
Oxidation behavior of multiphase Mo5SiB2 (T2)-based alloys at
high temperatures
WANG Fang(王 方), SHAN Ai-dang(单爱党), DONG Xian-ping(董显平), WU Jian-sheng(吴建生)
Key Laboratory of Ministry of Education for High Temperature Materials and Testing,
School of Materials Science and Engineering, Shanghai Jiao Tong University, Shanghai 200030, China
Received 15 July 2007; accepted 10 September 2007
Abstract: Two Mo5SiB2 (T2)-based alloys with nominal compositions of Mo-12.5Si-25B and Mo-14Si-28B (molar fraction, %) were prepared in an arc-melting furnace, and their oxidation kinetics from 1 000 to 1 300 ℃ were studied. The microstructures of the alloys were characterized by X-ray diffractometry(XRD) and scanning electron microscopy(SEM) with energy dispersive spectroscopy (EDS). The oxide scales of both alloys oxidized at 1 200 ℃ for 10 min, 2 h and 100 h were investigated by surface XRD and cross-sectional SEM-EDS. The results show that the matrix of both alloys consists of T2. The dispersions of Mo-12.5Si-25B alloy are Mo and Mo3Si, and the dispersions of Mo-14Si-28B alloy are Mo5Si3 (T1) and MoB. The cyclic oxidation kinetics data exhibit initial rapid mass loss followed by slow mass loss. The mass loss of Mo-12.5Si-25B alloy is much faster than that of Mo-14Si-28B alloy at 1 200 and 1 300 ℃. For 10 min exposure, both alloys form irregular and porous thin scale. For 2 h exposure, Mo-12.5Si-25B alloy forms irregular thin scale and the scale contains large cracks, and Mo-14Si-28B alloy forms sound and continuous scale. For 100 h exposure, Mo-12.5Si-25B and Mo-14Si-28B alloys form sound and continuous scale about 50-75 μm and 40-45 μm in thickness, respectively. The better oxidation resistance of Mo-14Si-28B alloy is due to a sound and continuous B-SiO2 layer formation in the early stage of oxidation.
Key words: intermetallics; oxidation; diffusion; microstructure; Mo5SiB2
1 Introduction
Molybdenum silicides based intermetallic compounds and composites have high potential for high temperature structural application, since they have high melting temperature, excellent strength, good creep resistance and oxidation resistance at high temperatures [1-3].
In the last few decades, molybdenum disilicide (MoSi2) has been extensively studied because of its excellent oxidation resistance at high temperatures (up to 1 700 ℃), but poor creep strength above 1 200 ℃[4], and poor oxidation resistance in the temperature range of 400-600 ℃ leading to pest disintegration[5-7], make MoSi2 unsuitable as a structural material.
The other alternative is Mo5Si3, which has better creep resistance than MoSi2 at high temperatures[8]. But Mo5Si3 has poor oxidation resistance at elevated temperatures (above 1 000 ℃)[9] and shows “pesting behavior” at moderate temperature (about 800 ℃). Recently B-doped Mo5Si3 (T1) has been reported to show oxidation resistance as good as MoSi2[10-11] while maintaining a good creep resistance. Thus extensive studies have been made on the oxidation resistance of T1-based alloys.
On the other hand, the single crystalline and polycrystalline Mo5SiB2 (T2) have been found to have higher elevated temperature strength and creep resistance than those of MoSi2 and T1 single crystals and their in-situ composites[12]. These results in the Mo-Si-B alloy system of previous works[12-13] suggested that T2 is a better component for mechanical properties. Furthermore, T2 has a high melting temperature (above 2 200 ℃). Thermal expansion coefficient (ac/aa) of the T2 phase is approximately 1.4 at 500 ℃, which is significantly smaller than 2.2 for Mo5Si3. YOSHIMI et al[14] investigated isothermal oxidation behavior of Mo5SiB2-based alloy at elevated temperature, at above 1 273 K, transient and steady state oxidation stages have been clearly observed, and the SiO2 glass scale formed on the Mo5SiB2-based alloy was more protective than as expected.
Although T2 is believed to have good oxidation resistance during oxidizing process, experimental data on the oxidation behavior of T2 are relatively scarce. Small variations in alloy compositions can result in a great difference in microstructure, e.g. the constitution or the relative amounts of each phase in multiphase alloys[15]. For the multiphase T2-based alloys, it has not been reported the effect of phase constitution on the oxidation resistance of T2-based alloys. The goal of this study is to examine the oxidation behavior of the T2-based multiphase alloys in the Mo-Si-B system and to find the effect of small variations in alloy compositions on oxidation resistance. Microstructures after oxidation for 10 min, 2 h and 100 h at 1 200 ℃ were observed. Finally, the role that individual phases play in the oxidation behavior was discussed.
