Ψυ‘»Μ·¥ΠάμΕ‘DC÷ΐ‘λ7X50¬ΝΚœΫπœ‘ΈΔΉι÷·ΚΆΝΠ―ß–‘ΡήΒΡ”Αœλ
ά¥‘¥ΤΎΩ·ΘΚ÷–Ιζ”–…ΪΫπ τ―ß±®2015ΡξΒΎ4ΤΎ
¬έΈΡΉς’ΏΘΚ¥‘ΗΘΙΌ ’‘ Η’ ΫΣ Ζφ Χο ξ« άν»πΖε
ΈΡ’¬“≥¬κΘΚ1027 - 1034
Key wordsΘΚ7X50 aluminum alloy; microstructural evolution; homogenization; residual phase; recrystallization
’Σ “ΣΘΚ≤…”ΟSEMΓΔTEMΓΔEDSΓΔDSCΓΔXRDΚΆά≠…λ Β―ι―–ΨΩ÷ΐΧ§7X50ΚœΫπΦΑΤδΨυ‘»Μ·¥ΠάμΙΐ≥ΧΒΡΉι÷·―ί±δΓΘΫαΙϊ±μΟςΘ§÷ΐΧ§7X50ΚœΫπœύΉι≥…÷ς“Σ”–S(Al2CuMg)ΓΔT(Al2Mg3Zn3)ΓΔMgZn2ΚΆ…ΌΝΩΒΡAl7Cu2FeΚΆAl3ZrœύΓΘΨυ‘»Μ·¥ΠάμΙΐ≥Χ÷–÷ΠΨßΆχΚΆ≤–Ντœύ÷πΫΞΦθ…ΌΘ§Ψ≠(470 ΓψCΘ§24 h)+(482 ΓψCΘ§12 h)Ψυ‘»Μ·¥Πάμ ±Θ§Tœύœϊ ßΘ§Sœύ”–ΈΔΝΩ≤–ΝτΘ§Al7Cu2FeœύΦΗΚθΟΜ”–±δΜ·ΓΘ÷ΐΧ§ΚœΫπΒΡDSC«ζœΏ÷–‘Ύ477.8 ΓψC¥Π”–“ΜΫœ«ΩΈϋ»»ΖεΘ§Ψ≠470 ΓψCΓΔ1 hΨυ‘»Μ·ΚσΚœΫπΒΡDSC«ζœΏ‘Ύ487.5 ΓψC¥Π≥ωœ÷“ΜΗω–¬ΒΡΈϋ»»ΖεΘ§ΕχΨ≠482 ΓψCΓΔ24 hΨυ‘»Μ·¥ΠάμΚσΚœΫπ‘Ύ487.5 ΓψC¥ΠΒΡΈϋ»»ΖεΜυ±Ψœϊ ßΓΘ‘ΎXRDΤΉ÷–Έ¥≥ωœ÷T(Al2Mg3Zn3)œύΘ§’βΚΆTœύ”κS(Al2CuMg)ΦΑMgZn2œύœύΙΊΒΡΫα¬έœύΈ«ΚœΓΘ‘ΛΨυ‘»Μ·¥Πάμ÷Τ±ΗΒΡΑε≤Ρ÷–‘ΌΫαΨßΨßΝΘΖ÷ ΐΟςœ‘ΫΒΒΆΘ§ΩΙά≠«ΩΕ»ΚΆΕœΝ―»Ά–‘œύΕ‘≥ΘΙφΨυ‘»Μ·¥Πάμ÷Τ±ΗΒΡΑε≤ΡΖ÷±πΧαΗΏ‘Φ15 MPaΚΆ3.3 MPaΓΛm1/2ΓΘ
Abstract: The evolution of the eutectic structures in the as-cast and homogenized 7X50 aluminum alloys was studied by scanning electron microscopy (SEM), transmission electron microscopy (TEM), energy dispersive spectrometer (EDS), differential scanning calorimetry(DSC), X-ray diffraction(XRD) and tensile test. The results show that the main phases are S(Al2CuMg), T(Al2Mg3Zn3) and MgZn2, with a small amount of Al7Cu2Fe and Al3Zr in the as-cast 7X50 alloy. The volume fraction of the dendritic-network structure and residual phase decreases gradually during the homogenization. After homogenization at 470 ΓψC for 24 h and then 482 ΓψC for 12 h, the T(Al2Mg3Zn3) phase disappears and minimal S(Al2CuMg) phase remains, while almost no change has happened for Al7Cu2Fe. There is a strong endothermic peak at 477.8 ΓψC in the DSC curve of as-cast alloy. A new endothermic peak appears at 487.5 ΓψC for the sample homogenized at 470 ΓψC for 1 h. However, this endothermic peak disappears after being homogenized at 482 ΓψC for 24 h. The T(Al2Mg3Zn3) phase cannot be observed by XRD, which is consistent with that T phase is the associated one of S(Al2CuMg) phase and MgZn2 phase. The volume fraction of recrystallized grains is substantially less in the plate with pre-homogenization treatment. The strength and fracture toughness of the plate with pre-homogenization treatment are about 15 MPa and 3.3 MPaΓΛm1/2 higher than those of the material with conventional homogenization treatment.
