金刚石/铜复合材料与氧化铝陶瓷的Ag-Cu-Ti活性钎焊
来源期刊:中国有色金属学报(英文版)2013年第6期
论文作者:吴 茂 曹车正 Rafi-ud-din 何新波 曲选辉
文章页码:1701 - 1708
关键词:润湿;金刚石/铜复合材料;Ag-Cu-Ti钎料;活性钎焊;剪切强度
Key words:wetting; diamond/Cu composites; Ag-Cu-Ti brazing filler; reactive brazing; shear strength
摘 要:金刚石/铜复合材料具有低膨胀系数和高热导率等优异性能,使其成为一种理想的电子封装材料。采用97%(72Ag-28Cu)-3%Ti活性钎料对金刚石/铜复合材料和氧化铝陶瓷进行钎焊。发现活性钎料在氧化铝陶瓷和金刚石薄膜表面均具有良好的润湿性,在两者表面的平衡润湿角均小于5°。讨论了主要钎焊条件(如钎焊温度和保温时间等)对接头性能的影响。发现钎焊过程中Ti元素聚集在金刚石颗粒的表面形成TiC化合物,且TiC化合物的形貌与钎焊接头的剪切强度具有紧密联系。推测合适的TiC化合物层厚度可改善钎焊接头的剪切强度,而颗粒状的TiC化合物及过厚的TiC化合物层却会损害钎焊接头的性能。获得的最大剪切强度为117 MPa。
Abstract: The novel properties of diamond/Cu composites such as low thermal expansion coefficient and high thermal conductivity have rendered the composites a valuable packaging material. The reactive brazing of diamond/Cu composites and alumina was performed using the 97%(72Ag-28Cu)-3%Ti alloy. The reactive brazing alloy displays good wettability with alumina and diamond film, and the equilibrium contact angle on both the substrates is found to be less than 5o. The influence of main bonding conditions such as peak heating temperature and holding time was investigated in detail. It is found that Ti element concentrates at the surface of diamond particle resulting in the formation of TiC compound. The morphology of TiC compound exhibits a close relationship with the shear strength of brazing joint. It is surmised that an optimal thickness of TiC layer on the diamond particle surface can ameliorate the shear strength of brazing joint. However, on the contrary, the particle-like shaped TiC compound or a thicker TiC compound layer can impair the shear strength. The maximum shear strength is found to be 117 MPa.
Trans. Nonferrous Met. Soc. China 23(2013) 1701-1708
Mao WU1, Che-zheng CAO1, Rafi-ud-din2, Xin-bo HE1, Xuan-hui QU1
1. State Key Laboratory for Advanced Metals and Materials, Beijing Key Laboratory for Powder Metallurgy and Particulate Materials, University of Science and Technology Beijing, Beijing 100083, China;
2. Chemistry Division, PINSTECH, P.O. Box, Nilore, Islamabad, Pakistan
Received 22 October 2012; accepted 30 November 2012
Abstract: The novel properties of diamond/Cu composites such as low thermal expansion coefficient and high thermal conductivity have rendered the composites a valuable packaging material. The reactive brazing of diamond/Cu composites and alumina was performed using the 97%(72Ag-28Cu)-3%Ti alloy. The reactive brazing alloy displays good wettability with alumina and diamond film, and the equilibrium contact angle on both the substrates is found to be less than 5°. The influence of main bonding conditions such as peak heating temperature and holding time was investigated in detail. It is found that Ti element concentrates at the surface of diamond particle resulting in the formation of TiC compound. The morphology of TiC compound exhibits a close relationship with the shear strength of brazing joint. It is surmised that an optimal thickness of TiC layer on the diamond particle surface can ameliorate the shear strength of brazing joint. However, on the contrary, the particle-like shaped TiC compound or a thicker TiC compound layer can impair the shear strength. The maximum shear strength is found to be 117 MPa.
