
Effect of Ni on glass-forming ability of Cu-Ti-based amorphous alloys
LIN Tao(林 涛)1, JIANG Jiang(姜 江)1, BIAN Xiu-fang(边秀房)1, DONG Ying(董 瑛)2
1. Key Laboratory of Structure and Heredity of Materials, Ministry of Education, Shandong University,
Ji’nan 250061, China;
2. Quality and Technology Supervision Bureau, Weifang 261031, China
Received 13 August 2005; accepted 14 December 2005
Abstract: Ribbons of amorphous Cu-Ti-Ni alloys were prepared by the melt spinning method. The amorphous structure of these ribbons was confirmed by X-ray diffractametry and transmission electron microscopy. The effect of Ni on the glass-forming ability of Cu-Ti-based alloys was studied by differential scanning calorimetry(DSC). It is found that the supercooled liquid region, ΔTx value shows the maximum value of 61℃ at x=10 in the Cu50Ti50-xNix (x=0, 5, 10, 15 mole fraction, %) system. And the reduced glass transition temperature, Trg, is smaller than 0.45. The glass forming ability(GFA) of Cu-Ti alloy is not effectively promoted by Ni addition.
Key words: Cu-Ti-based alloys; glass-forming ability; melt-spinning method; deep eutectics
1 Introduction
Recently, a number of bulk metallic glassy alloys have been reported, such as La-Al-Ni[1], Cu-Ti-Zr-Ni [2, 3], Cu-Ti-Zr-Ni-Be[4, 5], Cu-Ti-Zr-Ni-Si[6-8] and Cu-Ti-Zr-Ni-Sn[9]. Because of the exceptional good glass-forming ability, these multicomponent alloy systems attracted much attention. It is noted that all these alloy systems contain the element Ni, which plays an important role in obtaining these bulk amorphous alloys.
There have been many studies on binary amorphous Cu-Ti [10] and Ti-Ni [11] alloys, but the effect of Ni on GFA of Cu-Ti-based alloys is not studied further. In order to obtain some useful information in alloy design for achieving new bulk glassy alloys, we have prepared ternary Cu50Ti50-xNix glassy ribbons by the melt spinning method and investigated the effect of Ni on the glass- forming ability of the Cu-Ti-based alloys.
2 Experimental
The alloy ingots of nominal compositions Cu50Ti50-xNix were prepared by arc melting mixtures of Cu (99.9%), Ti (99.9%), Ni (99.9%) in a Ti-gettered argon high-purity atmosphere. The ribbons sample of approximately 40 μm in thickness and 2 mm in width were prepared by remelting the ingots in quartz tubes, and ejecting under pure argon through a nozzle onto a copper wheel rotating with a surface velocity of 44 m/s. The amorphous structure of the ribbons was confirmed by X-ray diffractometry(XRD, D/max-rB), using a monochromatic Cu Kα radiation of 10?-60? and by transmission electron microscopy(TEM, HITACHI-800). Thermal analysis of the ribbon samples was carried out to determine the glass transition temperature, Tg, the crystallization temperature, Tx, solidus temperature, Tm, and liquidus temperature, Tl , by differential scanning calorimetry (DSC, Netzsch404).
3 Results and discussion
Fig.1 shows the X-ray diffraction patterns of the melt-spun ribbons of the Cu50Ti50-xNix alloys. Each pattern shows a broad diffraction peak at the position of about 2θ=42? and no diffraction peaks from crystalline phases are detected. The Cu50Ti50 and Cu50Ti45Ni5 alloys consist of a single glassy phase. But there are some nanocrystals in Cu50Ti40Ni10 and Cu50Ti35Ni15, which is reflected by the small peaks located at about 2θ=59? on the XRD patterns.
As show in Fig.2 the glassy structure of the meltspun Cu50Ti50 ribbon was further confirmed by TEM analysis. The bright field TEM image shows no crystalline phase, and the selected area diffraction pattern (SADP) shows a diffuse halo ring, which is a characteristics of amorphous phase.

