Vanadium extraction from vanadium-bearing titanomagnetite by selective chlorination using chloride wastes (FeClx)
来源期刊:中南大学学报(英文版)2017年第2期
论文作者:郑海燕 孙瑜 卢金文 董建宏 章苇玲 沈峰满
文章页码:311 - 317
Key words:vanadium-bearing titanomagnetite; vanadium extraction; selective chlorination; thermodynamic analysis; chloride wastes
Abstract: Vanadium extraction of vanadium-bearing titanomagnetite was investigated by selective chlorination. Thermodynamics analyses on the interactive reactions among related species in the system were made before the experiments. Some fundamental experiments for extracting vanadium by FeClx as chlorinating agent were conducted over the temperature range of 900-1300 K under air or oxygen atmosphere. The results show that vanadium can be extracted by the selective chlorination, using FeClx, based on thermodynamic analysis and experiment. Vanadium extraction ratio first increases with the increase of temperature, and then decreases with the increase of temperature over the range of 900-1300 K under air or oxygen atmosphere. The higher molar ratio of FeCl3 to oxides (nchl:noxd) reacting with FeCl3, the higher ratio of vanadium extraction. Under oxygen atmosphere, the vanadium extraction ratio is up to 32% at 1100 K for 2 h by using FeCl3 as chlorinating agent.
Cite this article as: ZHENG Hai-yan, SUN Yu, LU Jin-wen, DONG Jian-hong, ZHANG Wei-ling, SHEN Feng-man. Vanadium extraction from vanadium-bearing titanomagnetite by selective chlorination using chloride wastes (FeClx) [J]. Journal of Central South University, 2017, 24(2): 311-317. DOI: 10.1007/s11171-017-3432-5.
J. Cent. South Univ. (2017) 24: 311-317
DOI: 10.1007/s11171-017-3432-5
ZHENG Hai-yan(郑海燕)1, SUN Yu(孙瑜)1, LU Jin-wen(卢金文)2, DONG Jian-hong(董建宏)1,
ZHANG Wei-ling(章苇玲)1, SHEN Feng-man(沈峰满)1
1. School of Materials & Metallurgy, Northeastern University, Shenyang 110819, China;
2. Northwest Institute for Non-ferrous Metal Research, Xi’an 710016, China
 Central South University Press and Springer-Verlag Berlin Heidelberg 2017
Central South University Press and Springer-Verlag Berlin Heidelberg 2017
Abstract: Vanadium extraction of vanadium-bearing titanomagnetite was investigated by selective chlorination. Thermodynamics analyses on the interactive reactions among related species in the system were made before the experiments. Some fundamental experiments for extracting vanadium by FeClx as chlorinating agent were conducted over the temperature range of 900-1300 K under air or oxygen atmosphere. The results show that vanadium can be extracted by the selective chlorination, using FeClx, based on thermodynamic analysis and experiment. Vanadium extraction ratio first increases with the increase of temperature, and then decreases with the increase of temperature over the range of 900-1300 K under air or oxygen atmosphere. The higher molar ratio of FeCl3 to oxides (nchl:noxd) reacting with FeCl3, the higher ratio of vanadium extraction. Under oxygen atmosphere, the vanadium extraction ratio is up to 32% at 1100 K for 2 h by using FeCl3 as chlorinating agent.
Key words: vanadium-bearing titanomagnetite; vanadium extraction; selective chlorination; thermodynamic analysis; chloride wastes
1 Introduction
Vanadium-bearing titanomagnetite deposits are abundant and widely-distributed in China. The ores of the titanomagnetite deposits are complex, with a wide range of titanium(Ti), vanadium(V), and iron(Fe) proportions; they may be essentially titaniferrous or essentially iron-vanadium ones. Indeed, 98% of the world’s total vanadium reserve is found in vanadium- bearing titanomagnetite deposits. Besides, recoverable amounts of scandium(Sc), copper(Cu), cobalt(Co), nickel(Ni), gold(Au), platinum(Pt), lead(Pd) and others have been found in some deposits. Hence, it is essential to comprehensively utilize vanadium-bearing titanomagnetite for sustainable development in the future [1-6].