Trans. Nonferrous Met. Soc. China 25(2015) 1027-1034
Fu-guan CONG1,2, Gang ZHAO1, Feng JIANG3, Ni TIAN1, Rui-feng LI1
1. Key Laboratory for Anisotropy and Texture of Materials, Ministry of Education, Northeastern University, Shenyang 110004, China;
2. Northeast Light Alloy Co., Ltd., Harbin 150060, China;
3. School of Materials Science and Engineering, Central South University, Changsha 410083, China
Received 5 May 2014; accepted 20 October 2014
Abstract: The evolution of the eutectic structures in the as-cast and homogenized 7X50 aluminum alloys was studied by scanning electron microscopy (SEM), transmission electron microscopy (TEM), energy dispersive spectrometer (EDS), differential scanning calorimetry(DSC), X-ray diffraction(XRD) and tensile test. The results show that the main phases are S(Al2CuMg), T(Al2Mg3Zn3) and MgZn2, with a small amount of Al7Cu2Fe and Al3Zr in the as-cast 7X50 alloy. The volume fraction of the dendritic-network structure and residual phase decreases gradually during the homogenization. After homogenization at 470 ΓψC for 24 h and then 482 ΓψC for 12 h, the T(Al2Mg3Zn3) phase disappears and minimal S(Al2CuMg) phase remains, while almost no change has happened for Al7Cu2Fe. There is a strong endothermic peak at 477.8 ΓψC in the DSC curve of as-cast alloy. A new endothermic peak appears at 487.5 ΓψC for the sample homogenized at 470 ΓψC for 1 h. However, this endothermic peak disappears after being homogenized at 482 ΓψC for 24 h. The T(Al2Mg3Zn3) phase cannot be observed by XRD, which is consistent with that T phase is the associated one of S(Al2CuMg) phase and MgZn2 phase. The volume fraction of recrystallized grains is substantially less in the plate with pre-homogenization treatment. The strength and fracture toughness of the plate with pre-homogenization treatment are about 15 MPa and 3.3 MPaΓΛm1/2 higher than those of the material with conventional homogenization treatment.
Key words: 7X50 aluminum alloy; microstructural evolution; homogenization; residual phase; recrystallization
1 Introduction
The mechanical properties of aluminum alloys depend largely on the alloying elements and their presence in the solid solution. During the solidification of aluminum alloys, a large portion of alloying elements may segregate into liquid and form coarse particles in the grain boundary (GB) regions or inside the grains. The chemical composition, casting and homogenization processes will influence the volume fraction, morphology, and intrinsic characteristics of these coarse particles [1-3]. These particles can deteriorate the hot workability especially when they are in the GB regions, and limit the range of applicable process parameters during subsequent hot deformation [4]. Usually, the residual coarse particles (>1 ΠΧm) such as Fe-rich or Cu-rich phases will deteriorate the toughness and fatigue properties [1,4-8]. The microstructures of the as-cast and homogenized Al-Zn-Mg-Cu alloys have been studied in recent years. Most of the investigations are focused on the nature, evolution and distribution of Al2Mg3Zn3 (T), Al2CuMg (S), MgZn2 (Π«), (AlCuZn)2Mg, Al2Cu (Π»), Al7Cu2Fe and Al13Fe4 phases during homogenization [9-21]. ZHAO et al [21] and RANGANATHA et al [22] studied the effects of homogenization treatment on the microstructural changes of AA7075 and 7049 alloys. LIM et al [23] studied the effects of constitutional changes and preheating conditions on the evolution of constituent particles, the M, T, and S phases, and dispersoids in AA7175 and AA7050 alloys. LI and STARINK [9] and FAN et al [10] studied the evolution of microstructure in an Al-Zn-Mg-Cu alloy during homogenization. 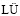 et al [11] and MONDAL and MUKHOPADHYAY [20] studied the phases in the as-cast and homogenized 7055 aluminum alloy, and revealed that the major residual phases were Π«(MgZn2), T(Al2Mg3Zn3), S(Al2CuMg) and Π»(Al2Cu). The microstructure of the as-cast 7050 alloy consists of dendrites, high angle grain boundaries, and inter- dendritic eutectic regions containing phases such as Al2CuMg, MgZn2 and Mg2Si [14,15]. ROKHLIN et al [16] and LI and STARINK [17] studied the effects of Zr, Sc and Cr on microstructures and intermetallic phases in Al-Zn-Mg-Cu aluminium alloys.
et al [11] and MONDAL and MUKHOPADHYAY [20] studied the phases in the as-cast and homogenized 7055 aluminum alloy, and revealed that the major residual phases were Π«(MgZn2), T(Al2Mg3Zn3), S(Al2CuMg) and Π»(Al2Cu). The microstructure of the as-cast 7050 alloy consists of dendrites, high angle grain boundaries, and inter- dendritic eutectic regions containing phases such as Al2CuMg, MgZn2 and Mg2Si [14,15]. ROKHLIN et al [16] and LI and STARINK [17] studied the effects of Zr, Sc and Cr on microstructures and intermetallic phases in Al-Zn-Mg-Cu aluminium alloys.
