Trans. Nonferrous Met. Soc. China 23(2013) 3330-3336
One-step hydrothermal synthesis of Cu-SAPO-34/cordierite and its catalytic performance on NOx removal from diesel vehicles
Jian-cheng WANG1, Ying CHEN1, Lei TANG1, Wei-ren BAO1, Li-ping CHANG1, Li-na HAN1,2
1. Key Laboratory of Coal Science and Technology, Ministry of Education and Shanxi Province, Taiyuan University of Technology, Taiyuan 030024, China;
2. College of Materials Science and Engineering, Taiyuan University of Technology, Taiyuan 030024, China
Received 22 October 2012; accepted 30 January 2013
Abstract: Cu-SAPO-34/cordierite catalysts were prepared via one-step hydrothermal synthesis method and their performances to remove NOx from the diesel vehicle exhaust were evaluated. The morphology, structure, Cu content and valence state were characterized by SEM, XRD, ICP and XPS, respectively. The experimental results show the active component Cu of the catalysts via in situ synthesis could significantly improve the selective catalytic reduction (SCR) activities of NOx and the optimal Cu content is in the range of 0.30%-0.40% (mass fraction). No N2O is detected by gas chromatograph (GC) during the evaluation process, which implies that NOx is almost entirely converted to N2 over Cu-SAPO-34/cordierite catalyst. The conversion rate of NOx to N2 by NH3 over catalyst could almost be up to 100% in the temperature range of 300-670 ��C with a space velocity of 12000 h-1 and it is still more than 60% at 300-620 ��C under 36000 h-1. The catalysts also show the good hydrothermal and chemical stability at the atmosphere with H2O.
Key words: in situ synthesis; Cu-SAPO-34/cordierite; NOx; selective catalyst reduction; NH3; diesel vehicle exhaust
1 Introduction
The number of diesel vehicles is gradually increased due to the advantages of their strong motivation, excellent economization and trace amounts of HC and CO emissions. However, the removal of NOx by conventional three-way catalysts is difficult because of the high content of oxygen in the diesel engine exhaust. Nowadays, the selective catalytic reduction technology with CHx or NH3 has been widely researched for NOx removal of the exhaust gases from diesel vehicles [1-4]. It has been reported that Cu-SAPO-34 powder has higher catalytic activity and hydrothermal stability in the selective reduction of NOx by CHx [5, 6]. However, the effective utilization rate of active component is lower, and the high bed layer resistance always occurs. It is known that catalyst carrier has a great effect on the activities and service-life of the catalyst. Cordierite (2MgO��2Al2O3��5SiO2) monolith is widely used due to its superior hydrothermal stability, low thermal expansivity and low price [7]. The honeycomb catalysts prepared via the traditional coating technology, such as the Al2O3 coating, are easy to lose the coating and active components, resulting in the decrease of the catalytic efficiency [8]. The molecular sieve coatings of the honeycomb cordierite catalysts prepared via in situ synthesis technology have large specific surface area, and the internal and external surfaces of the coatings may be fully used [9,10]. Meanwhile, the metal-molecular sieves synthesized through directly adding the active component precursors into the molecular sieve mother liquor presents a good dispersion of metal in the structure. Moreover, the metal atoms are good activity centers to the oxidation or reduction [11-13].
In this work, Cu-SAPO-34/cordierite catalysts were prepared via one-step hydrothermal synthesis method. The experiments for removal of NOx from the simulate diesel vehicle exhaust were carried out. The effects of Cu content and water vapor on the catalyst activities were mainly investigated. The catalysts before and after reaction or aging treatment were analyzed by XRD, SEM and XPS, respectively.
2 Experimental
2.1 Preparation of Cu-SAPO-34/cordierite
Cordierite honeycomb was cut into square-shaped samples with the dimensions of 16 mm��16 mm��16 mm. Subsequently, the samples were pretreated for 2 h with 15% (mass fraction) nitric acid solutions through continuous stirring at 80 ��C, washed with deionized water, dried for 2 h at 110 ��C, calcined for 2 h at 700 ��C, and then stored in a drier for the later use.