Key words: wetting; diamond/Cu composites; Ag-Cu-Ti brazing filler; reactive brazing; shear strength
1 Introduction
With the rapid development of microelectronic technology, the density and overall efficiency of microelectronic devices are significantly enhanced, resulting in the continual increase of power density of the integrated circuit (IC) chips. Most of power consumed by ICs is transformed into heat within the package [1,2]. It has been predicted that advanced chips can generate heat fluxes as high as ten of watts per square centimeter. Therefore, the high thermal conductivity and excellent match of coefficient of thermal expansion (CTE) are perquisites for effective heat sink materials [3,4].
The diamond/Cu composites are considered new generation heat sink materials in the field of electronic packing because of their potential thermal conductivity exceeding 500 W/(m・K-1), and lower CTE matching to chip [5,6]. The diamond/Cu composites can be prepared by the methods of powder metallurgy and metal infiltration, both methods have been widely employed in the production of other heat sink materials, such as SiCp/Al and SiCp/Cu composites [7]. Most of researches about diamond/Cu composites revolve around its production techniques and less have been carried out about its post-processing techniques such as brazing.
The present study aims to utilize the diamond/Cu composites as heat sink materials for ceramic packaging applications, involving the brazing of composites with alumina. The standard industrial process for joining alumina to alumina or to metallic solids involves the Mo-Mn metallization and subsequent Ni plating of alumina surfaces prior to brazing. Nevertheless, this manufacturing technique, involving metallization, is complex and expensive. Therefore, various attempts underway to exclude the metallization step using braze containing active alloying elements such as Ti [8]. The presence of such elements modifies the wetting and adhesion by altering the chemistry of in situ interfacial reaction with the alumina surfaces [9].
In the present work, the Ag-Cu-Ti active brazing filler is employed to join the diamond/Cu composites with alumina. The microstructures of brazing joint are investigated in detail. Moreover, the interface evolution and elemental diffusion mechanism of the diamond/Cu composite/Ag-Cu-Ti joint are analyzed. The mechanical properties of the joints are also discussed in association with the evolution of interfacial layer.
2 Experimental
SPS system (Mod. 1050, Sumitomo Coal Mining Co. Ltd., Japan) was used to synthesize diamond/Cu composites. Copper powder (99.9% in purity) that passed through 500 mesh sieve was used as the composite matrix. The reinforcements were synthetic MBD6 type diamonds with an average diameter of 70 μm. The copper powders, with 50% (volume fraction) of diamond particles, were dry mixed at room temperature using a three-dimensional vibratory mill at 1400 r/min for 60 min. The powder mixture was put into a cylindrical graphite die with an inner diameter of 10 mm. The sample was heated by passing alternating DC current through the die and punches from room temperature to 820-940 °C and held for 5 min. The heating rate was 100 °C/min, and a pressure of 30 MPa was applied from the start to the end of the sintering.
The brazing filler metal was the Ag-Cu-Ti system alloy with the chemical composition of 96.5%(72Ag- 28Cu)-3.5%Ti, and the initial thickness of brazing foil was about 200 μm.
Contact angle measurement system (OCA 15 LHT-SV, Dataphysics Co. Ltd., Germany) was used to evaluate the wettability of Ag-Cu-Ti brazing alloy with alumina and diamond film Alumina disk (d15 mm, 2 mm thickness), and the diamond film (d 8 mm, 1 mm thickness) substrates respectively using the sessile drop method. The brazing filler sample was placed on the surface of the substrate, and inserted into the heated zone of vacuum furnace (10-4 Pa). Time counting started after the filler had melted. The wetting process was filmed by a camera (100 flames per second) connected to a computer, enabling automatic image analysis. The characteristic dimensions of the drop (drop base diameter D and visible contact angle) were acquired with an accuracy of ±2% for the contact angle and ±2% for D. After cooling, the selected specimens were cut perpendicular to the interface through the middle for scanning electron microscopy (SEM). The experiments were performed at 1173-1233 K for up to 4000 s time interval.
The brazing process was performed in a vacuum furnace. The process variables were as follows: vacuum degree of 10-4 Pa before heating in the furnaces, heating rate 5 °C/min. After brazing, all the specimens were furnace-cooled to room temperature. The peak heat temperatures were 900, 915, 930, 945 and 960 °C, respectively. The holding times were 5, 10, 15, 30 min, respectively.
