


Trans. Nonferrous Met. Soc. China 22(2012) 2459-2464
Synthesis and photoluminescence properties of single-crystal ZnO hexagonal pyramids by PEG400-assisted thermal decomposition route
LIU Jin-song1,2, ZHANG Shuo1, LI Zi-quan1, ZHU Kong-jun2, CHEN Jian-kang1,
QIU Jin-hao2, WANG Chun-hua3, GAO Xue-qin3, WANG Li-ping3
1. College of Material Science and Technology, Nanjing University of Aeronautics and Astronautics,Nanjing 210016, China;
2. State Key Laboratory of Mechanics and Control of Mechanical Structures,
Nanjing University of Aeronautics and Astronautics, Nanjing 210016, China;
3. Wuxi Hengye Electrical Heater Equipment Co. Ltd., Wuxi 214161, China
Received 9 July 2012; accepted 6 August 2012
Abstract: Large-scale synthesis of ZnO hexagonal pyramids was achieved by a simple thermal decomposition route of precursor at 240 oC in the presence of PEG400. The precursor was obtained by room-temperature solid-state grinding reaction between Zn(CH3COO)2・2H2O and Na2CO3. Crystal structure and morphology of the products were analyzed and characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM) and high-resolution transmission electron microscopy (HRTEM). The results of further experiments show that PEG400 has an important role in the formation of ZnO hexagonal pyramids. Difference between the single and double hexagonal pyramid structure may come from the special thermal decomposition reaction. The photoluminescence (PL) spectra of ZnO hexagonal pyramids exhibit strong near-band-edge emission at about 386 nm and weak green emission at about 550 nm. The Raman-active vibration at about 435 cm-1 suggests that the ZnO hexagonal pyramids have high crystallinity.
Key words: ZnO hexagonal pyramids; thermal decomposition route; formation mechanism; photoluminescence property
1 Introduction
As known, the shape and size of nanomaterials tremendously affect their physical and chemical properties. Zinc oxide is an important wide-band-gap semiconductor, and has a variety of applications including varistors, transparent conductors, transparent UV-protection films and chemical sensors [1,2]. To date, well-defined ZnO with different morphologies have been successfully synthesized by various methods [3-8]. However, it seems that either complex process control or long synthesis time might be required for these routes.
Solid-state synthesis is an effective approach to obtain nanoparticles, and it exhibits many advantages such as solvent-free, simplicity and low cost, which has been extensively used in the synthesis of many nanomaterials [9]. To obtain other nanomaterials with different morphologies, surfactant-assisting or microwave- assisting solid-state method has been developed [10-12], which extends its application in the field of synthesizing nanomaterials. ZnO hexagonal pyramids are special structures with different shapes and exposed surfaces, and it is not reported to synthesize through solid state thermal decomposition reactions.
In this work, a novel synthetic method of hexagonal pyramid ZnO nanostructures was developed and the surfactant was introduced into solid-state thermal decomposition route. Hexagonal pyramid ZnO was synthesized by simple thermal decomposition route at about 240 ℃ in the presence of PEG400. The synthesized products were single-crystalline ZnO hexagonal pyramid. The effect of various factors on the products was studied, and the photoluminescence properties of the products were investigated.
2 Experimental
All the chemical reagents used in the experiment were of analytical grade. The detailed synthetic process was as follows. In a typical synthesis, 0.005 mol hydrated zinc acetate [Zn(CH3COO)2・2H2O] was ground for 5 min before mixing with 2.0 mL polyethylene glycol (PEG400). Afterward, 0.005 mol sodium carbonate (Na2CO3) was added to the mixture. After 20 min of grinding, the mixture was washed with distilled water to remove any by-product, and then filtrated. It was transferred to a 100 mL Teflon container with 40 mL PEG400. The mixture was then heated at 240 ℃ for 1 h. The product was washed and dried in air.
