凝固冷却速率对铸态A356铝合金固溶热处理的影响
来源期刊:中国有色金属学报(英文版)2015年第10期
论文作者:杨长林 李远兵 党波 吕贺宾 刘峰
文章页码:3189 - 3196
Key words:A356 aluminum alloy; solution heat treatment; eutectic silicon; cooling rate
摘 要:通过对凝固冷却速度分别为96 K/s和3 K/s的阶梯模具制备的A356铝合金在540 °C下进行固溶热处理,观察和分析该合金经过不同时间固溶处理后共晶硅形貌和拉伸性能的变化,研究凝固冷却速率对A356铝合金固溶热处理的影响。结果表明,凝固过程的冷速不仅能有效地改变合金中共晶硅的形貌,而且高的凝固冷速能缩短合金的固溶热处理时间、加速共晶硅形貌的演变和提高拉伸性能。而A356铝合金中共晶硅的断裂、球化和粗化行为在固溶热处理过程中通过先后两个阶段完成:首先主要是共晶硅的断裂和球化,随后是共晶硅的粗化。
Abstract: The effect of cooling rate of the solidification process on the following solution heat treatment of A356 alloy was investigated, where the cooling rates of 96 K/s and 3 K/s were obtained by the step-like metal mold. Then the eutectic silicon morphology evolution and tensile properties of the alloy samples were observed and analyzed after solution heat treatment at 540 °C for different time. The results show that the high cooling rate of the solidification process can not only reduce the solid solution heat treatment time to rapidly modify the eutectic silicon morphology, but also improve the alloy tensile properties. Specially, it is found that the disintegration, the spheroidization and coarsening of eutectic silicon of A356 alloy are completed during solution heat treatment through two stages, i.e., at first, the disintegration and spheroidization of the eutectic silicon mainly takes place, then the eutectic silicon will coarsen.
Trans. Nonferrous Met. Soc. China 25(2015) 3189-3196
Chang-lin YANG, Yuan-bing LI, Bo DANG, He-bin  , Feng LIU
, Feng LIU
State Key Laboratory of Solidification Processing, Northwestern Polytechnical University, Xi’an 710072, China
Received 19 November 2014; accepted 30 January 2015
Abstract: The effect of cooling rate of the solidification process on the following solution heat treatment of A356 alloy was investigated, where the cooling rates of 96 K/s and 3 K/s were obtained by the step-like metal mold. Then the eutectic silicon morphology evolution and tensile properties of the alloy samples were observed and analyzed after solution heat treatment at 540 °C for different time. The results show that the high cooling rate of the solidification process can not only reduce the solid solution heat treatment time to rapidly modify the eutectic silicon morphology, but also improve the alloy tensile properties. Specially, it is found that the disintegration, the spheroidization and coarsening of eutectic silicon of A356 alloy are completed during solution heat treatment through two stages, i.e., at first, the disintegration and spheroidization of the eutectic silicon mainly takes place, then the eutectic silicon will coarsen.
Key words: A356 aluminum alloy; solution heat treatment; eutectic silicon; cooling rate
1 Introduction
A356 alloys are available for casting components of complex geometry in automatic and aircraft industries due to good castability, corrosion resistance, and weldability [1-4]. However, the plate-like silicon is unfavorable for the desired better mechanical properties [5]. Then, it is important and necessary for the as-cast A356 alloys to modify the eutectic silicon morphology from plate-like to particle or sphericity by the solution heat treatment process in order to improve the alloy strength and ductility.
However, the modification of eutectic silicon during solution heat treatment is affected strongly by solution heat treatment temperature and morphology and size of eutectic silicon in the as-cast condition [6]. Moreover, it has been shown that the morphology and size of eutectic silicon are depended on cooling rate in the as-cast conditions [7-9], which indicates that the cooling rates in the solidification process have an influence on the solid solution heat treatment. Considering that the relationship between the solidification process and the solid solution treatment is not still clear, in order to further improve the effect of the solution treatment and reduce production cost, in the present work, the unmodified as-cast A356 alloys prepared by different cooling rates were chosen to investigate the influence of cooling rate on the evolution of morphology of eutectic silicon in the solution heat treatment in detail, and then to discuss the tensile properties change.