Regarding the comprehensive utilization of vanadium-bearing titanomagnetite, many efforts have been made by lots of researchers [7-13]. Currently, the major process for comprehensive utilization of vanadium-bearing titanomagnetite in China is blast furnace (BF) process [14-15]. However, this BF process is not enough to satisfy the demand for effectively recovering the useful elements, such as V and Ti, in the ore. Therefore, attention has been drawn to comprehensive utilization of the titanomagnetite deposits with a new method. Based on the abovementioned background, the objective of this project is to extract vanadium from the ore first, then to reduce the ore to produce direct reduction iron (DRI), and finally to separate metal from the titanium-rich slag and obtain DRI with high quality.
Some chloride wastes, such as iron chlorides (FeClx, x=2, 3), are generated from the chlorination sub-process in the Kroll process for Ti smelting because the TiO2 feed contains impurities such as Fe [16-17]. Currently, there is no process that efficiently recycles or treats the chloride wastes generated from the Kroll process; therefore, they are discarded after chemical treatment. The treatment of chloride wastes involves several problems, e.g., disposal costs and environmental issues. Furthermore, an additional amount of Cl2 gas has to be purchased in order to compensate for the chlorine loss caused in the generation of the chloride wastes. In order to minimize the generation of chloride wastes, rutile ore or upgraded ilmenite (UGI) comprised of approximately 95% TiO2 is currently used as the raw material in the
Kroll process. In the future, the amount of chloride wastes will increase as the production volume of Ti increases and the quality of the ore feed decreases.
Due to the abovementioned factors, a novel process for comprehensively recovering V, Ti, and Fe from vanadium-bearing titanomagnetite by selective chlorination using chloride wastes (FeClx) is proposed, as shown in Fig. 1.
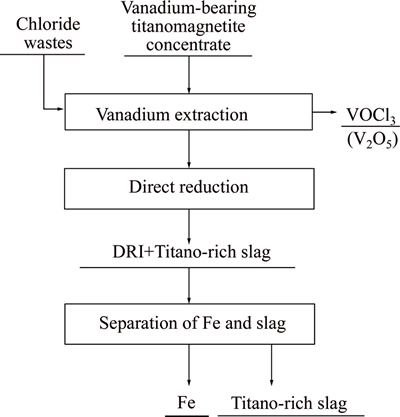
Fig. 1 Proposed concept of new smelting process for vanadium-bearing titanomagnetite
2 Concept of vanadium extraction by selective chlorination
The proposed novel process as shown in Fig. 1 involves three parts: 1) selectively chloridizing vanadium-bearing ore and extracting vanadium by the form of vanadium trichloride oxide gas (Melting point: 196 K, boiling point: 400 K) from the ore through controlling the chemical potential in the system; 2) reducing the vanadium free pellets after roasting for producing DRI; 3) separating the DRI from titano-rich slag. The left titano-rich slag can be used as the alternative of raw material for the production of titanium oxide or titanium smelting process. The advantages of this process are that it is possible to utilize vanadium- bearing resources rationally and recover vanadium, iron, and titanium efficiently; moreover, it is a simple, high- speed, semi-continuous process, and easy to scale-up; the chloride wastes can also be efficiently recovered by this process, which not only can reduce the environmental burden but also decrease the chlorine loss due to the generation of chloride wastes. This work reports the selective chlorination of vanadium-bearing titanomagnetite using chloride wastes. If this new process is feasible, not only can vanadium be extracted but it would also be possible to effectively recover the chlorine in the chloride wastes generated from Ti smelting or any other process. Another benefit of this process is that low-grade Ti ore can be used in the Kroll process if the chlorine can be efficiently recovered.
3 Thermodynamic calculations on selective chlorination
Prior to the experimental works, the feasibilities of
vanadium extraction from vanadium-bearing titanomagnetite by selective chlorination were discussed from the thermodynamic viewpoint.

Fig. 2 Combined chemical potential diagrams for Fe-V-Cl-O system (a) and Ti-V-Cl-O system (b) at 1100 K 
Some species related in the system are listed as C, O2, Cl2, Fe, Ti, V, CaO, CaCl2, CaTiO3, FeO, FeCl2, FeCl3, VO, V2O3, V2O4, V2O5, VCl2, VCl4, VOCl3, TiO, Ti2O3, Ti3O5, Ti4O7, TiO2, TiCl, TiCl2, TiCl3, TiCl4, CO and CO2. The reactions among them are discussed by using the thermodynamic information obtained from previous study [18].