2 Experimental
Mo-Si-B alloys with the nominal composition of Mo-12.5Si-25B and Mo-14Si-28B (molar fraction, %) were prepared in a non-consumable tungsten arc-melting furnace under an argon atmosphere. The raw materials of Mo, Si, and B were 99.9%, 99.999 9%, and 99.9% (mass fraction) pure, respectively. The arc-melted alloys were remelted 6 times to ensure chemical homogeneity. These alloys were annealed at 1 800 ℃ for 2 h in Ar for homogenization.
The samples for oxidation tests with a size about 13 mm×4 mm×1.5 mm were cut from the button by electro-discharge machine. All samples for cyclic oxidation were metallographically polished. The samples were cleaned ultrasonically in acetone and alcohol, and then dried in air. The mass and size of the sample were measured carefully before oxidation exposure for mass change. Cyclic oxidation tests were performed at 1 000, 1 200 and 1 300 ℃ in air. During cyclic oxidation tests, the samples were withdrawn from the furnace and cooled to room temperature, weighed and put back in the furnace. The mass change of the samples during exposure was measured by an electron balance with a sensitivity of 10-4 g.
Microstructures of the annealed alloys were observed by scanning electron microscopy (SEM, JEOL JSM-6460) with energy dispersive spectroscopy(EDS), and constituent phases were identified by X-ray diffractometry(XRD, Rigaku D/max 2000PC). The oxide scales at 1 200 ℃ oxidized for 10 min, 2 h and 100 h were examined by surface XRD and cross-sectional SEM-EDS.
3 Results and discussion
3.1 Microstructures of alloys
Fig.1 shows backscattered electron(BSE) images of annealed Mo-12.5Si-25B and Mo-14Si-28B alloys. Although Mo-12.5Si-25B and Mo-14Si-28B alloys have similar composition, they show significantly different microstructures. The microstructure of Mo-12.5Si-25B alloy consists of a 3-phase Mo-T2-Mo3Si eutectic embedded in T2 matrix with discrete Mo particles of the order of 1 μm distributed in the matrix, as shown in Fig.1(a). The microstructure of Mo-14Si-28B alloy is composed of T2 as matrix with Mo5Si3 (T1) and MoB as dispersions shown in Fig.1(b). The existence of these phases was also identified by EDS and XRD.

Fig.1 SEM backscattered electron(BSE) images of Mo-12.5Si- 25B (a) and Mo-14Si-28B (b) alloys
3.2 Oxidation kinetics
Fig.2 shows cyclic mass change of Mo-12.5Si-25B and Mo-14Si-28B alloys oxidized for 100 h from 1 000 to 1 300 ℃. The cyclic oxidation kinetics data exhibit initial rapid mass loss followed by slow mass loss. This mass change behavior named “transient and steady state oxidation” was discussed in Refs.[10,14,16]. The occurrence of the transient and steady state oxidation is interpreted in terms of rapid volatilization of MoO3 and B2O3 under ambient O2 pressure at the initial stage and the passive oxidation after completely sealing the sub-
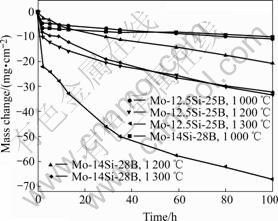
Fig.2 Cyclic mass change curves of Mo-12.5Si-25B and Mo- 14Si-28B alloys oxidized for 100 h
strate by silicate glass[14]. The mass loss curves of Mo-12.5Si-25B and Mo-14Si-28B alloys at 1 000 ℃ are very close, and the mass loss after 100 h is found to be about 10 mg/cm2. As temperature increases, the extent of mass loss is accelerated and the mass loss of Mo-12.5Si-25B alloy is much faster than that of Mo-14Si-28B alloy. The total mass losses at 1 300 ℃ are about 67 and 32 mg/cm2 for Mo-12.5Si-25B and Mo-14Si-28B alloys, respectively, oxidized for 100 h.