To characterize the microstructures of the joints, specimen was cut and subsequently processed by standard metallographic procedure prior to inspection. The cross-sections of the brazed joints were examined by SEM (LEO-1450) equipped with an energy dispersive spectrometer (EDS, KEVEX Sigma). The shear test was performed at room temperature using RG3000A microtester with a speed of 0.2 mm/min, and the average strength of three joints brazed under the same conditions was used. Fracture surface and cross-section perpendicular to solder joint were also characterized by optical microscopy (OM) and SEM.
3 Results and discussion
3.1 Wettability of Ag-Cu-3.5Ti with alumina and diamond film
Ag-Cu-Ti/Al2O3 and Ag-Cu-Ti/diamond wetting systems are typical reactive wetting systems. The reactive wetting is characterized by the spreading velocity of liquid metals, which is controlled by the chemical reaction rate at the triple line, and the spreading time lying between a few seconds to several hours [10,11]. Figure 1 depicts the variation of contact angle versus time for molten Ag-Cu-Ti brazing alloy spreading on alumina or diamond film substrates at various temperatures. It is obvious that the contact angle is decreased with the increase of wetting time, and the ultimate contact angles, calculated at the temperatures of 930 °C and 960 °C for 1 h, are less than 5°. The spreading time for Ag-Cu-Ti brazing alloy to achieve its equilibrium contact angle is 20-30 min. It can also be found that the peak temperature has significant effect on the equilibrium contact angle, and moreover, the contact angle at 900 °C is much higher than that at 930 °C or 960 °C, which can be attributed to the high viscosity of Ag-Cu-Ti brazing alloy at relatively low temperature. It is note worthy that the equilibrium contact angle at the temperature of 930 °C and 960 °C is almost similar for the same substrates.
The controversy exists about the effects of reaction product and the intensity of interfacial reaction on the dynamics of wetting in reactive metal/ceramic systems. AKSAY et al [12] believed that the negative Gibbs free energy for the reaction (ΔG) is the key point for reactive wetting. EUSTATHOPOULOS [13] proposed the “Reaction Product Control” (RPC) model, and argued that the molten brazing alloy spreads on the reaction product layer rather than substrate itself. Thus, the ultimate contact angle is controlled by the properties of reaction product. In the present work, for the Ag-Cu-Ti/Al2O3 or Ag-Cu-Ti/diamond wetting system, the ultimate contact angle is the same despite holding at different peak temperatures, which is in agreement with the RPC model. However, the interfacial reaction is found to be more violent at higher temperature, rendering the wetting system to achieve equilibrium state more quickly, which is consistent with the AKSAY’s theory. The relevant wetting mechanism here needs to be further studied.
Figure 2 shows the variation in contact angle (θ) and drop base diameter (D) observed in Ag-Cu- Ti/Al2O3 system at 900 °C. It is found that the contact angle is further decreased by further prolonging the holding time compared to that in Fig. 1(a). The D(t) and θ(t) curves consist of three kinetic stages. In the first kinetic stage, between time t0≈ 0 and t1≈ 600 s, the velocity of the triple line decreases rapidly from θ0≈70° to θ1≈ 30°. The sharp decrease in contact angle during the early stages is ascribed to the maximum interfacial reaction rate caused by the interaction of liquid Ag-Cu-Ti brazing alloy with the fresh, unreacted alumina surface. Thereafter, at time t1
In the reactive wetting system, two limiting cases are defined depending on the rate of chemical reaction at the triple line compared to the rate of diffusion of reactive solute from the drop bulk to triple line (or of a solute reaction product from the triple line to drop bulk).
In the first limit case, the chemical kinetics at the triple line is rate-limiting because transport within the droplet is comparatively rapid (or not needed when the drop is made of a pure reactive metal). In this case, the reaction does not significantly change the global drop composition, implying that the chemical environment of the triple line remains constant with time, and a steady configuration is established at the triple line during wetting. In both cases, the rate of reaction and hence the triple line velocity remain constant with time [14]:
 (1)
(1)
where R0 is the initial drop base radius and K is a system constant, independent of the drop volume Vd.