2 Experimental
The commercial as-cast A356 aluminum alloy was chosen as an object alloy and the chemical composition is listed in Table 1. Firstly, the commercial A356 alloy ingots were melted at 720 °C in a resistance furnace. After the degassing process using dry argon for 15 min and C2Cl6 for 10 min, the alloy melt was held at 680 °C for 20 min to homogenize temperature. Then, the alloy melt was poured into two metal molds with the same height of 50 mm (one is copper mold (CM) with an inner diameter of 15 mm cooled by the cycled water; the other is stainless steel mold (SSM) with an inner diameter of 60 mm without cycled cooling water) to prepare the alloy samples with two different cooling rates. During the alloy melt solidification, two K-type thermocouples were placed in the center of the mold to record the cooling curve, and the average cooling rates were calculated as 96 K/s for CM sample and 3 K/s for SSM sample. Thirdly, the flat-like tensile test specimens with 20 mm in gauge length and a cross section of 2 mm × 5 mm were cut from the as-solidified sample. Subsequently, the tensile test specimens were annealed at 540 °C for different time from 5 min to 50 h, and then the specimens were rapidly quenched into 20 °C water to complete solution heat treatment. After quenching, the tensile tests were immediately carried out at room temperature on Shimadzu apparatus (AutoGRAPH AG-X 5 kN). Herein, the duration between quenching and tensile test was not longer than 15 min, and the tensile data were taken from the average value of 3 tensile specimens.
Table 1 Composition of A356 alloy (mass fraction, %)
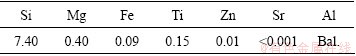

Fig. 1 Cooling curves of two step-like molds
After the alloy samples were etched by 0.5% HF, silicon morphologies were observed and measured from more than eight microstructure photos with different fields by Zeiss optical microscope and software ImageJ, in which the surface area of every photo is between 54000 μm2 and 60000 μm2. Then, the eutectic silicon morphology was characterized by three parameters of aspect ratio, circularity, and mean area. Herein, the aspect ratio is an average ratio of length to width of all the measured eutectic silicon, which can represent the disintegration degree of plate-like silicon. The circularity is a numerical representation of the eutectic silicon roundness. The schematic diagram of circularity is shown in Fig. 2, the larger the circularity, the rounder the silicon particles. The mean area represents the size of silicon phase, which is an average section area of the eutectic silicon phase. Additionally, in order to describe the number change of eutectic silicon, the number density of eutectic silicon is also measured. In the present work, the circularity is defined as follows: [10]
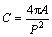 (1)
(1)
where C is the circularity, A and P, which are shown in Fig. 2, are the area and the perimeter of eutectic silicon phase, respectively. Mean area and aspect ratio are calculated as follows [11]:
 (2)
(2)
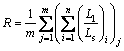 (3)
(3)
where Am is the mean area, R is the aspect ratio, m is the number of viewing fields in one sample, n is the eutectic silicon number in one viewing field, Ll and Ls are the length and width of eutectic silicon phase, respectively.
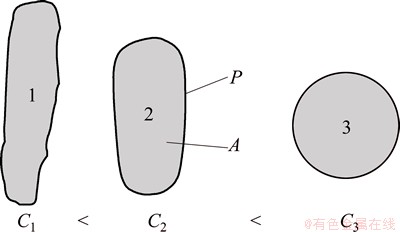
Fig. 2 Schematic diagram of circularity and parameters
3 Results
3.1 Microstructures of solution heat treated A356 alloy
The as-cast microstructures of the CM samples (cooling rate of 96 K/s) and the SSM samples (cooling rate of 3 K/s) are shown in Fig. 3, in which the white dendrite phase is α(Al) and the dark grey plate-like phase which is marked by black arrow is the eutectic silicon. In terms of dendrite spacing depending on cooling rate, it can be found that the α(Al) phase and the eutectic silicon of the CM sample are finer due to a higher cooling rate compared with the microstructure of the SSM sample. Moreover, the eutectic silicons are not all plate-like, some of them are coral-like in the CM sample.
