��е�Ͻ��Ʊ�����þ���Ͻ�����ȶ���
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2016���2��
�������ߣ�M. RAJABI R. M. SEDIGHI S. M. RABIEE
����ҳ�룺398 - 405
�ؼ��ʣ�þ���Ͻ����ȶ��ԣ�������������е�Ͻ�
Key words��Mg-based alloys; thermal stability; grain growth; mechanical alloying
ժ Ҫ���о���е�Ͻ��Ʊ�������Mg-6Al-1Zn �� Mg-6Al-1Zn-1Si �Ͻ�����ȶ��Ժ;�����������ѧ���о���Ԫ�ط�ĩ��ʼ��ʹ���˸��ַ����ֶΣ�������ʾɨ�����ȷ�(DSC)��X��������(XRD)��ɨ��羵����羵�����������о������˻�����еľ�����������ѧ��XRD����������������ֲ�����ϵ�ľ����ߴ����˻��¶ȵ����߶����ӣ�����350 ��C�˻�1 h��Si��ϵ���и�С�ľ����ߴ�(60 nm)����Mg-6Al-1Zn��ϵ�ľ����ߴ�Ϊ72 nm��Mg-6Al-1Zn-1Si��ϵ�ڵ����˻�������γɵĵڶ�������仯����Mg2Si����Ӱ���ܣ�����Ӱ�춯��ѧ���̵�ָ������Si��ϵ��Ӳ�ȸ��������ڽ�������Mg2Si���γɡ�
Abstract: The thermal stability and the kinetics of grain growth of nanocrystalline Mg-6Al-1Zn and Mg-6Al-1Zn-1Si alloys prepared via mechanical alloying were investigated. It started with elemental powders, using a variety of analytical techniques including differential scanning calorimetry (DSC), X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM) and energy dispersive spectrometry. The kinetics of grain growth in isothermal annealing was investigated. The XRD results show that, although the grain sizes of both material systems increase as the annealing temperature rises, the Si-containing system displays a relatively smaller grain size, i.e., 60 nm compared with 72 nm in Mg-6Al-1Zn system, after being exposed to 350 ��C for 1 h. The second-phase intermetallic particle Mg2Si formed during the isothermal annealing of Mg-6Al-1Zn-1Si system could influence not only the activation energy but also the exponent of kinetic equation. Higher hardness values obtained in the Si-containing system would be due to the formation of Mg2Si intermetallic phase. Key words:Mg-based alloys; thermal stability; grain growth; mechanical alloying
Trans. Nonferrous Met. Soc. China 26(2016) 398-405
M. RAJABI, R. M. SEDIGHI, S. M. RABIEE
Department of Material Science and Engineering, Faculty of Mechanical Engineering, Babol Noshirvani University of Technology, P. O. Box 47148-71167, Shariati Street, Babol, Iran
Received 3 March 2015; accepted 20 November 2015
Abstract: The thermal stability and the kinetics of grain growth of nanocrystalline Mg-6Al-1Zn and Mg-6Al-1Zn-1Si alloys prepared via mechanical alloying were investigated. It started with elemental powders, using a variety of analytical techniques including differential scanning calorimetry (DSC), X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM) and energy dispersive spectrometry. The kinetics of grain growth in isothermal annealing was investigated. The XRD results show that, although the grain sizes of both material systems increase as the annealing temperature rises, the Si-containing system displays a relatively smaller grain size, i.e., 60 nm compared with 72 nm in Mg-6Al-1Zn system, after being exposed to 350 ��C for 1 h. The second-phase intermetallic particle Mg2Si formed during the isothermal annealing of Mg-6Al-1Zn-1Si system could influence not only the activation energy but also the exponent of kinetic equation. Higher hardness values obtained in the Si-containing system would be due to the formation of Mg2Si intermetallic phase.
Key words: Mg-based alloys; thermal stability; grain growth; mechanical alloying
1 Introduction
Mg alloys have been regarded as promising materials over the years because of their low density and high specific strength and stiffness [1,2]. However, Mg alloys are used limitedly in high performance applications due to their low mechanical properties [3]. Refining the grain size to nanostructure and in-situ alloying have been used in order to improve the mechanical properties [4,5]. Mechanical alloying (MA) is an appealing solid-state technique to synthesize powders with controlled microstructure. This technique can produce considerably fine grain sizes down to nano-scales with flexibility in alloying [6,7].
