Oxidation behavior of single crystal TMS-82+ superalloy in air
来源期刊:中国有色金属学报(英文版)2007年增刊第1期
论文作者:武英 宋桂明 周少雄 T. NARITA
文章页码:136 - 140
Key words:single crystal Ni-based superalloy; oxidation behavior; microstructure; scale
Abstract: The oxidation behavior of the single crystal Ni-based superalloy TMS-82+ was studied under cyclic condition at 900 and 1 000 ℃ for 200 h in air. The oxidation kinetics for the superalloy at both exposure temperatures was explained by the subparabolic relationship. The results show that increasing the exposure temperature from 900 to 1 000 ℃, the amounts of α-Al2O3, (Ni, Co)Al2O4 and NiCr2O4 increase, resulting in a higher mass gain. The oxides consist of (Ni, Co)O, CrTaO4, AlTaO4, Cr2O3, θ-, α-Al2O3 and (Ni, Co)Al2O4 in the specimen at 900 ℃, but NiCr2O4 spinel forms for the specimen exposed at 1 000 ℃ except the above mentioned oxides. A continuous α-Al2O3 layer is responsible for a slow growth rate of the scale after an initial rapid oxidation of NiO.

WU Ying(武英)1, 2, 3, SONG Gui-ming(宋桂明)4, ZHOU Shao-xiong(周少雄)1, 2, T. NARITA 3
1. Central Iron and Steel Research Institute, Beijing 100081, China;
2. Advanced Technology and Materials Co Ltd, Beijing 100081, China;
3. Research Group of Interface Control Engineering, Graduate School of Engineering,
Hokkaido University, Kita-13, Nishi-8, Kita-ku, Sapporo 060-8628, Japan;
4. Fundamentals of Advanced Materials, Delft University of Technology,
Kluyverweg 1, 2629 HS Delft, Netherlands
Received 15 July 2007; accepted 10 September 2007
Abstract: The oxidation behavior of the single crystal Ni-based superalloy TMS-82+ was studied under cyclic condition at 900 and 1 000 ℃ for 200 h in air. The oxidation kinetics for the superalloy at both exposure temperatures was explained by the subparabolic relationship. The results show that increasing the exposure temperature from 900 to 1 000 ℃, the amounts of α-Al2O3, (Ni, Co)Al2O4 and NiCr2O4 increase, resulting in a higher mass gain. The oxides consist of (Ni, Co)O, CrTaO4, AlTaO4, Cr2O3, θ-, α-Al2O3 and (Ni, Co)Al2O4 in the specimen at 900 ℃, but NiCr2O4 spinel forms for the specimen exposed at 1 000 ℃ except the above mentioned oxides. A continuous α-Al2O3 layer is responsible for a slow growth rate of the scale after an initial rapid oxidation of NiO.
Key words: single crystal Ni-based superalloy; oxidation behavior; microstructure; scale
1 Introduction
Single crystal (SC) Ni-based superalloys for gas turbine have been developed to improve the thermal efficiency in industrial and jet engines due to their superior strength and better oxidation resistance. Several SC Ni-based superalloys have been developed by the modification of chemical composition. Elements such as Al, Ti and Ta are known to form a γ′-precipitate in a γ-Ni matrix, while Mo, W and Ta are added to improve the mechanical properties by solid-solution hardening[1-4]. The alloy contains refractory elements such as Mo, W, Ta, Cr and Co to prevent local hot corrosion, and Re-addition improves the oxidation resistance[5-6]. The oxidation behavior of the SC Ni-based superalloys has been studied by some researchers[7-11]. LI et al[7-8] have reported that the oxidation resistance of a SC Ni-based superalloy exposed at 900-1 150 ℃ is higher than that at 800 ℃ because of the formation of a protective α-Al2O3 layer. LIU et al[11] have studied the oxidation of the SC Ni-base superalloy DD32 with Re-addition at 900 and 1 000 ℃. They have reported that two oxidation steps appear in the oxidation kinetics and the first one is controlled by NiO growth and the second by α-Al2O3 growth in the scale. However, most results are conflicting, especially on the oxidation kinetics. It is important to study the high-temperature oxidation resistance of SC superalloys because of their actual working environments. In a previous paper, WU and NARITA[12] have studied the effect of water vapor on the oxidation behavior of the SC Ni-based superalloy TMS-82+ at 900 ℃. The purpose of the present work is, therefore, to study the cyclic oxidation resistance of the SC Ni-based superalloy TMS-82+ at 900 and 1 000 ℃ in air, focusing on the microstructural development of the oxide scale.