3.1 Isothermal chemical potential diagrams for Fe-Ti- V-Cl-O system
Figure 2(a) shows the combined isothermal chemical potential diagram for the Fe-Cl-O (solid lines) and V-Cl-O (dashed lines) systems at 1100 K as a function of 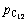 and
and 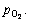 The hatched region A in Fig. 2(a) is the chemical potential region in which Fe2O3 and VOCl3 are thermodynamically stable, which indicates that when the chemical potential is controlled in the hatched region A, vanadium in the titanomagnetite ore is chlorinated to form VOCl3 and the chlorination of FeOx does not proceed. The related possible chlorination may emerge as below when FeCl3 is used as chlorinating agent.
The hatched region A in Fig. 2(a) is the chemical potential region in which Fe2O3 and VOCl3 are thermodynamically stable, which indicates that when the chemical potential is controlled in the hatched region A, vanadium in the titanomagnetite ore is chlorinated to form VOCl3 and the chlorination of FeOx does not proceed. The related possible chlorination may emerge as below when FeCl3 is used as chlorinating agent.
1/2V2O5(l)+FeCl3(g)=VOCl3(g)+1/2Fe2O3(s) (1)
1/2V2O4(s)+FeCl3(g)+1/4O2(g)=VOCl3(g)+1/2Fe2O3(s) (2)
Figure 2(b) shows the combined isothermal chemical potential diagram for the Ti-Cl-O (dotted lines) and V-Cl-O (dashed lines) systems at 1100 K as a function of 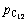 and
and 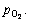 The hatched region B in Fig. 2(b) is the chemical potential region in which TiO2 and VOCl3 are thermodynamically stable, which indicates that when the chemical potential is controlled in the hatched region B, vanadium in the titanomagnetite ore is chlorinated to form VOCl3 and the chlorination of TiO2 does not proceed. The related possible chlorination reactions are listed as reactions (1), (3) and (4) when FeCl3 is used as chlorinating agent.
The hatched region B in Fig. 2(b) is the chemical potential region in which TiO2 and VOCl3 are thermodynamically stable, which indicates that when the chemical potential is controlled in the hatched region B, vanadium in the titanomagnetite ore is chlorinated to form VOCl3 and the chlorination of TiO2 does not proceed. The related possible chlorination reactions are listed as reactions (1), (3) and (4) when FeCl3 is used as chlorinating agent.
1/2V2O3(g)+FeCl3(g)+(2x-1)/4O2(g)=VOCl3(g)+FeOx(s) (3)
1/2V2O4(s)+FeCl3(g)+(x-1)/2O2(g)=VOCl3(g)+FeOx(s) (4)
Because region A is included in region B, so when the chemical potential is controlled in the hatched region A, the VOCl3 can be generated by using FeCl3 as chlorinating agent and released from the ore, thus contributing to V extraction and the acquisition of vanadium-free titanomagnetite.
3.2 Effects of temperature on chemical potential diagrams for Fe-Ti-V-Cl-O system
Figure 3 shows the combined chemical potential diagrams for the Fe-Cl-O system (solid lines), Ti-Cl-O system (dotted lines), and V-Cl-O system (dashed lines) at 900 K, 1100 K, and 1300 K, respectively. As shown in Fig. 3, there are the hatched zones in which VOCl3, FeOx, and TiOx are stable and coexist over the temperature range of 900-1300 K, and this indicates that it is possible to remove V in the form of VOCl3 over the temperature range of 900-1300 K. Furthermore, it is found that the hatched zone shrinks with the increase of
temperature and indicating that higher oxygen partial pressure is necessary and the control of potential into the hatched zone is harder at higher temperature. Therefore, elevated temperature is unfavorable to the generation of VOCl3 in terms of chlorine potential control.