Figure 4 shows the microstructure of the CM samples and SSM samples after solution heat treatment, in which Figs. 4(a)-(c) correspond to solution treatments of CM samples for 0.5, 2 and 8 h, respectively, and Figs. 4(d)-(f) correspond to solution heat treatment of SSM samples for 0.5, 4 and 20 h, respectively. Compared with as-cast microstructure in Fig. 3, most of the plate-like eutectic silicon phases disintegrated into particle (Fig. 4), except for the large ones that only the edges of some of them became smooth and round, and the mean length also decreased obviously. Especially, for the CM samples with increasing solution treatment time (Figs. 4(a)-(c)), not only the distribution of the eutectic silicon phase was more uniform, the number density of the eutectic silicon phase and the size of plate-like silicon phase decreased, but also silicon particles became rounder and bigger. These similar phenomena were also found in the SSM samples, but it took more time for the disintegration of the plate-like eutectic silicon. For the same time of solution heat treatment, as shown in Figs. 4(a) and (d), the eutectic silicon phases were almost globular in Fig. 4(a), whereas, in the Fig. 4(d), only a few of the small plate-like eutectic silicon phases broke down, and the most still kept plate-like similar to as-cast state except that the edges were smoothed. Even for 20 h solution heat treatment, there were still some large and plate-like eutectic silicon particles in Fig. 4(f). However, only after 8 h for the CM samples, almost all the eutectic silicon phases have become globular particles.

Fig. 3 Optical microstructures of as-cast A356 aluminum alloy

Fig. 4 Optical microstructures of A356 alloys after solution heat treatment at 540 °C for different time
3.2 Tensile properties of solution heat treated A356 alloy
Figure 5 shows the ultimate tensile strength (UTS) and the elongation of the prepared A356 alloys by different solidification cooling rates after solution heat treatment at 540 °C for different time. It is evident that for both of CM and SSM samples, the elongation increases rapidly just after a short time solution and then it always increases with the increase of solution time. The elongation increases to about 2 times that of the as-cast state for just 0.5 h solution time for both alloys. This indicates that the disintegration and roundness of plate-like silicon can reduce the stress concentration largely, and thus unfavorable for the crack generation, and contribute to a large elongation. Additionally, the corresponding UTS is determined by the shape of eutectic silicon and the number density of reinforcement eutectic silicon. Consequently, the UTS values of the two alloy samples increase firstly, they continue for about 1 h for CM samples and for 6 h for SSM samples, and then they decrease slowly with increasing the solution time (see Fig. 5). This indicates that the modification of eutectic silicon can improve the ductility of A356 alloy and affect the strength significantly. What’s more, the effect of solution heat treatment on tensile properties can be accelerated by high cooling rate, i.e., the tensile properties of CM samples change more rapidly than those of SSM samples.
