����������а������Ҷˮ����ȡ��� �����港ʴ�������õ�Ӱ��
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2014���8��
�������ߣ�K. KRISHNAVENI J. RAVICHANDRAN
����ҳ�룺2704 - 2712
�ؼ��ʣ�����������ʧ���ḯʴ�����ƣ������
Key words��aluminium; mass loss; acid corrosion; inhibition; Morinda tinctoria
ժ Ҫ���о�����������а������Ҷˮ����ȡ��������港ʴ�������õ�Ӱ�졣����������ʧ���͵绯ѧ�����о����ں������ֲ����Ҷ��ȡ���0.5 mol/L HCl��Һ�еĻ�ʴ���á��Ի�ʴ��Ũ�ȡ���Ũ�ȡ��¶Ⱥ�±��������Ũ�ȵȲ��������Ż����������������¶Ⱥ����Ũ�Ȼή�ͻ�ʴ�������ܡ�����ѧ����������������Ļ�ʴ�����ӵ���������Langmuir���������ߡ�
Abstract: The effect of Morinda tinctoria (MT) leaves extract on the corrosion inhibition of Al in acid medium was studied. The inhibition studies were carried out on Al in 0.5 mol/L HCl with the extract of leaves of MT using mass loss and electrochemical techniques. Parameters, such as concentration of the inhibitor, concentration of the acid, temperature and concentration of halide ions, were varied and optimized. Increase of the temperature and concentration of acid can decrease the performance of the inhibitor. Thermodynamic parameters show that the physisorption of the inhibitor molecules on Al surface obeys Langmuir isotherm.
Trans. Nonferrous Met. Soc. China 24(2014) 2704-2712
K. KRISHNAVENI, J. RAVICHANDRAN
Post Graduate and Research Department of Chemistry, Sri Ramakrishna Mission Vidyalaya College of Arts and Science, Coimbatore-641020, Tamilnadu, India
Received 9 October 2013; accepted 13 January 2014
Abstract: The effect of Morinda tinctoria (MT) leaves extract on the corrosion inhibition of Al in acid medium was studied. The inhibition studies were carried out on Al in 0.5 mol/L HCl with the extract of leaves of MT using mass loss and electrochemical techniques. Parameters, such as concentration of the inhibitor, concentration of the acid, temperature and concentration of halide ions, were varied and optimized. Increase of the temperature and concentration of acid can decrease the performance of the inhibitor. Thermodynamic parameters show that the physisorption of the inhibitor molecules on Al surface obeys Langmuir isotherm.
Key words: aluminium; mass loss; acid corrosion; inhibition; Morinda tinctoria
1 Introduction
The corrosion inhibition process helps in reducing the safety and economic input of corrosion damage. Each metal is subjected to its own unique corrosion process. Aluminium (Al) is the most reactive metal compared to copper and steel. The significant property of Al is its tendency to form strongly bonded passivating oxide film on its surface. The thickness of the oxide film may vary as a function of temperature, environment and alloy elements. If the oxide film is damaged by scratch, new oxide film will immediately form on the bare metal. For this reason, it offers excellent resistance to corrosion and provides years of maintenance free service in natural atmosphere. As a result, Al and its alloys are used extensively both in industrial and domestic applications. The stability of the oxide film is dependent upon the pH value of the environment. Since the oxide film is stable within the pH range of about 4 to 8, it is disturbed and gets damaged when the Al metal is in contact with acidic medium of pH of less than 4 and in alkaline medium with pH of more than 9 [1]. The corrosion of metals in acid solutions can be inhibited by a wide range of substances, which may be synthetic or natural inhibitors. Synthetic compounds containing multiple bonds and hetero atoms are effective inhibitors, but at the same time the processing time, cost and their toxic nature have compelled the researchers to look for eco-friendly, non- toxic and low cost inhibitors for the corrosion protection of metals. Many corrosion prevention works have been carried out using extracts of various plants as corrosion inhibitors [2-7]. The use of plants extracts has been found to be viable alternative. The bioactive compounds in the plant extract are as effective as synthetic inhibitors. These bioactive compounds act as inhibitors in acid solution which interact with metals and affect the corrosion reaction in a number of ways, some of which may occur simultaneously. Morinda tinctoria (MT) which belongs to the family of Rubiaceae is a plant which is used extensively in therapeutic purposes. It is found to contain phytochemical constituents like alkaloids, flavonoids, terpenoids, steroids and amino acids [8,9]. The extract of this plant have been used successfully for the corrosion prevention of mild steel in HCl and H2SO4, and it has been found very effective [10,11]. Hence, in the present work, an effort has been made to explore the inhibition characteristics of aqueous extract (AE) of MT on Al corrosion in 0.5 mol/L HCl.
