
Crystallization kinetics of bulk Cu58.1Zr35.9Al6 alloy
WANG Shao-xu(王韶旭)1, 2, ZHU Chun-lei(朱春雷)1, QUAN Shi-guang(全世光)1, LI Yan- hui(李艳辉)1,
WANG Ying-min(王英敏)1, WANG Qing(王 清)1, DONG Chuang(董 闯)1
1. Key Laboratory of Materials Modification by Laser, Ion and Electron Beams,
Dalian University of Technology, Dalian 116024, China;
2. College of Environmental Science and Engineering, Dalian Jiaotong University, Dalian 116028, China
Received 10 August 2009; accepted 15 September 2009
Abstract: The crystallization kinetics of the bulk amorphous Cu58.1Zr35.9Al6 alloy was examined by differential scanning calorimetry under continuous heating and isothermal annealing. During continuous heating, the activation energy of crystallization was determined to be 383 kJ/mol by Kissinger method. However, on the isothermal annealing, the activation energy was determined to be 459.2 kJ/mol by the Arrhenius method, which was much larger than that obtained from the Kissinger method. The different temperatures at which crystallization occurs are responsible for the discrepancy in the activation energy. The average Avrami exponent of about 3.5 implies that the crystallization process of the bulk amorphous Cu58.1Zr35.9Al6 alloy is diffusion-controlled with a nucleation rate decreasing with time.
Key words: Cu58.1Zr35.9Al6 alloy; different scanning calorimetry (DSC); crystallization kinetics
1 Introduction
Among a large number of amorphous alloys, Cu-based alloys have attracted much attention due to their higher fracture strength, elastic modulus, and lower cost. Therefore, great efforts have been devoted to the development of Cu-based amorphous alloys. In recent years, INOUE et al[1-4] has succeeded in fabricating new Cu-based BMGs in Cu-(Zr, Hf)-Ti and Cu-(Zr, Hf)-Al systems containing more than 50% Cu(molar fraction). Much recently, the authors have developed a new Cu-Zr-Al amorphous alloy system with a good glass forming ability[5]. The optimum glass-forming composition in this alloy system was found to occur in the Cu58.1Zr35.9Al6 alloy.
Crystallization kinetics is very important for the development of amorphous alloys. Study of crystallization kinetics has significant influence on understanding mechanism of non-equilibrium phase transformation and evaluation of glass forming ability (GFA). However, too limited information is available to date for understanding the crystallization kinetics of Cu-based BMGs. Therefore, an attempt has been made to study the crystallization kinetics of Cu58.1Zr35.9Al6 alloy. Differential scanning calorimetry (DSC) has been employed for such an investigation under non-isothermal and isothermal conditions.
2 Experimental
Master ingot of the Cu58.1Zr35.9Al6 alloy was prepared by using arc melting the mixtures of constituent elements under argon atmosphere. The purities of elements are 99.99% for Cu, 99.9% for Zr and 99.999% for Al, respectively. Alloy rod with a diameter of 3 mm was prepared by means of copper mould suction casting, and the details on the samples preparation were described in Ref.[6]. Structural identification of the alloy was carried out by means of X-ray diffractometry (XRD) on the bottom of the rods using the Cu Kα radiation. Both isochronal and isothermal differential scanning calorimetry (DSC) measurements were carried out using a TA Q100. In the case of continuous heating, rates of 10, 20, 30, 40 K/min were adopted to heat the samples, respectively. For the isothermal DSC analysis, the amorphous samples were first heated to a fixed temperature between 768 and 788 K at a rate of 100 K/min, and then held for a certain period until the crystallization was completed.
3 Results and discussion
X-ray diffraction result shown in Fig.1 confirmed that the as-cast Cu58.1Zr35.9Al6 alloy was amorphous.

Fig.1 XRD pattern of bulk amorphous Cu58.1Zr35.9Al6 alloy
3.1 Non-isothermal analysis
Fig.2 shows the continuous heating DSC curves for the Cu58.1Zr35.9Al6 alloy at heating rates of 10, 20, 30 and 40 K/min. An exothermic event corresponding to crystallization of the amorphous sample was observed for each heating rate. It is obvious that an increase in the heating rate results in an increase in the transformation temperature. The activation energy for crystallization was evaluated by the Kissinger equation[7]:
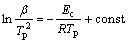 (1)
(1)
where β is the heating rate; Tp is the peak temperature and R is the gas constant. A straight line was obtained by fitting  vs 1/Tp, as shown in Fig.3. The activation energy calculated for crystallization of the Cu58.1Zr35.9Al6 alloy was 383 kJ/mol.
vs 1/Tp, as shown in Fig.3. The activation energy calculated for crystallization of the Cu58.1Zr35.9Al6 alloy was 383 kJ/mol.