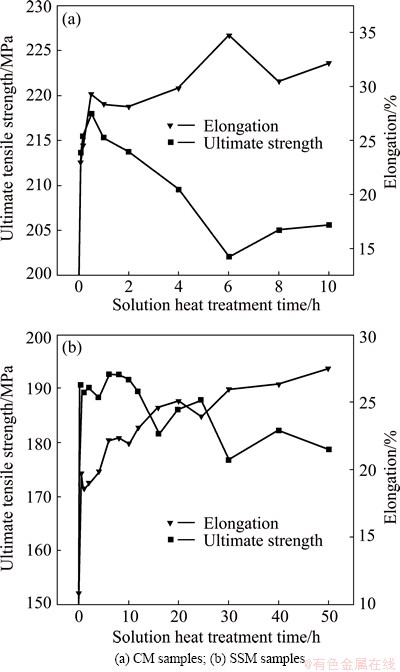
Fig. 5 Ultimate tensile strength and elongation of A356 alloys after solution heat treatment for different time
3.3 Characteristics of eutectic silicon morphology
Figure 6 shows the evolution of aspect ratio of the eutectic silicon particles as a function of solution time. The aspect ratio of silicon particles in CM samples rapidly decreases to about 2 just after 1 h of solution heat treatment. Subsequently, it changes very slowly and there is a plateau from 2 h to 8 h. For SSM samples, the aspect ratio of silicon particles is almost similar to that of the CM samples with solution time elongating. Nevertheless, the aspect ratio of the eutectic silicon in SSM samples is larger and the decreasing rate is slower than that in CM samples, i.e., the aspect ratio reaches about 2 only after 1 h solution treatment of CM samples while it takes 40 h for SSM samples to reach about 2.2. So, it is evident that fast solidification cooling rate can shorten a lot of solution time.

Fig. 6 Aspect ratio of eutectic silicon particles as function of solution heat treatment time
The evolutions of circularity of eutectic silicon particles of CM samples and SSM samples with increasing solution time are shown in Fig. 7. It is obvious that the evolutions of circularity of both of the CM samples and SSM samples are similar. It increases very fast firstly, almost linearly within 1 h for CM samples, while it is 6 h for the SSM samples. Subsequently, there is a slow increase and stable stage, the circularity of CM samples is about 0.8, larger than that of SSM which is about 0.7. This infers that the modification of eutectic silicon mainly takes place at the early several hours of solution time and the effect of following extending the solution time is slight. And then the eutectic silicon particles prepared by high cooling rate alloy are easy to disintegrate and spheroidize.
Figure 8 shows the mean area of the eutectic silicon as function of solution time of CM samples (Fig. 8(a)) and SSM samples (Fig. 8(b)), respectively. During solution heat treatment, the small particles will dissolve and large ones grow, which are controlled by Oswald mechanism. It is obvious that the mean area of eutectic silicon of both alloys becomes larger and larger with solution time increasing. The number density decreases with solution time increasing, which is shown in Fig. 9. And both of the mean area and number density undergo a fast change stage within short solution time, less than 1 h for CM samples and shorter than 6 h for SSM samples, and then a relatively slow evolution stage. CM samples have smaller size of eutectic silicon and larger number density compared with the SSM samples. Herein, it is clear that the growth and dissolution of eutectic silicon in high cooling rate samples (CM) are faster than those of SSM samples.

Fig. 7 Circularity change of eutectic silicon with solution heat treatment time

Fig. 8 Mean area of eutectic silicon as function of solution heat treatment time

Fig. 9 Number density change of eutectic silicon with solution heat treatment time
4 Discussion
The research results have shown that the plate-like eutectic silicon phases in unmodified as-cast A356 alloy are interconnected by the nodes, and the cross section area of the nodes is related to the silicon plates morphology [7,10]. Then, the eutectic silicon undergoes three successive processes: disintegration, spheroidization and coarsening [12,13]. In the present work, as described above, after the solution heat treatment, the morphology change trend of the eutectic silicon phase in the two alloys prepared by different cooling rates is almost the same, i.e., the initial of solution heat treatment resulted in the nodes disintegrating, the plates disconnecting and the edges of plates rounding. Then, the finer plates begin to disintegrate, while the coarser plates start to form bays with a high curvature, which stretched from the periphery into the plates. With the increase in solution time, the bays become deep and the plates disintegrate to round-like particles [14]. While, the small particles disintegrated from plates will dissolve and the large ones become larger and rounder by diffusion.
