J. Cent. South Univ. Technol. (2009) 16: 0513-0519
DOI: 10.1007/s11771-009-0085-4
Selective catalytic reduction of NO with NH3 over sol-gel-derived CuO-CeO2-MnOx/γ-Al2O3 catalysts
ZHAO Qing-sen(赵清森), XIANG Jun(向 军), SUN Lu-shi(孙路石),
SHI Jin-ming(石金明), SU Sheng(苏 胜), HU Song(胡 松)
(State Key Laboratory of Coal Combustion, Huazhong University of Science and Technology, Wuhan 430074, China)
Abstract: Granular CuO-CeO2-MnOx/γ-Al2O3 catalysts were synthesized by the sol-gel method. The performance of the CuO-CeO2-MnOx/γ-Al2O3 catalysts for the selective catalytic reduction (SCR) was studied in a fixed bed system. Preliminary tests were carried out to analyze the behavior of NH3 and NO over catalyst in the presence of oxygen. The optimum temperature range for SCR over the CuO-CeO2-MnOx/γ-Al2O3 catalysts is 300-400 ℃. The catalysts maintain nearly 100% NO conversion at 350 ℃. The NH3 oxidation experiments show that both NO and N2O are produced gradually with the increase of temperature. The catalysts in this experiment have a stronger oxidation property on NH3, which improves the denitrification activity at low temperature. The over-oxidation of NH3 at high temperature is the main cause leading to a decrease in the NO conversion. The NH3 and NO desorption experiments show that NH3 and NO can be adsorbed on CuO-CeO2-MnOx/γ-Al2O3 granular catalysts. The transient response of NH3 and NO indicates that the SCR reaction proceeds in accordance with the Eley-Rideal mechanism. The adsorbed NO has little influence on the denitrification activity in SCR process.
Key words: sol-gel method; CuO-CeO2-MnOx/γ-Al2O3; NH3; NO; conversion; selective catalytic reduction (SCR)
1 Introduction
Sulfur dioxide (SO2) and nitrogen oxides (NOx) are the major air pollutants, which have to be removed before emitting flue gas into the atmosphere. Various processes are under operation to remove SO2 and NOx from flue gas. Numerous researches on the simultaneous removal of SO2 and NOx were carried out to find the cost-effective catalysts. In simultaneous removal of SO2 and NOx, the selective catalytic reduction (SCR) process with CuO/γ-Al2O3 has distinct advantages to remove SO2 and NOx in flue gas because of the high desulfurization and denitrification efficiency in the same temperature range (300-400 ℃) and easy regeneration under the reductant gas atmosphere [1-3].
The characteristic of CuO/γ-Al2O3 catalysts was applied in fixed-, moving-, and fluidized-bed reactors [4-7]. However, the poor mechanical strength of the catalyst available at the time presented a major problem for the practical application of the department of energy (DOE) copper oxide process [8]. The catalyst used did not have sufficiently high attrition resistance, which impeded the operation of the moving-bed process and substantially increased the operational costs of the copper oxide process. Thus, the development of an alumina catalyst support with good mechanical properties is of practical importance for many industrial processes.
QI and YANG [9-10] found that the manganese- cerium catalyst yielded nearly 100% NO conversion at low temperature (120-150 ℃). TANG et al [11] also reported that the manganese oxide catalyst had excellent denitrification activity at 80 ℃.
Recently, BUELNA et al [12-13] developed a sol-gel process to prepare γ-alumina granular support and catalyst. The sol-gel-derived alumina granules have a large surface area, large pore volume, uniform pore size distribution, and excellent mechanical properties (attrition resistance and crush strength). However, the denitrification performance of the CuO-CeO2-MnOx/ γ-Al2O3 granular catalysts prepared by the sol-gel method has not been reported yet.
Therefore, in this work, the manganese oxides and cerium oxide as the promoters were added into the CuO/γ-Al2O3 catalysts. The NO-removal performance of the sol-gel-derived CuO-CeO2-MnOx/γ-Al2O3 granular catalysts and the influence of NH3 and NO oxidation in the SCR process were determined in a fixed flow reactor. To obtain more information about the behaviors of NH3 and NO in this system, temperature programmed desorption(TPD) method and transient behaviors of catalysts were investigated.