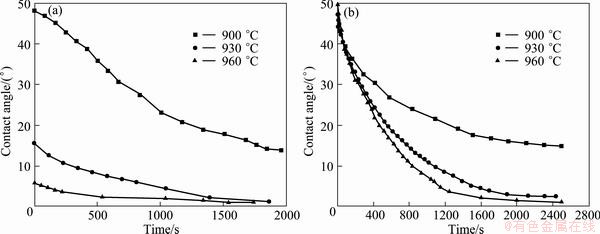
Fig. 1 Contact angle versus time for molten Ag-Cu-Ti at various temperatures on alumina (a) and diamond film (b) substrates

Fig. 2 Contact angle and drop base diameter versus time of Ag-Cu-Ti/Al2O3 wetting system at 900 °C
Compared with the reaction-limited spreading, the second one is transport-limited spreading. As the local reaction rates reach comparatively high, the rate of lateral growth of reaction product at the triple line is limited by the supply of reactant from the drop bulk to the triple line. Because the contact angle decreases continuously during wetting, therefore, the reduction in transport field will lead to a continuous decrease in the reaction rate and, that will ultimately decrease the rate of movement of the triple line itself. Therefore, the time-dependent spreading rates are expected in this case, and the drop spreading kinetics can be expressed by the following equation [15]:
 (2)
(2)
where K is a constant for a given system, involving temperature, drop volume and concentration C0.
From the above analysis, it can be surmised that the Ag-Cu-Ti/Al2O3 wetting system belongs to the reaction- limited spreading, and the velocity of triple line is controlled by the rate of interfacial reaction.
3.2 Microstructure of brazing joint
Figure 3 shows the microstructure of diamond/Cu composites/Ag-Cu-Ti/alumina brazing joint at different peak temperatures of 900, 915, 930, 945, and 960 °C for 30 min. It is obvious in Fig. 3 that an interlayer of alumina exists between diamond/Cu composites and alumina. The joint interfaces are microstructurally sound, well bonded, and devoid of any type of imperfections such as cracks and voids.

Fig. 3 Microstructures of brazing joint brazed at different temperatures for 30 min
Figure 3 also depicts that some Ti and Ag atoms in the brazing alloy diffuse to diamond/Cu composites sides during brazing process, culminating in an increase in the diffusion distance with the increase of peak temperature. Meanwhile, the Ti element reacts with diamond particle and forms TiC compounds. It is found that the molten braze tends to separate into two parts during the brazing process. One part is rich in Ag, and the other part is rich in both Cu and Ti. Therefore, during the following cooling process, the Ag phase (white) and the Ti-Cu phase (grey) distribute into interlayer. MANDAL et al [16] demonstrated that titanium has greater affinity towards copper in comparison to silver, and most of the titanium is associated with the copper rich phase in Ag-Cu-Ti filler metal. The Ti exhibits a solubility of 67% at 1150 °C in copper and it remains as intermetallic phase, which does not normally play a significant role during the brazing process. The presence of silver facilitates titanium to dissociate from the intermetallics, thereby, increasing the activity of the Ag-Cu-Ti alloy for brazing ceramic materials. The titanium renders the molten silver-copper alloy to separate into two fractions, one with a composition of Ag-27Cu-2Ti and the other with Ag-66Cu-22Ti. The 22% (mass fraction) titanium is more than adequate to ensure the wetting of even highly refractory ceramics, such as alumina, silicon carbide, diamond particle or graphite. Similarly, the activity of alloy falls sharply with the reduction of titanium concentration below 2%.
The brazing temperature exhibits a significant effect on the thickness of TiC compound layer. Figure 4 presents the microstructural evolution of TiC layer holding at different temperatures for 30 min. It is found that the TiC layer is a discontinuous layer while holding at 900 °C for 30 min, compared with the continuous TiC layer at 930 °C for 30 min. Moreover, the thickness of TiC layer increases with the increase of peak temperature. This phenomenon may be ascribed to relatively high diffusion rate of Ti element at higher temperature.