Fig.1 XRD patterns of melt-spun Cu50Ti50-xNix ribbons

Fig.2 Bright field TEM image and selected area diffraction pattern of Cu50Ti50 ribbon
Fig.3 shows DSC traces obtained from melt-spun ribbons during continuous heating with a heating rate of 20 K/min. The amplified DSC trace of Cu50Ti45Ni5 ribbon showing glass transition is also shown as an inset. With increasing x from 0 to 15, there is nearly no change in glass transition temperature, Tg. Crystallization temperature is assumed to be the onset temperature of the first exothermic peak. The crystallization temperature, Tx, increases from 409 ℃ to 445 ℃ with x value increasing from 0 to 10, and then decreases to 415 ℃ with x value further increasing to 15. Therefore the supercooled liquid region, ΔTx=Tx-Tg, exhibiting the same trend as Tx, increases from 24 ℃ to 61 ℃, and then decreases to 29 ℃.
In Fig.4 the DSC traces of melt-spun Cu50Ti50 and Cu50Ti45Ni5 alloys are shown with a heating rate of 20 K/min from 700 to 1 100 ℃. The solidus temperature (Tm) and liquidus temperature(Tl) are marked with arrows. With increasing x from 0 to 5, Tm decreased from 898 to 896 ℃ and Tl decreased form 945 to 933 ℃. Both Tm and Tl increased with further increase of x.
Fig.5 shows the variations of Tg, Tx and Tm of the melt-spun Cu50Ti50-xNix (x=0, 5, 10, 15) alloys. It can be seen that the maximum value for ΔTx is at x=10.
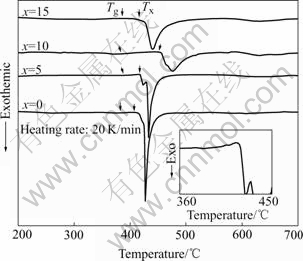
Fig.3 DSC traces of Cu50Ti50-xNix (x=0, 5, 10, 15) amorphous alloys and amplified DSC trace of Cu50Ti45Ni5 ribbon (inset)

Fig.4 DSC traces of melt-spun Cu50Ti50 and Cu50Ti45Ni5 amorphous alloys
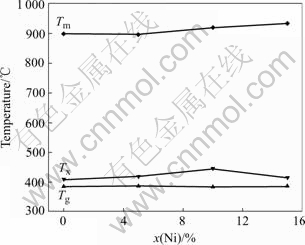
Fig.5 Thermal analysis of melt-spun Cu50Ti50-xNix (x=0, 5, 10, 15) alloys showing variations of Tg, Tx and Tm as function of x
Table 1 lists the supercooled liquid region and the reduced glass transition temperature of the four Cu-Ti-Ni melt-spun amorphous alloys. It can be seen that the reduced glass transition temperature, Trg=Tg/Tm, changes a little and is not larger than 0.45 with increasing value x.
Table 1 Thermal parameter of Cu50Ti50-xNix (x=0, 5, 10, 15) amorphous alloys with heating rate of 20 K/min

Bulk glassy alloys may be obtained for the alloys with Trg above 0.60 by copper mold casting method[12]. The addition of Ni to Cu-Ti alloy can not promote its glass forming ability essentially.
There are three empirical rules suggested to get a high glass-forming ability for metallic alloys[13] as follows. 1) Multicomponent systems with more than three components; 2) A significant difference in atomic size ratios above 12% among the three main constituent elements; 3) Large negative heats of mixing in the liquid. The atomic radius is 0.128 nm and 0.125 nm for Cu and Ni respectively, leading a difference of only 2.3% in atomic size ratio between Cu and Ni atoms. On the other hand, the enthalpy of mixing between Cu and Ni is 4 kJ/mol[14]. These two factors are not favourable to forming an amorphous structure in Cu-Ti-Ni alloys. In order to obtain a glass transition during solidification, the nucleation and growth of a crystalline phase should be prevented. If the crystal nucleus has a different composition from that of the undercooled liquid, the srystallization can occur only when the composition of a local liquid region and the size of a critical crystalline nucleus satisfy the composition requirements of the crystalline phase[2]. The small atomic size ratio of Cu and Ni determines that the addition of Ni to Cu-Ti alloy does not play an important role in preventing rearrange- ment of different species of atoms and nucleation.
A number of previous observations demonstrate that high GFA often occurs around deep eutectics. A deep eutectic means that the liquid state is energetically favored over ordered solid state in a large temperature range above the eutectic temperature. Compared with the case of noneutectic, the energy difference between these two states (i.e. the driving force for crystal nucleation and growth) is relatively small below the eutectic temperature. So it is easier to quench the liquid to an amorphous state before a detectable fraction of ordered crystals can be formed[15]. With x value increasing from 0 to 5, Tl is only lowered by 12 ℃, and the liquidus temperature of Cu50Ti40Ni10 and Cu50Ti35Ni15 is higher than that of Cu50Ti50. The deviation from deep eutectics leading by increasing of Ni content is another reason that results in the poor GFA of Cu-Ti-Ni alloys.