The thermal stability of nanocrystalline materials is of noteworthy scientific interest and of technological importance as well [8]. Since the grain growth of nanocrystalline materials occurs during the service or secondary process to form bulk alloys, recently, a wide range of studies have been carried out in this respect [8,9]. Research findings about the stability of grain sizes have been reported in various nanocrystalline Mg alloys, for example Mg-12.1Cu [3], Mg-5Al-10.3Ti [9] (mass fraction, %) and Mg-25%Si (mole fraction) [1]. Different mechanisms of nanograin stability were reported including remnant pore drag, grain boundary segregation, solute drag, second phase (Zener) drag and chemical ordering, all of which lead to the inhibition of grain growth [8,10,11]. To improve the heat resistance of Mg-based alloys, developing an alloy containing thermal stable intermetallic phases would be one of the most effective methods, which could suppress grain boundary sliding and their dislocation motion [1,12]. The intermetallic compound Mg2Si is a thermally stable phase with low density and high specific strength [1]. The production of Mg2Si by conventional methods tends to result in heterogeneous microstructure with undesirable coarse grains [4,12]. Solid-state synthesis of Mg2Si by mechanical alloying of elemental powders was also investigated [4,13]. However, it is usually difficult to synthesize Mg2Si by direct mechanical milling due to the stable oxide films formed on the Mg particles [4].
The present study focuses on the effect of Si addition on the thermal stability and kinetics of grain growth of nanocrystalline Mg-6Al-1Zn alloy prepared via mechanical alloying, being initiated by elemental powders.
2 Experimental
Elemental powders of Mg (>97%, Merck), Al (>98%, Merck), Zn (>99.9%, Merck) and Si (>98%, Sigma) were used as raw materials. The powders with nominal compositions of Mg-6Al-1Zn and Mg-6Al-1Zn-1Si (mass fraction, %) were mechanically alloyed in a Retsch PM100 planetary ball mill at room temperature for 50 h. The mass ratio of ball to powder of 20:1 was selected and the rotation speed was adjusted to 250 r/min. In order to prevent a large temperature increment during the process, the experiment was periodically stopped every 25 min for 5 min. Weighing, filling and handling the powder were performed in a glove box under argon atmosphere.
The thermal stability of milled powder was determined using a differential scanning calorimeter (DSC, NETZSCH STA 409 PC/PG) in argon atmosphere at five different heating rates of 5, 7.5, 10, 15 and 20 ��C/min. Based on the DSC results, MA-processed alloy powders were isothermally annealed at 350, 400 and 450 ��C for 1 h. To investigate the kinetics of grain growth, the mass ratio of e first 5 days isothermal annealing at 350, 400 and 450 ��C over 30, 60, 90, 120 and 180 min was used to calculate the grain growth rates. The phase constituents of as-milled powder before and after annealing were analyzed by X-ray diffraction (XRD, X/Pert Pro MPD, PANalytical) with Cu K�� radiation. The grain size of the powder was estimated by broadening XRD peaks using Williamson�CHall method [14]:
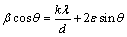 (1)
(1)
where �� is the full width at half maximum of a diffraction peak, �� is the Bragg angle, �� is the used X-ray wavelength and �� is the microstrain. The microstructures and morphologies of milled and annealed samples were characterized using scanning electron microscopy (SEM, KYKY EM-3200). The analyses of grain sizes and their chemical compositions within the microstructure of MA-processed powder particles were also done using TEM (Philips CM200 FEG) operated at 200 kV. In the TEM analysis, bright field image technique, selected area diffraction (SAD) and energy dispersive spectrometry (EDS) were used to characterize the powder particles. Hardness was measured by Vickers diamond indenter in a microhardness tester at the load of 0.098 N. The cross section of powder particles was provided by mounting a small amount of powder in a resin by conventional grinding and polishing methods. A total of 10 measures were taken for each sample and the average values were reported.