The reaction zone between the alumina and interlayer is shown in Fig. 5. A reaction layer with the thinkness of 4-6 μm has formed, and the chemical composition of reaction layer obtained by EDXS analysis of areas free of metallic inclusions is: xO=17.1% (mole fraction), xAl=1.9%, xAg=0.3%, xTi=41.5%, and xCu= 39.2%, which corresponds to the Ti3(Cu,Al)3O phase in accordance with KOZLOVA’s study [8].
3.3 Shear strength and fracture behavior
The fracture behavior of the solder joints is influenced by the interfacial reaction during the solid-state reaction. Owing to the presence of Ti, the formation of additional layers, such as the TiC and Ti3(Cu,Al)3O may influence the fracture behavior of the brazing joint. The excessive growth of these compounds may be detrimental to the reliability of the brazing joint because of the brittleness of the compound layer, and the stress concentration generated from the volume change [17]. The shear strength of Ag-Cu-Ti brazing joint as a function of holding time is shown in Fig. 6. It is found that the shear strength is increased with the time prolonging at 930 °C. The shear strength increases first and then decreases with the elevation of temperature. The shear strength of diamond/Cu composites/ Ag-Cu-Ti brazing joint reaches maximum of 117 MPa at 930 °C for 30 min.
The fracture surface morphology of the joints is shown in Fig. 7 for different peak heating temperatures.

Fig. 4 Microstructures evolution of TiC layer holding at different temperatures for 30 min

Fig. 5 Microstructure of Ag-Cu-Ti/Al2O3 brazing joints
Figure 7(a) shows the fracture surface morphology of a joint brazing at 900 °C for 30 min. It has been observed that fracture occurs at the interface of Ag-Cu-Ti brazing alloy and diamond/Cu composites. As the joint is brazed at elevated temperatures, the brazing joint deteriorates both into the bulk brazing alloy as well as at the composites/brazing alloy interface, even inside the diamond/Cu composites, as depicted in Figs. 7(b), (c) and (d). The brazing joint brazed at 930 °C for 30 min shows the highest shear strength, and fails almost inside the diamond/Cu composites.

Fig. 6 Shear strength at different brazing conditions
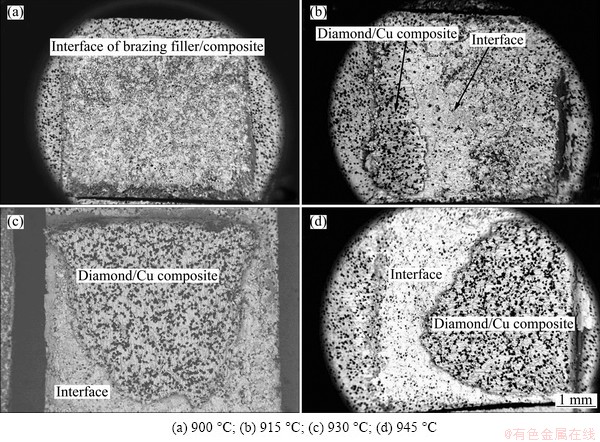
Fig. 7 Fracture surfaces of brazing joint brazed at different temperatures for 30 min
The relatively low brazing temperature leads to a lower diffusion rate of Ti element as well as lower growth rate of TiC compound layer. Figure 8 presents the morphology of TiC compound at the surface of diamond particle. It is conspicuous that the TiC compound is in granular shape brazed at 900 °C for 30 min, and becomes a continuous TiC layer at 960 °C for 30 min. The difference in CTE values of diamond particles, compact TiC compounds layer and brazing alloy generates thermal mismatch and stress at the interface of brazing joints. That thermal mismatch and stress may be high enough to initiate cracks at hard and brittle TiC phase or may cause pre-existing cracks to grow. Thus, the growth of TiC layer and formation of cracks in the compound layer are the major sources of failure.