The X-ray diffraction (XRD) analysis was carried out on a Bruker D8-Advance X-ray diffractometer with Cu Kα radiation. The transmission electron microscopy (TEM) images were taken using an FEI Tecnai-20 transmission electron microscope. The scanning electron microscopy (SEM) images were taken with an LEO1530VP field emission scanning electron microscope. The Raman spectra were recorded on an HR-800 spectrometer. The photoluminescence (PL) spectra were recorded on a Varian Cary-Eclipse spectrometer.
3 Results and discussion
3.1 XRD pattern analysis
The crystallinity and phases of the product were confirmed by X-ray diffraction. Figure 1 shows the XRD pattern of the final product obtained by the thermal decomposition route at 240 ℃ for 1 h in the presence of PEG400. In contrast to the standard diffraction peaks (JCPDS card No. 36-1451, a=0.3249 nm, c=0.5206 nm), the pronounced ZnO peaks for (100), (002), (101), (102), (110), (103), (200), (112), (201), (004) and (202) appear at 2θ=31.8, 34.5, 36.3, 47.6, 56.6, 62.9, 66.3, 68.0, 69.2, 72.5 and 76.9°, respectively. These results indicate that the ZnO obtained has a hexagonal wurtzite structure with high crystallinity. In addition, it can be seen that only ZnO diffraction peaks are found, and the characteristic peaks of other impurities such as Zn(OH)2 are not observed in the figure.
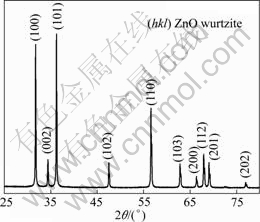
Fig. 1 XRD pattern of ZnO nanostructure
3.2 SEM and TEM analysis
The typical SEM images of as-obtained wurtzite ZnO nanostructure are shown in Fig. 2, as well as the TEM images and SAED pattern for detail illustration. Figure 2(a) is a low-magnification SEM image that shows the high yield of the hexagonal pyramid ZnO. The product consists of different morphologies with single and double hexagonal pyramids. The high-magnification SEM image (Fig. 2(b)) shows the hexagonal bottom and pyramid-like surface of the ZnO. The bottom has a hexagonal structure and the side length is about 500 nm. Figures 2(c) and (e) present the typical TEM images of the ZnO nanostructure. The product has a hexagonal pyramid shape which is in good agreement with the SEM results. The electron diffraction patterns shown in Figs. 2(d) and (f) indicate that the product has a single- crystal structure.
High-resolution TEM (HRTEM) could provide the structural details of the products. Figure 3 reveals the HRTEM image of the ZnO nanostructure. An interplanar spacing of the crystalline stripes is about 0.261 nm, corresponding to the separation between the (002) planes of ZnO. It indicates that the growing plane and the growing direction of the ZnO are along the (002) plane and the [002] direction, respectively.
3.3 Further experiments and analysis
To explore the formation mechanism of the ZnO hexagonal pyramid, further experiments were designed and more information was obtained. Figure 4(a) shows the XRD pattern of the precursor. Based on the comparison with the JCPDS cards, the diffraction peaks of the precursor are consistent with Zn4CO3(OH)6・H2O. The product obtained is not ZnO but Zn4CO3(OH)6・H2O when the reaction temperature is 200 ℃ in the presence of PEG400 (Fig. 4(b)). This shows that the precursor does not decompose to ZnO at 200 ℃. Only ZnO nanoparticles are required by heating the precursor at 240 ℃ in the absence of PEG400 (Fig. 4(c)). The broadness of the diffraction peaks indicates that the dimensions of the ZnO nanoparticles are very small. The hexagonal pyramid ZnO products can be obtained at 240 ℃ in the presence of PEG400 for different durations, namely, 0.5, 2 and 4 h, respectively (Figs. 4(d)-(f)).