Fig. 3 Combined chemical potential diagrams for Fe-Ti-V-Cl-O system at 900 K (a), 1100 K (b) and 1300 K (c) 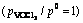
3.3 △Gor in chlorination reactions of oxides with FeClx
Figure 4 shows 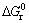 of chlorination reactions of some oxides with FeCl3. As shown in Fig. 4, it is feasible to extract V from the slag by chlorination with FeCl3 (reaction (1)) and the order of chlorination is CaO>MnO>V2O5>MgO over the temperature range of 900-1300 K in which FeCl3 does not react with Al2O3, Cr2O3, SiO2 and TiO2. The mass amount of FeCl3 added into the system is obtained by the mass balance of the above oxides (CaO, MnO and V2O5) reacting with FeCl3.
of chlorination reactions of some oxides with FeCl3. As shown in Fig. 4, it is feasible to extract V from the slag by chlorination with FeCl3 (reaction (1)) and the order of chlorination is CaO>MnO>V2O5>MgO over the temperature range of 900-1300 K in which FeCl3 does not react with Al2O3, Cr2O3, SiO2 and TiO2. The mass amount of FeCl3 added into the system is obtained by the mass balance of the above oxides (CaO, MnO and V2O5) reacting with FeCl3.
FeCl2 can not directly react with V2O5 based on the thermodynamics calculation. However, FeCl2 can react with V2O5 under the condition of oxygen as shown in reaction (5). Figure 5 shows  of chlorination reactions of some oxides with FeCl2 and indicates that it is feasible to extract V from the slag by chlorination with FeCl2 under enough oxygen partial pressure and the order of chlorination is CaO>MnO>V2O5>MgO over the temperature range of 900-1300 K in which FeCl2 does not react with Al2O3, Cr2O3, SiO2 and TiO2. The mass amount of FeCl2 added into the system is obtained by the mass balance of the above oxides (CaO, MnO, and V2O5) reacting with FeCl2.
of chlorination reactions of some oxides with FeCl2 and indicates that it is feasible to extract V from the slag by chlorination with FeCl2 under enough oxygen partial pressure and the order of chlorination is CaO>MnO>V2O5>MgO over the temperature range of 900-1300 K in which FeCl2 does not react with Al2O3, Cr2O3, SiO2 and TiO2. The mass amount of FeCl2 added into the system is obtained by the mass balance of the above oxides (CaO, MnO, and V2O5) reacting with FeCl2.
FeCl2(g)+1/3V2O5(s)+1/4O2(g)=1/2Fe2O3(s)+ 2/3VOCl3(g) (6)
4 Selective chlorination experiment
In order to oxidize V2O3 in the ore and remove the free water or crystal water, vanadium-bearing titanomagnetite concentrate is roasted at 1173 K (900 °C) for 2 h in an electric furnace. Based on the thermodynamic analysis and mass balance calculation, feed pellets of d20 mm×8 mm are fabricated from a mixture prepared by mixing the roasted vanadium- bearing titanomagnetite and FeClx as chlorinating agent with a presser under 40 MPa pressure. Prior to the experiments, the fabricated pellet is put into a special reactor sealed with a special cover, and then oxygen is filled into the reactor at the flow rate of 10 L/min for 5 min in some experiments in order to keep oxygen full of the reactor. As shown in Fig. 6, the reactor containing the sample, together with oxygen, is placed in an electric furnace and maintained at an elevated temperature for several hours. After the experiment, nitrogen was flowed into the reactor by the flow rate of 10 L/min for 5 min in order to flow the VOCl3 out of the reactor. The solid residues in the reactor is recovered and subjected to analyze by X-ray diffraction analysis (XRD) and chemical analysis. The chemical compositions of the raw ore after roasting and chlorinating agents used in this work are shown in Tables 1 and 2, respectively. Table 3 shows the experimental conditions.
Fig. 4 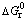 of chlorination reactions with FeCl3
of chlorination reactions with FeCl3
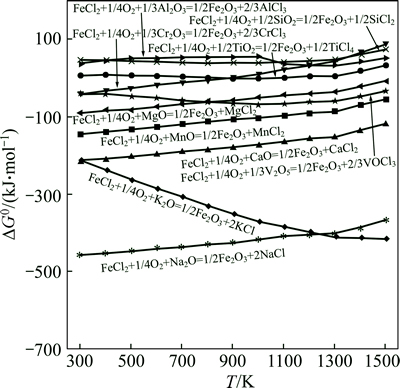
Fig. 5 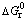 of chlorination reactions with FeCl2
of chlorination reactions with FeCl2

Fig. 6 Schematic illustration of experimental apparatus
Table 1 Composition of vanadium-bearing titanomagnetite after roasting (Mass fraction, %)
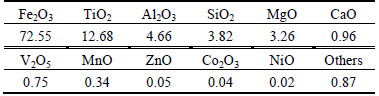
Table 2 Parameters of chlorinating agent

5 Results and discussion
5.1 Phases in samples (XRD patterns)
The samples before and after roasting are analyzed by XRD. As shown in Fig. 7, Fe2O3 and Fe2TiO5(Fe2O3・TiO2) are detected in the samples after roasting, which is attributed to that Fe3O4 and FeTiO3(FeO・TiO2) are oxidized respectively in the atmosphere as shown in reactions (6) and (7). It is found that vanadium oxides are not detected in Fig. 7 because the amounts of vanadium oxides in the ores are small (<0.8%). It is known that vanadium in the ore exists as the form of FeO・V2O3. Figure 8 shows the Ellingham diagrams of Fe3O4 (reaction (6)) and V2O3 (reactions (8)). As shown, oxidization of V2O3 is easier than that of Fe3O4, so oxidization of V2O3 would happen during the roasting experiment before the appearance of Fe2O3 and Fe2TiO5(Fe2O3・TiO2), that is, reaction (9) would happen in the experiment and the chlorination of vanadium oxide(V2O5) should be expressed as shown in reaction (1).