Morpholine, copper oxide, phosphoric acid, aluminum hydroxide and silica gel were used as the template, active metal source, phosphorus source, aluminum source and silicon source, respectively. The typical composition (molar ratio) of the crystallization solution is xCuO (x=0, 0.04, 0.08 and 0.12):0.2 SiO2:0.46 Al2O3: 0.45 P2O5:1.25 C4H9NO:50 H2O. The catalyst samples were synthesized as follows. CuO was added into the diluted orthophosphoric acid solution and stirred at 80 ��C until CuO was dissolved completely. Secondly, Al(OH)3 was added to form a homogeneous gel by stirring. And then, silica sol and morpholine were added into the gel to obtain the initial gel. Finally, the 35 mL initial gel with a piece of cordierite was placed into an autoclave (volume of 50 mL) and statically crystallized for 7 d at 190 ��C. The products were washed by deionized water, dried in air at 110 ��C for 3 h, and then calcined for 5 h in an O2 atmosphere of 700 ��C. The detailed synthesis procedures can be found in our previous work [14]. The catalysts prepared from 0, 0.04, 0.08 and 0.12 mol/L Cu precursor solution were named F0.00, F0.04, F0.08 and F0.12.
2.2 Characterization of Cu-SAPO-34/cordierite
The samples were examined in the form of powder by XRD (Rigaku D/max2500 diffractometer, Japan) with a graphite monochromator and a Cu K�� radiation source (��=0.154056 nm). The total Cu contents of the samples were analyzed by the inductively coupled plasma-Auger electron spectroscope (ICP-AES, Atomcan-16, America). The morphology of the catalysts was examined by SEM (Jeol JSM-6700F). The samples were covered with a thin gold layer before detection, and the accelerating voltage was 10 kV. XPS surface analysis was conducted to determine the Cu content and valence of catalysts. The spectra were acquired on an ESCALAB 250 spectrometer (VG Scientific Ltd., UK) equipped with an Al K�� radiation source (h��=1486.6 eV). The data smoothing routine was applied to analyze the results.
2.3 Activity evaluation of Cu-SAPO-34/cordierite
The NH3-SCR activities of Cu-SAPO-34/cordierite catalysts on NOx reduction under the atmosphere pressure were evaluated with a fixed-bed flow reactor (inner diameter of 20 mm). The typical simulated diesel vehicle exhaust consisted of 0.05% NOx, 0.05% NH3, 7% O2, 10 % H2O (g), and He as the balance gas. The total flow rate is 420 mL/min. The steam was from the gasification of liquid water controlled by a trace pump. A piece of Cu-SAPO-34/cordierite was put into the quartz reactor for each run. The aging treatment was carried out with 15% H2O, 0.02% SO2 and N2 as balance gas under the space velocity of 12000 h-1. One of the aging condition (denoted: A10.08) is 650 ��C for 10 h, the other (denoted: A20.08) is 720 ��C for 15 h. The inlet concentration of NO, NO2 and O2 were analyzed by a flue gas analyzer (British, Kane-9106). The effluent gas composition was analyzed online using a gas chromatograph (GC98090A, Shanghai LingHua YiQi, China) equipped with two TCD detectors and two serial columns (a Porapak Q column served for detection of N2O, and a 5A Molecular Sieve column for detection of N2).
The conversion rate of NOx to N2 was calculated with the equation y=(C0-C1)/C0��100%, where y was the conversion rate of NOx to N2; C0 and C1 were the concentration of NOx and N2 in the inlet and outlet gases of reactor, respectively.