3 Results
3.1 Characteristics of powder
The XRD patterns of the powder mixture before and after milling for 50 h are presented in Fig. 1. As can be seen, after 50 h milling, all diffraction peaks expand and decrease evidently due to their mechanically-induced lattice strain and crystallite size reduction [15]. The absence of Zn and Si peaks in the XRD pattern could be due to their low concentrations. The peak shifting of Mg reflections to higher 2�� values was also observed. This suggests the formation of Mg-based solid solution during milling. Since the atomic sizes of Al, Zn and Si elements are smaller than that of Mg element, the dissolution of these elements is expected to reduce the lattice parameter of Mg. The appearance of XRD peaks of MgAl2O4 determines the oxidation of Mg during the process. Weak Al peaks are noticeable in both MA-processed samples, which suggests the incomplete solubility of Al in the Mg matrix. Figure 2 shows the sizes and morphologies of powder particles unmilled and milled for 50 h. Based on Fig. 2, after 50 h milling, the particles fragment become considerably small in size and granular in shape. No significant difference in the size and morphology between the Si-containing powder and Mg-6Al-1Zn is seen.

Fig. 1 XRD patterns of Mg-6Al-1Zn powder mixture before milling (a), milled Mg-6Al-1Zn (b) and Mg-6Al-1Zn-1Si (c)

Fig. 2 SEM images of Mg-6Al-1Zn powder mixture before milling (a), milled Mg-6Al-1Zn (b) and Mg-6Al-1Zn-1Si (c)
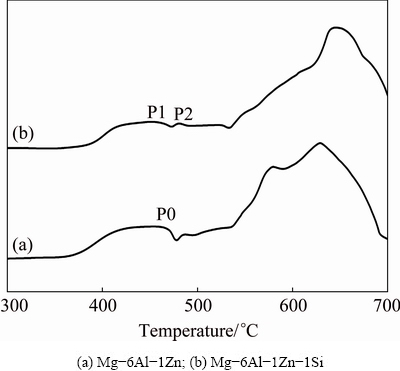
Fig. 3 DSC curves of as-milled powders with heating rate of 10 ��C/min
3.2 Structural evolution at elevated temperatures
For a thorough study of thermal behavior, the DSC analyses of as-milled powders were carried out at five different heating rates, 5, 7.5, 10, 15 and 20 ��C/min. Figure 3 compares the DSC curves of as-milled powders with a heating rate of 10 ��C/min. Considering the peaks observed at temperatures less than 500 ��C, it should be mentioned that in both DSC curves, broad exothermic peaks (P0 and P1) occur between 360 and 470 ��C. The onset and peak temperatures shift to higher temperatures with the increase of heating rate which is not shown in Fig. 3. This exothermic peak, observed in many MA-processed powder systems, may originate from the grain growth, strain relaxation and decomposition of metastable solid solution into component elements [16,17]. In Mg-6Al-1Zn-1Si system, there is an exothermic peak (P2) at ~480 ��C, which is not observed in Mg-6Al-1Zn system.
To understand the reactions noticed in the DSC curves shown in Fig. 3, isothermal annealing was performed. Figure 4 shows the XRD patterns after annealing at 350, 400 and 450 ��C for 1 h. For comparison, the corresponding pattern of as-milled powder is also shown. As can be seen, as the annealing temperature increases, the breadth of fundamental diffraction peaks decreases compared with the result of as-milled powder. In Mg-6Al-1Zn-1Si system, new diffraction peaks corresponding to Mg2Si intermetallic phase are also considerable. Figure 5 shows the typical XRD patterns of MA-processed powders after isothermal annealing at 350 ��C for different durations. It can be notified that the diffraction peaks of Mg17Al12 are evident in both systems after 30 min. Additionally, the characteristics peaks of MgAl2O4 are also observed, with their intensities increasing at longer annealing duration. In Mg-6Al-1Zn-1Si system, the peaks corresponding to Mg2Si phase are detected.