4 Conclusions
With increasing Ni content(x) from 0 to 15, the supercooled liquid region ΔTx reaches the maximum value of 61 ℃ at x=10, and the reduce glass transition temperature, Trg reaches the maximum value of 0.432 at x=5. The small value of Trg determined that the GFA of Cu-Ti-Ni alloys was not effectively promoted by Ni addition.
References
[1] Inoue A, Zhang T, Masumoto T. Al-La-Ni Amorphous alloys with a wide supercooled liquid region [J]. Materials Transactions JIM, 1989, 30: 965-972.
[2] Lin X H , Johnson W L. Formation of Ti-Zr-Cu-Ni bulk metallic glasses [J]. Journal of Applied Physics, 1995, 78(11): 6514-6519.
[3] FANG J D, GAO Y Q, LI S Z. Crystallization behavior of Ti61.67Zr17.15Ni14.80Cu6.38 glass-forming alloy [J]. Trans Nonferrous Met Soc China, 2004, 14(1): 156-160.
[4] Peker A, Johnson W L. A highly processable metallic glass: Zr41.2Ti13.8Cu12.5Ni10.0Be22.5 [J]. Applied Physics Letters, 1993, 63(17): 2342-2344.
[5] Waniuk T A, Schroers J, Johnson W L. Critical cooling rate and thermal stability of Zr-Ti-Cu-Ni-Be alloys [J]. Applied Physics Letters, 2001, 78(9): 1213-1215.
[6] Inoue A, Zhang T. Preparation of Ti-Cu-Ni-Si-B amorphous alloys with a large supercooled liquid region [J]. Materials Transactions JIM, 1999, 40: 301-306.
[7] Choi-Yim H, Busch R, Johnson W L. The effect of silicon on the glass forming ability of the Cu47Ti34Zr11Ni8 bulk metallic glass forming alloy during processing of composites [J]. Journal of Applied Physics, 1998, 83(12): 7993-7997.
[8] Park E S, Lim H K, Kim W T, et al. The effect of Sn on the glass-forming ability of Cu-Ti-Zr-Ni-Si metallic glass alloys [J]. Journal of Non-Crystalline Solids, 2002, 298: 15-22.
[9] Li C F, Saida J J, Kiminami M, et al. Dynamic crystallization process in a supercooled liquid region of Cu40Ti30Ni15Zr10Sn5 amorphous alloy [J]. Journal of Non-Crystalline Solids, 2000, 261: 108-114.
[10] Takemoto R, Nagata M, Mizubayashi H. Effect of passing electric current on the elastic property of amorphous Cu50Zr50 and Cu50Ti50 [J]. Acta Materialia, 1996, 44(7): 2787-2795.
[11] Chen J Z, Wu S K. Crystallization temperature and activation energy of rf-sputtered near-equiatomic TiNi and Ti50Ni40Cu10 thin films [J]. Journal of Non-Crystalline Solids, 2001, 288: 159-165.
[12] Inoue A, Zhang W, Zhang T, et al. Cu-based bulk glassy alloys with high tensile strength of over 2000 MPa [J]. Journal of Non-Crystalline Solids, 2002, 304: 200-209.
[13] Inoue A, Takeuchi A, Zhang T. Ferromagnetic bulk amorphous alloys [J]. Metall Mater Trans A, 1998, 29: 1779-1793.
[14] Takeuchi A, Inoue A. Calculations of mixing enthalpy and mismatch entropy for ternary amorphous alloys [J]. Materials Transactions JIM, 2000, 41: 1372-1387.
[15] Xu D, Duan G, Johnson W L. Unusual glass-forming ability of bulk amorphous alloys based on ordinary metal copper [J]. Physical Review B, 2004, 92(24): 245504.
Foundation item: Project(50471052) supported by the National Natural Science Foundation of China; Project(Z2004F02) supported by the Natural Science Foundation of Shandong Province, China; Project(2005422024) supported by the Specialized Research Fund for the Doctoral Program of Higher Education
Corresponding author: BIAN Xiu-fang; Tel:+86-531-88395015; E-mail: xfbian@sdu.edu.cn
(Edited by LONG Huai-zhong)