2 Experimental
2.1 Material preparation
The Al specimen of 1 cm �� 5 cm �� 0.15 cm in size with area of 11.7 cm2 was used for study of the mass loss, and specimen with exposed area of 0.95 cm2 was used for electrochemical study. The surfaces of the specimens were mechanically polished with different grades (800, 1000 and 2000) of emery papers and then degreased with acetone. The chemical composition (mass fraction) of the specimen is: 0.20% Si, 0.42% Fe, 0.11% Cu, 0.027% Mn and the balance of Al. The AE was prepared by refluxing 5 g of dried and powdered leaves of MT for 3 h at 60 ��C in 100 mL of distilled water. The solution was allowed to cool to room temperature, filtered and stored. Proper care was taken to maintain the concentration of the acid as constant in the reaction system. Distilled water and analar grade chemicals were used for the study.
2.2 Mass loss method
The pre-cleaned and pre-weighed Al specimens in triplicate were suspended in 100 mL test solution without and with different concentrations of AE in 0.5 mol/L HCl for a period of 1-4 h. After that, the specimens were taken out, washed with distilled water, dried and weighed. From the mass loss data, inhibition efficiency (��) was calculated and the optimum concentration of the inhibitor was identified from the immersion of 1 h. To predict the effectiveness of the inhibitor at high acid concentration range, the concentration of the acid increased from 0.5 to 1.5 mol/L. The synergistic influence of halide ions was studied by adding the halide additives (1% KCl, KBr and KI) to the test solution. All the above reactions were carried out in the temperature of 30��1 ��C. The influence of temperature on the corrosion behavior of Al in the presence of AE of MT was studied by varying the temperature from 303 to 333 K. The value of inhibition efficiency was calculated from the equation as follows:
��=[(mB-mI)/mB]��100% (1)
where mB and mI are the mass loss of the Al specimens in the absence and in the presence of inhibitor, respectively. The corrosion rate RC (mg��cm-2��h-1) was calculated employing the equation [12] as follows:
RC=��m/(St) (2)
where ��m is the difference in the mass loss of the specimens before and after immersion (mg); S is the area of specimen (cm2) and t is the exposure time (h).
2.3 Electrochemical method
Electrochemical studies were carried out using electrochemical analyzer of HCH Instruments (Model 608D). The experiments were carried out in three- electrode cell assembly with platinum wire mesh electrode and saturated calomel electrode (SCE) used as auxiliary and reference electrodes, respectively. Al specimen was used as the working electrode. AC impendence studies were conducted in the frequency range of 10000-1 Hz at the rest potential using 0.02 V sine wave as the excitation signal. Rct and Cdl values were obtained from the Nyquist plots. The �� was calculated as
��=[(Rict�CRoct)/Rict]��100% (3)
where Roct and Rict are the charge transfer resistance values in the absence and in the presence of inhibitor, respectively. Potentiodynamic polarization studies were carried out in the potential range from -0.3 to -1.2 V at the scan rate of 0.01 V/s. The electrochemical parameters such as corrosion current density (Jcorr), corrosion potential (��corr), anodic and cathodic slopes (ba and bc) were obtained from Tafel plots and the �� was determined using the formula as follows:
��=[(Jocorr-Jicorr)/Jocorr ]��100% (4)
where Jocorr and J icorr are the corrosion current densities in the absence and in the presence of inhibitor, respectively.
2.4 Surface analysis
The effect of inhibitor on corrosion process of Al was examined using scanning electron microscope (SEM, JOEL-JSM 6390). The surface analysis of the Al for fresh, inhibited and uninhibited systems was investigated.
3 Results and discussion
3.1 Mass loss method
Table 1 shows the calculated values of RC and �� of the inhibitor at different concentrations on Al surface in 0.5 mol/L HCl solution. The table also gives the data corresponding to the studies on the change in immersion time from 1 to 4 h. The data clearly indicate that the �� increases with increasing concentration of inhibitor from 0.5% to 7% (volume fraction). The maximum �� observed for 1 h is 88.89% and the efficiency gradually increases up to 2 h immersion time. At 2 h immersion period, the maximum efficiency is 96.72%, when the concentration of inhibitor is 7%. After 2 h immersion, the �� starts decreasing for 3 and 4 h. The maximum inhibition efficiencies for the highest concentration (7%) of extract at different immersion periods (1, 2, 3 and 4 h) are found to be 88.89%, 96.72%, 76.01% and 70.66%, respectively, indicating that the maximum efficiency is achieved with 2 h immersion. The increase of �� with increasing of inhibitor concentration indicates the adsorption of the inhibitor molecules on the aluminium metal surface in 0.5 mol/L HCl medium. Similarly, the decrease in the efficiency at higher immersion period indicates desorption of the inhibitor molecules from the metal surface.