2 Experimental
2.1 Catalysts preparation
Spherical CuO-CeO2-MnOx/γ-Al2O3 granular catalysts were prepared by sol-gel method, as described in Ref.[14]. Stable boehmite sols were synthesized from hydrolysis and condensation of aluminum isopropoxide. Stable 2 mol/L boehmite sol was mixed with nitrate solutions (Cu, Ce and Mn) at a predetermined concentration according to the required loading after aging at 70 ℃ for 0.5 h.
The doped boehmite reached the gelation point and was dropped and aged in an ammonia solution (10% NH3, volume fraction). The gel particles were then dried in air at 40 ℃ for 48 h. The precursors of the active species were coated on the surface of the boehmite primary particles and converted to CuO, CeO2 and MnOx by calcinating at 550 ℃ for 6 h. In this work, two catalysts synthesized by sol-gel method are denoted as S(1) and S(2), respectively. The mass fractions of Cu, Mn and Ce for S(1) are 6%, 2% and 3%, and those for S(2) are 6%, 2% and 13%, respectively.
2.2 Experimental procedure
The SCR activity measurement was carried out in a quartz tubular reactor (0.02 m in inner diameter) in which 7 mL of catalyst was placed. When the system reached the steady state at a desired temperature, the premixed simulated gases (NO, O2, N2 and NH3) were introduced into the upper part of the reactor with a desired volume velocity. The concentrations of NO, N2O, NO2 and NH3 were measured by gas analyzers using an FT-IR method (Bomen, MB154 model GASMET FTIR Dx4000). The O2 concentration was measured by flue gas analyzer (KM900). All of the data were obtained after 30-60 min when the SCR reaction reached the steady state.
The experiments were performed under atmospheric pressure at 100-500 ℃. The typical composition (volume fraction) of the reactant gas was as follows: 7×10-4 NO, 9×10-4 NH3, 5%O2, and balanced N2. And the gas hourly space velocity (vGHSV) was 12 800 h-1. The gas inlet composition (volume fraction) for ammonia oxidation was: 9×10-4 NH3, 5%O2 and balanced N2. For the oxidation of nitric oxide the gas composition was the same but using 7×10-4 NO instead of NH3.
2.3 Desorption experiment
The TPD method was used to test desorption properties of NH3 and NO on the catalysts. The catalysts were heated from room temperature to 500 ℃ under N2 atmosphere to eliminate any adsorbed water. After cooling to room temperature under the same atmosphere, NH3 (9×10-4) and N2 were introduced to the catalyst. After 2 h, the physisorpted NH3 on the catalyst was purged with N2 flow until outlet NH3 could not be detected by FT-IR. Finally, the samples were heated from room temperature to 500 ℃ at a rate of 7 ℃/min under 1.44 L/min N2 flow, and the outlet NH3 was detected. The operation process of NO desorption was almost the same but using NO instead of NH3.
2.4 Catalyst characterization
The Brunauer-Emmett-Teller (BET) surface area, pore volume and average pore diameter of the samples were measured by nitrogen adsorption porosimetry (Micromeritics ASAP-2020). The crystalline structure of the powdered catalysts and the extent of dispersion of the active species were examined by X-ray diffraction (XRD, X′ Pert PRO).
3 Results and discussion
3.1 Effect of temperature on catalytic activity of CuO-CeO2-MnOx/γ-Al2O3
The variations of the NO conversion and the N2O outlet as a function of the temperature for the two catalysts are shown in Fig.1. For CuO-CeO2-MnOx/ γ-Al2O3 catalysts the denitrification activity is improved with temperature increasing. With higher loading of Ce, the SCR activity increases. Clearly, high catalytic activities are achieved in a lower temperature region (100-300 ℃). The NO conversion reaches nearly 100% at 300-400 ℃. The SCR activity decreases gradually when the temperature is higher than 400 ℃. Compared with the activity of S(1), that of S(2) decreases quickly. The NO conversion of S(2) decreases to 50% at 500 ℃. N2O formation during SCR reaction is detected at the
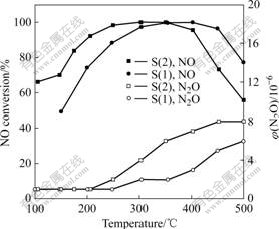
Fig.1 Effect of reaction temperature on NO conversion over CuO-CeO2-MnOx/γ-Al2O3 catalysts (Reaction conditions: φ(NO)=7×10-4; φ(NH3)=9×10-4; φ(O2)=5%; vGHSV =12 800 h-1)
temperature above 200 ℃ for S(2). For S (1), N2O is formed when the temperature is high than 250 ℃.
