
Effects of electrolytes on properties of anodic coatings formed on
AZ91HP magnesium alloy
ZHANG Shu-fang(张淑芳)1, WANG Fang-yuan(王方圆)2, ZHANG Rong-fa(张荣发)1, LI Ming-sheng(李明升)1,
ZHAO Fang(赵 芳)3, ZHOU Chong(周 冲)3, HU Chang-yuan(胡长员)1
1. Jiangxi Key Laboratory of Surface Engineering, Jiangxi Science and Technology Normal University,Nanchang 330013, China;
2. Jinhua College of Profession and Technology, Jinhua 321007, China;
3. College of Material Science and Engineering, Jiangxi Science and Technology Normal University, Nanchang 330013, China
Received 15 July 2007; accepted 10 September 2007
Abstract: In a basic solution containing 10 g/L NaOH, the thickness, morphology and corrosion resistance of anodic coatings formed in solutions without and with additions of Na2SiO3, Na2CO3, Na2B4O7 and Na2SnO3 were separately determined and evaluated. The results show that the electrolytes used above take part in the coating formation and the obtained anodic coatings contain elements from the electrolytes. These electrolytes, especially Na2SiO3 and Na2B4O7, contribute to the film thickness. The coating uniformity obtained by addition of Na2SiO3 is the best, while Na2CO3, Na2B4O7 and Na2SnO3 decrease the number of pores per area on the coatings surface though they worsen the uniformity of the anodic coatings. In addition, the anodic coatings formed in solutions with addition of Na2CO3 or Na2SnO3 exhibit loose and net-like structure. Na2SiO3 and Na2B4O7 can significantly improve the corrosion resistance of the anodic coatings while Na2CO3 and Na2SnO3 have minor effects on improving the coating corrosion resistance.
Key words: magnesium alloy; anodic coatings; electrolyte; effect
1 Introduction
Because of the lightest density among the practical metallic materials and high specific strength, magnesium and its alloy offer various possibilities as regards applications in the automotive, electronic and aeronautical industries[1-2]. However, their poor corrosion and wear resistance restrict their practical applications. Surface treatment methods, such as electrochemical plating[3], conversion coatings[4], anodizing[5] and organic coatings[6], are applied to improve their properties in these areas prior to service. Micro arc oxidation is an effective method to improve the corrosion resistance of magnesium alloy and the properties of anodic coatings are mainly decided by the compositions and concentrations of the electrolytes[5]. According to the pH value of bath solutions, the anodizing process can be sorted into the acid and basic baths. The most favorable acid and basic solutions are separately DOW 17 and HAE. However, they contain harmful electrolytes such as chromate, phosphate and/or fluoride[7-8]. At present, developing environmentally friendly anodizing processes for magnesium alloy has received much interest and some processes have been successfully developed in the basic baths[9-10].
In this work, in a base solution containing 10 g/L NaOH, the effects of oxysalt such as Na2SiO3, Na2CO3, Na2B4O7 and Na2SnO3 on the thickness, morphologies and corrosion resistance of anodic coatings formed on AZ91HP magnesium alloy were separately determined and evaluated by an eddy current instrument, scanning electron microscope (SEM) and salt spray test.
2 Experimental
An ingot of AZ91HP magnesium alloy was employed and its chemical compositions were as follows (mass fraction, %): Al 8.93, Zn 0.47, Mn 0.22, Si 0.03,Cu 0.002, Ni 0.001, Fe 0.001, Mg balance.
The samples for anodization were masked with sealant leaving an area of 5 cm×6 cm exposed. A hole with 3 mm in diameter was drilled to connect with a copper rod, which was further connected with the anode. These samples were placed in a desiccator after they were polished successively on SiC paper up to 1000 grit finish, degreased by acetone, washed with distilled water and dried in a cool air stream. The samples were anodized by homemade micro-arc oxidation equipment, which consists of a MAOI-050 power supply, a stainless steel barrel and a stirring and cooling system that automatically controls the electrolyte temperature at (20±0.5) ℃. The power supply can provide unipolar pulse or bipolar pulses and electric parameters, such as current density, frequency and duty cycle, can be adjusted independently to obtain excellent anodic coatings. In this experiment, unipolar current was used under constant current mode. The same electric parameters, namely, current density 40 mA/cm2, frequency 2 000 Hz, duty cycle 20% and anodizing time 3 min, were used for different processes. The base solution was 10g/L NaOH and Na2SiO3, Na2CO3, Na2B4O7 and Na2SnO3 were separately added in order to investigate their effects on the coating properties. The compositions and concentrations of different anodizing processes are list in Table 1.
