled in a tube with volume capacity of 50mL. Air was flowed from the bottom by a small glass pipe when the temperature of activation solution was 20℃ and airflow was 20L/h (WM-2H gas compressor). In this paper, a stability time is used to measure the stability of salt-based colloid palladium. The stability time is the time when the brown dark solution disappears completely and becomes transparent since air flows[16].
2.4 Backlight lever test
According to above-mentioned steps, copper was deposited on a piece of PCB by electroless plating for 10min.The test piece was cut along the center of holes according to Fig.1.Then the hole side of the cut piece was polished by sand papers. Backlight levers were determined according to the picture of 10-lever system.

Fig.1 Picture of backlight test (mm)
2.5 Granularity analysis of salt-based colloid palladium
After adding 3mL salt-based colloid palladium solution into 400mL methanol, the particle size of the colloid palladium was measured by Master Sizer2000 Particle Meter(Malvern, English).
2.6 Adhesion force test of electroless copper plating layer
Adhesion force was measured according to GB5270―85 of China standard.
3 RESULTS AND DISCUSSION
3.1 Impact of mole ration of Sn to Pd on activation and stability of salt-based colloid palladium
Salt-based colloid palladium was prepared according to experiment step 2.1. The impacts of n(Sn)/n(Pd) on the activation and stability of the colloid palladium are shown in Figs.2 and 3 when PdCl2 concentration was 1.6g/L, NaCl concentration was 200g/L and other components were fixed.

Fig.2 Impact of n(Sn)/n(Pd) on activation of salt-based colloid palladium

Fig.3 Impact of n(Sn)/n(Pd) on stability of salt-based colloid palladium
In this paper, t1 stands for the time of starting hydrogen-deposition, t2 stands for the time of completely coating copper on a substrate, t3 stands for the stability time. It is presented in Figs.2 and 3 that the time of starting hydrogen-deposition 12, 9, 12, 11, 10, 11, 10s, the time of completely coating copper on a substrate was 172, 168, 170, 167, 162, 170, 164s, the stability time of salt-based colloid palladium was 10, 15, 25, 35, 50, 54, 70h when n(Sn)/n(Pd) was 10, 15, 20, 25, 30, 50, 70 respectively. It was testified that n(Sn)/n(Pd) had less effect on the activation of salt-based colloid palladium, but the stability of salt-based colloid palladium increased with the increases of n(Sn)/n(Pd).
Because the concentration of activation solution was 0.1g/L(PdCl2), the concentration of colloid palladium particles was almost the same and the quantity of Pd particle deposited on a unit area of a substrate was nearly uniform after activation. Therefore, the time of starting hydrogen-deposition and the time of completely coating copper on a substrate were almost equal. The structure of colloid palladium is
{(Pd0)m・nSn2+}・x Cl-・(2n-x) Cl-
Sn2+ in colloid palladium particles and in the solution are oxided into Sn4+ when air is aerated into the colloid solution, and colloid palladium loses stability to precipitate. When palladium concentration in a solution was fixed, Sn2+ concentration increased with the increases of n(Sn)/n(Pd). The more the concentration of Sn2+, the more stable the colloid palladium. The stability time of salt-based colloid palladium was 54h when n(Sn)/n(Pd) was 50∶1, which was equal to that of acid-based colloid palladium ZL-14 prepared by the author and applied in industry widely. Therefore, it can satisfy the requirement of industry application when n(Sn)/n(Pd) is 50∶1 in the preparation of salt-based colloid palladium.
3.2 Impact of NaCl concentration on activation and stability of salt-based colloid palladium
The impacts of NaCl concentration on the activation and stability of the colloid palladium are shown in Figs.4 and 5 when PdCl2 concentration was 1.6g/L, n(Sn)/n(Pd) was 50∶1 and other components were fixed.
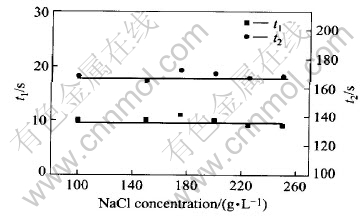
Fig.4 Impact of NaCl concentration on activation of salt-based colloid palladium
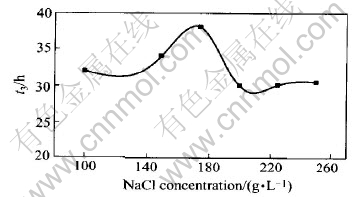
Fig.5 Impact of NaCl concentration on stability of salt-based colloid palladium
In Fig.4, the time of starting hydrogen-deposition is about 10s and the time of completely coating copper on a substrate is 170s approximately when NaCl concentration is in the range of 100-250g/L. Obviously, the impact of NaCl concentration on slat-based colloid palladium is little. There are some impacts of NaCl concentration on the stability time according to Fig.5.When NaCl concentration is 175g/L, the stability time is up to 38h.
Because the concentration of activation solution is 0.1g/L(PdCl2), the concentration of colloid palladium particle was almost same and the nearly same quantity of Pd was deposited on a test piece after activation. So the time of starting hydrogen-deposition and the time of completely coating copper on a substrate are almost invariable. From the structure of colloid palladium, Cl- can increase the stability of colloid palladium. The stability of salt-based colloid palladium increases with the increases of sodium chloride concentration. However, when NaCl concentration is more than 175g/L, the stability time of salt-based colloid palladium decreased with the increases of sodium chloride concentration. Because NaCl is a strong electrolyte, the double-charge-layer of colloid is compressed by Cl- when NaCl concentration is more than 175g/L, ζ voltage of a colloid palladium decreases. As a result, the stability of salt-based colloid palladium falls down.
3.3 Impact of PdCl2 concentration on activation and stability of salt-based colloid palladium
The impacts of PdCl2 concentration on the activation and the stability of the colloid palladium are respectively shown in Figs.6 and 7 when NaCl concentration is 175g/L, n(Sn)/n(Pd) is 50∶1 and other components are fixed.