The SCR activity as a function of reaction temperature is correlated with the oxidation degree of NH3. The equations in the NO-NH3-O2 reaction system are as follows. There are two competitive reactions in this system. In Reaction (1), NO is reduced to N2 with NH3. In Reactions (2), (3) and (4), NH3 is oxidized to N2O, NO and N2 directly.
4NO+4NH3+O2→4N2+6H2O (1)
2NH3+2O2→N2O+3H2O (2)
4NH3+5O2→4NO+6H2O (3)
4NH3+3O2→2N2+6H2O (4)
To study the oxidation property of NH3 on the catalyst, separate ammonia oxidation experiments were carried out. The variation of NH3 conversion, and volume fractions of NO, N2O formed during reaction as a function of the temperature for the two catalysts are represented in Figs.2(a) and (b), respectively. NH3 is oxidized gradually with temperature increasing. The NH3
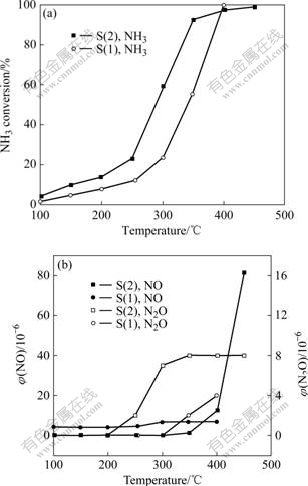
Fig.2 NH3 conversion, volume fraction of NO and N2O as function of operating temperature: (a) NH3 conversion; (b) Volume fractions of NO and N2O (Reaction conditions: φ(NH3)=9×10-4; φ(O2)=5%; vGHSV = 12 800 h-1)
conversion of S(1) reaches 55% at 350 ℃. The conversion of S(2) is up to 92% at the same temperature. Thus, these results indicate that the oxidation degree of NH3 is strengthened gradually with the increase of Ce content on catalysts.
The NH3 oxidation degree is enhanced with increasing temperature. When the temperature is higher than 400 ℃, the NH3 oxidation degrees of two catalysts maintain nearly 100%. In Fig.2(b), NO formation increases quickly at temperatures above 400 ℃ during ammonia oxidation reaction. This indicates that ammonia is over-oxidized to N2 (Reaction (1)), but also a small quantity of ammonia is over-oxidized to NO according to Reaction (3). N2O is also detected in the outlet (Reaction (2)), and the amount is smaller than that during the SCR reaction.
RAMIS et al [15-16] reported that the activity was not only dependent upon the oxidation property of NH3 but also in relation to the adsorption capacity of NH3. The oxidation property is improved with the addition of Ce. The higher the loading is, the more the NH3 is oxidized. It can also be seen that the SCR activity is improved at low temperature. The activity of S(2) and S(1) decreases gradually at temperatures above 400 ℃. The over-oxidation property of NH3 reduces the SCR activity in high temperature range (400-500 ℃). S(1) still maintains activity nearly 96% at 450 ℃. This may be related to the adsorption capacity of NH3.
3.2 Desorption of NH3 and NO on CuO-CeO2-MnOx/ γ-Al2O3
The SCR system has the gas-solid multiphase reactions and the adsorption of NH3 and NO may play an important role in the reaction. The TPD method was used to test desorption properties of NH3 and NO on the catalysts.
3.2.1 Desorption properties of NH3
The NH3 desorption as a function of operating temperature is shown in Fig.3. There is an obvious NH3
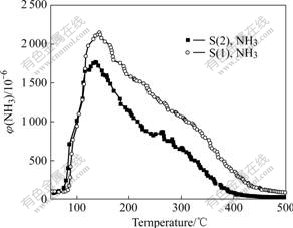
Fig.3 NH3 desorption as function of operating temperature
desorption peak at 100-150 ℃ for the two samples. Most of NH3 is desorbed from the catalysts when the temperature increases to 450 ℃. It can be seen that NH3 can be adsorbed on the catalysts and then desorbed when the temperature increases.