As described above, in the solution heat treatment process, the aspect ratio of the eutectic silicon (Fig. 6) decreases with solution time, i.e., the length of the eutectic silicon decreases and the width increases, which is in accordance with disintegration stage. Furthermore, the number density (Fig. 9) of the eutectic silicon particles always decrease, while the mean area and the circularity of the eutectic silicon particles increase (Figs. 7 and 8). So, it is obvious that the three processes take place at the same time. However, in terms of the evolution curves of the eutectic silicon morphology, it seems more reasonable to be divided into two stages, i.e., a rapid change (the first stage) and a slow change (the second stage). In the first stage, the plate-like silicon disintegration, the fine silicon particles dissolving and big silicon particles round can take place rapidly, but for the alloys with high cooling rate, the time of the first stage is shorter, due to fine grain size (shorter diffusion distance) and eutectic silicon forming in the as-cast microstructure and bigger driving force of silicon diffusing provided by the supersaturation matrix for high cooling rate CM samples. As shown in Figs. 6-9, the first stage time for CM alloy is shorter (about 1 h) than that for SSM alloy (about 6 h). In addition, there are some small fluctuations in the end of the first stage, especially obvious for SSM alloy. During this time, the majority of plate-like silicon particles have completed disintegrating, and more fine silicon particles are formed, while the remaining large plate-like silicon particles are hard to disintegrate, dissolve and coarsen, inducing the fluctuation of silicon morphology characteristics. Then, as solution heat treatment goes on, the evolution of silicon particles steps into the second stage, in which the morphology changes slightly even for a long period of solution heat treatment. That is because coarsening by diffusion is the main mechanism in this stage and it is only affected by temperature and distance.
For the tensile property of A356 alloy, the grain size, the eutectic silicon morphology, size and distribution of silicon are the main effect factors. Especially, the eutectic silicon in A356 is an important reinforcement phase for the strength improvement of A356 alloy. In terms of the curves of the tensile properties after the solution heat treatment for different time (Fig. 5), the two stages can also be found which are similar to the evolution of the eutectic silicon morphology, i.e., the first stage corresponds to a rapid increase for the elongation and the ultimate strength in a short time, while the second stage corresponds to a slow elongation increase and a slow ultimate strength decrease, respectively. During solution heat treatment, the plate-like eutectic silicon disintegrates to particle-like silicon, spheroidizes and then grows, which will cause the aspect ratio decreasing and circularity increasing. Near-spherical shaped and small eutectic silicon particles inhibit crack nucleation and resist plastic deformation, which improves the strength and ductility [15-17]. After that, in the second stage, the number density decreases and eutectic silicon still spheroidize and grow larger at slow rate. There are less reinforcement eutectic silicon particles and the larger the eutectic particles, the lower the fracture stress because of larger defects [15]. Then, the UTS decreases slightly and ductility increases slightly. Combination with the evolution of eutectic silicon during solution heat treatment, the optimum solution time, which is 1 h for CM samples and about 6 h for SSM samples, would cover the entire first stage of the evolution of eutectic silicon only because the saturated and homogeneous solid solution matrix could be obtained for A356 alloy just after 8-15 min solution at 540 °C [18].
Similarly, for CM alloy samples, not only the first stage time is shorter due to a rapid change of the eutectic silicon morphology, but also the strength and the elongation are both higher due to the formation of more fine eutectic silicon particles and matrix grains, which are favorable to improve the strength and the elongation in terms of the reinforcement theory. Whereas, in the second stage, most of the plate-like silicon particles have broken down, only the silicon particles spheroidization and coarsening take place. Though the silicon particles spheroidization is favorable to improve the strength and the elongation, the coarsening of the eutectic silicon phases including remaining plate-like silicon reduces the strength. Thus, combined with the evolution of the eutectic silicon morphology, it can be obtained that the lager silicon number density, the finer size of silicon, the smaller aspect ratio and the larger circularity contribute to a higher ultimate strength, while high cooling rate can reduce the solution heat treatment time without decreasing the tensile property of A356 alloy. This indicates that, in the practical production, the thin-wall castings or some small components which are solidified with higher cooling rate have the finer grain and eutectic silicon. Therefore, considering improving the mechanical properties and production efficiency, it is necessary and important to reduce solution heat treatment time for these castings with higher cooling rate.