2 Experimental
The chemical composition of the SC TMS-82+ superalloy was Al: 12.24, Co: 8.12, Cr: 5.42, Hf: 0.05, Mo: 1.14, Re: 0.82, Ta: 2.20, W: 2.90, Y: 004, Zr: 0.01, Ni: Bal(mole fraction, %). The specimens were prepared by cutting into d 12 mm×1 mm plates from the rods, and the surface of the specimens was ground by SiC papers up to No. 1200 mesh finish followed by ultrasonic cleaning with acetone. Cyclic oxidation tests were carried out in air at 900 and 1 000 ℃ for 200 h. The specimens were suspended with a platinum wire in Al2O3 crucibles, and put into the furnace to expose and then regularly removed from the furnace at intervals of 0-10 h, followed by air cooling to room temperature. The mass change of specimens including exfoliated oxides was measured using a balance with 0.01 mg accuracy.
Phase identification of the oxides was performed by X-ray diffraction (XRD). Surface morphology was observed using a JEOL field-emission gun scanning electron microscope (SEM, JSM-6500). The cross- sectional morphology and the elemental concentration profiles of the oxides were investigated quantitatively by electron probe microanalysis (EPMA, JEOL JXA-8900M WD/ED) at 15 kV and 3.0×10-8 A.
3 Results and discussion
The cyclic oxidation tests demonstrate that the oxidation rate is accelerated by increasing exposure temperature from 900 to 1 000 ℃. Fig.1(a) shows the mass gains per unit surface area for the SC Ni-based superalloy after cyclic oxidation in air at 900 and 1 000 ℃ for 200 h. The overall mass gain for the superalloy exposed at 1 000 ℃ is about 7.5 g/m2, which is a little higher than that at 900 ℃ (6.9 g/m2). In an initial exposure period, the oxidation rate is very fast. The mass gains of the superalloy exposed at 900 and 1 000 ℃ under 0 cycle are 2.8 and 3.4 g/m2, respectively. After the initial kinetics period, the rate of mass gain gradually decrease with extended exposure times. As reported in previous studies[7, 12-13], the kinetics can be described by a subparabolic relationship. The logarithm plots of mass gain per unit area (lg Δm) as a function of exposure time (lg t) for oxidation at 900 and 1 000 ℃ in air are shown in Fig.1(b). The slope of a lgΔm-lgt plot gives the n value, which provides an indication of the oxidation mechanism. The n values of about 0.154 and 0.097 were obtained for the superalloy exposed at 900 and 1 000 ℃ above 1 h, respectively, indicating a subparabolic time dependence of growth rate. The result of present study is not in agreement with the that of LIU et al[11], the reason is probably due to higher Al content (6.0% mass fraction) in the latter.