Fig. 2 SEM images (a, b), TEM images (c, e) and corresponding SAED patterns(d, f) of ZnO nanostructure

Fig. 3 HRTEM image of ZnO nanostructure
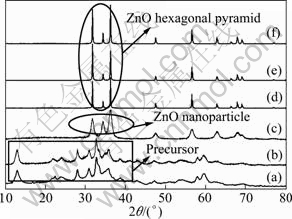
Fig. 4 XRD patterns of precursor (a), products obtained at 200 ℃ in the presence of PEG400 (b), at 240 ℃ in the absence of PEG400 (c), at 240 ℃ in presence of PEG400 for 0.5 h (d), 2 h (e) and 4 h (f)
The SEM image of the product obtained at 240 ℃ without PEG400 for 4 h (Fig. 5(a)) shows that the typical morphology of the products consists of small spheres. The SEM images of the ZnO products (Figs. 5(b)-(d)) obtained by heating for 0.5, 2 and 4 h in the presence of PEG400 show hexagonal pyramid ZnO nanostructures instead of spherical particles typical of the common thermal decomposition reaction [13].
3.4 Growth mechanism
Based on the above experiments, a possible growth mechanism was proposed. Two steps in the formation process of ZnO hexagonal pyramid nanostructure were employed, as shown in Fig. 6. First, the solid-state reaction between Zn(CH3COO)2・2H2O and Na2CO3 can occur in the process of grinding, and then the precursor nanoparticles (Zn4CO3(OH)6・H2O) form. PEG400 is a uniform and ordered chain structure and it is easily adsorbed on the surface of reactant, and it will modify the growth kinetics of the growing colloids, which finally leads to anisotropic growth of the crystals. When the precursor is combined with PEG400, the surface of the precursor colloid adsorbs PEG400, which greatly decreases the activities of the colloid [14]. This confines the growth rate to some directions [15]. Therefore, in the presence of PEG400, the precursor is decomposed and ZnO hexagonal pyramids form. Difference between the single and double hexagonal pyramids may come from the special thermal decomposition reaction in the presence of PEG400. When the suitable lattice plane between the particles is close enough to each other [16], the simultaneous growth starts during thermal decomposition, which is responsible for the formation of the double hexagonal pyramid.

Fig. 5 SEM images of products obtained at 240 ℃ without PEG400 for 4 h (a), in the presence of PEG400 heated for 0.5 h (b), 2 h (c) and 4 h (d)
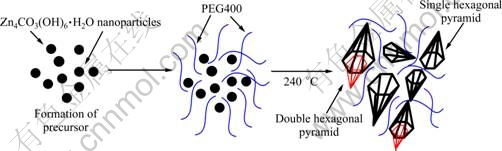
Fig. 6 Schematic illustration of formation mechanism of ZnO hexagonal pyramid
3.5 PL spectra analysis
The corresponding PL spectra of the products are shown in Fig. 7(a). All the spectra have a relatively sharp ultraviolet emission centered at about 386 nm. The strong near-band-edge emission at 386 nm is similar to the ZnO bulk band gap at 380 nm which originates from the recombination of free excitons [17,18]. The weak green emission band at about 550 nm is attributed to a singly charged oxygen vacancy, which results from the recombination of a photo-generated hole with a charge state of the specific defect, or from the surface deep traps [19,20]. The results illustrate that ZnO hexagonal pyramids possess high crystallinity and few oxygen vacancies. The PL emission intensities of the products increase with increasing reaction time at 240 ℃, which suggests that the optical property of the products could be tuned by the reaction parameter. LI et al [15] reported the similar results of PL emission coming from different morphologies of ZnO. To further understand the crystalline structure, the product obtained at 240 ℃ heated for 1 h in the presence of PEG400 was characterized by Raman spectroscopy (Fig. 6(b)). The Raman-active vibration of the product was detected at about 435 cm-1, which is characteristic of the high-frequency E2 mode of the wurtzite structure [21]. The strong E2 peak centered at about 435 cm-1 suggests that the ZnO nanostructures are of good crystalline, which is consistent with the PL observation.