Table 1 Compositions and concentrations of anodizing electrolytes

The morphologies of anodized samples obtained were observed after they were rinsed with distilled water and then dried in a cool air stream before coating with gold. The coating thickness was measured by a 6000-FN1 eddy current instrument (Made in Hong Kong), which was calibrated with the same base metal. The measurements were carried out 10 times and the readings were averaged to yield the coating thickness value. The corrosion resistance of the anodized samples was evaluated after 120 h salt spray test in 5% (mass fraction) NaCl solution.
3 Results and discussion
3.1 Voltage―time responses of five processes
Fig.1 shows the curves of voltage vs time of the five anodizing processes.
According to Fig.1, in the original anodizing stage, the working voltage increases fast in all processes. With prolonging anodizing, voltage increases with a small slope especially for process 1 and 3. After anodizing for 3 min, the final voltage for processes 1, 2, 3, 4, 5 are separately 155, 338, 196, 324 and 225 V. Therefore, Na2SiO3, Na2CO3, Na2B4O7 and Na2SnO3 additions to the base solution resulted in higher final voltage, especially for Na2SiO3 and Na2B4O7. The final voltage is an important electric parameter and it is connected with the coating properties such as porous size, thickness and corrosion resistance of the anodic coatings[11-12]. In the solutions with additions of Na2SiO3 and Na2B4O7, the sample can be anodized to higher final voltage, which is generally helpful to achieve excellent anodic coatings.
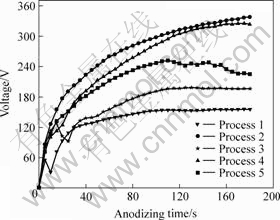
Fig.1 Curves voltage vs time by different anodizing processes
3.2 Coating thickness
The thickness of anodic coatings formed by different anodizing processes is shown in Fig.2.

Fig.2 Coating thickness formed by different anodizing processes
The anodization in sodium hydroxide (Process 1), with no other additional electrolyte, produces a coating with thickness of 2 mm. After addition of Na2SiO3, Na2CO3, Na2B4O7 and Na2SnO3 to the base solution, the anodic coatings are thickened more or less. It is obvious that only some electrolytes, such as Na2SiO3 and Na2B4O7, contribute to the layers with significant thickness, which is in agreement with the results obtained by BARTON et al[13]. Other electrolytes, such as Na2CO3 and Na2SnO3, can not thicken the anodic coatings significantly.
3.3 Appearance and surface morphologies of anodized samples
The appearance of anodized samples by different processes is first observed with bare eyes after they are rinsed with water and dried in a cool air stream. The result of a visual examination of anodized samples is listed in Table 2. It is obvious that the certain electrolytes, such as Na2SiO3 and Na2SnO3, contribute to the film color.
The surface morphologies of the anodic coatings obtained by different processes are shown in Fig.3. No coating forms on the magnesium surface by Process 1 (Fig.3(a)). Scratches on the sample surface can be clearly seen. The result is contrary with the thickness of 2 μm by
Table 2 Visual characteristics of anodized samples
eddy current method, which may result from the precision errors. It is worthy to note that on the sample surface anodized by Process 1, some areas are very rough and many small pits appear probably resulting from spark breakdown[14]. Na2SiO3 addition results in porous and uniform anodic coatings, generally 0.6-3.0 μm in diameter and 1.8-3.2 μm from each other (Fig.3(b)). However, after additions of Na2CO3 and Na2SnO3, especially for Na2SnO3, the coatings become net-like and loose (Fig.3(c) and 3(e)). The coating achieved with Na2B4O7 addition is compact and the distance between the neighbor pores is very large, about 2-6 μm (Fig.3(d)). Compared with Na2SiO3,Na2CO3, Na2B4O7 and Na2SnO3 decrease the number of pores per area on the coatings surface though they worsen the uniformity of anodic coatings.