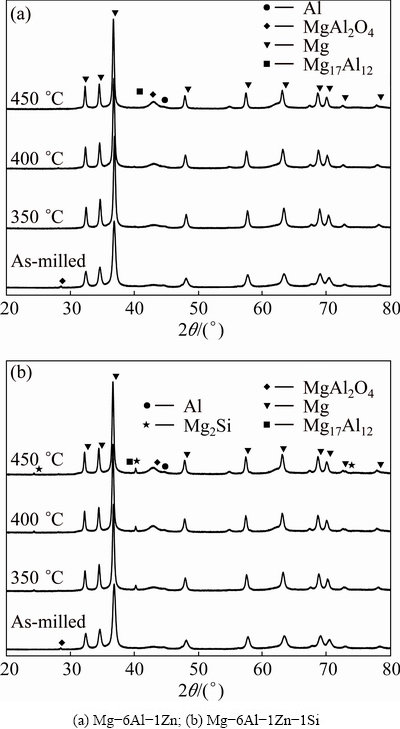
Fig. 4 XRD patterns of MA-processed powders as function of annealing temperature
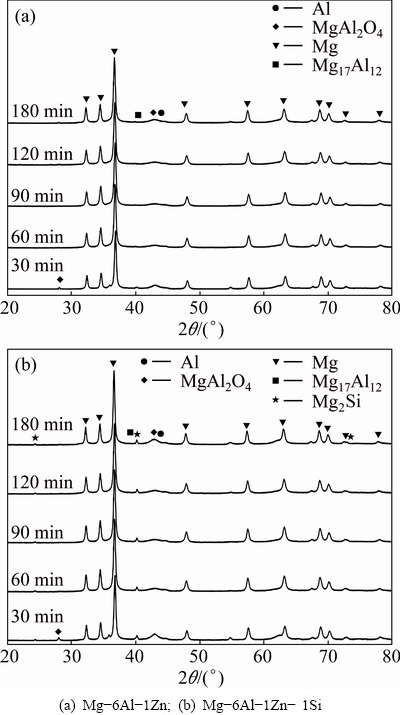
Fig. 5 XRD patterns of MA-processed powders as function of annealing time at 350 ��C
The hardness values of MA-processed powder particles derived from annealing temperature are shown in Fig. 6. It can be seen that the hardness decreases as the annealing temperature increases. However, the Si-containing system shows a relatively higher hardness value at all annealing temperatures compared with Al-6Al-1Zn system.
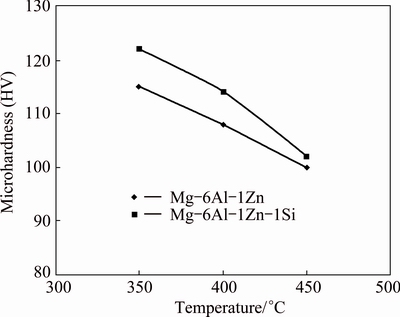
Fig. 6 Microhardness values of MA-processed powders as function of annealing temperature
4 Discussion
4.1 Mechanism of Mg2Si formation
At equilibrium state, the intermetallic phase of Mg2Si formed by adding Si to Mg [18,19]. The results of the present investigation illustrate that Mg2Si intermetallic compound does not form in as-milled Mg-Al-Zn-Si system, which is in agreement with the previous reports [4,20]. The possible reasons include the limitation of XRD in detecting the phases at low volume fractions (<5% [21]), and the formation of stable oxide films on the Mg particles which prevents Si from being alloyed directly with Mg. In general, the direct synthesis of Mg2Si by mechanical milling at temperatures much lower than the melting point of Mg would be difficult, as stated by WANG and QIN [4] for the formation of nanocrystalline Mg2Si through solid-state reaction. However, the mechanical milling is able to refine microstructures and represent defects, which would facilitate the subsequent solid-state reaction of Mg and Si. On the other hand, as found out, the mechanical milling can facilitate the reaction of solid-state to form Mg2Si in the subsequent annealing. According to the DSC and XRD results (Figs. 3 and 4), the exothermic peak P0 would be related to the formation of Mg17Al12 phase in Mg-6Al-1Zn system, whereas the exothermic peak P1 in Mg-6Al-1Zn-1Si system is more likely to be attributed to the formation of Mg2Si and Mg17Al12 phases. Moreover, the exothermic peak P2 in Mg-6Al-1Zn-1Si system could be related to the completion of formation of Mg2Si due to the reaction of residual Mg and Si at higher temperature, as no formation of new phase is observed in the XRD patterns. It is shown that the reaction of Mg and Si and the formation of Mg2Si would take place at very high temperatures due to the oxide films (MgO and/or SiO2) covering the particles [4]. However, as known, the processes such as mechanical alloying change the formation temperature of Mg2Si. SUN et al [1] concluded that at 545 ��C, the synthesis of in-situ Mg2Si compound from Mg and Si elemental powders could be formed. According to WANG and QIN [4], after 60 h milling, the formation temperature of Mg2Si decreases from ~460 ��C to ~180��C in Mg-37%Si system. As the powder mixtures were processed through high-energy milling, the followings would happen: 1) grain refinement and defect generation; 2) the crack of oxide films; 3) in-situ alloying of the elements. When the oxide films crack as a result of milling, the fresh surfaces would lead to direct contact of Mg with Si, consequently, Mg would begin to react with Si at lower temperature for the milled powder.