3.3 Microstructures of oxide scales
Fig.3 shows the cross-sectional microstructures of Mo-12.5Si-25B and Mo-14Si-28B alloys oxidized at 1 200 ℃ for 10 min, 2 h and 100 h, respectively. For 10 min exposure, Mo-12.5Si-25B alloy forms irregular and porous thin scale as shown in Fig.3(a). The oxide scale consists mainly of crystalline and amorphous SiO2, which is proved by XRD and EDS results, as shown in Figs.4(b) and (e). The scale also contains many bright particles; the large grained particles adjacent to the matrix consist of MoO2. However, the smaller grained particles that are too small to be analyzed individually by EDS. When the bright particles are probed, Mo together

Fig.3 Cross-sectional SEM backscattered electron(BSE) micrographs of Mo-12.5Si-25B and Mo-14Si-28B alloys oxidized at 1 200℃ for 10 min (a, b), 2 h (c, d) and 100 h (e, f)
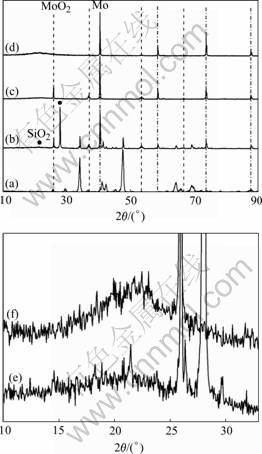
Fig.4 Surface XRD patterns of Mo-12.5Si-25B alloy oxidized at 1 200 ℃ for different times: (a) 0 min; (b) 10 min; (c) 2 h; (d) 100 h; (e) and (f) Local magnification patterns of (b) and (c), respectively (Dot markers identify MoO2 peaks and dash dot markers identify Mo peaks within scale. Unmarked peaks correspond to silicides phases from underlying alloy.)
with Si that comes from the surrounding oxide of SiO2 is detected. Thus, these bright particles are likely to be Mo, which are detected during XRD analysis. At the scale/substrate interface, the regions containing the 3-phase eutectic is the preferential oxidation sites; these are somewhat similar to the previously reported result [17]. EDS analysis indicates that the preferentially oxidized regions are composed primarily of Mo and Si as well as a small amount of O, the preferentially oxidized products are mixed oxides (MoOx and SiOy) or ternary oxide (Mo-Si-O). According to a previous work[18], the Mo-Si-O oxide formed at the initial stage of oxidation is unstable and decomposes into MoO3 and SiO2 when further oxidized. Mo-14Si-28B alloy also forms irregular thin scale with large pores and cracks as shown in Fig.3(b). The matrix of the oxide scale also consists of crystalline and amorphous SiO2 and the dispersions are Mo and molybdenum oxides, XRD also confirmed all these phases, as shown in Figs.5(b) and (e).
For 2 h exposure, Mo-12.5Si-25B alloy remains
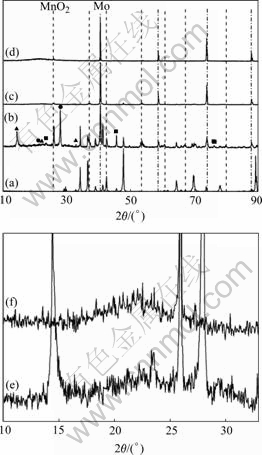
Fig.5 Surface XRD patterns of Mo-14Si-28B alloy oxidized at 1 200 ℃ for different times: (a) 0 min; (b) 10 min; (c) 2 h; (d) 100 h; (e) and (f) Local magnification patterns of (b) and (c), respectively (Dot markers identify MoO2 peaks and dash dot markers identify Mo peaks within scale. Unmarked peaks correspond to silicides phases from underlying alloy.) ▲ Mo4O41; ■ MoO2; ● SiO2
irregular thin scale and the scale contains large cracks, as shown in Fig.3(c). XRD (Figs.4(c) and (f)) and EDS results indicate that the oxide scale consists of amorphous SiO2, there is a MoO2 layer adjacent to the SiO2 oxide scale, which suggests that after formation of a glass layer, oxygen partial pressure at oxide/alloy interface must be reduced to a level so that only partial oxidation of Mo occurs. A Mo solid solution (Mo(ss)) interlayer is observed adjacent to substrate (Fig.3(c)). At the scale/substrate interface, the regions containing the 3-phase Mo-T2-Mo3Si eutectic are still preferentially oxidized. Mo-14Si-28B alloy forms sound and continuous scale, as shown in Fig.3(d). XRD (Figs.5(c) and (f)) and EDS results indicate that the matrix of the oxide scale consists of amorphous SiO2 and the dispersion is Mo. There is also a Mo(ss) interlayer between SiO2 oxide scale and substrate, as shown in Fig.3(d).