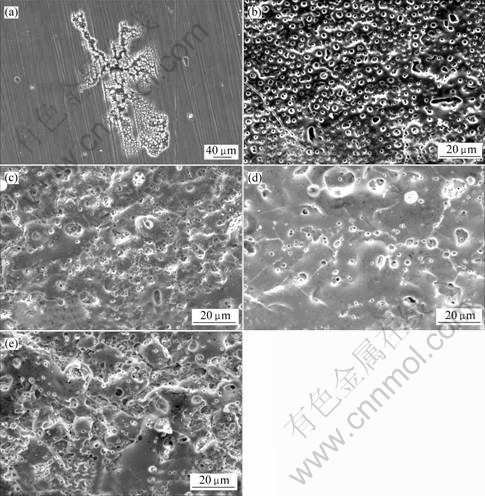 Fig.3 Surface morphologies of AZ91HP anodized by different processes: (a) Process 1; (b) Process 2; (c) Process 3; (d) Process 4; (e) Process 5
Fig.3 Surface morphologies of AZ91HP anodized by different processes: (a) Process 1; (b) Process 2; (c) Process 3; (d) Process 4; (e) Process 5
EDX analysis show that the coating contains Mg, Al, O and other elements from the electrolytes, which indicates that the added electrolytes take part in the coating formation.
3.4 Corrosion resistance of anodized samples by different processes
After 120 h salt spray test, the corrosion resistance of anodized samples were evaluated and the sequence is as follows: Process 2>Process 4>Process 3>Process 5>Process 1. About 20% and 80% of sample surfaces by Process 2 and Process 5 is separately eroded.
The properties of anodic coating on magnesium depend on many factors, such as electrolyte[15], electric parameters[16] and substrate[17]. The electrolyte plays a main role in determining the coating properties, such as voltage―time responses, coating thickness, sample color, surface morphology and corrosion resistance. The corrosion resistance of anodic coatings depends on a lot of factors such as the coating composition, coating thickness, porous number and size (namely porosity). The results show that the corrosion of anodized samples could be classified into three stages[11, 18]. In the first stage, the water and electrolyte penetrates into the oxide film through the pores in outer porous layer, reaching the interface between the inner barrier and outer porous layer. In the second stage, it is suggested that MgO is converted to the lower density Mg(OH)2 film. In the third stage, small pores or microcracks in the barrier layer form and corrosion initiates. Therefore, the corrosion resistance of the anodized samples is mainly decided by coating thickness and porosity and it is improved with the coating thickening and decrease of coating porosity. The anodic coating obtained by addition of Na2SiO3 is uniform and the thickest, which can impede the corrosive ions such as chloride ions penetrating into the coating and therefore the best corrosion resistance is achieved. On the contrary, the coating by addition of Na2SnO3 is loose and the thinnest and its corrosion resistance is the worst.
4 Conclusions
1) Under the same electric parameters, the additions of Na2SiO3, Na2CO3, Na2B4O7 and Na2SnO3 into the base solution result in higher final voltage, especially for Na2SiO3 and Na2B4O7.
2) Na2SiO3 and Na2B4O7 can significantly thicken the anodic coatings.
3) The uniformity of anodic coatings obtained by addition of Na2SiO3 and Na2B4O7 is separately the best and the worst, while the coatings by Na2B4O7 and Na2SnO3 are separately the best compact and the loosest.
4) The effect order of electrolytes on corrosion resistance of anodized samples is ranked as Na2SiO3>Na2B4O7>Na2CO3>Na2SnO3.
References
[1] MORDIKE B L, EBERT T. Magnesium properties-applications- potential[J]. Mater Sci Eng A, 2001, 302: 37-45.