4.2 Kinetics of grain growth
For the study of grain growth, the grain size of ��-Mg affected by temperature annealing for 1 h was estimated using Williamson-Hall method, as shown in Fig. 7. Although the grain sizes of both material systems increase as the annealing temperature rises, the Si- containing system shows a smaller grain size. To investigate the behavior of grain growth meticulously, TEM was used. The TEM bright-field images of MA-processed alloys after being exposed to 350 ��C for 1 h are shown in Fig. 8. The average grain sizes of Mg-6Al-1Zn and Mg-6Al-1Zn-1Si systems are about 95 and 83 nm, respectively. These average grain size values are relatively similar to those determined by XRD being 72 and 60 nm in Mg-6Al-1Zn and Mg-6Al-1Zn-1Si systems, respectively. However, the size differences in both methods could be attributed to the influence of strain on the XRD results.
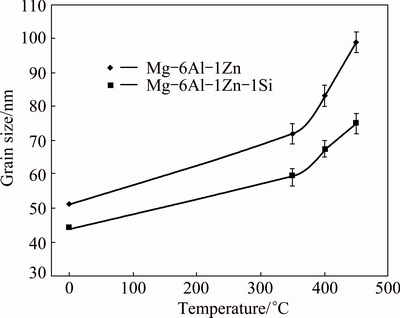
Fig. 7 Grain size of ��-Mg as function of temperature annealing for 1 h

Fig. 8 TEM bright-field images of MA-processed Mg alloys after being exposed to 350 ��C for 1 h
Figure 9 shows the grain sizes as a function of annealing time at different temperatures. It should be noted that the grain size of ��-Mg changes relatively largely up to 60 min, but starts to decrease gradually after 60 min, and finally reaches 114 and 87 nm after being exposed to 450 ��C over 180 min in Mg-6Al-1Zn and Mg-6Al-1Zn-1Si systems, respectively.
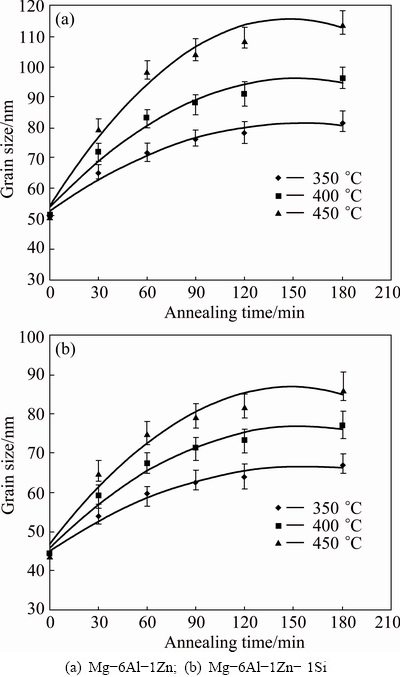
Fig. 9 Grain size of ��-Mg as function of annealing time at different temperatures

Fig. 10 Plot of lg(dD/dt) vs lg D for Mg-6Al-1Zn (a) and Mg-6Al-1Zn-1Si (b)
The parabolic kinetic equation of grain growth which can be used in some nanocrystalline materials for isothermal annealing is [22]
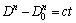 (2)
(2)
where D is the grain size, D0 is the initial grain size at the annealing time t=0, t is the annealing time, n is the grain growth exponent and c is a constant. The logarithm of grain growth rates extracted from the tangents of the respective curves in Fig. 9 was plotted against the logarithm of D (Fig. 10). The linear relationships are obtained with slopes of -8.39, -7.45, -7.29 for Mg- 6Al-1Zn and -9.47, -8.46, -8.40 for Mg-6Al-1Zn-1Si system annealed at 350, 400 and 450 ��C, respectively. The rate of grain growth can be calculated by
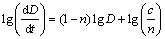 (3)
(3)
The grain growth exponent n at 350, 400 and 450 ��C can be estimated to be 9, 8, 8 in Mg-6Al-1Zn system and 10, 9, 9 in Mg-6Al-1Zn-1Si system, respectively. Figure 11 shows the lg(D9-D09) versus lg t plot in Mg-6Al-1Zn system and lg(D10-D010) versus lg t plot in Mg-6Al-1Zn-1Si at 350 ��C. As seen, the linear relationships are observed and the grain growth can be represented as Dn�CD0n =ct.