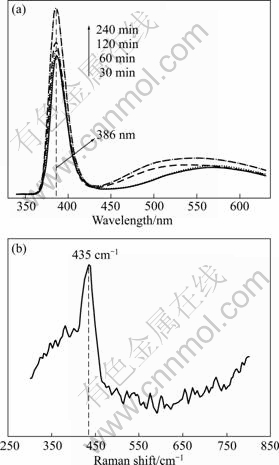
Fig. 7 PL (a) and Raman spectra (b) of product obtained at 240 ℃ for 1 h in the presence of PEG400
4 Conclusions
1) A simple PEG400-assisted thermal decompo- sition route of the precursor to synthesize ZnO hexagonal pyramids is successfully developed. The precursor Zn4CO3(OH)6・H2O nanoparticle is obtained by room-temperature solid-state grinding reaction between Zn(CH3COO)2・2H2O and Na2CO3.
2) The results of XRD, SEM, TEM, SAED and HRTEM characterizations indicate that the product is hexagonal wurtzite ZnO with hexagonal pyramid morphology.
3) Further experiments show that PEG400 has an important role in the formation of ZnO hexagonal pyramids. Difference between the single and double hexagonal pyramids comes from the special thermal decomposition reaction.
4) The PL spectra exhibit strong near-band-edge emission at about 386 nm and weak green emission at about 550 nm, and their intensities could be tuned by the reaction time. The Raman-active vibration at about 435 cm-1 suggests that the ZnO hexagonal pyramid has high crystallinity.
References
[1] WU J, XIE C S, BAI Z K, ZHU B L, HUANG K J, WU R. Preparation of ZnO-glass varistor from tetrapod ZnO nanopowders [J]. Mater Sci Eng B, 2002, 95: 157-161.
[2] CAO H, XU J Y, ZHANG D Z, CHANG S H, HO S T, SEELIG E W, LIU X, CHANG R P H. Spatial confinement of laser light in active random media [J]. Phys Rev Lett, 2000, 84: 5584-5587.
[3] DONG Lin, JIA Xiao-lin, TONG Yan-hong, MA Ying, LIU Jing-wei. Growth of ZnO nanorod arrays on soft substrates by hydrothermal process [J]. Transactions of Nonferrous Metals Society of China, 2007, 17(s1): s721-s724.
[4] HAMADA T, FUJII E, CHU D W, KATO K, MASUDA Y. Aqueous synthesis of single-crystalline ZnO prisms on graphite substrates [J]. J Cryst Growth, 2011, 314: 180-184.
[5] SHI Wen-tao, GAO Guo, XIANG Lan. Synthesis of ZnO whiskers via hydrothermal decomposition route [J]. Transactions of Nonferrous Metals Society of China, 2010, 20(6): 1049-1052.
[6] LI Y, LIU C S. Hydro/solvo-thermal synthesis of ZnO crystallite with particular morphology [J]. Transactions of Nonferrous Metals Society of China, 2009, 19(2): 399-403.
[7] WANG Yi-ming, LI Jian-hua, HONG Ruo-yu. Large scale synthesis of ZnO nanoparticles via homogeneous precipitation [J]. Journal of Central South University, 2012, 19(4): 863-868.
[8] LI Yan, FENG Hui-yun, ZHANG Nan, LIU Chun-sheng. Solvo-thermal synthesis and characterization of nest-like zinc oxide [J]. Transactions of Nonferrous Metals Society of China, 2010, 20(1): 119-122.
[9] ZHOU Yi-ming, XIN Xin-quan. Synthetic chemistry for solid state reaction at low-heating temperatures [J]. Chinese J Inorg Chem, 1999, 19(3): 273-292. (in Chinese)
[10] LIU J S, CAO J M, LI Z Q, KE X F. Micorwave-assisted solid-state synthesis of ZnO nanorods [J]. Acta Chimica Sinica, 2007, 65: 1476-1480.
[11] LIU J S, CAO J M, LI Z Q, JI G B, DENG S G, ZHENG M B. Low-temperature solid-state synthesis and phase-controlling studies of CdS nanoparticles [J]. J Mater Sci, 2007, 42: 1054-1059.
[12] WANG W Z, ZHAN Y J, WANG G H. One-step, solid-state reaction to the synthesis of copper oxide nanorods in the presence of a suitable surfactant [J]. Chem Commun, 2001(8): 727-728.