Fig.6 Impact of PdCl2 concentration on activation of salt-based colloid palladium

Fig.7 Impact of PdCl2concentration on stability of salt-based colloid palladium
In Fig.7, the stability time of salt-based colloid palladium is 31, 54, 85, 96, 98, 100h when palladium chloride concentration is 0.8, 1.6, 2.4, 3.2, 3.6, 4.0g/L respectively. It turns out that PdCl2 concentration has less effect on the activation of salt-based colloid palladium, but the stability of salt-based colloid palladium increases as PdCl2 concentration increases.
Because the palladium chloride concentration in an activation solution is 0.1g/L (PdCl2), the concentration of colloid palladium particles is almost the same, the quantity of Pd deposited on test piece after activation is nearly uniform. As a result, the time of starting hydrogen-deposition and the time of completely coating copper on a substrate are almost the same.
The higher the concentration of PdCl2 is, the faster the colloid palladium cores form. In this way, colloid palladium particles are very fine and colloid particles contain more Sn2+. So the stability of colloid palladium increases with the increases of PdCl2 concentration. But Pd separates directly out of the solution because colloid palladium cores forms too fast when the PdCl2 concentration was equal to or more than 4.0g/L.
In experiment, it is found that the grass green solution with little activation was produced when the surplus SnCl2 solution was added too early or too late in a PdCl2 solution. The reaction occurs as follows in the solution at first.
PdCl2+2SnCl2→[PdSn2]6++6Cl-
[PdSn2]6+ is unstable and it decomposes to fine palladium particles , the reaction is
[PdSn2]6+→Pd0+Sn4++Sn2+
If the surplus SnCl2 solution was added too early, the reaction occurs
[PdSn2]6++Sn2+→[PdSn3]8+
[PdSn3]8+ is a grass green ion in a solution, its activation is very little. Therefore, the activation of colloid palladium is bad. When the surplus SnCl2 solution is added too late, colloid particles become too large, so the activation and stability is also bad. Thereof the surplus SnCl2 solution should be added into the solution in a proper time in the preparation of salt-based colloid palladium.
3.4 Impact of reaction temperature on activation of salt-based colloid palladium
Impact of reaction temperature on the activation of salt-based colloid palladium is described in Fig.8 when PdCl2 concentration is 1.6g/L, n(Sn)/n(Pd) is 50∶1, NaCl concentration is 175g/L and other components are fixed.
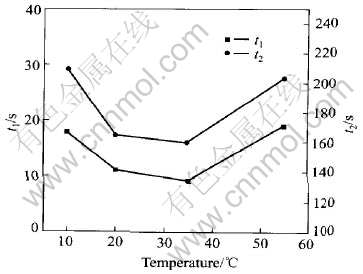
Fig.8 Impact of reaction temperature on activation of salt-based colloid palladium
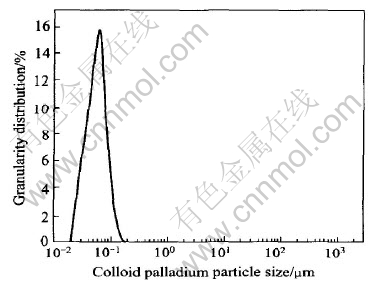
Fig.9 Granularity distribution of salt-based colloid palladium
In Fig.8, the time of starting hydrogen-deposition is 18, 11, 9, 19s and the time of completely coating copper on a substrate is 210, 165, 163, 208s when the reaction temperature is 10, 20, 35, 55℃ respectively. Obviously, the activation of slat-colloid palladium is the best when the reaction temperature is in the range of 20-35℃.
The speed of forming colloid particles was slower when the reaction temperature was lower. When the reaction temperature was too high, colloid particle formed would congregate and become large because of faster reaction and heat movement. Therefore, a colloid palladium with good activation could be only obtained when the reaction temperature is 20-35℃.
4 CONCLUSIONS