5 Conclusions
1) The eutectic silicon of as-cast A356 alloy could be modified from large plate-like eutectic with slow cooling rate of 3 K/s to coral-like and fine plate-like eutectic coexisted with high cooling rate of 96 K/s. During solution heat treatment, the disintegration, sphroidization and coarsening of eutectic silicon of high cooling rate (CM) samples were modified much faster than that of SSM samples and high cooling rate can reduce the time of solution heat treatment.
2) With the solution heat treatment time increasing, the evolution of eutectic silicon morphology of A356 alloy can be divided into a rapid stage (the first stage) and a slow stage (the second stage). While the disintegration and spheroidization of the eutectic silicon mainly take place in the first stage, in the second stage the coarsening of the eutectic silicon is dominant. What’s more, modification of eutectic silicon mainly takes place in the first stage, so the optimum solution time should be chosen at the end of the first stage, i.e., 1 h for CM samples and about 6 h for SSM samples.
References
[1] ZHANG L Y, JIANG Y H, MA Z, SHAN S F, JIA Y Z, FAN C Z, WANG W K. Effect of cooling rate on solidified microstructure and mechanical properties of aluminum-A356 alloy [J]. Journal of Materials Processing Technology, 2008, 207(1): 107-111.
[2] YOUN A W, KANG C G. Characterization of age-hardening behavior of eutectic region in squeeze-cast A356-T5 alloy using nanoindenter and atomic force microscope [J]. Materials Science and Engineering A, 2006, 425: 28-35.
[3] SEBAIE O E, SAMUEL A M, SAMUEL F H, DOTY H W. The effects of mischmetal, cooling rate and heat treatment on the eutectic Si particle characteristics of A319.1, A356.2 and A413.1 Al-Si casting alloys [J]. Materials Science and Engineering A, 2008, 480 (S1-S2): s342-s355.
[4] KUNTONGKUM S, WISUTMETHANGOON S, PLOOKPHOL T. WANNASIN J. Influence of heat treatment processing parameters on the hardness and the microstructure of semi-solid aluminum alloy A356 [J]. Journal of Metals, Materials and Minerals, 2008, 18: 93-97.
[5] WANG Q G, C H CACERES C H, GRIFFITHS J R. Damage by eutectic particle cracking in aluminum casting alloys A356/357 [J]. Metallurgical and Materials Transactions A, 2003, 34: 2901-2912.
[6]  E, SEIFEDDINE S. The heat treatment of Al-Si-Cu-Mg casting alloys [J]. Journal of Materials Processing Technology, 2010, 210: 1249-1259.
E, SEIFEDDINE S. The heat treatment of Al-Si-Cu-Mg casting alloys [J]. Journal of Materials Processing Technology, 2010, 210: 1249-1259.
[7] MAKHLOUF M M, GUTHY H V. The aluminum-silicon eutectic reaction mechanisms and crystallography [J]. Journal of Light Metals, 2001, 1: 199-218.
[8] XU C L, WANG H Y, QIU F, YANG Y F, JIANG Q C. Cooling rate and microstructure of rapidly solidified Al-20 wt. % Si alloy [J]. Materials Science and Engineering A, 2006, 417(1-2): 275-280.
[9] CHEN Rui, SHI Yu-feng, XU Qing-yan, LIU Bai-cheng. Effect of cooling rate and microstructure of Al-7Si-0.3Mg-0.15Fe alloys [J]. Transactions of Nonferrous Metals Society of China, 2014, 24(6): 1645-1652.
[10] LASAGNI F, LASAGNI A, MARKS E, HOLZAPFEL C,  F, DEGISCHER H P. Three-dimensional characterization of ‘as-cast’ and solution-treated AlSi12(Sr) alloys by high-resolution FIB tomography [J]. Acta Materialia, 2007 55(11): 3875-3882.