For 100 h exposure, Mo-12.5Si-25B alloy forms sound and continuous scale about 50-75 μm in thickness as shown in Fig.3(e). Mo-14Si-28B alloy forms sound and continuous scale about 40-45 μm in thickness as shown in Fig.3(f). The oxide scales of Mo-12.5Si-25B and Mo-14Si-28B alloys are composed mainly of amorphous SiO2, XRD results confirm this as shown in Fig.4(d) and Fig.5(d). There is a MoO2 layer adjacent to SiO2 oxide scale. Between the oxide scale and substrate there is an Mo(ss) interlayer, and the interlayer of Mo(ss) provides evidence that the oxidation interface is enriched in molybdenum due to the selective oxidation of silicon over the substrate during passivating scale growth.
The data on cyclic oxidation kinetics and the oxide scale microstructures indicate two distinct oxidation states, which correspond to two stages. One stage is transient state; the oxidation is most likely to be controlled by the formation and volatilization of primarily MoO3. The other is steady state; the formation of a continuous B-SiO2 scale begins to provide a degree of protection, which increases with increasing oxidizing time. This transient and steady state oxidation behavior has also been observed in Mo5Si3- and Mo5SiB2-based alloys [10,14].
When the alloy is exposed to air at 1 200 ℃, at the beginning, oxygen immediately diffuses through grain boundaries and reacts with the Mo, T2, Mo3Si, T1 and MoB phases, forming SiO2, B2O3 and molybdenum oxides (e.g., MoO3 and MoO2). With time increasing, the mass of the samples rapidly decreases due to the volatilization of MoO3 and B2O3 in the transient oxidation period. Simultaneously, a porous B-SiO2 scale is left on the surface. For the alloys in the present investigation, the T2 and MoB phases are the source of B, which can significantly lower the viscosity and increase the flow of the resulting glass silicate. Hence, it appears that the more boron-added Mo-14Si-28B alloy easily forms a protective scale at high temperatures. If viscous flow occurs to close pores, a coherent passivating layer does form, i.e., the change of transient to steady state occurs, and then the steady state oxidation rate will be controlled by the diffusion rate of O2 through the passivating layer. If there is a sound and continuous silica scale on the surface, the partial pressure of oxygen at the interface between the oxide scale and substrate is considerably reduced from that in the ambient atmosphere. Under a low partial pressure of oxygen, selective oxidation of Si occurs and the oxide scale is protective.
According to MEYER and AKINC[11], a higher B/Si molar ratio in the Mo-Si-B alloys is beneficial to the non-porous protective oxide scale at elevated temperatures. In the present study, the molar ratio of B/Si is 2 for both alloys. Since there is no difference in the B/Si molar ratio of the presently studied alloys, the different oxidation behaviors of both alloys can be considered from the individual phase components of each alloy. The matrix of both alloys is composed of T2. However, Mo-12.5Si-25B alloy contains dispersive Mo and Mo3Si, while Mo-14Si-28B alloy contains Mo5Si3 and MoB as dispersions. It has been reported that B-doped Mo5Si3 has good oxidation resistance comparable to MoSi2[10]. MEYER et al[11] have also found that compositions with more Mo3Si in the microstructure have a larger degree of molybdenum oxide formation in the scale. As a result, the scales contain more pores than those of samples with little or no Mo3Si in the microstructure. Therefore, the protective scale consisting of B-SiO2 is easily formed on the surface of Mo-14Si-28B alloy in comparison with Mo-12.5Si-25B alloy.
4 Conclusions
1) The microstructure of Mo-12.5Si-25B alloy consists of a 3-phase Mo-T2-Mo3Si eutectic embedded in T2 matrix with discrete Mo particles of the order of 1 μm distributed in the matrix. The microstructure of Mo-14Si-28B alloy is composed of T2 as matrix with T1 and MoB as dispersions.
2) The cyclic oxidation kinetics data exhibit that the oxidation resistance of the Mo-12.5Si-25B and Mo-14Si-28B alloys at 1 000 ℃ is very close. However, Mo-14Si-28B alloy has better oxidation resistance than Mo-12.5Si-25B alloy at 1 200 and 1 300 ℃.
3) For 10 min exposure, Mo-12.5Si-25B and Mo-14Si-28B alloys form irregular and porous thin scale, and the oxide scale is composed mainly of crystalline and amorphous SiO2. For 2 h exposure, Mo-12.5Si-25B alloy forms irregular thin scale and the scale contains large cracks; while Mo-14Si-28B alloy forms sound and continuous scale. After 100 h exposure, Mo-12.5Si-25B and Mo-14Si-28B alloys form sound and continuous scale about 50-75 μm and 40-45 μm in thickness, respectively. The oxide scales of both alloys consist of amorphous SiO2, there is an Mo solid solution interlayer between oxide scale and substrate.