To obtain the activation energy, the DSC results from different scanning rates are analyzed using Kissinger equation [23]:
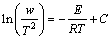 (4)
(4)
where w is the heating rate in the DSC scanning, T is the peak temperature of exotherm, E is the activation energy, R is the gas constant and C is a constant. By the linear fit of the experimental data to Eq. (4), the activation energies corresponding to P0, P1 and P2 would be 168.2, 356.7 and 309.8 kJ/mol, respectively. Comparing the activation energies corresponding to P0 and P1, a considerable increase of activation energy of Si-containing alloy can be observed. According to SUN et al [1], the activation energy of 376 kJ/mol would cause the solid state synthesis of Mg2Si at 445 ��C in Mg-25%Si alloy. The value of (190��5) kJ/mol at 540 ��C was reported by WANG and QIN [4] for the synthesis of Mg2Si from blended Mg and Si powders according to the stoichiometric composition of Mg2Si. In general, higher value of grain growth exponent, n, and the sharp increase of activation energy in Mg-6Al-1Zn-1Si system compared with those in Mg-6Al-1Zn can be due to the presence of Mg2Si intermetallic phase, which would disperse homogeneously in the Mg matrix, as shown in Fig. 12. It can be noticed that at higher temperature, the grain growth takes place, which would lead to the release of grain boundary energies and contribute partly to the DSC peaks [4]. However, the effect of grain boundary energies on the DSC peaks is withdrawn.
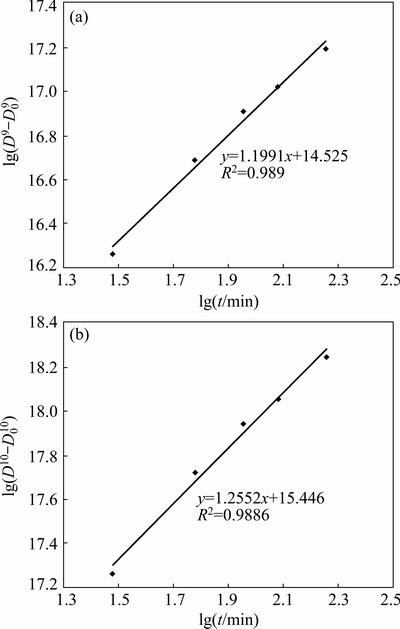
Fig. 11 Plot of lg(Dn-D0n) vs lg t at 350 ��C for Mg-6Al-1Zn (a) and Mg-6Al-1Zn-1Si (b)
The grain growth inhibition associated with dispersed second-phase particles has been reported in a wide variety of non-ferrous metals. The typical examples can be Mg and Al alloys [8,9]. According to ZENER [24], the resistance force, P, of secondary phase against the grain growth is proportional to the volume fraction of intermetallic particles, ��, and converse to the size of dispersoids, r, as
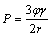 (5)
(5)
where is the interfacial energy.
is the interfacial energy.
Therefore, the dispersion of second-phase reduces the rate of grain growth because the driving pressure of the grain growth is compensated by the pinning force of particles [25,26]. A relatively higher hardness value is achieved in Mg-6Al-1Zn-1Si system compared with that in Mg-6Al-1Zn system at all annealing temperatures due to finer grain sizes and the formation of Mg2Si intermetallic phase.