The results obtained in Table 2 clearly indicate that, the RC increases and the �� decreases with increasing the acid concentration. Higher concentration of acid increases the rate of chemical reaction and controls the adsorption power of inhibitor by ionization of active species in the corrosion process [13]. The other possibility is that the bond between plant compounds and metal surface (bio compounds of Al) may be ruptured by acid molecules at higher concentration to form hydrogen- metal bond. This results in the higher coverage of the active sites by acid molecules which prevent the penetration of inhibitor molecules to the surface of the metal. However, the increase of RC is more prominent at 1 mol/L acid concentration than that at higher acid concentrations. This might be due to the fact that at this pH, the interaction between the inhibitor molecules and the corrosion product is less on Al surface. This leads to lower surface coverage and higher exposure of the metal to acid.
Table 3 lists the influence of halide additives on the corrosion inhibition of Al using MT. The effect of halide ions on the inhibition efficiency of various organic compounds and natural products on Al corrosion was studied in Refs. [14-18], and it was observed that the synergistic effect increased in the order of Cl-��Br-��I-.
According to the literature, the halide ions facilitate the adsorption of organic compounds in plant extracts during Al corrosion in acidic medium. The addition of halide ions improves the inhibitive action of inhibitor and decreases the amount of inhibitor usage.
The addition of 1% KCl to MT solution of different concentrations (0.5%-7%) increases the ��. The synergistic influence of KCl enhances the inhibition efficiency of MT in the concentration range of 0.5%-3%. The synergistic or co-operative mechanism observed on the addition of KCl to acid solution containing MT is due to the increased surface coverage which results from the ion-pair interaction between the organic cations and anions.
Table 1 Data from mass loss studies for corrosion inhibition of Al in 0.5 mol/L HCl solution with various concentrations of AE at different immersion time
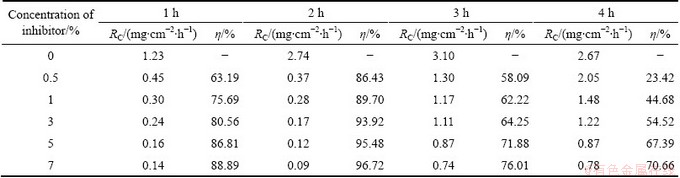
Table 2 Data from mass loss studies for corrosion inhibition of Al at different concentrations of inhibitor and acid
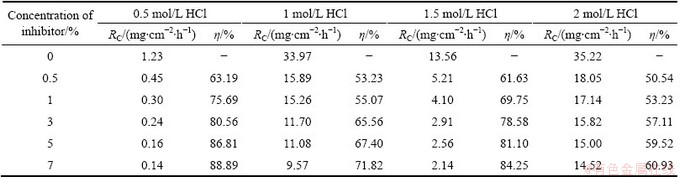
Table 3 Influence of halide ions on corrosion of Al in 0.5 mol/L HCl solution with different concentrations of AE
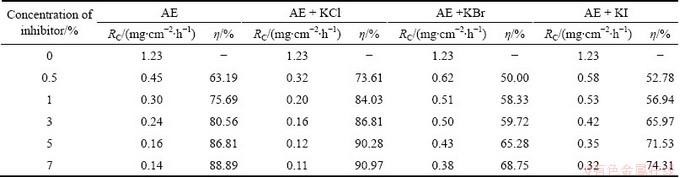
Table 3 also shows that the addition of 1% KBr and KI with different concentrations of inhibitor (0.5%-7%) decreases the inhibition efficiency. The antagonistic or competitive mechanism observed during the addition of KBr and KI to acid solution containing inhibitor molecules which blunt the Al corrosion is due to the weaker interaction between the inhibitor molecules and the halide (KBr and KI) additives [19,20].