3 Results and discussion
3.1 SCR activities of different Cu content catalysts
It is generally acknowledged that the loading amount of active component has important influence on the property of the monolithic catalyst. The loading amount of Cu-SAPO-34 prepared with different Cu precursor solution were listed in Table 1. They are 20.6%, 21.8%, 22.5%, and 21.3% corresponding to F0.00, F0.04, F0.08, and F0.12 samples, respectively, and all the Cu-SAPO-34 loading amounts are more than 20% after one synthesis operation. In our previous work, it has been reported that the repeatability synthesis was very good and the mass loss was minor. The metal Cu content characterized by ICP is also shown in Table 1. It can be seen that the Cu content of fresh F0.04, F0.08 and F0.12 catalyst is 0.30%, 0.38%, 0.45%, respectively.
Table 1 Cu content and loading amount of Cu-SAPO-34 in different catalysts

The NH3-SCR activities of Cu-SAPO-34/ cordierites with different Cu content are shown in Fig. 1. The highest conversion rate of NOx on Cu-SAPO-34/ cordierite (Fig. 1(a)) is only about 60% in temperature range of 600-670 ��C. But the SCR activity on Cu-SAPO-34/cordierite increase rapidly and the conversion rate on F0.04 and F0.08 catalysts can reach almost 100% in the range of 280-680 ��C when the space velocity is 12000 h-1. It proves that the copper active component in the catalyst can significantly improve the SCR activity of NOx in the simulation diesel vehicle exhaust. It can also be seen from Fig. 1(a) that the NOx conversion rate increases slightly in the low temperature range of 200-350 ��C with increasing molar ratio of Cu precursor from F0.04 to F0.08 and then decreases when Cu content is further increased to F0.12, which indicates that excessive amount of active component copper is not good for improving catalytic activity. So the catalyst F0.08 with proper copper content has the optimal SCR activities of NOx.
The effect of space velocity on SCR activity is shown in Figs. 1(b) and (c). It can be found that the conversion rate of NOx can keep at a higher level over a broader range of space velocity from 24000 to 36000 h-1, although it decreased slightly with the increase of space velocity. When the space velocity is 36000 h-1, the conversion rate of NOx still maintains above 75% in the temperature range of 350-620 ��C for F0.08 catalyst. It should be noted that no N2O was detected by GC with the Porapak Q column, which indicates that the catalyst can be well suitable for the removal of NOx from the diesel vehicle exhaust.
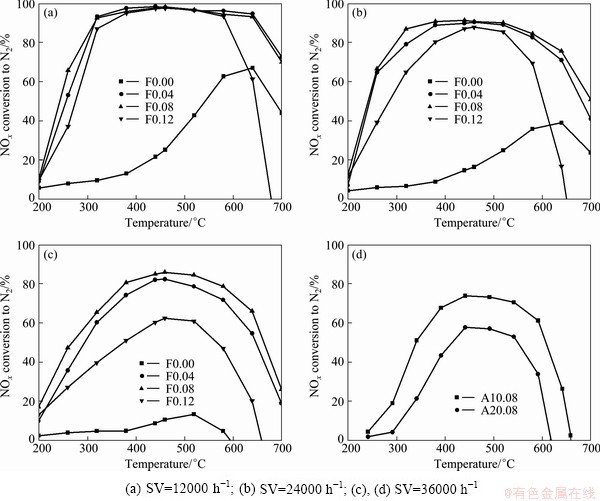
Fig. 1 Effect of Cu content, space velocity and aging treatment on SCR activity of Cu-SAPO-34/cordierite
The SCR activities of F0.08 catalyst after the two kinds of aging treatment are presented in Fig. 1(d). It can be seen that the de-NOx activity of A10.08 (650 ��C, 10 h) is decreased and the conversion rate maintains at 70% in the temperature range of 350-650 ��C when the space velocity is 36000 h-1. And another catalyst (named A20.08) was heated to 720 ��C and exposed to 15% H2O and 0.02% SO2 feeding gas for 15 h. The conversion rate of NOx is decreased to 50% in the temperature range of 400-550 ��C when the space velocity is 36000 h-1. These results show that the catalyst still has some SCR activity for NOx removal in the diesel exhaust gases after high temperature aging treatment.