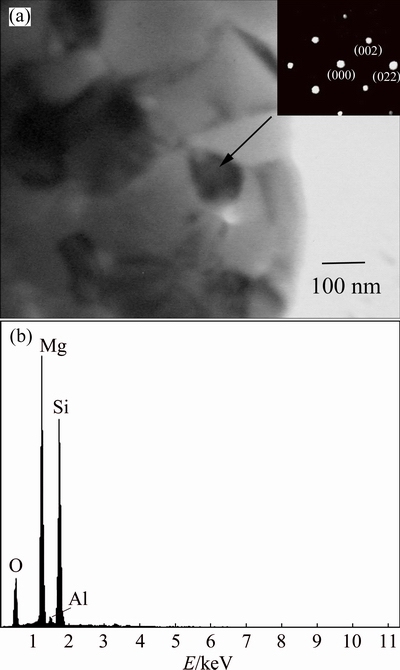
Fig. 12 TEM bright field image (a) (where inset shows SAD pattern) and EDS spectrum (b) of intermetallic phase for 50 h-milled powder annealed at 450 ��C for 1 h
5 Conclusions
1) The grain sizes of both material systems increase as the annealing temperature increases.
2) The grain growth behavior can be described by the parabolic kinetic equation of grain growth.
3) Higher value of grain growth exponent, n, and a spectacular increase of activation energy are observed in Mg-6Al-1Zn-1Si system compared with those in Mg-6Al-1Zn system due to the presence of Mg2Si intermetallic phase. This would lead to an increase of hardness of Si-containing alloy at all annealing temperatures.
References
[1] SUN B, LI S, IMAI H, UMEDA J, KONDOH K. Synthesis kinetics of Mg2Si and solid-state formation of Mg-Mg2Si composite [J]. Powder Technology, 2012, 217: 157-162.
[2] HUANG Zheng-hua, QI Wen-jun, XU Jing, CAI Chang. Microstructures and mechanical properties of Mg-Al-Sm series heat-resistant magnesium alloys [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(1): 22-29.
[3] LU L, THONG K K, GUPTA M. Mg-based composite reinforced by Mg2Si [J]. Composite Science and Technology, 2003, 63(5): 627-632.
[4] WANG L, QIN X Y. The effect of mechanical milling on the formation of nanocrystalline Mg2Si through solid-state reaction [J]. Scripta Materialia, 2003, 49(3): 243-248.
[5] XU Yan, HU Lian-xi, SUN Yu, JIA Jian-bo, JIANG Ju-fu, MA Qing-guo. Microstructure and mechanical properties of AZ61 magnesium alloy prepared by repetitive upsetting-extrusion [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(2): 381-388.
[6] FATHY A, WAGIH A, EL-HAMID A A, HASSAN A A. The effect of Mg add on morphology and mechanical properties of Al-xMg/10Al2O3 nanocomposite produced by mechanical alloying [J]. Advanced Powder Technology, 2014, 25(4): 1345-1350.
[7] WANG C, JI W, FU Z. Mechanical alloying and spark plasma sintering of CoCrFeNiMnAl high-entropy alloy [J]. Advanced Powder Technology, 2014, 25(4): 1334-1338.
[8] SHAW L, LUO H, VILLEGAS J, MIRACLE D. Thermal stability of nanostructured Al93Fe3Cr2Ti2 alloys prepared via mechanical alloying [J]. Acta Materialia, 2003, 51(9): 2647-2663.
[9] THEIN M A, LU L, LAI M O. Kinetics of grain growth in nanocrystalline magnesium-based metal�Cmetal composite synthesized by mechanical alloying [J]. Composite Science and Technology, 2006, 66(3-4): 531-537.
[10] MORRIS D G, MORRIS M A. Microstructure and strength of nanocrystalline copper alloy prepared by mechanical alloying [J]. Acta Metallurgica et Materialia, 1991, 39(8): 1763-1770.
[11] MALOW T R, KOCH C C. Grain growth in nanocrystalline iron prepared by mechanical attrition [J]. Acta Materialia, 1997, 45(5): 2177-2186.
[12] GUANGYIN Y, MANPING L, WENJIANG D, INOUE A. Mechanical properties and microstructure of Mg-Al-Zn-Si-base alloy [J]. Materials Transaction, 2003, 44(4): 458-462.
[13] XIONG Wei, QIN Xiao-ying, KONG Ming-guang, CHEN Li. Synthesis and properties of bulk nanocrystalline Mg2Si through ball-milling and reactive hot-pressing [J]. Transactions of Nonferrous Metals Society of China, 2006, 16(5): 987-991.
[14] KLUG H P, ALEXANDER L E. X-ray diffraction procedures for polycrystalline and amorphous materials [M]. 2nd ed. New York: John Wiley and Sons Inc, 1974.