4Fe3O4(s)+O2(g)=6Fe2O3 (s) (6)
4FeO・TiO2(s)+2Fe2O3(s)+O2(g)=4Fe2O3・TiO2(s) (7)
V2O3(s)+O2(g)=V2O5(s) (8)
4FeO・V2O3(s)+2Fe2O3(s)+5O2(g)=4Fe2O3・V2O5(s) (9)
5.2 Factors on vanadium extraction
Vanadium extraction ratio is introduced and determined by formula in order to check the effect of vanadium extraction. The physical meaning of vanadium extraction ratio is the ratio of vanadium mass loss by the generation of VOCl3 to the vanadium mass in the raw material.
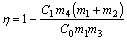 (10)
(10)
where C0 is the concentration of vanadium in the ore after roasted, %; C1 is the concentration of vanadium in the sample after chlorination experiment, %; m1 is the mass of the ore in original mixture, g; m2 is the mass of chlorinating agent in original mixture, g; m3 is the total mass of a feed pellet, g; m4 is the total mass of the pellet obtained after chlorination experiment, g.
Figure 9 shows that vanadium extraction ratio initially increases and then decreases with an increase of
temperature under air atmosphere. The chlorination of oxides by FeCl3 is influenced by both thermodynamics and kinetics aspects. Reaction (1) is an exothermic reaction  kJ/mol [15] at 1100 K), therefore elevated temperature is unfavorable to extracting vanadium in terms of thermodynamics and chlorine potential control as mentioned above. However, it is beneficial to extract vanadium at elevated temperatures in terms of kinetics because reaction rate and mass transport are faster at elevated temperatures. Figure 9 shows the coupled results of thermodynamics and kinetics aspects, indicating that the extraction ratio of vanadium is maximum of 32% at 1100 K for 2 h. The chlorination by FeCl2 is similar with the chlorination by FeCl3. The vanadium extraction ratio is higher by using FeCl3 than that by using FeCl2. Figure 10 also shows that vanadium extraction ratio increases with the increase of temperature first and then decreases with the increase of temperature under oxygen atmosphere. By comparing Fig. 9 and Fig. 10, it is found that air atmosphere and oxygen atmosphere have a minor influence on the vanadium extraction ratio; whereas the temperature has a great influence due to the previous result [19].
kJ/mol [15] at 1100 K), therefore elevated temperature is unfavorable to extracting vanadium in terms of thermodynamics and chlorine potential control as mentioned above. However, it is beneficial to extract vanadium at elevated temperatures in terms of kinetics because reaction rate and mass transport are faster at elevated temperatures. Figure 9 shows the coupled results of thermodynamics and kinetics aspects, indicating that the extraction ratio of vanadium is maximum of 32% at 1100 K for 2 h. The chlorination by FeCl2 is similar with the chlorination by FeCl3. The vanadium extraction ratio is higher by using FeCl3 than that by using FeCl2. Figure 10 also shows that vanadium extraction ratio increases with the increase of temperature first and then decreases with the increase of temperature under oxygen atmosphere. By comparing Fig. 9 and Fig. 10, it is found that air atmosphere and oxygen atmosphere have a minor influence on the vanadium extraction ratio; whereas the temperature has a great influence due to the previous result [19].