1) When n(Sn)/n(Pd) is fixed, the stability of salt-based colloid palladium increased as PdCl2 concentration increases. When PdCl2 concentration is constant, the stability of salt-based colloid palladium increases as n(Sn)/n(Pd) increases. The stability of salt-based colloid palladium increases as NaCl concentration increases when NaCl concentration is 100-175g/L.
2) The salt-based colloid palladium has a high activation when reaction temperature is 20-35℃.
3) The optimal preparation conditions of the salt-based colloid palladium are that PdCl2 concentration is 3.6g/L, n(Sn)/n(Pd) is 50∶1, NaCl concentration is 175g/L, the reaction temperature is 20-35℃ and urea, ascorbic acid and vanillin are added in a proper amount.
4) Salt-based colloid palladium is prepared. The time of starting hydrogen-deposition is 9s, the time of completely coating on a substrate is 2min. The stability time of activation solution (0.1g/L PdCl2) is 98h when solution temperature is 20℃, the particle sizes of salt-colloid palladium is 81nm, the backlight lever of electroless copper plating layer is 10th grade and the adhesion force of the copper layer is up to GB5270―85 of China stand- ard after electroless copper plating for 10min.
REFERENCES
[1]JIN Hong, CHEN Sen. Printed circuit technology[M]. Beijing: Chemical Industry Publishing Company, 2003. 12-20.
[2]ZHANG Yong-feng, MA Ling-jun, GUO Wei-min. Activation technology of nonmetal electroless plating[J]. Material Exploitation and Utility, 2004, 15(2): 30-34.
[3]LI Bing, WEI Xi-wen, ZHANG Zhao-yang. Study on activation technique of electroless nickel-plating on nonmetal material[J]. Material Protect, 2001. 2, (34)2: 17-18.
[4]JIANG Xiao-xia, SHEN Wei. Electroless Plating Theory and Practice[M]. Beijing: National Defence Industry Publishing Company, 2000. 6.
[5]ZHENG Ya-jie, GONG Zhu-qing, CHEN Bai-zhen. Hole metalization of printed circuit board and path of technology improvement[J]. Material Report, 2003,17(4): 11-13.
[6]Gulla M, Conlan W. Catalyst Solution for Electroless Deposition of Metal on Substrate[P]. US 3874882,1975.
[7]Hidber P C, Helbig W, Kim E. Microcontact printing of palladium colloids: Micro-scale patterning by electroless deposition of copper[J]. Langmuir, 1996, 12(5): 1375-1380.
[8]Gulla M, Conlan W Jr. Catalyst Solution for Electroless Metal Deposition on a Substrate[P]. US 3904792,1975.
[9]GONG Fan, WANG Bin-sheng. Development of the activation technology of electroless plating[J]. Application Science, 2002, (29)3: 59-60.
[10]CHEN Da-hong. Printed circuit hole metalization[J]. Electro Technology, 2001. 5, (22)3: 102-105.
[11]Brandow S, Chen M S, Wang T. Size-controlled colloidal Pd(Ⅱ) catalysts for electroless Ni deposition in nanolithography applications[J]. Electrochem Soc, 1997, 12, (144)10: 3425-3433.
[12]Gulla M, Dutkewych O B, Blandon J J. Catalyst Metal of Reduced Particle Size[P]. US 4634468, 1987.
[13]YU Wei-yong, LIU Man-hong, LIU Han-fan. Preparation of polymer-stabilized noble metal colloids[J]. Journal of Colloid and Interface Science, 1999, (210): 218-221.
[14]Cohen R L, Meek R L. Process for Electroless Plating Using Colloid Sensitization and Acid Rinse[P]. US 4008343, 1977.
[15]Kanoh O, Yoshida Y, Ogiso Y. Activating Catalytic Solution for Electroless Plating and Method for Electroless Plating[P]. US 5989787, 1999.
[16]ZHENG Fu-yang, MA Yan-chun. Impact of several chloride on stability and activation of salt-based colloid palladium[J]. Plating and Finishing, 1990, 12(3): 9-12.
Foundation item: Project supported by Hubei Daye Nonferrous Metal Corporation of China
Received date:2004-06-07; Accepted date: 2004-11-08
Correspondence: ZHENG Ya-Jie, Professor, PhD; E-mail: ZZYYJJ01 @yahoo.com.cn