High temperature not only decreases the NH3 adsorption capacity but also accelerates and deepens the over-oxidation of NH3. From Fig.2, most of NH3 is oxidized to N2, NO and N2O at temperatures above 400 ℃. The reduced adsorption capacity and the over- oxidation of NH3 are the main causes of bad SCR activity for the CuO-CeO2-MnOx/γ-Al2O3 catalyst at high temperature (500 ℃).
The two samples have high NH3 oxidation degree at 400 ℃. The SCR activity depends on the adsorption capacity of NH3 in high temperature range. As shown in Fig.3, the desorption amount of NH3 with S(1) is larger than that with S(2). This difference may be related to the catalyst structure and the amount of Ce. The high adsorption amount of NH3 ensures that S(1) has higher activity than S(2) at 400-500 ℃.
3.2.2 Desorption properties of NO
The variations of the NO desorption, and volume fraction of NO2 formed during desorption process as a function of the temperature are illustrated in Fig.4. There
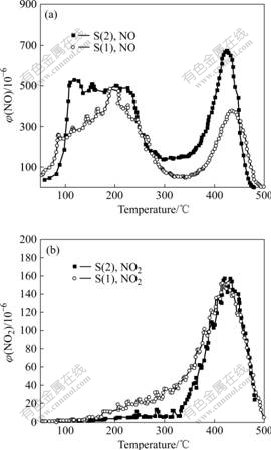
Fig.4 NO desorption and volume fraction of NO2 as function of operating temperature: (a) NO desorption; (b) Volume fraction of NO2
are two peaks of NO desorption on the two samples when the temperature increases. The desorption peak at low temperature is rather wide, which may be the superposition of two or more peaks. These desorption peaks indicate that there are multi-activity sides with adsorbed NOx on the CuO-CeO2-MnOx/γ-Al2O3 catalysts [1]. Most of the adsorbed NO on the catalysts is desorbed when the temperature is high than 475 ℃.
NO2 is detected during the NO desorption process. The formation rate of NO2 increases with the increase of temperature. The amount of NO2 reaches the maximum value at 425 ℃, and then it decreases quickly. The formation of NO2 is difficult at high temperature. The formation of NO2 can be explained by NO reacting with oxygen from the lattice.
The desorption processes of two samples are almost the same, the desorption amount of NO with S(2) is larger than that with S(1). But the desorption capacity of NH3 with S(2) is smaller than that with S(1) in Fig.3. Therefore, the adsorption mechanisms of NH3 and NO on the two samples are different, and NO is more weakly adsorbed than NH3. The active sites of NH3 and NO adsorption and the effect of the active site need further study.
3.3 Transient response of NH3 and NO on CuO-CeO2-MnOx/γ-Al2O3
3.3.1 Transient response of NH3
In order to find the role of adsorption with NH3 and NO in the SCR process the transient process experiments were studied. The transient response process of NH3 as a function of time is shown in Fig.5. At first, the SCR reaction remains relatively stable at 150 ℃ for S(1) samples, and then NH3 is turned off after 10 min. NO concentration at the outlet increases slowly and reaches a high value after about 90 min reaction. It can be inferred that NH3 is consumed completely.

Fig.5 Transient response on CuO-CeO2-MnOx/γ-Al2O3 catalysts upon switching off and on NH3 (Reaction conditions: temperature 150 ℃, φ0(NO)=7×10-4; φ0(NH3)=9×10-4; φ0(O2)=5%; vGHSV=12 800 h-1)
Switching NH3 back on results in an immediate decrease in NO concentration, and the original conversion is restored quickly. But the concentration of NH3 is kept in low value at this time. The NH3 concentration increases gradually and recovers to the primary level for a long time. This indicates that NH3 is not only used for reacting with NO, but also adsorbed on the catalysts.
3.3.2 Transient response of NO
The transient response process of NO as a function of time is shown in Fig.6. At first, the SCR reaction remains relatively stable at 150 ℃, then NO is turned off after 8 min, and NH3 concentration at the outlet increases. The concentration of NH3 reaches a high value after a period of time. It can be inferred that NH3 reaches the equilibrium on the catalysts.