Table 3 Experimental conditions

Fig. 7 XRD patterns of samples before and after roasting

Fig. 8 Ellingham diagrams of Fe3O4 and V2O3
Fig. 9 Effect of temperature on vanadium extraction ratio under air atmosphere
Fig. 10 Effect of temperature on vanadium extraction ratio under oxygen atmosphere
Figures 11 and 12 show the effects of nchl:noxd on vanadium extraction ratio by FeCl2 and FeCl3, respectively. As shown, vanadium extraction ratio increases with the increase of temperature first and then decreases with the increase of temperature under oxygen atmosphere; vanadium extraction ratio is higher by using FeCl3 than that by using FeCl2; vanadium extraction ratio with nchl:noxd of 2:1 is higher than that with nchl:noxd of 1:1, which indicates that high nchl:noxd is beneficial to extracting vanadium, that is, the higher ratio of nchl:noxd, the higher ratio of vanadium extraction, and it is in a good agreement with thermodynamic analyses.
Fig. 11 Effect of nchl:noxd ratio on vanadium extraction ratio by FeCl2 under oxygen atmosphere

Fig. 12 Effect of nchl:noxd ratio on vanadium extraction ratio by FeCl3 under oxygen atmosphere
6 Conclusions
When the chlorine potential 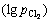 and oxygen potential
and oxygen potential 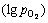 in the system are controlled in certain region, VOCl3 can be generated and extracted from the ore whereas FeOx and TiOx still remain in the ore in terms of thermodynamics over the temperature range of 900-1300 K and the stable region of VOCl3 will shrink gradually with chlorination temperature rising. Therefore, vanadium extraction by selective chlorination using FeClx from vanadium-bearing resources is feasible based on the thermodynamic analyses. Fundamental investigation is conducted in this work. It is indicated that vanadium extraction ratio increases with the increase of temperature first and then decreases with the increase of temperature over the range of 900-1300 K under air or oxygen atmosphere; the higher ratio of nchl:noxd, the higher ratio of vanadium extraction; when the chlorination condition is 1100 K for 2 h with nchl:noxd of 2:1 under oxygen atmosphere, vanadium extraction ratio reaches maximum of 32%.
in the system are controlled in certain region, VOCl3 can be generated and extracted from the ore whereas FeOx and TiOx still remain in the ore in terms of thermodynamics over the temperature range of 900-1300 K and the stable region of VOCl3 will shrink gradually with chlorination temperature rising. Therefore, vanadium extraction by selective chlorination using FeClx from vanadium-bearing resources is feasible based on the thermodynamic analyses. Fundamental investigation is conducted in this work. It is indicated that vanadium extraction ratio increases with the increase of temperature first and then decreases with the increase of temperature over the range of 900-1300 K under air or oxygen atmosphere; the higher ratio of nchl:noxd, the higher ratio of vanadium extraction; when the chlorination condition is 1100 K for 2 h with nchl:noxd of 2:1 under oxygen atmosphere, vanadium extraction ratio reaches maximum of 32%.
References
[1] FU Wen-zhang. Features of vanadium-bearing titanomagnetite and analyses on its comprehensive utilization [J]. Multipurpose Utilization of Mineral Resources, 1996, (1): 27-34. (in Chinese)
[2] MOSKALYK R R, ALFANTAZI A M. Processing of vanadium: A review [J]. Minerals Engineering, 2003, 16: 793-805.
[3] HAO Zi-guo, FEI Hong-cai, LIU Lian, TURNER S. Comprehensive utilization of vanadium-titanium magnetite deposits in China has come to a new level [J]. Acta Geologica Sinica (English Edition), 2012, 87(1): 286-287.
[4] LIU Ke-min. Comprehensive utilization and process flow of Panxi vanadium titanomagnetite [J]. Sichuan Metallurgy, 1990, (1): 3-17. (in Chinese)
[5] DU He-gui. Principle of blast furnace smelting vanadium-bearing titanomagnetite [M]. Beijing, China: Science Press, 1996: 1-16. (in Chinese)
[6] CAPPUYNS V, SWENNEN R. Release of vanadium from oxidized sediments: insights from different extraction and leaching procedures [J]. Environ Sci Pollut Res, 2014, 21: 2272-2282.