Fig. 8 Morphology of TiC compound on surface of diamond particle
The main difference among all the diamond/Cu composites/Ag-Cu-Ti brazing joints, holding at different peak temperatures, is the morphology of TiC compounds. Therefore, it can be conjectured that the morphology of TiC compound has a close relationship with the shear strength of brazing joint. Moreover, it is surmised that a suitable thickness of TiC layer on the diamond particle surface can ameliorate the shear strength of brazing joint. However, on the contrary, the particle-like shaped TiC compound or a thicker TiC compound layer can impair the shear strength.
4 Conclusions
Ag-Cu-3.5Ti alloy is used was braze the diamond/Cu composites with alumina for microelectronic packaging application. The Ag-Cu-Ti brazing alloy exhibits good wettability with alumina and diamond film, and the equilibrium contact angle on both the substrates is less than 5°. For the Ag-Cu-Ti/Al2O3 wetting system, the local reaction kinetics is rate-limiting, and the diffusion of reactive element within the droplet is comparatively fast.
During the brazing process, the brazing alloy reacts with alumina and diamond particles and forms TiC and Ti3(Cu,Al)3O compounds, ensuring the good bonding strength of brazing joint. It is found that peak heating temperature and holding time have significant effect on the morphology of TiC compounds, which exhibits a close relationship with the shear strength of brazing joint. It is surmised that a suitable thickness of TiC layer on the diamond particle surface can ameliorate the shear strength of brazing joint. However, on the contrary, the particle-like shaped TiC compound or a thicker TiC compound layer can impair the shear strength. The maximum shear strength is found to be 117 MPa.
References
[1] ZWEBEN C. Thermal materials solve power electronics challenges [J]. Power Electronics Technology, 2006, 32(2): 40-47.
[2] YOSHIDA K, MORIGAMI H. Thermal properties of diamond/copper composite material [J]. Microelectronics Reliability, 2004, 44(2): 303-308.
[3] SERGIO N, HELMUT J B.Modeling of the thermal conductivity and thermomechanical behavior of diamond reinforced composites [J].International Journal of Engineering Science, 2008, 46(6): 606-619.
[4] XIA Yang, SONG Yue-qing, LIN Chen-guang, CUI Shun, FANG Zhen-zheng. Effect of carbide formers on microstructure and thermal conductivity of diamond-Cu composites for heat sink materials [J]. Transactions of Nonferrous Metals Society of China, 2009, 19(5): 1161-1166.
[5] REN Shu-bin, SHEN Xiao-yu, GUO Cai-yu, LIU Nan, ZANG Jian-bing, HE Xin-bo, QU Xuan-hui. Effect of coating on the microstructure and thermal conductivities of diamond-Cu composites prepared by powder metallurgy [J]. Composites Science and Technology, 2011, 71(13): 1550-1555.
[6] NOGALES S,  H J. Modeling of the thermal conductivity and thermomechanical behavior of diamond reinforced composites [J]. International Journal of Engineering Science, 2008, 46(6): 606-619.
H J. Modeling of the thermal conductivity and thermomechanical behavior of diamond reinforced composites [J]. International Journal of Engineering Science, 2008, 46(6): 606-619.
[7] WU Mao, HE Xin-bo, Rafi-ud-din, REN Shu-bin, QIN Ming-li, QU Xuan-hui. Effect of various Ni plating layers and aging on microstructure and shear strength of Sn-2.5Ag-2.0Ni solder joint [J]. Surface and Coatings Technology, 2009, 203(20-21): 3011-3018.
[8] KOZLOVA O, BRACCINI M, VOYTOVYCH R, EUSTATHOPOULOS N, MARTINETTI P, DEVISMES M F. Brazing copper to alumina using reactive CuAgTi alloys [J]. Acta Materialia, 2010, 58(4): 1252-1260.
[9]  The rapidly quenched Ag-Cu-Ti ribbons for active joining of ceramics [J]. Materials Science and Engineering A, 2001, 304-306: 569-573.