3.2 Structure analyses of catalysts by XRD
XRD spectra of the fresh samples with different copper content are shown in Fig. 2. The diffraction peaks at 9.6��, 16.2��, 20.9��, 25.9��, 31.2�� corresponding to the characteristic peaks of SAPO-34 [15] show that SAPO-34 crystallites have been loaded on the cordierite substrates. It should be noted that the characteristic peaks of Cu species have not been detected in all Cu-SAPO-34/cordierite catalysts, which could possibly be attributed to the uniform distribution of Cu in the samples or the migration of some Cu to the skeleton of the molecular sieves during the crystallization process [13,16]. The effective load of SAPO-34 crystallites and distribution of Cu in the catalyst should contribute to the good de-NOx performance of Cu-SAPO-34/cordierite catalysts. F0.08 catalyst has the highest relative crystallinity among 4 different catalysts, which may explain its best SCR activity of NOx. XRD characteristics of the catalyst after reaction and aging treatment are also shown in Fig. 2. SAPO-34 characteristic peaks of R0.08 (denoted as the catalyst which is F0.08 after 4 times of the SCR reactions) are little changed compared with fresh F0.08 catalyst, which suggests that SAPO-34 crystallites loaded on the cordierite substrates have excellent chemistry stability. The results above suggest that the deactivation caused by the irreversible physical change of the copper sintering and zeolite dealuminizing in Cu-zeolites catalyst [17,18] is not presented in the F0.08 catalyst after a series of complicated SCR reactions of NOx in the simulative diesel vehicle exhaust. But the slight decrease of SAPO-34 characteristic patterns of A10.08 can be observed. It suggests that the SAPO-34 crystallites can be destroyed partially and it can be attributed the decrease of the activity for removal NOx.

Fig. 2 XRD patterns of cordierite, catalysts with different Cu content and samples after reaction or aging treatment F0.08 catalyst
3.3 Morphology analyses of catalysts by SEM
SEM images of F0.08, R0.08, A10.08 and A20.08 samples are shown in Fig. 3. It indicates that the uniform and compact morphology of the crystalloids of prepared catalyst are almost unchanged after reactions compared with F0.08 and R0.08. But the SEM images of A10.08 and A20.08 indicate that the crystalloids had been destroyed slightly after the aging treatments. It can be deduced that the decrease of the de-NOx activities is mainly caused by the change of the morphology and crystallite structure on the surface of catalyst during the process of aging treatment. To improve the performance of the catalyst, its structure stability will be our focused point in the future.
3.4 Chemical state analyses of catalysts by XPS
The valence state of Cu in F0.08 catalyst is very important for the NOx SCR [5]. Therefore, in this part, the chemical states of the surface Cu species of F0.08, R0.08, A10.08 and A20.08 catalysts were investigated using XPS and the results are shown in Fig. 4. The Cu 2p3/2 peak around 934.9 eV and the satellite peak around 940-948 eV indicate that the surface copper is largely presented as Cu2+ (note: the satellite peak is usually used as a characteristic to determine Cu2+), the second Cu 2p3/2 peak around 932.2 eV could be assigned to Cu+ due to the related Cu auger spectra at 916.8 eV [19,20]. The former should be assigned to Cu2+ in CuO, and the latter is tentatively assigned to Cu2O [19]. The increase of Cu 2p3/2 peak at 934.9 eV of R0.08 and the obvious decrease of this peak of A10.08 and A20.08 samples are observed. These indicates that the changes of the Cu valence state are obvious.