The constituents of oxides depend on the exposure temperature. XRD patterns of the oxides formed in the SC Ni-based superalloy at 900 and 1 000 ℃ after 200 h cyclic exposure are shown in Figs.2(a) and (b), respectively. At both exposure temperatures, complex oxide products including predominately NiO, some CrTaO4, AlTaO4, Cr2O3, α-Al2O3 and a minor of θ-Al2O3 are detected. However, the amounts of α-Al2O3 and (Ni, Co)Al2O4 spinel increase distinctly in the specimen exposed at 1 000 ℃, especially a new peak of NiCr2O4 spinel is observed, indicating an accelerated formation of NiCr2O4 and (Ni, Co)Al2O4 spinels as well as α-Al2O3 at 1 000 ℃. Increasing exposure temperature, the inward diffusion rate of oxygen and outward diffusion rate of alloy elements are improved, the growth of all oxides is thus enhanced. Although NiO and Cr2O3 also form at 900 ℃, the amount of these oxides is too small to form NiCr2O4 spinel. In the case of specimen at 1 000 ℃, the amounts of NiO and Cr2O3 are enough to form NiCr2O4 spinel. From the values of ![]() of reaction of NiO and Cr2O3 calculated by RUDNYI[14] and MULLER and KLEPPA[15], the values of
of reaction of NiO and Cr2O3 calculated by RUDNYI[14] and MULLER and KLEPPA[15], the values of ![]() at 900 and 1 000 ℃ are about -5.2 and -32 kJ/mol, respectively. The lower value of
at 900 and 1 000 ℃ are about -5.2 and -32 kJ/mol, respectively. The lower value of ![]() at 1 000 ℃ indicates the reaction of NiO and Cr2O3 is easier to form NiCr2O4. Therefore, a subscale just below the outermost (Ni, Co)O layer of predominatel NiCr2O4 and (Ni, Co)Al2O4 spinels forms, while CrTaO4 and AlTaO4 and other oxides form mainly further in the inner layer. Similar study was reported in the Ni-Cr-Al alloy[16], various alloy constituents were oxidized to form the (Ni, Co)O and (Ni, Cr)Al2O4 together with Cr2O3 and Al2O3.
at 1 000 ℃ indicates the reaction of NiO and Cr2O3 is easier to form NiCr2O4. Therefore, a subscale just below the outermost (Ni, Co)O layer of predominatel NiCr2O4 and (Ni, Co)Al2O4 spinels forms, while CrTaO4 and AlTaO4 and other oxides form mainly further in the inner layer. Similar study was reported in the Ni-Cr-Al alloy[16], various alloy constituents were oxidized to form the (Ni, Co)O and (Ni, Cr)Al2O4 together with Cr2O3 and Al2O3.

Fig.1 Oxidation kinetics of SC TMS-82+ superalloy at 900 and 1 000 ℃ for 200 h under cyclic oxidation in air: (a) Mass gain vs exposure time; (b) Logarithm of mass gain per unit area vs logarithm of exposure time
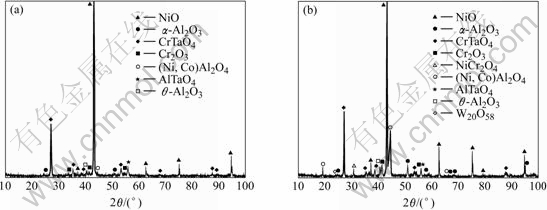
Fig.2 X-ray diffraction patterns of oxide scales formed at 900 (a) and 1 000 ℃ (b) after 200 h exposure for SC TMS-82+ superalloy in air
With increasing exposure temperature, the growth rate of NiO is enhanced. Fig.3 shows SEM micrographs of the surface morphology of the oxide scales of the SC Ni-based superalloy at 900 and 1 000 ℃ after 200 h cyclic exposure. For the both cases, entire surfaces of the specimens are covered by coarse-grained oxides. The results of EDS analysis show that these blocky-type oxides consist mainly of oxygen and Ni with some amount of Co, which is a typical NiO blocky morphology. The grain size of the oxides at 1 000 ℃ is about 2 times than that at 900 ℃ (2-6 μm in diameter). Some pores are observed in the specimen at 1 000 ℃(see Fig.3(b)).