[7] LIANG Jing-dong, GAO Ren-wan, QIU Liang-bang, LI Yao, YUE Qing-feng, YANG Zhuang-hua, ZHENG Han-liang. Study on sodium sulfate roasting and oxidizing roasting of concentrate pellets of vanadium-bearing titaniferrous magnetite in rotary kiln followed by the recovery of vanadium from the pellets by water leaching [J]. Mining and Metallurgical Engineering, 1983, 3(2): 35-38, 44. (in Chinese)
[8] GUO Mu-sun. Fluidizing reduction on Panzhihua vanadium-bearing titanomagnetaite concentrate [C]// The Collection of S&T Reports of Panzhihua Resource Comprehensive Utilization―The New Process Flow of Comprehensive Utilization. Panzhihua, 1985: 700-738. (in Chinese)
[9] MA Ji-tang. Direct reduction of vanadium-bearing titanomagnetite pellets containing carbon in coke oven [C]// The Collection of S&T Reports of Panzhihua Resource Comprehensive Utilization―The New Process Flow of Comprehensive Utilization. Panzhihua, 1985: 793-830. (in Chinese)
[10] ZHENG Hai-yan, XUE Xun, WEI Guo, SHEN Feng-man. Thermodynamics analyses on the comprehensive utilization of vanadium-bearing titanomagnetite [C]// Proceedings of the 5th International Congress on the Science and Technology of Ironmaking. Shanghai, 2009: 1208-1210.
[11] AARABI-KARASGANI M, RASHCHI F, MOSTOUFI N, VAHIDI E. Leaching of vanadium from LD converter slag using sulfuric acid [J]. Hydrometallurgy, 2010, 102(1-4): 14-21.
[12] FU Wei-guo, WEN Yong-cai, XIE Hong-en. Development of intensified technologies of vanadium-bearing titanomagnetite smelting [J]. Journal of Iron and Steel Research, International, 2011, 18(4): 7-10, 18.
[13] LIU Gong-guo. Study on applied technology of vanadium titanium magnetite based on rotary hearth furnace direct reduction process [J]. Research on Iron and Steel, 2012, 40(2): 4-7. (in Chinese)
[14] MA Jia-yuan. Smelting of vanadium-bearing titanomagnetite in BF process [M]. Beijing, China: Metallurgical Industry Press, 2001. (in Chinese)
[15] DENG Jun, XUE Xun, LIU Gong-Guo. Current situation and development of comprehensive utilization of vanadium-bearing titanomagnetite at Pan Gang [J]. Journal of Materials and Metallurgy, 2007, 6(2): 83-86, 93. (in Chinese)
[16] FUKUYAMA T, KOIZUMI M, HANAKI M, KOSEMURA S. Production of titanium sponge and ingot at Toho Titanium Co. Ltd. [J]. J Mining and Mater Processing Inst of Japan, 1993, 109: 1157-1163.
[17] MORIYA A, KANAI A. Titanium sponge production at sumitomo Sitix Xorporation [J]. J Mining and Mater Processing Inst of Japan, 1993, 109: 1164-1169.
[18] BARIN. Thermochemical data of pure substances [M]. 3rd ed. Weinheim: VCH Verlagsgesellschaft mbH, 1997.
[19] ZHENG Hai-yan, SUN Yu, DONG Yue, SHEN Feng-man. Fundamental study on vanadium extraction from vanadium-bearing titanomagnetite by selective chlorination using chloride wastes [C]// Proceedings of Metallurgical Reaction Engineering in China. Guiyang, China, 2012: 190-195. (in Chinese)
(Edited by FANG Jing-hua)
Cite this article as: ZHENG Hai-yan, SUN Yu, LU Jin-wen, DONG Jian-hong, ZHANG Wei-ling, SHEN Feng-man. Vanadium extraction from vanadium-bearing titanomagnetite by selective chlorination using chloride wastes (FeClx) [J]. Journal of Central South University, 2017, 24(2): 311-317. DOI: 10.1007/s11171-017-3432-5.
Foundation item: Projects(51374061, 51074040) supported by the National Natural Science Foundation of China; Project(201202064) supported by the Natural Science Foundation of Liaoning Province, China; Project(N120402004) supported by the Fundamental Research Funds for the Central Universities, China
Received date: 2015-01-12; Accepted date: 2015-12-10
Corresponding author: ZHENG Hai-yan, PhD, Associate Professor; Tel: +86-24-83691266, E-mail: zhenghy@smm.neu.edu.cn