F, DEGISCHER H P. Three-dimensional characterization of ‘as-cast’ and solution-treated AlSi12(Sr) alloys by high-resolution FIB tomography [J]. Acta Materialia, 2007 55(11): 3875-3882.
[11] PEDERSEN L, ARNBERG L. The effect of solution heat treatment and quenching rates on mechanical properties and microstructures in AlSiMg foundry alloys [J]. Metallurgical and Materials Transactions A, 2001, 32(3): 525-532.
[12] APELIAN D, SHIVKUMAR S, SIGWORTH G. Fundamental aspects of heat treatment of cast Al-Si-Mg alloys [J]. AFS Transactions, 1989, 97: 727-742.
[13] LADOS D A, APELIAN D, WANG L B. Solution treatment effects on microstructure and mechanical properties of Al-(1 to 13 pct)Si-Mg cast alloys [J]. Metallurgical and Materials Transactions B, 2003, 42(1): 171-180.
[14] RAO Y, YAN H, HU Z. Modification of eutectic silicon and β-Al5FeSi phases in as cast ADC12 alloys using samarium addition [J]. Journal of Rare Earths, 2013, 31(9): 916-922.
[15] WANG Q G. Microstructural effects on the tensile and fracture behavior of aluminum casting alloys A356/357 [J]. Metallurgical and Materials Transactions A, 2003, 34(12): 2887-2899.
[16] OGIRS E, WAHLEN A,  H, UGGOWITZER P J. On the silicon spheroidization in Al-Si alloys [J]. Journal of Light Metals, 2002, 2: 263-269.
H, UGGOWITZER P J. On the silicon spheroidization in Al-Si alloys [J]. Journal of Light Metals, 2002, 2: 263-269.
[17]  C H, WANG Q G. Solidification conditions, heat treatment and the tensile ductility of Al-7Si-0.4Mg casting alloys [J]. AFS Transactions, 1996, 104: 1039-1043.
C H, WANG Q G. Solidification conditions, heat treatment and the tensile ductility of Al-7Si-0.4Mg casting alloys [J]. AFS Transactions, 1996, 104: 1039-1043.
[18] ROMETSCH P A, ARNBERG L, ZHANG D L. Modelling dissolution of Mg2Si and homogenisation in Al-Si-Mg casting alloys [J]. International Journal of Cast Metals Research, 2009, 12: 1-8.
杨长林,李远兵,党 波,吕贺宾,刘 峰
西北工业大学 凝固技术国家重点实验室,西安 710072
摘 要:通过对凝固冷却速度分别为96 K/s和3 K/s的阶梯模具制备的A356铝合金在540 °C下进行固溶热处理,观察和分析该合金经过不同时间固溶处理后共晶硅形貌和拉伸性能的变化,研究凝固冷却速率对A356铝合金固溶热处理的影响。结果表明,凝固过程的冷速不仅能有效地改变合金中共晶硅的形貌,而且高的凝固冷速能缩短合金的固溶热处理时间、加速共晶硅形貌的演变和提高拉伸性能。而A356铝合金中共晶硅的断裂、球化和粗化行为在固溶热处理过程中通过先后两个阶段完成:首先主要是共晶硅的断裂和球化,随后是共晶硅的粗化。
关键词:A356铝合金;固溶处理;共晶硅;冷却速率
(Edited by Yun-bin HE)
Foundation item: Project (3102014KYJD002) supported by the Fundamental Research Funds for the Central Universities of China; Projects (50901059, 51431008, 51134011) supported by the National Natural Science Foundation of China; Project (2011CB610403) supported by the National Basic Research Program of China; Project (51125002) supported by the China National Funds for Distinguished Young Scientists; Project (JC20120223) supported by the Fundamental Research Fund of Northwestern Polytechnical University, China
Corresponding author: Chang-lin YANG; Tel: +86-29-88460622; E-mail: ycl@nwpu.edu.cn
DOI: 10.1016/S1003-6326(15)63952-8