3.2 Effect of temperature
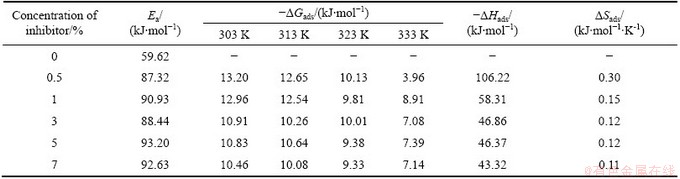
Table 4 lists the details about the effect of temperature on the corrosion of Al in free acid and in the presence of different concentrations of AE in the temperature range of 30-60 ��C. It clearly shows that the increase of temperature increases the mass loss of the metal in the corrosion medium. Temperature favors the reactivity of the active constituents of the corrosion medium and also increases the chemical reactions thereby decreases the ��. The activation energy (Ea) for Al corrosion reaction at 7% inhibitor concentration was found out from the slope of the Arrhenius plot (lg RC vs 1/T) (Fig. 1), where the slope is Ea/(2.303R); RC is the corrosion rate; R is the gas constant; T is the temperature in absolute scale. These values are listed in Table 5 and it is seen from the table that the calculated values of Ea for the inhibited system are found to be higher than those for the uninhibited system. This is an indication of spontaneous adsorption of the inhibitor molecules on Al surface and is attributed to physisorption [21]. An increase in the activation energy of inhibited system is due to the adsorption of the inhibitor molecules on active sites.

Fig. 1 Arrhenius plot (lg RC vs 1/T) for Al corrosion in 0.5 mol/L HCl in presence and absence of AE
3.3 Adsorption isotherm
Table 4 Data from mass loss studies for effect of temperature on corrosion of Al with AE
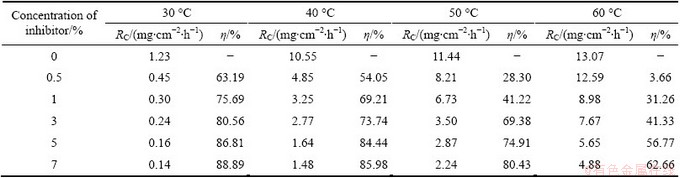
Table 5 Thermodynamic parameters for adsorption of AE on Al in 0.5 mol/L HCl solution at different temperatures
The nature of interaction between the inhibitor and metal surface can be clearly explained by the adsorption isotherm. This plays an important role in determining the adsorption process. The surface coverage values obtained from the mass loss method was tested with different adsorption isotherms. Among various adsorption isotherms tested, Langmuir isotherm gives the best fit in the temperature range of 303-333 K. The Langmuir isotherm is represented by
��/(1-��)=KadsC (5)
where Kads is the adsorption equilibrium constant and is related to the free energy of adsorption. The Langmuir isotherm is obtained by plotting lg [��/(1-��)] vs lg C, as shown in Fig. 2. The free energy of adsorption (��Gads) at various concentrations of inhibitor at different temperatures is calculated using the following equation:
��Gads=-RTln(55.5K) (6)
where K=��/Cinh(1-��); �� is the surface coverage, Cinh is concentration of inhibitor, and the constant value of 55.5 represents the concentration of water in solution. Generally, the value of ��Gads less negative than -20 kJ/mol signifies physisorption and the value more negative than about -40 kJ/mol indicates chemisorption [22]. Table 5 shows that the values of ��Gads are less negative than -20 kJ/mol, indicating that the process of inhibition is through physisorption. Further, the negative values of ��Gads point out the stability of the adsorbed layer and the spontaneity of adsorption. Table 5 also shows that the values of ��Hads are negative and the values of ��Sads are positive. The negative value of ��Hads and the positive value of ��Sads indicate the feasibility of the reaction. Further the ��Gads, ��Hads and ��Sads values indicate that the process of adsorption of AE is spontaneous and exothermic in nature.
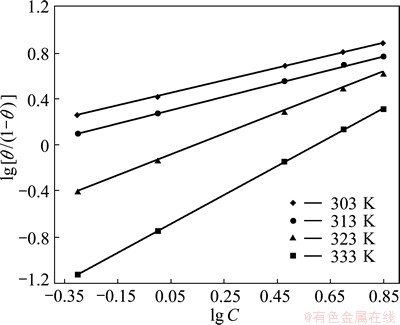
Fig. 2 Langmuir adsorption isotherm for Al corrosion in 0.5 mol/L HCl in presence of AE
3.4 Electrochemical method
Impedance studies were carried out by varying the concentration of inhibitor, concentration of acid, temperature and the concentration of added halide ions. The Randles equivalent circuit used for impedance studies [23,24] is shown in Fig. 3, where Rs is the solution resistance, Cdl is the double layer capacitance, and Rct is the charge transfer resistance. The impedance data obtained from Nyquist plots for the above mentioned systems are listed in Tables 6-13. The representative Nyquist plots for various concentrations of inhibitor AE is shown in Fig. 3. The values of Rct increases with increasing in concentrations of the inhibitor AE. The impedance diagrams obtained are approximately elliptical in shape. Similar plots have been reported by in Refs. [25-28] for aluminium corrosion in acid medium. Generally, this type of spectra is always observed for aluminium in hydrochloric acid [29,30]. Figure 3 shows that the impedance spectra consist of one capacitive loop at high frequencies followed by one inductive loop at low frequencies. The capacitive loop at high frequencies is assigned to the formation of oxide film on Al surface, and the inductive loop at low frequencies is attributed to the relaxation of the charged intermediates adsorbed on the electrode surface [23,26-28]. The size of the loop increases with increasing the concentration of inhibitor, and the shape is maintained throughout the experiment, suggesting the protective layer formation on the metal surface and corrosion of Al is controlled by charge transfer process. The imperfect semicircle (elliptical) is attributed to the frequency dispersion as a result of the roughness and inhomogeneity of electrode surface.