Although some researches have been reported on the microstructure in as-cast condition and the evolution of the eutectic phases during homogenization in the case of Al-Zn-Mg-Cu series alloys, the information on the 7X50 aluminum alloys is still rarely seen in literature. Moreover, there is no comparative study on homogenized microstructure. The objectives of this work were to determine the effect of homogenization treatment on the microstructural evolution. The transformation from eutectic structures to coarse particles was also investigated.
2 Experimental
Cubic samples with dimensions of 20 mm ΓΝ 20 mm ΓΝ 20 mm were cut from the center of a semi- continuous casting AA7X50 ingot with the dimensions of 520 mm ΓΝ 1600 mm. The chemical composition of this alloy is shown in Table 1. Single-stage homogenization treatment was performed at 470 ΓψC for 1, 8, 24, 72, and 240 h, respectively. Double-stage homogenization treatment was performed at 470 ΓψC for 24 h and then at 482 ΓψC for 1, 2, 6, 24, and 72 h, respectively. Pre-homogenization treatment was performed at 400 ΓψC for 10 h and then at 470 ΓψC for 24 h. The homogenized samples were machined with dimensions of 100 mm ΓΝ 250 mm ΓΝ 300 mm. The blocks were rolled with seven passes up to 30 mm in thickness after being reheated at 380 ΓψC for 4 h. Solution treatment of hot-rolled plates was carried out at 480 ΓψC for 1 h and the samples were quenched immediately in water.
Table 1 Chemical composition of 7X50 aluminium alloy (mass fraction, %)
The microstructures of different homogenized samples etched using KellerΓ·s etchant (2 mL HF + 3 mL HCL + 5 mL HNO3 + 190 mL H2O) were analyzed by an Olympus GX71 optical microscope(OM) and a Hitachi S-4700 scanning electron microscope(SEM). A Finder 1000 energy dispersive spectrometer (EDS) was used for analyzing the constituents. The melting temperatures of the eutectic phases in different homogenized samples were determined by a Netzsch STA 449C differential scanning calorimetry (DSC). An X'Pert MPD diffractometer (XRD) was used to identify the phases.
All the samples under different homogenization conditions were ground, polished and etched by BarkerΓ·s etchant for optical microscope observation. The OM images were analyzed using the imaging software. Three samples in the same condition were prepared, and analysis was performed on the three images. The SEM images of the samples after different homogenization treatments were analyzed to investigate the dissolution of the eutectic phases during homogenization. The SEM was operated at 20 kV, and the samples were prepared as the same as that of OM. The DSC experiments were performed with high purity aluminium as a reference. 80-100 mg specimens were prepared using an electro-discharge machine. The heating rate used in the DSC was 10 ΓψC/min. X-ray diffraction (XRD) measurements were performed using a Rigaku D/max-rB X-ray diffractometer with Cu KΠΝ radiation. The scanning from 2Π»=10Γψ to 100Γψ was performed to record the XRD pattern.
3 Results and discussion
3.1 As-cast microstructure
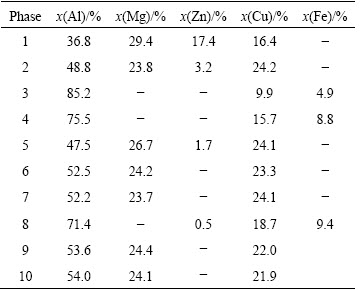
Lower and higher magnification SEM images of the microstructure of the as-cast 7X50 aluminum alloy are shown in Fig. 1. The constitutive eutectic phases elongated along the GB can be clearly seen. Different types of eutectic phases are illustrated with arrows in Fig. 1(b). The results of the image analysis indicate that the average spacing of the second dendrite arm is 140 ΠΧm, the average width of these eutectic phases is about 2.5 ΠΧm and the volume fraction of the eutectic phases is close to 9.2%. The lamellar eutectic structure is shown in Fig. 1(b), and the average width of the lamellas is about 0.8 ΠΧm. In order to determine the compositions of different eutectic phases, EDS analysis was performed on the phases with the same morphology. The results show that most of the eutectic phases have the same composition, and the results of EDS analysis of different morphology phases illustrated with arrows in Fig. 1(b) are listed in Table 2. In the as-cast alloy, the gray phase of the fine particles in Al matrix is MgZn2. The deep gray needle-like phase contains Cu and Fe, which is close to Al7Cu2Fe phase in composition (spot A in Fig. 1(b)), while the gray bone-like phase is T(Al2Mg3Zn3) phase (spot B and in Fig. 1(b)), and the lamellar eutectic phase is S(Al2CuMg) phase (spot C in Fig. 1(b)). The gray eutectic phase is also considered to be T(Al2Mg3Zn3) phase with solute of Cu [10-14,24].