4) The better oxidation resistance of Mo-14Si-28B alloy is due to a sound and continuous B-SiO2 layer formation in the early stage of oxidation.
References
[1] SAKIDJA R, RIOULT F, WERNERB J, PEREPEZKO J H. Aluminum pack cementation of Mo-Si-B alloys [J]. Scripta Materialia, 2006, 55: 903-906.
[2] SCHNEIBEL J H, RITCHIE R O, KRUZIC J J, TORTORELLI P F. Optimization of Mo-Si-B intermetallic alloys [J]. Metallurgical and Materials Transactions, 2005, A36(3): 525-531.
[3] MITRA R, SRIVASTAVA A K, PRASAD N E, KUMARI S. Microstructure and mechanical behaviour of reaction hot pressed multiphase Mo-Si-B and Mo-Si-B-Al intermetallic alloys [J]. Intermetallics, 2006, 14: 1461-1471.
[4] SADANANDA K, FENG C R, MITRA R, DEEVI S C. Creep and fatigue properties of high temperature silicides and their composites [J]. Mater Sci Eng A, 1999, A261: 223-238.
[5] ZHANG F, ZHANG L T, SHAN A D, WU J S. Oxidation of stoichiometric poly- and single-crystalline MoSi2 at 773 K [J]. Intermetallics, 2006, 14: 406-411.
[6] LIU Y Q, SHAO G, TSAKIROPOULOS P. On the oxidation behaviour of MoSi2 [J]. Intermetallics, 2001, 9: 125-136.
[7] CHOU T C, NIEH T G. Kinetics of MoSi2 pest during low- temperature oxidation [J]. J Mat Res, 1993, 8(7): 1605-1610.
[8] ANTON D, SHAW D. High temperature properties of refractory intermetallics [J]. Mat Res Soc Symp Proc, 1991, 213: 733-739.
[9] BARTLETT R W, MCCAMONT J W, GAGE P R. Structure and chemistry of oxide films thermally grown on molybdenum silicides [J]. J Am Ceram Soc, 1965, 48(11): 551-558.
[10] MEYER M K, AKINC M. Oxidation behavior of boron-modified Mo5Si3 at 800-1 300 ℃[J]. J Am Ceram Soc, 1996, 79(4): 938-944.
[11] MEYER M K, THOM A J, AKINC M. Oxide scale formation and isothermal oxidation behavior of Mo-Si-B intermetallics at 600- 1 000 ℃[J]. Intermetallics, 1999, 7: 153-162.
[12] ITO K, IHARA K, TANAKA K, FUJIKURA M, YAMAGUCHI M. Physical and mechanical properties of single crystals of the T2 phase in the Mo-Si-B system [J]. Intermetallics, 2001, 9: 591-602.
[13] HAYASHIA T, ITO K, IHARA K, FUJIKURA M, YAMAGUCHI M. Creep of single crystalline and polycrystalline T2 phase in the Mo-Si-B system [J]. Intermetallics, 2004, 12: 699-704.
[14] YOSHIMI K, NAKATANI S, SUDA T, HANADA S, HABAZAKI H. Oxidation behavior of Mo5SiB2-based alloy at elevated temperatures [J]. Intermetallics, 2002, 10: 407-414.
[15] NUNES C A, SAKIDJA R, DONG Z, PEREPEZKO J H. Liquidus projection for the Mo-rich portion of the Mo-Si-B ternary system. Intermetallics, 2000, 8: 327-337.
[16] MENDIRATTA M G, PARTHASARATHY T A, DIMIDUK D M. Oxidation behavior of aMo-Mo3Si-Mo5SiB2 (T2) three phase system [J]. Intermetallics, 2002, 10: 225-232.
[17] SUPATARAWANICH V, JOHNSON D R, LIU C T. Effects of microstructure on the oxidation behavior of multiphase Mo-Si-B alloys [J]. Mater Sci Eng, 2003, A344: 328-339.
[18] CHOU T C, NIEH T G. Mechanism of MoSi2 pest during low temperature oxidation [J]. J Mat Res, 1993, 8(1): 214-225.
(Edited by LI Xiang-qun)
Foundation item: Project(2003AA305750) supported by the National High Technology Research and Development Program of China; Project (NCET- 04-0386) supported by the New Century Excellent Talents in University, China
Corresponding author: SHAN Ai-dang; Tel: +86-21-34203092; E-mail: adshan@sjtu.edu.cn