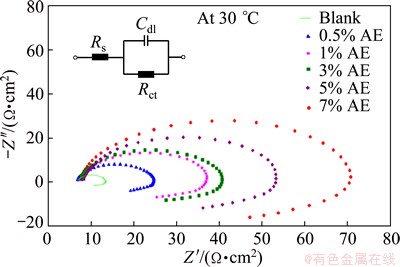
Fig. 3 Nyquist plots for corrosion of Al in 0.5 mol/L HCl solution with various concentrations of AE at (30��1) ��C
Figure 4 shows the typical cathodic and anodic Tafel polarisation curves for Al in 0.5 mol/L HCl solution in the absence and presence of different concentrations of inhibitor. Similar curves were obtained for the other systems (varying the acid medium, addition of halide ions and varying the temperature). The results of polarisation studies for Al in 0.5 mol/L HCl under different conditions are listed in Tables 6-13. It is seen from these tables that the corrosion current (Jcorr) decreases and the �� increases with increasing the concentration of inhibitor which is due to the increase in the surface coverage by the inhibitor molecules on the metal surface. The corrosion potential (��corr) is found to be high in the absence of inhibitor for all the studied systems. Competitive adsorption mechanism directs the corrosion potential of blank solution to more negative values. The addition of inhibitor produces a positive shift (less negative) in the ��corr values which is an indication of the interaction of the molecules with metal surface [31]. The result also reveals that in inhibited system ��corr values are shifted towards less negative values representing the nobility. Further, there has not been much change in the ��corr of the inhibited test solutions indicating the mixed nature of the inhibitor, which prevents both anodic and cathodic reactions. It is further confirmed by the variation of ba and bc values of blank solution from that of the inhibited solution. It shifts the anodic curves to more positive potentials and shifts the cathodic curves to more negative potentials. However, the shift in the cathodic slope values is found to be considerable compared to the shift in the anodic slopes. Tables 8-13 show that the �� calculated from Jcorr values of polarisation studies is not equal to that obtained from impedance studies but the trend is similar. This is due to the fact that the real polarisation resistance or the charge transfer resistance is lower than polarisation resistance obtained from EIS method. This deviation is the result of the resistance of the diffuse layer [29,32].
Table 6 Electrochemical parameters for corrosion of Al in 0.5 mol/L HCl solution in absence and presence of different concentrations of AE at (30��1) ��C
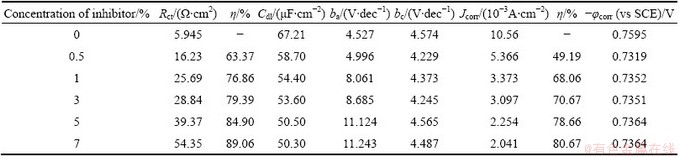
Table 7 Electrochemical parameters for corrosion of Al with AE and KCl
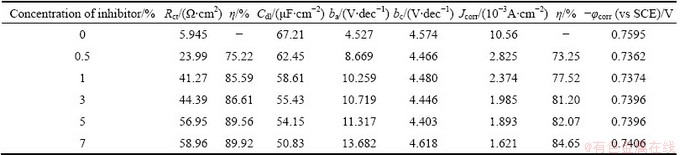
Table 8 Electrochemical parameters for corrosion of Al with AE and KBr
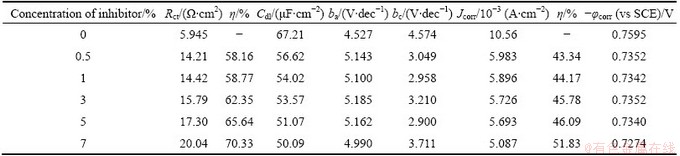
Table 9 Electrochemical parameters for corrosion of Al with AE and KI
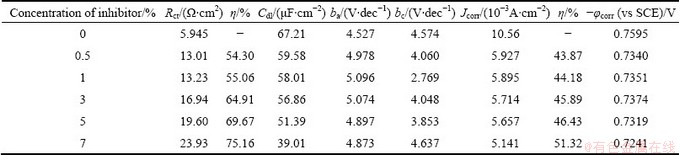