Fig. 1 SEM micrographs of as-cast alloy
Table 2 Chemical compositions of secondary phases in Fig. 1(b)

3.2 Microstructure of homogenized alloys
Figure 2 shows the TEM images of the alloy after different homogenization treatments. The spherical white dispersoids in Fig. 2 are metastable Al3Zr. TEM images in Fig. 2 show that a relatively large number and high density of Al3Zr dispersoids (15-30 nm in diameter) can be observed in the samples homogenized at 400 ΓψC for 10 h and then at 470 ΓψC for 24 h. However, when the samples are homogenized at 470 ΓψC for 24 h, Al3Zr dispersoids are 20-50 nm in diameter. It was reported that the nucleation rate for the formation of Al3Zr dispersoids is dependent on the Zr content and temperature [25,26]. The rates of maximum nucleation and growth increase with increasing Zr content and homogenization temperature. Furthermore, it is suggested that at certain Zr content, a high homogenization temperature is required to obtain big Al3Zr dispersoids and high growth rate. In this work, with 0.10% Zr, the results suggest that homogenization at 400 ΓψC results in both high density and dispersive Al3Zr phase.

Fig. 2 TEM images showing Al3Zr dispersoids after being homogenized at 400 ΓψC for 10 h and then at 470 ΓψC for 24 h (a) and at 470 ΓψC for 24 h (b)
The evolution of eutectic structures during homogenization was investigated. Figure 3 shows the microstructures of specimens homogenized at 470 ΓψC for different time, and at 470 ΓψC for 24 h and then at 482 ΓψC for different time. The EDS analysis results of different morphology phases after different homogenization treatments are listed in Table 3.
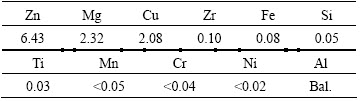
Figure 3 shows that the volume fraction of the dendritic-network structure decreases gradually, and the residual phases become smaller and sparser with increasing the homogenization time. The non- equilibrium phases at the grain boundaries also dissolve gradually with increasing the homogenization time. It can be seen from Fig. 3(a) and Table 3 that the area fraction of the eutectic phases decreases to 5.6% and the residual eutectic phases are mainly T(Al2Mg3Zn3), S(Al2CuMg) and Al7Cu2Fe. When the time increases to 24 h at 470 ΓψC, it can be observed from Fig. 3(b) and Table 3 that the T(Al2Mg3Zn3) phase disappears, and the S(Al2CuMg) phase decreases significantly and the dissolution of Al7Cu2Fe is not obvious. It can be seen from Figs. 3(b) and (c) that the S(Al2CuMg) phase is increasingly dissolved with time going on. The microstructure has no significant change compared with that homogenized at 470 ΓψC for 48 h (Fig. 3(c)) and 96 h (Fig. 3(d)). Figures 3(e) and (f) show the microstructures after homogenization at 470 ΓψC for 24 h and then at 482 ΓψC for 12 h and 24 h, respectively. It can be seen from Fig. 3 and Table 3 that the continuous residual phases along the grain boundaries have turned out to be discontinuous ones and presented to be round or elliptic in shape. When the temperature of the second homogenization stage is 482 ΓψC, the S(Al2CuMg) phase becomes smaller and almost no change has happened for the Al7Cu2Fe [10]. It is also found that, even after homogenization at a higher temperature (482 ΓψC), some of the residual phases do not dissolve completely in the structure. These particles retained are mostly S(Al2CuMg) and Al7Cu2Fe suggested by the EDS analysis in Table 3. It can be concluded that the suitable process of homogenization is 470 ΓψC for 24 h and then at 482 ΓψC for 12 h.