The oxides formed in air show a complex structure. Fig.4(a) shows a SEM micrograph of the cross-sectional morphology of the SC Ni-based superalloy after 200 h cyclic oxidation at 900 ℃ in air. The corresponding concentration profiles of the elements by EPMA are shown in Fig.4 (b). The thickness of oxide scale is 5-9 μm. Main elements of Ni, O and Al together with other elements such as Cr, Ta, Co, Mo, Re and W are also detected in the scale. Particularly a wide Al depletion zone, approximately 10 μm thickness, close to the substrate is observed, indicating a great deal of Al depletion on the substrate. A total of three layers with different chemical compositions are identified for the oxide scale. Starting from the substrate, the oxide scales consist of Al-rich layer, mixed layer and Ni-rich layer. The existence of the outermost layer of NiO is common to the Ni-based alloys. The intermediate layer contains main elements of Ni, Al and O with trace elements of Cr, Ta, Co, Mo, Re and W. Together with the result of XRD, the mixture oxides consist mainly of CrTaO4, AlTaO4, Cr2O3, spinels and Al2O3. The innermost layer is α-Al2O3-rich layer.

Fig.3 SEM micrographs of surface morphologies of oxide scales formed at 900 ℃ (a) and 1 000 ℃ (b) after 200 h exposure for SC TMS-82+ superalloy in air
The structure of the scale in the specimen exposed at 1 000 ℃ is similar to that at 900 ℃ although NiCr2O4 spinel in the intermediate layer and a considerable amount of α-Al2O3 in the innermost layer forms in former. Fig.5(a) shows a SEM micrograph of the cross-sectional morphology of the SC Ni-based superalloy cyclically oxidized at 1 000 ℃ for 200 h. The corresponding concentration profiles of the elements by EPMA are shown in Fig.5(b). The thickness of the oxide scale is not much thicker than that at 900 ℃ due to the overall low growth rate of the scale, which is in accordance with the results of the mass gain. Main elements of Ni, O and Al together with other elements such as Cr, Ta, Co, Mo, Re and W are detected in the scale. Co content of about mole fraction of 6% is detected in NiO layer, indicating a small amount of CoO in solution coexisting with NiO. The Al-depletion zone, approximately 5 μm thickness, is also observed. A strong peak in the Al-content next to the substrate of the scale layer indicates the formation of a continuous α-Al2O3 layer in the scale. Therefore, the outermost layer of the scale is NiO, the intermediate layer consists of CrTaO4, AlTaO4, Cr2O3, Al2O3, (Ni,Co)Al2O4 and NiCr2O4 spinels, and the innermost layer is α-Al2O3-rich layer, which is responsible for slow oxidation of the SC superalloy exposed at 1 000 ℃.

Fig.4 SEM micrograph of cross-sectional morphology (a) and concentration profiles of elements (b) for SC TMS-82+ superalloy after 200 h cyclic oxidation at 900 ℃ in air
The oxidation kinetics of the superalloy shows a rapid oxidation during the initial exposure stage. (Ni, Co)O formed on the surface of the specimen due to fast diffusion of Ni indicates that the oxidation kinetics of the superalloy at the initial period is controlled by the NiO growth. With thickening of the oxide scale, the inward flux of oxygen is reduced further, which is beneficial to the preferential oxidation of Al, leading to a layer of α-Al2O3 formed next to the substrate. This protective oxide of α-Al2O3 provides a diffusion barrier to the reactive species and, therefore, the overall growth rate is stable. As shown in Fig.1, a low oxidation rate is obtained.

Fig.5 SEM micrograph of cross-sectional morphology (a) and concentration profiles of elements (b) for SC TMS-82+ superalloy after 200 h cyclic oxidation at 1 000 ℃ in air
4 Conclusions
1) Regardless of the exposure temperature, time- dependence of the growth rate of the scale for the SC Ni-based superalloy TMS-82+ is fitted by the subparabolic relationship. Increasing the exposure temperature from 900 to 1 000 ℃, the amounts of α-Al2O3, (Ni,Co)Al2O4 and NiCr2O4 spinels increase, resulting in a higher mass gain.
2) The constituents of the oxides depend on the exposure temperature. The outermost (Ni,Co)O layer is common. The intermediate mixed layer consists of CrTaO4, AlTaO4, Cr2O3, θ-, α-Al2O3 and (Ni,Co)Al2O4 in the specimen oxidized at 900 ℃, but NiCr2O4 spinel forms in the specimen exposed at 1 000 ℃ except the above oxides. A continuous α-Al2O3 layer next to the substrate is responsible for a slow growth rate of the scale after an initial rapid oxidation of NiO.