[13] WANG Z J, ZHANG H M, ZHANG L G, YUAN J S, YAN S G, WANG C Y. Low-temperature synthesis of ZnO nanoparticles by solid-state pyrolytic reaction [J]. Nanotechnology, 2003, 14: 11-15.
[14] DOBRYSZYCKI J, BIALLOZOR S. On some organic inhibitors of zinc corrosion in alkaline media [J]. Corros Sci, 2001, 43: 1309-1319.
[15] LI Z Q, XIONG Y J, XIE Y. Selected-control synthesis of ZnO nanowires and nanorods via a PEG-assisted route [J]. Inorg Chem, 2003, 42: 8105-8109.
[16] JIE J S, WANG G Z, HAN X H, HOU J G. Synthesis and characterization of ZnO: In nanowires with superlattice structure [J]. J Phys Chem B, 2004, 108: 17027-17031.
[17] WONG E M, SEARSON P C. ZnO quantum particle thin films fabricated by electrophoretic deposition [J]. Appl Phys Lett, 1999, 74: 2939-2941.
[18] MONTICONE S, TUFEU R, KANAEV A V. Complex nature of the UV and visible fluorescence of Colloidal ZnO nanoparticles [J]. J Phys Chem B, 1998, 102: 2854-2862.
[19] LI Y, CHENG G S, ZHANG L D. Fabrication of highly ordered ZnO nanowire arrays in anodic alumina membranes [J]. J Mater Res, 2005, 15: 2305-2308.
[20] HU J Q, LI Q, WONG N B, LEE C S, LEE S T. Synthesis of uniform hexagonal prismatic ZnO whiskers [J]. Chem Mater, 2002, 14: 1216-1219.
[21] YAN J, FANG X S, ZHANG L D, BANDO Y, GAUTAM U K, DIERRE B, SEKIGUCHI T, GOLBERG D. Structure and cathodoluminescence of individual ZnS/ZnO biaxial nanobelt heterostructures [J]. Nano Lett, 2008, 8: 2794-2799.
单晶ZnO六棱锥的PEG400辅助热分解合成及其光致发光性能
刘劲松1,2,张 朔1,李子全1,朱孔军2,陈建康1,裘进浩2,王春花3,高雪琴3,王莉萍3
1. 南京航空航天大学 材料科学及技术学院,南京 210016;
2. 南京航空航天大学 机械结构力学及控制国家重点实验,南京 210016;
3. 无锡恒业电热电器有限公司,无锡 214161
摘 要:将由Zn(CH3COO)2・2H2O和Na2CO3通过室温研磨反应获得的前驱体在PEG400存在下于240 ℃热分解获得大量的ZnO六棱锥产物。用X射线衍射仪(XRD)、扫描电子显微镜(SEM)、透射电子显微镜(TEM)和高分辨透射电子显微镜(HRTEM)表征产物的晶体结构和形貌。进一步的实验结果表明:PEG400在ZnO六棱锥形成过程中发挥着重要作用,单六棱锥和双六棱锥的结构差异来自于热分解反应。光致发光谱(PL)测试表明:ZnO六棱锥在386 nm处展示强的近带隙发射,在550 nm处展示较弱的绿光发射。435 cm-1处的拉曼振动表明ZnO六棱锥具有良好的晶体质量。
关键词:ZnO六棱锥;热分解路线;形成机理;光致发光性能
(Edited by FANG Jing-hua)
Foundation item: Project (BK2009379) supported by the Natural Science Foundation of Jiangsu Province, China; Project (1006-56XNA12069) supported by the Nanjing University of Aeronautics and Astronautics Research Funding, China; Projects (51172108, 91023020) supported by the National Natural Science Foundation of China; Project (IRT0968) supported by the Program for Changjiang Scholars and Innovative Research Team in University, China; Project (NCET-10-0070) supported by the Program for New Century Excellent Talents in University, China; Project supported by the Priority Academic Program Development of Jiangsu Higher Education Institutions, China
Corresponding author: LIU Jin-song; Tel/Fax: +86-25-84895871; E-mail: jsliu@nuaa.edu.cn
DOI: 10.1016/S1003-6326(11)61485-4