Fig. 3 SEM images of different sample surfaces
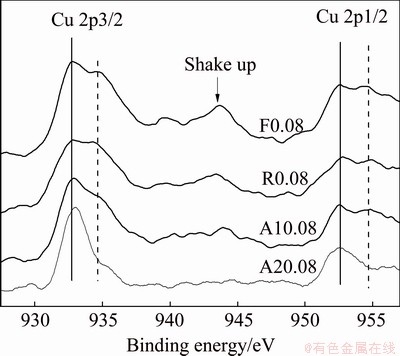
Fig. 4 Cu 2p XPS spectra of F0.08, R0.08, A10.08 and A20.08 catalysts
The peaks of Cu 2p3/2 are fitted for further discussing the effect of the reaction and aging treatments of catalyst on Cu valence state on the surface of the catalysts. Gaussian�CLorentzian fit to Cu 2p3/2 indicates that the main peak is composed of two peaks at 932.7 and 935 eV, respectively, as shown in Fig. 4. The fitting results and the Cu/Al atomic ratios of different catalysts are listed in Table 2. The similar Cu/Al atomic ratio of F0.08, R0.08, A10.08 and A20.08 catalysts suggests that there is almost no change of the surface elements. The relative content of Cu2+ and Cu+ according to the fitting results can be easily calculated and the results are listed in Table 2. It can be seen that the relative contents of Cu2+ and Cu+ before and after reaction have no obvious difference, but the relative contents of Cu2+ and Cu+ of A10.08 and A20.08 change greatly. There is an oxidation reduction cycle between Cu2+ and Cu+, in which Cu2+ is usually used as the active center [21]. Active species are formed through NH3 adsorption on Cu2+ and then react with NO into N2, Cu2+ change into Cu+ and then being oxidized into Cu2+. So, less Cu2+ on the surface of the aging sample leads to the performance changes for NOx removal in a certain degree, because the mixed valence state of Cu+ and Cu2+ is useful for the catalysts to maintain the de-NOx activities [18,22].
Table 2 Fitting results of Cu 2p3/2 XPS curve and Cu/Al atomic ratios in different catalysts

4 Conclusions
1) Cu-SAPO-34/cordierite catalysts with the good hydrothermal stability and higher efficiency for the removal of NOx from the simulated diesel vehicle exhaust were prepared via one-step hydrothermal synthesis procedure.
2) The active component copper, which is evenly migrated into the structure of molecular sieves in the process of hydrothermal synthesis, can significantly improve the de-NOx activities and the optimal Cu content is in the range of 0.30%-0.40%.
3) The conversion rate of NOx to N2 by NH3 over Cu-SAPO-34/cordierite can almost be up to 100% in the temperature range of 300-670 ��C with the space velocity of 12000 h-1 and it is still more than 60% from 300 to 620 ��C corresponding to 36000 h-1.
4) Almost no N2O can be detected by GC during the evaluation process, which shows that NOx is entirely converted to N2 and the prepared catalyst is suitable for the removal of NOx from the diesel vehicle exhaust.
5) The catalysts also show a good hydrothermal and chemical stability in the atmosphere with H2O and SO2. It will be our focused point to improve the structure stability of the catalyst in the future.
References
[1] BALLE P, GEIGER B, KURETI S. Selective catalytic reduction of NOx by NH3 on Fe/HBEA zeolite catalysts in oxygen-rich exhaust [J]. Applied Catalysis B: Environmental, 2009, 85(3-4): 109-119.
[2] KIM M H, NAM I S. New opportunity for HC-SCR technology to control NOx emission from advanced internal combustion engines [J]. Catalysis, 2005, 18: 116-185.
[3] KELLY J F, STANCIULESCU M, CHARLAND J P. Evaluation of amines for the selective catalytic reduction (SCR) of NOx from diesel engine exhaust [J]. Fuel, 2006, 85(12-13): 1772-1780.
[4] WANG J C, TIAN D, HAN L N, CHANG L P, BAO W R. In situ synthesized Cu-ZSM-5/cordierite for reduction of NO [J]. Transactions of Nonferrous Metals Society of China, 2011, 21(2): 353-358.