Table 10 Electrochemical parameters for corrosion of Al in 1 mol/L HCl solution in absence and presence of different concentrations of AE
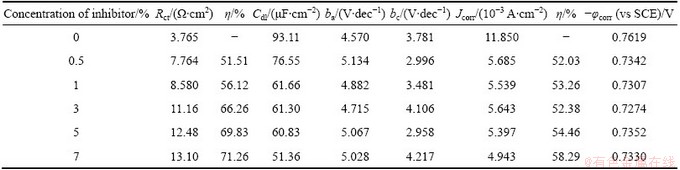
Table 11 Electrochemical parameters for corrosion of Al in 1.5 mol/L HCl solution in absence and presence of different concentrations of AE
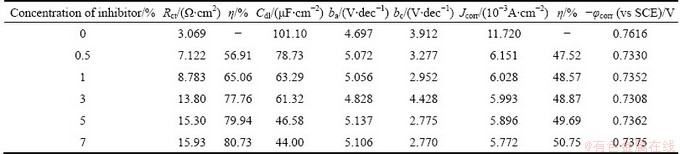
Table 12 Electrochemical parameters for corrosion of Al in presence of AE at (40��1) ��C
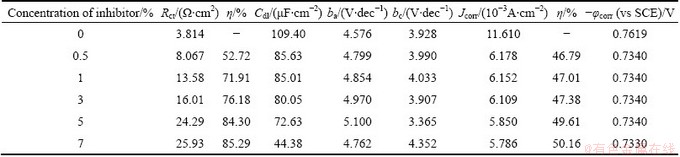
Table 13 Electrochemical parameters for corrosion of Al in presence of AE at (50��1) ��C
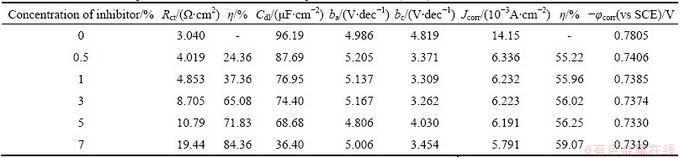
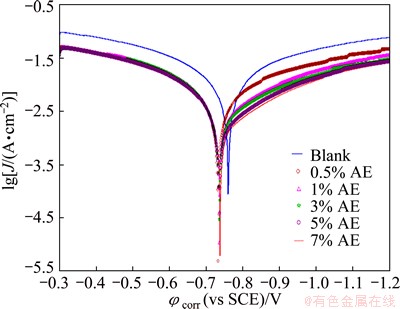
Fig. 4 Tafel plots for corrosion of Al in 0.5 mol/L HCl solution with various concentrations of AE at (30��1) ��C
4 Surface analysis
The SEM images of fresh specimen, specimen in acid and specimen in inhibited acid are shown in Fig. 5. Figure 5 shows that the Al surface is damaged more in the absence of inhibitor and less in the presence of inhibitor, and the image shows good protective film formation by the adsorbed bioactive species on the metal surface.

Fig. 5 SEM images of different Al specimens
5 Conclusions
1) Aqueous extract of Morinda tinctoria acts as good inhibitor for the corrosion of aluminium in 0.5 mol/L HCl solution.
2) Increasing concentration of inhibitor increases the inhibition efficiency, and the efficiency decreases with increasing the concentration of acid as well as temperature. The inhibitor gives high efficiency of about 96.72% with immersion of 2 h.
3) The addition of KCl exerts synergistic effect, and antagonistic effect is observed with KBr and KI addition.
4) Negative values of ��Gads indicate the spontaneous adsorption of inhibitor molecules on Al surface. The negative values of ��Hads and the positive values of ��Sads predict that the reaction is exothermic in nature and the process of adsorption of the inhibitor is through physisorption.
5) The calculated values of Ea are found to be higher for inhibited system than that for uninhibited system advocating the physisorption. The adsorption of AE on Al follows Langmuir isotherm.
6) Large capacitive loop followed by inductive loop is obtained in EIS method. Potentiodynamic polarisation studies indicate the mixed nature of the inhibitor. Surface analysis reveals the adsorption of inhibitor molecule on the metal surface.
Acknowledgement
The authors wish to acknowledge the Post Graduate and Research Department of Chemistry, Sri Ramakrishna Mission Vidyalaya College of Arts and Science, Coimbatore-20 for providing the facilities.
References
[1] BRANZOI V, GOLGOVICI F, BRANZOI F. Aluminium corrosion in hydrochloric acid solutions and the effect of some organic inhibitors [J]. Materials Chemistry Physics, 2002, 78(1): 122-131.