Fig. 3 SEM images of specimens homogenized at different temperatures for different time
Table 3 Chemical compositions of secondary phases in Fig. 3
3.3 DSC curves of homogenized alloys
The DSC curves of the as-cast ingots and the specimens after homogenization at 470 ΓψC for different time are shown in Fig. 4. Figure 5 shows the enthalpies of peaks 1 (477.8 ΓψC) and 2 (487.5 ΓψC) on different curves of DSC analysis for as-cast alloy, and the specimens homogenized for different time in Fig. 4. For Curve 1 in Fig. 4, a relatively high endothermic peak can be observed when the sample is heated to 477.8 ΓψC and the enthalpy associated with it is 5.935 J/g. This indicates that the constituents will melt at 477.8 ΓψC and the over-burning occurs if the ingots are quickly heated to 477.8 ΓψC after being cast [10,14]. Hence, to avoid over- burning, the conventional homogenization temperature for the semi-continuous casting ingots of the alloy is below 470 ΓψC [14,27]. For Curves 2 and 3 in Fig. 4, the endothermic peak at 477.8 ΓψC becomes smaller after the sample is homogenized at 470 ΓψC for 1 h and 8 h, the enthalpy associated with peak 1 of the sample homogenized at 470 ΓψC for 8 h is only 0.594 J/g. Meanwhile, a new endothermic peak at 487.5 ΓψC (peak 2) is observed, which means that the majority of the constituents melted at 477.8 ΓψC disappear after homogenization at 470 ΓψC for 8 h, and some new constituents melted at 487.5 ΓψC are formed. After homogenization at 470 ΓψC for 8 h, the enthalpy of peak 2 (which is a new and endothermic one) of the sample is the highest. The endothermic peak 1 at 477.8 ΓψC disappears gradually and the enthalpy of the endothermic peak 2 at 487.5 ΓψC becomes smaller with increasing the homogenization time at 470 ΓψC. It may be related to the dissolution of some non-equilibrium phases during homogenization. By prolonging the homogenization treatment time, the constituents melted at 477.8 ΓψC will gradually decrease until they disappear at 470 ΓψC for 24 h, and the amount of those melted at 487.5 ΓψC will become smaller too, as shown on Curves 2-4 of Fig. 4. However, the constituents melted at 487.5 ΓψC still exist during homogenization at 470 ΓψC for 240 h, which means that they cannot be eliminated at this temperature.
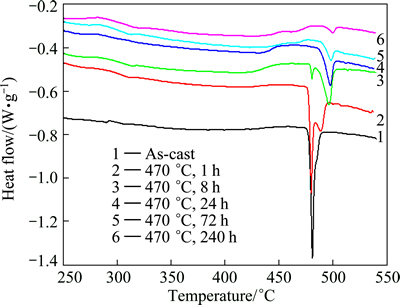
Fig. 4 DSC curves of as-cast ingot and specimens homogenized at 470 ΓψC for different time
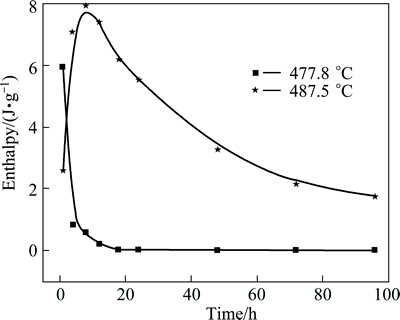
Fig. 5 Enthalpies of endothermic peaks of different DSC curves in Fig. 4
Figures 6 and 7 show the DSC curves and the enthalpies of specimens homogenized at 470 ΓψC for 24 h and then at 482 ΓψC for different time, respectively. The endothermic peak and enthalpy at 482 ΓψC for 1 h are the highest. The endothermic peak at 487.5 ΓψC disappears gradually and the enthalpy becomes smaller with increasing the homogenization time at 482 ΓψC, as shown on the curves 1-4 in Fig. 6. The majority of the constituents melted at 487.5 ΓψC disappear after homogenization at 482 ΓψC for 24 h, and the enthalpy associated with the peak at 487.5 ΓψC of the sample homogenized is only 0.476 J/g, which means that they are almost eliminated at 482 ΓψC for 24 h.

Fig. 6 DSC curves of specimens homogenized at 470 ΓψC for 24 h and then at 482 ΓψC for different time

Fig. 7 Enthalpies of endothermic peaks of different DSC curves in Fig. 6
3.4 XRD patterns of homogenized alloys
It is possible to identify these phases in the as-cast and as-homogenized materials by XRD analysis. The XRD patterns are shown in Fig. 8. In the as-cast alloy, XRD peaks arise from the (Al), MgZn2, Al2CuMg and Al3Zr phases. The gray phase of the fine particles in Al matrix is MgZn2 as shown in Fig. 1(b). In the as-homogenized alloy, XRD peaks are also related to the (Al), MgZn2, Al2CuMg and Al3Zr phases. The XRD peak of Al2CuMg in the alloy homogenized at 470 ΓψC for 48 h is smaller. The XRD peaks of Al2CuMg and MgZn2 are the smallest in the alloy homogenized at 470 ΓψC for 24 h and then at 482 ΓψC for 24 h. Small amounts of Fe-rich phases, Al2Cu and Mg2Si may exist. It can be concluded that the MgZn2, Al2CuMg and Al3Zr exist in the as-cast and as-homogenized alloys, but the T (Al2Mg3Zn3) phase cannot be observed, which means that the gray eutectic phases are considered to be associated with S (Al2CuMg) phase and MgZn2 phase [10,28].