Fig.2 DSC curves of bulk amorphous Cu58.1Zr35.9Al6 alloy at different heating rates
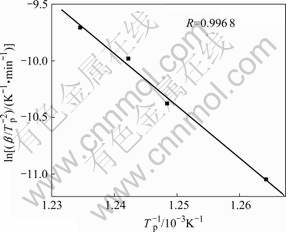
Fig.3 Plot of Kissinger equation for crystallization in bulk amorphous Cu58.1Zr35.9Al6 alloy
3.2 Isothermal analysis and crystallization kinetics
Based on the experimental results of the isochronal DSC measurements, we performed the isothermal DSC measurement of the as-cast Cu58.1Zr35.9Al6 alloy in the temperature range from 768 to 788 K. The results are shown in Fig.4(a). In all DSC traces, a single peak was observed after passing a certain incubation time. Thus, this transformation can be thought to be iso-kinetic.
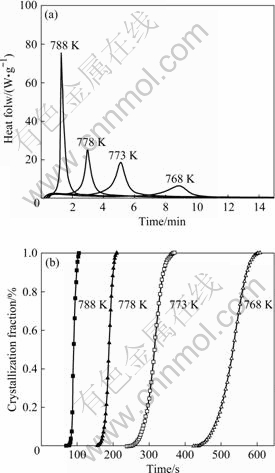
Fig.4 Isothermal DSC curves (a) and crystallization fraction vs annealing time curves (b) for bulk amorphous Cu58.1Zr35.9Al6 alloy at different annealing temperature
The isothermal crystallization kinetics of amorphous alloys is normally analyzed using the Johnson-Mehl-Avrami (JMA) equation[8-10]:
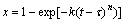 (2)
(2)
where x is the crystallized volume fraction, t the annealing time, τ the incubation time for nucleation, k the reaction rate constant which is a function of annealing temperature, and n the Avrami exponent which is related to the underlying crystallization mechanisms.
The crystallized volume fraction x can be accurately determined by measuring the partial area of peak. The results are exhibited in Fig.4(b). The crystallized volume fraction as a function of time displayed a classical sigmoidal type. The reaction rate constant k can be expressed as
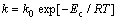 (3)
(3)
where k0 is the frequency factor, which is a measure of the probability that a molecule having energy Ec will participate in a reaction; and Ec is the apparent activation energy for crystallization. The value of k and n can be obtained as follows[11]:
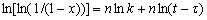 (4)
(4)
By plotting ln[ln(1/(1-x))] against ln(t-τ) at different annealing temperatures, the JMA plots were obtained, as shown in Fig.5. The data for 20%<x<80% gave a nearly straight line. The value of Avrami exponent n and the reaction constant k can be calculated from the slope and the intercept of the straight line in Fig.5. Table 1 presents the kinetic parameters of the Cu58.1Zr35.9Al6 alloy isothermally annealed in the supercooled liquid region. From Table 1, the Avrami exponents vary from 3.29 to 3.75 for different annealing temperatures. The average Avrami exponent of about 3.5 implied that the crystallization process of the bulk amorphous Cu58.1Zr35.9Al6 alloy was a diffusion-controlled continuous nucleation growth with constant nucleation rate[12]. Fig.6 shows the plot of ln k vs 1/T, which yields a straight line. According to Eq.(3), the activation energy for crystallization is Ec=459.2 kJ/mol. It is worth noting that the average activation energy obtained by using Arrhenius equation is much larger than that obtained by using Kissinger equation. YANG et al[13] have also observed this phenomenon and indicated that the activation energy derived from either Kissinger or Arrhenius plots might be temperature dependent. Compared with isothermal annealing, the sample subjected to continuous heating condition would crystallize at a relatively higher temperature. Crystallization at higher temperatures above onset temperature of crystallization makes it easier to transform from the amorphous state to crystalline phases.