Fig.6 Transient response on CuO-CeO2-MnOx/γ-Al2O3 catalysts upon switching off and on NO (Reaction conditions: temperature 150 ℃, φ0(NO)=7×10-4; φ0(NH3)=9×10-4; φ0(O2)=5%; vGHSV=12 800 h-1)
Switching NO back on results in an immediate increase in NO concentration, and the original NO conversion is restored quickly. NH3 concentration declines slowly and recoveres to primary level after a long period of time. This indicates that NO reacts with the adsorbed NH3 on the catalysts at first, and then NH3 is adsorbed on the catalysts gradually.
As shown in Fig.5, the equilibrium time of NO reaction is about 90 min when NH3 is turned off; NO concentration increases slowly after NH3 is turned off, and then followed by a fast growth. Upon switching on NH3, NO concentration rapidly decreases, but the NH3 concentration is still kept in low value. From Fig.6, it can be seen that NH3 reaction equilibrium time is about 40 min when NO is turned off, NH3 concentration increases quickly after NO is turned off. These results reveal that the amount of the adsorbed NO is smaller than that of NH3 on the catalyst. Switching NO back on results in an immediate increase of NO concentration, but NH3 concentration decreases slowly. The above results indicate that NO reacts with the adsorbed NH3 on the catalysts. NH3 is more strongly bound with the adsorption site of the catalysts. The NO adsorption has little influence on SCR activity in SCR process.
TANG et al [11] reported that the SCR reaction on MnOx catalysts proceeded in accordance with the Eley-Rideal mechanism and the Langmuir-Hinshelwood hypothesis at the same time. They only investigated the NO desorption process on MnOx catalysts, but they did not test the SCR reaction process in actual reaction gas including NH3 and NO. QI and YANG [9-10] found that the SCR reaction on the manganese-cerium oxide catalysts was zero order with respect to NH3 concentration and the first order with respect to NO. As a conclusion of this study, it is proposed that the SCR reaction can take place between the adsorbed NH3 and gaseous or weakly adsorbed NO species in actual SCR process. The adsorbed NO has little influence on SCR activity.
3.4 Reaction kinetics and characterization of CuO-CeO2-MnOx/γ-Al2O3
The experimental results show that the reaction proceeds between NO in the gas phase or weakly adsorbed species and the adsorbed NH3 on CuO/γ-Al2O3 catalysts. Following the Eley-Rideal mechanism, the reaction rate is proportional to the gas-phase concentration of NO and the fraction of the surface covered by the adsorbed NH3. The reaction rate is reduced to the first order for NO and zero order for NH3 when NH3 is strongly adsorbed on the catalysts [17]. Therefore, the intrinsic reduction rate of NO can be expressed as
r(NO)=Krc(NO) (5)
where r(NO) is the intrinsic reaction of NO, mol/(g?s); c(NO) is the concentration of NO at the entrance of the catalyst bed, mol/L; Kr is the intrinsic reaction rate constant, mol/(g?s). Kr can be calculated with an assumption of the gas phase flows in plug flow as
 (6)
(6)
and
Kr=K0exp[-Ea/(RT)] (7)
where V0 is total volumetric flow rate of the gas phase at the reactor entrance, m is the mass, x(NO) is the conversion of NO, K0 is the frequency factor of the intrinsic rate constant, T is the reaction temperature, and Ea is the activation energy of the reaction.
From Arrhenius plots (Kr vs reaction temperature) for S(1) and S(2) (Fig.7), Ea and K0 of the catalysts were derived. The intrinsic reaction rate constants of the two samples can be expressed as
Kr=1.75×106exp[-23.8×103/(RT)] for S(1) (8)
Kr=1.05×107exp[-24.5×103/(RT)] for S(2) (9)

Fig.7 Arrhenius plots of intrinsic reaction rate constants for CuO-CeO2-MnOx/γ-Al2O3 catalysts
The activation energies of S(1) and S(2) in the present study are lower than those in Refs.[2, 18]. This may be related with the pore property of the catalysts. As shown in Table 1, the CuO-CeO2-MnOx/γ-Al2O3 catalysts made by sol-gel method have larger surface area and pore volume than those made by wet-impregnation method [2]. The excellent pore structure favors pore diffusion of the reactant gas on the catalyst surface [19].