[15] VIJAY R, SUNDARESAN R, MAIYA M P, SRINIVASA MURTHY S. Comparative evaluation of Mg-Ni hydrogen absorbing materials prepared by mechanical alloying [J]. International Journal of Hydrogen Energy, 2005, 30(5): 501-508.
[16] ZENG Q, BAKER I, MCCREARY V, YAN Z. Soft ferromagnetism in nanostructured mechanical alloying FeCo-based powders [J]. Journal of Magnetism and Magnetic Materials, 2007, 318(1-2): 28-38.
[17] LUO Z, LI Z Q. A W�CIn solid solution synthesized by mechanical alloying [J]. Materials Science and Engineering A, 2007, 459(1-2): 47-51.
[18] YUAN G Y, LIU Z L, WANG Q D, DING W J. Microstructure refinement of Mg-Al-Zn-Si alloys [J]. Materials Letters, 2002, 56(1-2): 53-58.
[19] REN Yu-ping, QIN Gao-wu, PEI Wen-li, LI Song, GUO Yun, ZHAO Hong-da. Phase equilibria of Mg-rich corner in Mg-Zn-Al ternary system at 300 ��C [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(2): 241-245.
[20] KONDOH K, OGINUMA H, YUASA E, AIZAWA T. Solid-state synthesis of Mg2Si from Mg-Si mixture powder [J]. Materials Transaction, 2001, 42(7): 1293-1300.
[21] UNLU N, GENC A, OVECOGLU M L, ERUSLU N, FROES F H. Characterization investigations of melt-spun ternary Al-xSi-3.3Fe (x=10, 20 wt.%) alloys [J]. Journal of Alloys and Compounds, 2001, 322(1-2): 249-256.
[22] LU K. Nanocrystallinemetals crystallized from amorphous solids: Nanocrystallization, structure, and properties [J]. Journal of Materials Science and Engineering, 1996, R16(4): 161-221.
[23] KISSINGER H E. Reaction kinetics in differential thermal analysis [J]. Analytical Chemistry, 1957, 29(11): 1702-1706.
[24] ZENER C. Private communication to C.S. Smith [J]. Transactions of the Metallurgical Society of AIME, 1948, 175: 15-51.
[25] WANG Nan, WEN You-hai, CHEN Long-qing. Pinning force from multiple second-phase particles in grain growth [J]. Computational Materials Science, 2014, 93: 81-85.
[26] WEYGAND D, BRECHET Y, LEPINOUX J. Zener pinning and grain growth: A two-dimensional vertex computer simulation [J]. Acta Materialia, 1999, 47(3): 961-970.
M. RAJABI, R. M. SEDIGHI, S. M. RABIEE
Department of Material Science and Engineering, Faculty of Mechanical Engineering, Babol Noshirvani University of Technology, P. O. Box 47148-71167, Shariati Street, Babol, Iran
ժ Ҫ���о���е�Ͻ��Ʊ�������Mg-6Al-1Zn �� Mg-6Al-1Zn-1Si �Ͻ�����ȶ��Ժ;�����������ѧ���о���Ԫ�ط�ĩ��ʼ��ʹ���˸��ַ����ֶΣ�������ʾɨ�����ȷ�(DSC)��X��������(XRD)��ɨ��羵����羵�����������о������˻�����еľ�����������ѧ��XRD����������������ֲ�����ϵ�ľ����ߴ����˻��¶ȵ����߶����ӣ�����350 ��C�˻�1 h��Si��ϵ���и�С�ľ����ߴ�(60 nm)����Mg-6Al-1Zn��ϵ�ľ����ߴ�Ϊ72 nm��Mg-6Al-1Zn-1Si��ϵ�ڵ����˻�������γɵĵڶ�������仯����Mg2Si����Ӱ���ܣ�����Ӱ�춯��ѧ���̵�ָ������Si��ϵ��Ӳ�ȸ��������ڽ�������Mg2Si���γɡ�
�ؼ��ʣ�þ���Ͻ����ȶ��ԣ�������������е�Ͻ�
(Edited by Mu-lan QIN)
Corresponding author: M. RAJABI; Tel: +98-11-32334205; Fax: +98-11-32312268; E-mail: m.rajabi@nit.ac.ir
DOI: 10.1016/S1003-6326(16)64128-6