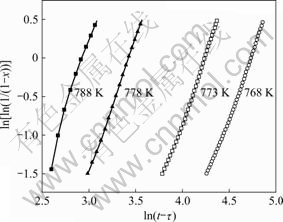
Fig.5 JMA plots for crystallization of bulk amorphous Cu58.1Zr35.9Al6 alloy at different annealing temperatures
Table 1 Kinetic parameters of bulk amorphous Cu58.1Zr35.9Al6 alloy isothermally annealed in supercooled liquid region
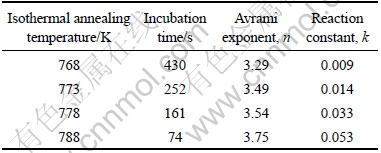
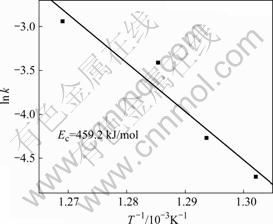
Fig.6 ln k vs 1/T curve to obtain activation energy of isothermal crystallization
The nucleation and growth do not remain constant during the whole crystallization process of an amorphous alloy, but exhibit different nucleation and growth behaviors at different stages. In order to investigate the details of the crystallization process, the local Avrami exponent n(x) has been calculated using the following equation[14]:
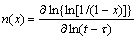 (5)
(5)
Fig.7 shows the value of local Avrami exponent at different temperatures with crystallization fraction. It can be seen that each curve displays obvious difference. At 788 K, the value n(x) is lower than 2 at the start, indicating one-dimensional growth with decreasing the nucleation rate; n(x) rapidly increases and reaches a maximum value of 6.8 at x=19%; then, it steadily decreases in the range of 20%<x<93% and reaches a value of about 3.0. This suggested that the nucleation is non-steady at 788 K. The three curves, corresponding to 778, 773 and 768 K, exhibit a similar tendency with crystallization fraction. By referring to the fact that n(x)≤2 at the beginning of the crystallization, the conclusion can be drawn that this crystallization process is mainly governed by one-dimensional growth with decreasing the nucleation rate and enormous number of short-range ordering clusters act as overcritical nuclei in the supercooled liquid region[15]. The growth of the overcritical nuclei and formation of new nuclei require simultaneous rearrangement of different species of atoms. Due to the dense randomly packed atomic configuration, diffusion of atoms is difficult, which retards the nucleation and growth, leading to a temporary decrease of nucleation rate. As the crystallization proceeds, n(x), increases and then falls in the range 2.0-4.0 until x reaches 90%, suggesting that crystallization is mainly controlled by continuous nucleation growth with constant nucleation rate. The growth of the initial nuclei changes the composition of the matrix surrounding the nuclei. The compositional fluctuation facilitates the nucleation adjacent to the growing nuclei, leading to a higher but nearly constant nucleation rate compared with the nucleation rate at the initial crystallization stage.
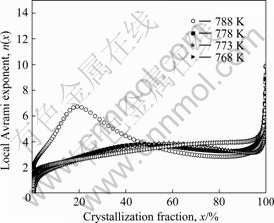
Fig.7 Local Avrami exponents with crystallization fraction for bulk amorphous Cu58.1Zr35.9Al6 alloy at different temperatures
The activation energy for different crystallization fractions is not constant in the whole transformation because of the difference of nucleation and growth behavior. Therefore, the local activation energy, Ec(x), has also been introduced, which can display the variation of activation energy at all crystallization stage[16]. In the case of isothermal annealing, the transformation time, t(x), is related to the annealing temperature by the Arrhenius equation[17]:
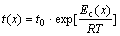 (6)
(6)
where t is the time required to crystallize to the volume fraction x, t0 is the time constant, and Ec is the activation energy for crystallization. The local activation energy can be obtained from the slope of ln[t(x)-τ] against 1/T. Fig.8 gives the relationship between crystallization fraction and local activation energy of amorphous Cu58.1Zr35.9Al6 alloy. It is seen that the Ec(x) steadily increases with x in the main crystallization process (0<x<65%) and reaches a maximum value of 466 kJ/mol at x=65%; then, it decreases slightly in the remainder crystallization process. The very large activation energy for crystallization in the supercooled liquid region under isothermal conditions again demonstrates the thermal stability of amorphous Cu58.1Zr35.9Al6 alloy is very high.
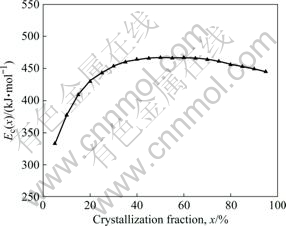
Fig.8 Relationship between crystallization fraction and Ec(x) of bulk amorphous Cu58.1Zr35.9Al6 alloy
4 Conclusions
1) During continuous heating, an increase in the heating rate resulted in an increase in the transformation temperature.