[5] XUE J, WANG X, QI G, WANG J, SHEN M, LI W. Characterization of copper species over Cu/SAPO-34 in selective catalytic reduction of NOx with ammonia: Relationships between active Cu sites and de-NOx performance at low temperature [J]. Journal of Catalysis, 2013, 297(0): 56-64.
[6] YE Q, WANG L, YANG R T. Activity, propene poisoning resistance and hydrothermal stability of copper exchanged chabazite-like zeolite catalysts for SCR of NO with ammonia in comparison to Cu/ZSM-5 [J]. Applied Catalysis A: General, 2012, 427-428: 24-34.
[7] 
 M J, de LECEA
M J, de LECEA  Z. Preparation of beta-coated cordierite honeycomb monoliths by in situ synthesis: Utilisation as Pt support for NOx abatement in diesel exhaust [J]. Applied Catalysis B: Environmental, 2005, 58(1-2): 1-7.
Z. Preparation of beta-coated cordierite honeycomb monoliths by in situ synthesis: Utilisation as Pt support for NOx abatement in diesel exhaust [J]. Applied Catalysis B: Environmental, 2005, 58(1-2): 1-7.
[8] MITRA B, KUNZRU D. Washcoating of different zeolites on cordierite monoliths [J]. Journal of the American Ceramic Society, 2008, 91(1): 64-70.
[9] LI L, XUE B, CHEN J, GUAN N, ZHANG F, LIU D, FENG H. Direct synthesis of zeolite coatings on cordierite supports by in situ hydrothermal method [J]. Applied Catalysis A: General, 2005, 292: 312-321.
[10] ZAMARO J M, MIR E E. Novel binderless zeolite-coated monolith reactor for environmental applications [J]. Chemical Engineering Journal, 2010, 165(2): 701-708.
[11] PANTAZIS C, TRIKALITIS P, POMONIS P. Highly loaded and thermally stable Cu-containing mesoporous silica active catalyst for the NO+CO reaction [J]. The Journal of Physical Chemistry B, 2005, 109(25): 12574-12581.
[12] DEKA U, LEZCANO-GONZALEZ I, WARRENDER S J, LORENA PICONE A, WRIGHT P A, WECKHUYSEN B M, BEALE A M. Changing active sites in Cu�CCHA catalysts: deNOx selectivity as a function of the preparation method [J]. Microporous and Mesoporous Materials, 2013, 166: 144-152.
[13]  R, MOLINER M, FRANCH C, KUSTOV A, CORMA A. Rational direct synthesis methodology of very active and hydrothermally stable Cu-SAPO-34 molecular sieves for the SCR of NOx [J]. Applied Catalysis B: Environmental, 2012, 127: 273-280.
R, MOLINER M, FRANCH C, KUSTOV A, CORMA A. Rational direct synthesis methodology of very active and hydrothermally stable Cu-SAPO-34 molecular sieves for the SCR of NOx [J]. Applied Catalysis B: Environmental, 2012, 127: 273-280.
[14] LIU Z, TANG L, CHANG L, WANG J, BAO W. In situ synthesis of Cu-SAPO-34/cordierite for the catalytic removal of NOx from diesel vehicles by C3H8 [J]. Chinese Journal of Catalysis, 2011, 32(3-4): 546-554.
[15] YAN Z, CHEN B, HUANG Y. A solid-state NMR study of the formation of molecular sieve SAPO-34 [J]. Solid State Nuclear Magnetic Resonance, 2009, 35(2): 49-60.
[16] FRACHE A, PALELLA B, CADONI M, PIRONE R, CIAMBELLI P, PASTORE H, MARCHESE L. Catalytic deNOx activity of cobalt and copper ions in microporous MeALPO-34 and MeAPSO-34 [J]. Catalysis Today, 2002, 75(1-4): 359-365.