Table 1 Pore structural parameters of fresh and used catalysts
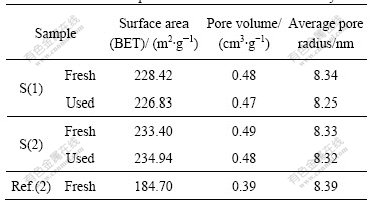
For S(2) catalysts, the surface area and pore volume increase with the loading of CeO2. ZHU et al [20] pointed out that CuO species had a stronger interaction with CeO2 than with γ-Al2O3, and the copper oxide species were preferentially dispersed on the surface of CeO2 if the loading of CuO species was lower than its dispersion capacity on the CeO2 support. Therefore, it can be concluded that CeO2 enhances the dispersion of CuO on the supports. The surface area and pore volume decrease slightly after 100 h SCR test, which indicates that the catalysts have a strong capability of anti-ablation. The samples can be recycled. This conclusion is consistent with the XRD study below.
The XRD patterns of S(1) and S(2) catalysts are shown in Fig.8. It can be seen that diffraction peaks are 37?, 47? and 67?, which belong to obviously γ-Al2O3 phase. No visible phase of Ce, Cu can be observed. Reunion of CuO does not take place either after 100 h SCR test; thus, the activity of the catalysis remains constant. It is inferred that the catalysts have good ability to be recyclable.
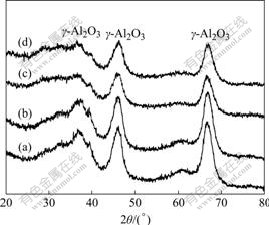
Fig.8 XRD patterns of different CuO-CeO2-MnOx/γ-Al2O3 catalysts: (a) S(1), fresh; (b) S(1), used; (c) S(2), fresh; (d) S(2), used
4 Conclusions
(1) The CuO-CeO2-MnOx/γ-Al2O3 catalysts made by the sol-gel method have higher activity and wider operating temperature range. The NO conversion of the catalysts maintains nearly 100% at 300-400 ℃. The higher active component Ce improves the SCR activity at low temperature.
(2) The oxidation property is improved when the loading of Ce increases. The SCR activity is improved at low temperature. The deep oxidation and desorption of NH3 at high temperature lead to a decrease in the NO removal activity.
(3) There are obvious adsorption phenomena of NH3 and NO on the catalysts. The SCR reaction mechanism can be proposed for the catalyst based on NH3 adsorption on the surface and the reaction of the adsorbed species with gaseous or weakly adsorbed NO species. The adsorbed NO has little influence on SCR activity in the actual SCR process.
(4) The changes of pore structure are neglectable after 100 h SCR test. There is no sintering phenomenon on the catalysts. Thus, the activity of the catalysts remains constant and catalysts can be recycled.
References
[1] CENTI G, PASSARINI N, PERATHONER S, RIVA A, STELLA G. Combined DeSOx/DeNOx reactions on a copper on alumina sorbent catalyst.3.DeNOx behavior as a function of the surface coverage with sulfate species [J]. Industrial and Engineering Chemistry Research, 1992, 31(8): 1963-1970.
[2] JEONG S M, JUNG S H, YOO K S, KIM S D. Selective catalytic reduction of NO by NH3 over a bulk sulfated CuO/γ-Al2O3 catalyst [J]. Industrial and Engineering Chemistry Research, 1999, 38(6): 2210-2215.
[3] XIANG Jun, ZHAO Qing-sen, HU Song, SUN Lu-shi, SU Sheng, FU Peng, ZHANG An-chao, QIU Jian-rong, CHEN Han-ping, XU Ming-hou. Experimental research and characteristics analysis of alumina-supported copper oxide sorbent for flue gas desulfurization [J]. Asia-Pacific Journal of Chemical Engineering, 2007, 2(3): 182-189.
[4] LI Wei, CHENG Hua. Synthesis and characterization of Cu-Cr-O nanocomposites [J]. Journal of Central South University of Technology, 2007, 14(3): 291-295.
[5] YEH J T, DEMSKI R J, STRAKEY J P, JOUBERTJ I. Combined SO2/NOx removal from flue gas: Detail discussion of a new regenerative fluidized-bed process [J]. Environmental Progress, 1985, 4(4): 223-228.