[2] MEHTA D S, MASOOD S H, SONG W Q. Investigation of wear properties of magnesium and aluminum alloys for automotive applications[J]. Journal of Materials Processing Technology, 2004, 155-156:1526-1531.
[3] LI J Z, SHAO Z C, ZHANG X, TIAN Y W. The electroless nickel-plating on magnesium alloy using NiSO4?6H2O as the main salt[J]. Surface and Coatings Technology, 2006, 200: 3010-3015.
[4] ZHAO M, WU S S, AN P, LUO J R, FUKUDA Y, NAKAE H. Microstructure and corrosion resistance of a chromium-free multi-elements complex coating on AZ91D magnesium alloy[J]. Materials Chemistry and Physics, 2006, 99: 54-60.
[5] CAI Q Z, WANG L S, WEI B K, LIU Q X. Electrochemical performance of microarc oxidation films formed on AZ91D magnesium alloy in silicate and phosphate electrolytes[J]. Surface and Coatings Technology, 2006, 200: 3727-3733.
[6] UMEHARA H, TAKAYA M, TSUKUBA T I. Corrosion resistance of the die casting AZ91D magnesium alloys with paint finishing[J]. Aluminium, 1999, 75(7/8): 634-641.
[7] Dow Chemical Co. Bath for and method of producing a corrosion resistant coating upon light metals. GB 762195[P]. 1956.
[8] EVANGELIDES H A. Method of electrolytically coating magnesium and electrolyte therefor. US 2723952[P]. 1955.
[9] ZHANG Y J, YAN C W, WANG F H, LOU H Y, CAO C N. Study on the environmentally friendly anodizing of AZ91D magnesium alloy[J]. Surface and Coatings Technology, 2002, 161:36-43.
[10] LUO S L, DAI L, ZHOU H H, CHAI L Y, KUANG Y F. Research on environmental-friendly anodizing process for magnesium alloys[J]. Journal of Hunan University (Natural Sciences), 2005, 32(3): 15-18. (in Chinese)
[11] BIRSS V, XIA S, YUE R, RATEICK R G. Characterization of oxide films formed on Mg-based WE43 alloy using AC/DC anodization in silicate solutions[J]. J Electrochem Soc, 2004, 151(1): B1-B10.
[12] HAO J M, CHEN H, ZHANG R J, JIANG B L. Corrosion resistance of magnesium alloy micro-arc oxidization ceramic coating[J]. The Chinese Journal of Nonferrous Metals, 2003, 13(4): 988-991. (in Chinese)
[13] BARTON T F, JOHNSON C B. The effect of electrolyte on the anodized finish of a magnesium alloy[J]. Plating and Surface Finishing, 1995, 82: 138-141.
[14] VERDIER S, BOINET M, MAXIMOVITCH S, DALARD F. Formation, structure and composition of anodic films on AM60 magnesium alloy obtained by DC plasma anodising[J]. Corrosion Science, 2005, 47: 1429-1444.
[15] MA Y, NIE X, NORTHWOOD D O, HU H. Systematic study of the electrolytic plasma oxidation process on a Mg alloy for corrosion protection[J]. Thin Solid Films, 2006, 494: 296-301.
[16] SHI Z M, SONG G L, ATRENS A. Influence of anodising current on the corrosion resistance of anodised AZ91D magnesium alloy[J]. Corrosion Science, 2006, 48(8): 1939-1959.
[17] SHI Z M, SONG G L, ATRENS A. Influence of the β phase on the corrosion performance of anodised coatings on magnesium- aluminium alloys[J]. Corrosion Science, 2005, 47: 2760-2777.
[18] XIA S J, YUE R, RATEICK R G Jr, BIRSS V I. Electrochemical studies of AC/DC anodized Mg alloy in NaCl solution[J]. J Electrochem Soc, 2004, 151(3): B179-B187.
(Edited by LONG Huai-zhong)
Foundation item: Project (0650034) supported by the Natural Science Foundation of Jiangxi Province, China
Corresponding author: ZHANG Rong-fa; Tel: +86-791-3831266; E-mail: rfzhang-10@163.com