[2] NOOR E A. Potential of aqueous extract of Hibiscus sabdariffa leaves for inhibiting the corrosion of aluminum in alkaline solutions [J]. Journal of Applied Electrochemistry, 2009, 39(9): 1465-1475.
[3] OBOT I B, OBI-EGBEDI N O. An interesting and efficient green corrosion inhibitor for aluminium from extracts of Chlomolaena odorata L. in acidic solution [J]. Journal of Applied Electrochemistry, 2010, 40(11): 1977-1984.
[4] ATING E I, UMOREN S A, UDOUSORO I I, EBENSO E E, UDOH A P. Leaves extract of ananas sativum as green corrosion inhibitor for aluminium in hydrochloric acid solutions [J]. Green Chemistry Letters and Reviews, 2010, 3(2): 61-68.
[5] OBOT I B, OBI-EGBEDI N O, UMOREN S A, EBENSO E E. Synergistic and antagonistic effects of anions and ipomoea invulcrata as green corrosion inhibitor for aluminium dissolution in acidic medium [J]. International Journal of Electrochemical Science, 2010, 5(7): 994-1007.
[6] ARAB S T, AL-TURKUSTANI A M, AL-DHAHIRI R H. Synergistic effect of Azadirachta Indica extract and iodide ions on the corrosion inhibition of aluminium in acid media [J]. Journal of Korean Chemical Society, 2008, 52(3): 281-294.
[7] ABDEL-GABER A M, KHAMIS E, ABO-ELDAHAB H, ADEEL S H. Inhibition of aluminium corrosion in alkaline solutions using natural compound [J]. Materials Chemistry and Physics, 2008, 109(2-3): 297-305.
[8] NISHA K, PRISCILLAPUSHPARANI V, YOGESHWARI R, SUBASHREE P, CHANDRAN M, HARIRAM S. Phytochemical screening of plant Morinda Tinctoria (Family Rubiaceae) using different solvents [J]. Journal of Pharmacognosy and Herbal Formulations, 2011, 1(6): 47-50.
[9] SHANTHI G, SARIDHA D, MARIAPPAN V. Pharmacognostical studies on Morinda Tinctoria Roxb [J]. International Journal of Pharmacy and Pharmaceutical Sciences, 2012, 4(2): 636-638.
[10] KRISHNAVENI K, RAVICHANDRAN J, SELVARAJ A. Effect of morinda tinctoria leaves extract on the corrosion inhibition of mild steel in acid medium [J]. Acta Metallurgica Sinica: English Letters, 2013, 6(3): 321-327.
[11] KRISHNAVENI K, RAVICHANDRAN J, SELVARAJ A. Inhibition of mild steel corrosion by Morinda tinctoria leaves extract in sulphuric acid medium [J]. Ionics, 2014, 20(1): 115-126.
[12] ABD EL HALEEM S M, ABD EL WANEES S, ABD EL AAL E E, FAROUK A. Factors affecting the corrosion behavior of aluminium in acid solutions. I.Nitrogen and/or sulphur containing organic compounds as corrosion inhibitors for Al in HCl solutions [J]. Corrosion Science, 2013, 68: 1-13.
[13] OMO-DUDU D U, OFORKA N C. Inhibition of the corrosion of mild steel in Tri oxonitrate(v)acid [J]. Nigerian Journal of Physics, 1999, 2: 148-153.
[14] OGUZIE E E, ONUCHUKWU A I, OKAFOR P C, EBENSO E E. Corrosion inhibition and adsorption behavior of ocimum basilicum extract on aluminum [J]. Pigment and Resin Technology, 2006, 35: 63-70.
[15] OGUZIE E E. Inhibition of acid corrosion of mild steel by telfaria occidentalis extract [J]. Pigment and Resin Technology, 2005, 34: 321.
[16] OGUZIE E E. Corrosion inhibition of aluminum in acidic and alkaline media by sansevieria trifaciata extract [J]. Corrosion Science, 2007, 49(3): 1527-1539.
[17] UMOREN S A, EBENSO E E. Studies of the anti�Ccorrosive effect of raphia hookeri exudates gum�Chalide mixtures for aluminum corrosion in acidic medium [J]. Pigment and Resin Technology, 2008, 37(3): 173-182.
[18] UMOREN S A, OGBOBE O, EBENSO E E. Synergistic inhibition of aluminum corrosion in acidic medium by gum arabic and halide ions [J]. Transactions of SAEST, 2006, 41: 74-81.