References
[1] ERICKSON G L. A new third generation single-crystal casting superalloy[J]. JOM, TMS, Warrendale, PA, 1995, 47(4): 36.
[2] DUHL D N. Directionally solidified superalloys[M]. In: SIMS C T, STOLOFF N S, HAGEL W C eds., Superalloys Ⅱ, New York: Wiley and Sons, Inc, 1987.
[3] FUCHS G E. Solution heat treatment response of a third generation single crystal Ni-base superalloy[J]. Mater Sci Eng A, 2001, 300: 52-60.
[4] ZHOU H, RO Y, HARADA H, AOKI Y, ARAI M. Deformation microstructures after low-cycle fatigue in a fourth-generation Ni-base SC superalloy TMS-138[J]. Mater Sci Eng A, 2004, 381: 20-27.
[5] FUCHS G E, BOUTWELL B A. Modeling of the partitioning and phase transformation temperatures of an as-cast third generation single crystal Ni-base superalloy[J]. Mater Sci Eng A, 2002, 333: 72-79.
[6] CZECH N, SCHMITZ F, STAMM W. Improvement of MCrAlY coatings by addition of rhenium[J]. Surf Coat Technol, 1994, 68/69: 17-21.
[7] LI M H, SUN X F, LI J G, ZHANG Z Y, JIN T, GUAN H R, HU Z Q. Oxidation behavior of a single-crystal Ni-base superalloy in air. I: At 800 and 900 ℃[J]. Oxid Met, 2003, 59: 591-605.
[8] LI M H, SUN X F, JIN T, GUAN H R, HU Z Q, LI M H, X F. Oxidation behavior of a single-crystal Ni-base superalloy in air. Ⅱ: At 1 000, 1 100, and 1 150 ℃[J]. Oxid Met, 2003, 60: 195-210.
[9] KUPPUSAMI P, MURAKAMI H. A comparative study of cyclic oxidized Ir aluminide and aluminized nickel base single crystal superalloy[J]. Surf Coat Technol, 2004, 186: 377-388.
[10] LIU C T, SUN X F, GUAN H R, HU Z Q. Effect of rhenium addition to a nickel-base single crystal superalloy on isothermal oxidation of the aluminide coating[J]. Surf Coat Technol, 2005, 194: 111-118.
[11] LIU C T, SUN X F, GUAN H R, HU Z Q. Oxidation of the single-crystal Ni-base superalloy DD32 containing rhenium in air at 900 and 1 000 ℃[J]. Surf Coat Technol, 2005, 197: 39-44.
[12] WU Y, NARITA T. Oxidation behavior of the single crystal Ni-based superalloy at 900 ℃ in air and water vapor[J]. Surf Coat Technol, 2007, 202: 140-145.
[13] WEINBRUCH S, ANASTASSIADIS A, ORTNER M, MARTINZ H P, WILHARTITZ P. On the mechanism of high-temperature oxidation of ODS superalloys: Significance of yttrium depletion within the oxide scales[J]. Oxid Met, 1999, 51: 111-128.
[14] RUDNYI E B, KAIBICHEVA E A, SIDOROV L N, VARSHAVSKII M T, MEN A N. (Ion+molecule) equilibrium technique applied to the determination of the activities of Cr2O3 and NiO―Standard molar Gibbs energy of formation of NiCr2O4[J]. J Chem Thermodyn, 1990, 22: 623-632.
[15] MULLER F, KLEPPA O J. Thermodynamics of formation of chromite spinels[J]. J Inorg Nucl Chem, 1973, 35: 2673-2678.
[16] GIGGINS C S, PETTIT F S. Oxidation of Ni-Cr-Al alloys between 1 000 and 1 200 ℃[J]. J Electrochem Soc, 1971, 118: 1782-1790.
Corresponding author: WU Ying; Tel: +86-10-58742810; Fax: +86-10-58742817; E-mail: yingwu2000@hotmail.com
(Edited by CHEN Wei-ping)