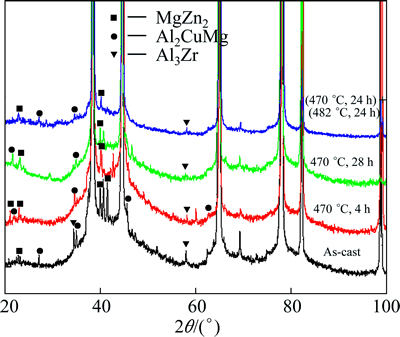
Fig. 8 XRD patterns of as-cast alloy and specimens after homogenization treatment
3.5 Microstructure of plate
Figure 9 shows optical micrographs of plate subjected to hot rolling, solution and aging treatment after pre-homogenization and conventional homogeniza- tion treatment. The unrecrystallized material is readily identified as the dark regions. It is apparent that the recrystallized fraction is substantially less in the material with pre-homogenization treatment. In the material with conventional homogenization treatment, continuous wide recrystallized bands form and the measured recrystallized fraction is 43%. In contrast, in the material subjected to a pre-homogenization treatment, the recrystallized regions are not interconnected and the measured recrystallized fraction is 18%. This is mainly because of the Al3Zr particles coherent to subgrain boundary and grain boundary migration hindrance, thus inhibiting recrystallization and growth. The pre-homogenization procedure involves using lower temperature hold to encourage Al3Zr dispersoid nucleation prior to conventional homogenization. The improved dispersoid distribution is more effective in suppressing recrystallization during subsequent processing, approximately halving the recrystallized fraction from that in the material subjected to conventional homogenization.

Fig. 9 Optical micrographs of heat-treated 7X50 alloy
3.6 Mechanical properties of plate
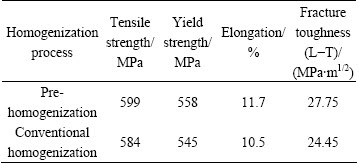
Table 4 shows the mechanical properties of the plate subjected to hot rolling, solution and aging treatment after different homogenization treatment. It is apparent that the strength and fracture toughness of the material with pre-homogenization treatment are higher. The strength and fracture toughness of the material with pre-homogenization treatment are about respectively 15 MPa and 3.3 MPaΓΛm1/2 higher than those of the material with a conventional homogenization treatment. The pre-homogenization procedure encourages Al3Zr dispersoid nucleation and the Al3Zr particle efficiently inhibits the recrystallization and growth. The fraction of low intensive recrystallization reduces and the substructure strengthening improves. The subgrain boundary can efficiently promote the nucleation and precipitation of the second phase particle, and therefore improve the strength of material.
Table 4 Mechanical properties of plate processed by different homogenization processes
4 Conclusions
1) Severe dendritic segregation exists in 7X50 alloy ingot, and the main phases are S(Al2CuMg), T(Al2Mg3Zn3), MgZn2, with small amounts of Al7Cu2Fe and Al3Zr phases. A relatively high endothermic peak at 477.8 ΓψC in as-cast 7X50 alloy can be observed and the enthalpy is 5.935 J/g.
2) The volume fraction of the dendritic-network structure decreases gradually and the residual phases become smaller and sparser by prolonging the homogenization time. The T(Al2Mg3Zn3) phase disappears, minimal S(Al2CuMg) phase remains, and almost no change has happened for the Al7Cu2Fe homogenized at 470 ΓψC for 24 h and then at 482 ΓψC for 12 h.
3) The DSC curve of the as-cast alloy has a relatively strong endothermic peak at 477.8 ΓψC. A new endothermic peak at 487.5 ΓψC appears for the samples homogenized at 470 ΓψC and the endothermic peak at 477.8 ΓψC gradually disappears. The majority of the constituents melted at 487.5 ΓψC disappear after homogenization at 482 ΓψC for 24 h.
4) The recrystallized fraction is substantially less in the material with pre-homogenization treatment. The strength and fracture toughness of the material with pre-homogenization treatment are about 15 MPa and 3.3 MPaΓΛm1/2 higher than those of the material with conventional homogenization treatment.
Acknowledgements
The authors are grateful to the Northeast Light Alloy Co., Ltd., for production and provision of the materials. The authors also appreciate LI Zhi-hui and ZHANG Yong-an of Beijing General Research Institute of Nonferrous Metals, and YIN Zhi-min and JIANG Feng of Central South University for valuable discussions and recommendations.
References
[1] ROBSON J D. Microstructural evolution in aluminium alloy 7050 during processing [J]. Materials Science and Engineering A, 2004, 382(1-2): 112-121.
[2] MARTIN J W. Precipitation hardening [M]. Oxford: Butterworth-Heinemann, 1998: 79-111.
[3] LALPOOR M, ESKIN D G., RUVALCABA D, FJAER H G., TEN CATE A, ONTIJT N, KATGERMAN L. Cold cracking inDC-cast high strengthaluminum alloyingots: An intrinsic problem intensified by casting process parameters [J]. Materials Science and Engineering A, 2011, 528(6): 2831-2842.
[4] ROBSON J D, PRANGNELL P B. Dispersoid precipitation and process modelling in zirconium containing commercial aluminium alloys [J]. Acta Materialia, 2001, 49(4): 599-613.
[5] ROBINSON J S. Influence of retrogression and reaging on fracture toughness of 7010 aluminium alloy [J]. Materials Science and Technology, 2003, 19(12): 1697-1704.