[19] EDDY N O, ODOEMELAM S A, ODIONGENYI A O. Joint effect of halides and ethanol extract of Lasianthera Africana on inhibition of corrosion of mild steel in H2SO4 [J]. Journal of Applied Electrochemistry, 2009, 39: 849-857.
[20] UMOREN S A, SOLOMON M M, UDOSORO I I, UDOH A P. Synergistic and antagonistic effects between halide ions and carboxymethyl cellulose for the corrosion inhibition of mild steel in sulphuric acid solution [J]. Cellulose, 2010, 17: 635-648.
[21] ASHASSI-SORKHABI H, SHAABANI B, SEIFZADEH D. Corrosion inhibition of mild steel by some schiff base compounds in hydrochloric acid [J]. Applied Surface Science, 2005, 239: 154-164.
[22] OBOT I B, OBI-EGBEDI N O. Adsorption properties and inhibition of mild steel corrosion in sulphuric acid solution by ketoconazole: Experimental and theoretical investigation [J]. Corrosion Science, 2010, 529: 198-204.
[23] GOPIRAMAN M, SELVAKUMARAN N, KESAVAN D, KARVEMBU R. Adsorption and corrosion inhibition behaviour of N-(Phenyl carbamothioyl) benzamide on mild steel in acidic medium [J]. Progress in Organic Coatings, 2012, 73: 104-111.
[24] EL REHIM S S, AMIN M A, MOUSSA S O, ELLITHY A S. The corrosion inhibition of aluminium and its copper alloys in 0.1 M H2SO4 solution using linear-sodium dodecyl benzene sulfonate as inhibitor [J]. Materials Chemistry and Physics, 2008, 112: 898-906.
[25] GARRIGUES L, PEBERE N, DABOSI F. An investigation of the corrosion inhibition of pure aluminum in neutral and acidic chloride solutions [J]. Electrochimica Acta, 1996, 41(7-8): 1209-1215.
[26] LI Xiang-hong, DENG Shu-duan, FU Hui. Inhibition by tetradecylpyridinium bromide of the corrosion of aluminium in hydrochloric acid solution [J]. Corrosion Science, 2011, 53(4): 1529-1536.
[27] LI Xiang-hong, DENG Shu-duan. Inhibition effect of Dendrocalamus brandisii leaves extract on aluminum in HCl, H3PO4 solutions [J]. Corrosion Science, 2012, 65: 299-308.
[28] DENG Shu-duan, LI Xiang-hong. Inhibition by Jasminum nudiflorum lindl leaves extract of the corrosion of aluminium in HCl solution [J]. Corrosion Science, 2012, 64: 253-262.
[29] YURT A, ULUTAS S, DAL H. Electrochemical and theoretical investigation on the corrosion of aluminium in acidic solution containing some Schiff bases [J]. Applied Surface Science, 2006, 253(2): 919-925.
[30] ZHANG Qi-bo, HUA Yi-xin. Corrosion inhibition of aluminum in hydrochloric acid solution by alkylimidazolium ionic liquids [J]. Materials Chemistry and Physics, 2010, 119(1�C2): 57-64.
[31] ASHASSI-SORKHABI H, GHASEMI Z, SEIFZADEH D. The inhibition effect of some amino acids towards the corrosion of aluminum in 1M HCl+1M H2SO4 solution [J]. Applied Surface Science, 2005, 249(1-4): 408-418.
[32] YURT A, MIHRICAN Y. Quantitative relationships between the structure of some thiol compounds and their inhibition efficiencies [J]. Anti-Corrosion Methods and Materials, 2008, 55(4): 195-203.
K. KRISHNAVENI, J. RAVICHANDRAN
Post Graduate and Research Department of Chemistry, Sri Ramakrishna Mission Vidyalaya College of Arts and Science, Coimbatore-641020, Tamilnadu, India
ժ Ҫ���о�����������а������Ҷˮ����ȡ��������港ʴ�������õ�Ӱ�졣����������ʧ���͵绯ѧ�����о����ں������ֲ����Ҷ��ȡ���0.5 mol/L HCl��Һ�еĻ�ʴ���á��Ի�ʴ��Ũ�ȡ���Ũ�ȡ��¶Ⱥ�±��������Ũ�ȵȲ��������Ż����������������¶Ⱥ����Ũ�Ȼή�ͻ�ʴ�������ܡ�����ѧ����������������Ļ�ʴ�����ӵ���������Langmuir���������ߡ�
�ؼ��ʣ�����������ʧ���ḯʴ�����ƣ������
(Edited by Chao WANG)
Corresponding author: J. RAVICHANDRAN; Tel: +91-4222692461; Fax: +91-4222693812; E-mail: jrsrmv@gmail.com
DOI: 10.1016/S1003-6326(14)63401-4