The rapidly quenched Ag-Cu-Ti ribbons for active joining of ceramics [J]. Materials Science and Engineering A, 2001, 304-306: 569-573.
[10] EUSTATHOPOULOS N. Dynamics of wetting in reactive metal/ ceramic systems [J]. Acta Materialia, 1998, 46(7): 2319-2327.
[11] MORTENSEN A, DREVET B, EUSTATHOPOULOS N. Kinetics of diffusion-limited spreading of sessile drops in reactive wetting [J]. Scripta Materialia, 1997, 36(6): 645-651.
[12] AKSAY I A, HOGE C E, PASK J A. Wetting under chemical equilibrium and nonequilibrium conditions [J]. The Journal of Physical Chemistry, 1974, 78(12): 1178-1183.
[13] EUSTATHOPOULOS N. Progress in understanding and modeling reactive wetting of metals on ceramics [J]. Current Opinion in Solid State and Materials Science, 2005, 9(4-5): 152-160.
[14] LANDRY K, EUSTATHOPOULOS N. Dynamics of wetting in reactive metal/ceramic systems: linear spreading [J]. Acta Materialia, 1996, 44(10): 3923-3932.
[15] EUSTATHOPOULOS N, GARANDET J P, DREVET B. Influence of reactive solute transport on spreading kinetics of alloy droplets on ceramic surfaces [J]. Philosophical Transactions of the Royal Society of London. Series A: Mathematical, Physical and Engineering Sciences, 1998, 356(1739): 871-884.
[16] MANDAL S, RAY A K, RAY A K. Correlation between the mechanical properties and the microstructural behaviour of Al2O3-(Ag-Cu-Ti) brazed joints [J]. Materials Science and Engineering A, 2004, 383(2): 235-244.
[17] WU Mao, HE Xin-bo, Rafi-ud-din, REN Shu-bin, QIN Ming-li, QU Xuan-hui. Effect of P and aging on microstructure and shear strength of Sn-2.5Ag-2.0Ni/Ni(P) solder joints [J]. Materials Chemistry and Physics, 2010, 121(1-2): 259-266.
吴 茂1,曹车正1,Rafi-ud-din2,何新波1,曲选辉1
1. 北京科技大学 新金属材料国家重点实验室,北京市粉末冶金重点实验室,北京 100083;
2. Chemistry Division, PINSTECH, P.O. Box, Nilore, Islamabad, Pakistan
摘 要:金刚石/铜复合材料具有低膨胀系数和高热导率等优异性能,使其成为一种理想的电子封装材料。采用97%(72Ag-28Cu)-3%Ti活性钎料对金刚石/铜复合材料和氧化铝陶瓷进行钎焊。发现活性钎料在氧化铝陶瓷和金刚石薄膜表面均具有良好的润湿性,在两者表面的平衡润湿角均小于5°。讨论了主要钎焊条件(如钎焊温度和保温时间等)对接头性能的影响。发现钎焊过程中Ti元素聚集在金刚石颗粒的表面形成TiC化合物,且TiC化合物的形貌与钎焊接头的剪切强度具有紧密联系。推测合适的TiC化合物层厚度可改善钎焊接头的剪切强度,而颗粒状的TiC化合物及过厚的TiC化合物层却会损害钎焊接头的性能。获得的最大剪切强度为117 MPa。
关键词:润湿;金刚石/铜复合材料;Ag-Cu-Ti钎料;活性钎焊;剪切强度
(Edited by Chao WANG)
Foundation item: Project (51204016) supported by the National Natural Science Foundation of China; Project (20120006120011) supported by the Specialized Research Fund for the Doctoral Program of Higher Education of China; Project (FRF-TP-12-154A) supported by the Fundamental Research Funds for the Central Universities, China; Project (11175020) supported by the National Science Foundation for Post-doctoral Scientists of China
Corresponding author: Mao WU; Tel: +86-10-82377286; E-mail: wumao@ustb.edu.cn
DOI: 10.1016/S1003-6326(13)62651-5