[6] LIU H S, QIAO X, CHEN Z H, JIANG R P, LI X Q. Effect of ultrasonic vibration during casting on microstructure and properties of 7050 aluminum alloy [J]. Journal of Materials Science, 2011, 86(11): 3923-3927.
[7] BELIN F E, FOURNEE V, MIZUTANI U, TAKEUCHI T, MULLER H. Investigation of Al and Mg occupied densities of states ofAl-Mg-Znalloys [J]. Materials Science and Engineering A, 2000, 294-296(12): 516-518.
[8] XU D K, ROMETSCH P A, BIRBILIS N. Improved solution treatment for an as-rolled Al-Zn-Mg-Cu alloy. Part I: Characterisation of constituent particles and overheating [J]. Materials Science and Engineering A, 2012, 534(1): 234-243.
[9] LI X M, STARINK M J. The effect of compositional variations on characteristics of coarse intermetallic particles in overaged 7000 aluminium alloys [J]. Materials Science and Technology, 2001, 17(11): 1324-1328.
[10] FAN X G, JANG D M, MENG Q C, LI Z. The microstructural evolution of an Al-Zn-Mg-Cu alloy during homogenization [J]. Materials Letters, 2006, 60(12): 1475-1479.
[11]  X Y, GUO E J, LI Z H, WANG G J. Research on microstructure in as-cast 7A55 aluminum alloy and its evolution during homogenization [J]. Rare Metals, 2011, 30(6): 664-668.
X Y, GUO E J, LI Z H, WANG G J. Research on microstructure in as-cast 7A55 aluminum alloy and its evolution during homogenization [J]. Rare Metals, 2011, 30(6): 664-668.
[12] EIVANI A R, AHMED H, ZHOU J, DUSZCZYK J. Evolution of grain boundary phases during the homogenization of AA7020 aluminum alloy [J]. Metallurgical and Materials Transactions A, 2009, 40(3): 717-728.
[13] HE L Z, LI X H, ZHU P, CAO Y H, GUO Y P, CUI J Z. Effects of high magnetic field on the evolutions of constituent phases in 7085aluminum alloy during homogenization [J]. Materials Characterization, 2012, 71(9): 19-23.
[14] DENG Y, YIN Z M, CONG F G. Intermetallic phase evolution of 7050 aluminum alloy during homogenization [J]. Intermetallics, 2012, 26(7): 114-121.
[15] XIE F Y, YAN X Y, DING L, ZHANG F, CHEN S L, HU M G, CHENG Y A. A study of microstructure and microsegregation of aluminum 7050 alloy [J]. Materials Science and Engineering A, 2003, 355(1-2): 144-153.
[16] ROKHLIN L L, DOBATKINA T V, BOCHVAR N R, LYSOVA E V. Investigation of phase equilibria in alloys of the Al-Zn-Mg-Cu- Zr-Sc system [J]. Journal of Alloys and Compounds, 2004, 367(1-2): 10-16.
[17] LI X M, STARINK M J. Identification and analysis of intermetallic phases in overaged Zr-containing and Cr-containing Al-Zn-Mg-Cu alloys [J]. Journal of Alloys and Compounds, 2011, 509(2): 471-476.
[18] LI Y X, LI P, ZHAO G, LIU X T, CUI J Z. The constituents inAl-10Zn-2.5Mg-2.5Cu aluminum alloy[J]. Materials Science and Engineering A, 2005, 397(1-2): 204-208.
[19] DONG J, CUI J Z, ZENG X Q, DING W J. Effect of low-frequency electromagnetic field on microstructures and macrosegregation of ΠΒ270 mm DC ingots of an Al-Zn-Mg-Cu-Zr alloy [J]. Materials Letters, 2005, 59(12): 1502-1506.
[20] MONDAL C, MUKHOPADHYAY A K. On the nature of T(Al2Mg3Zn3) and S(Al2CuMg) phases present in as-cast and annealed 7055 aluminum alloy [J]. Materials Science and Engineering A, 2005, 391(1-2): 367-376.
[21] ZHAO Z H, CUI J Z, DONG J, ZHANG B J. Effect of low-frequency magnetic field on microstructures and macrosegregation of horizontal direct chill casting 7075 aluminum alloy [J]. Journal of Materials Processing Technology, 2007, 182(1-3): 185-190.
[22] RANGANATHA R, KUMAR V A, NNANDI V S, NURALIDHARA B K. Multi-stage heat treatment of aluminum alloy AA7049 [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(6): 1570-1575.
[23] LIM S T, LEE Y Y, EUN I S. Microstructural evolution during ingot preheat in 7xxx aluminium alloys for thick semiproduct applications [J]. Materials Science Forum, 2006, 519-521(7): 549-554.
[24] CELENTANO D J. A thermomechanical model with microstructure evolution for aluminium alloy casting processes [J]. International Journal of Plasticity, 2002, 18(10): 1291-1335.
[25] ENGLER O. Nucleation and growth during recrystallization of aluminium alloys investigated by local texture analysis [J]. Materals Science and Technology, 1996, 12(10): 859-872.