[17] MEZ S G, CAMPERO A, FUENTES G. Changes in Cu2+ environment upon wet deactivation of Cu-ZSM-5 deNOx catalysts [J]. Applied Catalysis A: General, 2000, 197(1): 157-164.
[18] WILKEN N, WIJAYANTI K, KAMASAMUDRAM K, CURRIER N W, VEDAIYAN R, YEZERETS A, OLSSON L. Mechanistic investigation of hydrothermal aging of Cu-Beta for ammonia SCR [J]. Applied Catalysis B: Environmental, 2012, 111-112: 58-66.
[19] ZHOU J, XIA Q H, SHEN S C, KAWI S, HIDAJAT K. Catalytic oxidation of pyridine on the supported copper catalysts in the presence of excess oxygen [J]. Journal of Catalysis, 2004, 225(1): 128-137.
[20] VELU S, SUZUKI K, VIJAYARAJ M, BARMAN S, GOPINATH C S. In situ XPS investigations of Cu1-xNixZnAl-mixed metal oxide catalysts used in the oxidative steam reforming of bio-ethanol [J]. Applied Catalysis B: Environmental, 2005, 55(4): 287-299.
[21] LIU Fu-dong, SHAN Wen-po, SHI Xiao-yan, ZHANG Chang-bin, HONG H. Research progress in vanadium-free catalysts for the selective catalytic reduction of NO with NH3 [J]. Chinese Journal of Catalysis, 2011, 32(7): 1113-1128.
[22] PARK J H, PARK H J, BAIK J H, NAM I S, SHIN C H, LEE J H, CHO B K, OH S H. Hydrothermal stability of CuZSM5 catalyst in reducing NO by NH3 for the urea selective catalytic reduction process [J]. Journal of Catalysis, 2006, 240(1): 47-57.
Cu-SAPO-34/����ʯ��ԭλ�Ʊ�������ѳ����ͻ���β����NOx
������1���� Ӱ1���� ��1��������1������Ƽ1��������1,2
1. ̫ԭ������ѧ ú��ѧ�뼼����������ɽ��ʡ�ص�ʵ���ң�̫ԭ 030024��
2. ̫ԭ������ѧ ���Ͽ�ѧ�빤��ѧԺ��̫ԭ 030024
ժ Ҫ������ˮ�Ⱥϳɼ�����Cu-SAPO-34����ɸֱ�Ӻϳɵ�����״����ʯ�ϣ��о�ģ����ͻ���β����ѡ���Դ���ԭNOx������������SEM��XRD��ICP��XPS��������ʽ��������ò���ṹ��Cu�����ͼ�̬�仯�����������ԭλ�Ʊ������Ʊ���Cu-SAPO-34/����ʯ��������еĻ��Խ���Cu���кܸߵ�ѡ���Դ���ԭNOx�Ļ��ԣ�Cu�������������������Ϊ0.30%~0.40%����12000 h-1����������£���400~670 ��C֮�䣬�ɽ��ֵ�NOx����ԭ��N2����36000 h-1�Ŀ����£���300~620 ��C֮�䣬���ܽ�60%��NOxת����N2��ˮ���ϻ���Ĵ����Ϻõı��ִ����ԣ������������нϺõ�ˮ���ȶ��ԡ�
�ؼ��ʣ�ԭλ�ϳɣ�Cu-SAPO-34/����ʯ��NOx��ѡ���Դ���ԭ���������ͻ���β��
(Edited by Chao WANG)
Foundation item: Project (20906067) supported by the National Natural Science Foundation of China; Project (2011M500543) supported by the Postdoctoral Science Foundation of China; Project supported by the Program for the Top Young Academic Leaders of Higher Learning Institutions of Shanxi
Corresponding author: Li-ping CHANG; Tel/Fax: +86-351-6010482; E-mail: lpchang@tyut.edu.cn
DOI: 10.1016/S1003-6326(13)62872-1