2) In the isothermal annealing, the activation energy was determined to be 459.2 kJ/mol by the Arrhenius method, which was much larger than that obtained from the Kissinger method. The different temperatures at which crystallization occurs are responsible for the discrepancy in the activation energy.
3) The average Avrami exponent of about 3.5 implied that the crystallization process of the bulk amorphous Cu58.1Zr35.9Al6 alloy was diffusion-controlled with a nucleation rate decreasing with time.
References
[1] INOUE A, ZHANG Wei. Formation, thermal stability and mechanical properties of Cu-Zr-Al bulk glassy alloys [J]. Materials Transaction, 2002, 43(11): 2921-2925.
[2] ZHANG Wei, INOUE A. Thermal stability and mechanical properties of Cu-based bulk glassy alloys in Cu50(Zr1-xHfx)45Al5 system [J]. Materials Transaction, 2003, 44(10): 2220-2223.
[3] INOUE A, ZHANG Wei, ZHANG Tao, KUROSAKA K. High-strength Cu-based bulk glassy alloys in Cu-Zr-Ti and Cu-Hf-Ti ternary systems [J]. Acta Materialia, 2001, 49(14): 2645-2652.
[4] INOUE A, ZHANG Wei. Formation and mechanical properties of Cu-Hf-Al bulk glassy alloys with a large supercooled liquid region of over 90 K [J]. Journal of Materials Research, 2003, 18(6): 1435-1440.
[5] WANG Qing, DONG Chuang, QIANG Jian-bing, WANG Ying-min. Cluster line criterion and Cu-Zr-Al bulk metallic glass formation [J]. Mater Sci Eng A, 2007, 449: 18-23.
[6] QIANG Jian-bing, WANG Ying-min, WANG Dong-hai, KRAMER M, THIEL P, DONG Chuang. Quasicrystals in the Ti-Zr-Ni alloy system [J]. Journal of Non-Crystalline Solids, 2004, 334: 223-227.
[7] KISSINGER H E. Reaction kinetics in differential thermal analysis [J]. Analytical Chemistry, 1957, 29(11): 1702-1706.
[8] MITTEMEIJER E J. Analysis of the kinetics of phase transformations [J]. Journal of Materials Science, 1992, 27(15): 3977-3987.
[9] CALKA A, RADLINSKI A P. Decoupled bulk and surface crystallization in Pd85Si15 glassy metallic alloys: Description of isothermal crystallization by a local value of the Avrami exponent [J]. Journal of Materials Research, 1988, 3(1): 59-66.
[10] AVRAMI M. Kinetics of phase change. I General theory [J]. The Journal of Chemical Physics, 1939, 7(12): 1103-1112.
[11] CHRISTIAN J W. The theory of transformation in metals and alloys [M]. New York: Pergamon Press Inc, 1975: 542-550.
[12] YUAN Zi-zhou, CHEN Xue-ding, WANG Bin-xia, CHEN Zi-jiang. Crystallization kinetics of melt-spun Co43Fe20Ta5.5B31.5 amorphous alloy [J]. Journal of Alloys and Compounds, 2005, 399: 166-172.
[13] YANG Y J, XING D W, SHEN J, SUN J E, WEI S D, HE H J, MCCARTNEY D G. Crystallization kinetics of a bulk amorphous Cu-Ti-Zr-Ni alloy investigated by differential scanning calorimetry [J]. Journal of Alloys and Compounds, 2006, 415: 106-110.
[14] BUSCHOW K H. Stability and electrical transport properties of amorphous Ti1-xNix alloys [J]. Journal of Physics F-Metal Physics, 1983, 13(3): 563-572.
[15] CHRISTIAN J W. The theory of transformation in metals and alloys [M]. Oxford: Pergamon Press Inc, 1965: 489-500.
[16] LIU K T, DUH J G. Kinetics of the crystallization in amorphous NiTi thin films [J]. Journal of Non-Crystalline Solids, 2007, 353: 1060-1064.
[17] WANG Xiao-dong, LEE H, YI S. Crystallization behavior of preannealed bulk amorphous alloy Zr62Al8Ni13Cu17 [J]. Materials Letters, 2006, 60(7): 935-938.
Foundation item: Project(20080431141) supported by China Postdoctoral Science Foundation
Corresponding author: DONG Chuang; Tel: +86-411-84708389; E-mail: dong@dlut.edu.cn
DOI: 10.1016/S1003-6326(09)60078-9
(Edited by YANG Bing)