[6] XIE Guo-yong, LIU Zhen-yu, ZHU Zhen-ping, LIU Qing-ya, GE Jun, HUANG Zhang-geng. Simultaneous removal of SO2 and NOx from flue gas using a CuO/Al2O3 catalyst sorbent. I. Deactivation of SCR activity by SO2 at low temperatures [J]. Journal of Catalysis, 2004, 224(1): 36-41.
[7] XIE Guo-yong, LIU Zhen-yu, ZHU Zhen-ping, LIU Qing-ya, GE Jun, HUANG Zhang-geng. Simultaneous removal of SO2 and NOx from flue gas using a CuO/Al2O3 catalyst sorbent. II. Promotion of SCR activity by SO2 at high temperatures [J]. Journal of Catalysis, 2004, 224(1): 42-49.
[8] YEH J T, DRUMMOND C J, JOUBERT J I. Process simulation of the fluidized-bed copper-oxide process sulfation reaction [J]. Environmental Progress, 1987, 6(2): 44-50.
[9] QI G S, YANG R T. Low-temperature selective catalytic reduction of NO with NH3 over iron and manganese oxides supported on titania [J]. Applied Catalysis B: Environmental, 2003, 44(3): 217-225.
[10] QI G S, YANG R T. Performance and kinetics study for low- temperature SCR of NO with NH3 over MnOx-CeO2 catalyst [J]. Journal of Catalysis, 2003, 217(2): 434-441.
[11] TANG Xiao-long, HAO Ji-ming, XU Wen-guo, LI Jun-hua. Novel MnOx catalyst for low-temperature selective catalytic reduction of NOx with NH3 [J]. Chinese Journal of Catalysis, 2006, 27(10): 843-848. (inChinese)
[12] BUELNA G, LIN Y S, LIU L X, LITSTER J D. Structural and mechanical properties of nanostructured granular alumina catalysts [J]. Industrial and Engineering Chemistry Research, 2003, 42(3): 442-447.
[13] BUELNA G, LIN Y S. Characteristics and desulfurization- regeneration properties of sol-gel-derived copper oxide on alumina sorbents [J]. Separation and Purification Technology, 2004, 39(3): 167-179.
[14] ZHAO Qing-sen, SUN Lu-shi, XIANG Jun, HU Song. Preparation of CuO/γ-Al2O3 catalytic sorbent by sol-gel method [J]. Industrial Catalysis, 2008, 16(4): 24-29. (inChinese)
[15] RAMIS G, YI L, BUSCA G, TURCO M, KOTUR E, WILLEY R J. Adsorption, activation and oxidation of ammonia over SCR catalysts [J]. Journal of Catalysis, 1995, 157(2): 523-535.
[16] RAMIS G, LARRUBIA M A. An FT-IR study of the adsorption and oxidation of N-containing compounds over Fe2O3/Al2O3 catalyst [J]. Journal of Molecular Catalysis A: Chemical, 2004, 215(1/2): 161-167.
[17] KIEL J H A, EDELAAR A C S, PRINS W, SWAAIJ W P M. Performance of silica-supported copper oxide sorbents for SOx/NOx removal from flue gas. 2. Selective catalytic reduction of nitric oxide by ammonia [J]. Applied Catalysis B: Environmental, 1992, 1(1): 41-60.
[18] SIRDESHPANDE A R, LIGHTY J S. Kinetics of the selective catalytic reduction of NO with NH3 over CuO/γ-Al2O3 [J]. Industrial and Engineering Chemistry Research, 2000, 39(6): 1781-1787.
[19] JEONG S M, KIM S D. Enhancement of the SO2 sorption capacity of CuO/γ-Al2O3 sorbent by an alkali-salt promoter [J]. Industrial and Engineering Chemistry Research, 1997, 36(12): 5425-5431.
[20] ZHU Hai-yang, SHEN Ming-min, LIU Tian-dong, WEI Shu-ting, HU Yu-hai, DONG Lin, CHEN Yi. Surface interaction between copper oxide and CeO2-γ-Al2O3 mixed oxide support [J]. Chinese Journal of Catalysis, 2002, 23(4): 325-328. (inChinese)
(Edited by CHEN Wei-ping)
Foundation item: Projects (50776037, 50721005) supported by the National Natural Science Foundation of China
Received date: 2008-10-28; Accepted date: 2009-03-10
Corresponding author: XIANG Jun, Professor; Tel: +86-27-87542417-8509; E-mail: xiangjun@mail.hust.edu.cn