[26] ROBSON J D. Optimizing the homogenization of zirconium containing commercial aluminium alloys using a novel process model [J]. Materials Science and Engineering A, 2002, 338(1-2): 219-229.
[27] LI X M,STARINK M J. DSC study on phase transitions and their correlation with properties of overaged Al-Zn-Mg-Cu alloys [J]. Journal of Materials Engineering and Performance, 2012, 21(6): 977-984.
[28] WU L M, WANG W H, HSU Y F, TRONG S. Effects of homogenization treatment on recrystallization behavior and dispersoid distribution in an Al-Zn-Mg-Sc-Zr alloy [J]. Journal of Alloys and Compounds, 2008, 456(1-2): 163-169.
¥‘ΗΘΙΌ1,2Θ§’‘ Η’1Θ§ΫΣ Ζφ3Θ§Χο ξ«1Θ§άν»πΖε1
1. ΕΪ±±¥σ―ß ≤ΡΝœΗςœρ“λ–‘”κ÷·ΙΙΫΧ”ΐ≤Ω÷ΊΒψ Β―ι “Θ§…ρ―τ 110004ΘΜ
2. ΕΪ±±«αΚœΫπ”–œό‘π»ΈΙΪΥΨΘ§ΙΰΕϊ±θ 150060ΘΜ
3. ÷–Ρœ¥σ―ß ≤ΡΝœΩΤ―ß”κΙΛ≥Χ―ß‘ΚΘ§≥Λ…≥ 410083
’Σ “ΣΘΚ≤…”ΟSEMΓΔTEMΓΔEDSΓΔDSCΓΔXRDΚΆά≠…λ Β―ι―–ΨΩ÷ΐΧ§7X50ΚœΫπΦΑΤδΨυ‘»Μ·¥ΠάμΙΐ≥ΧΒΡΉι÷·―ί±δΓΘΫαΙϊ±μΟςΘ§÷ΐΧ§7X50ΚœΫπœύΉι≥…÷ς“Σ”–S(Al2CuMg)ΓΔT(Al2Mg3Zn3)ΓΔMgZn2ΚΆ…ΌΝΩΒΡAl7Cu2FeΚΆAl3ZrœύΓΘΨυ‘»Μ·¥ΠάμΙΐ≥Χ÷–÷ΠΨßΆχΚΆ≤–Ντœύ÷πΫΞΦθ…ΌΘ§Ψ≠(470 ΓψCΘ§24 h)+(482 ΓψCΘ§12 h)Ψυ‘»Μ·¥Πάμ ±Θ§Tœύœϊ ßΘ§Sœύ”–ΈΔΝΩ≤–ΝτΘ§Al7Cu2FeœύΦΗΚθΟΜ”–±δΜ·ΓΘ÷ΐΧ§ΚœΫπΒΡDSC«ζœΏ÷–‘Ύ477.8 ΓψC¥Π”–“ΜΫœ«ΩΈϋ»»ΖεΘ§Ψ≠470 ΓψCΓΔ1 hΨυ‘»Μ·ΚσΚœΫπΒΡDSC«ζœΏ‘Ύ487.5 ΓψC¥Π≥ωœ÷“ΜΗω–¬ΒΡΈϋ»»ΖεΘ§ΕχΨ≠482 ΓψCΓΔ24 hΨυ‘»Μ·¥ΠάμΚσΚœΫπ‘Ύ487.5 ΓψC¥ΠΒΡΈϋ»»ΖεΜυ±Ψœϊ ßΓΘ‘ΎXRDΤΉ÷–Έ¥≥ωœ÷T(Al2Mg3Zn3)œύΘ§’βΚΆTœύ”κS(Al2CuMg)ΦΑMgZn2œύœύΙΊΒΡΫα¬έœύΈ«ΚœΓΘ‘ΛΨυ‘»Μ·¥Πάμ÷Τ±ΗΒΡΑε≤Ρ÷–‘ΌΫαΨßΨßΝΘΖ÷ ΐΟςœ‘ΫΒΒΆΘ§ΩΙά≠«ΩΕ»ΚΆΕœΝ―»Ά–‘œύΕ‘≥ΘΙφΨυ‘»Μ·¥Πάμ÷Τ±ΗΒΡΑε≤ΡΖ÷±πΧαΗΏ‘Φ15 MPaΚΆ3.3 MPaΓΛm1/2ΓΘ
ΙΊΦϋ¥ ΘΚ7X50¬ΝΚœΫπΘΜΉι÷·―ί±δΘΜΨυ‘»Μ·¥ΠάμΘΜ≤–”ύœύΘΜ‘ΌΫαΨß
(Edited by Wei-ping CHEN)
Foundation item: Project (2011KJZX1-2) supported by the Science and Technology Development Fund of Aluminum Corporation of China
Corresponding author: Fu-guan CONG; Tel: +86-451-86564564; E-mail: cfgdy2010@163.com
DOI: 10.1016/S1003-6326(15)63694-9

