J. Cent. South Univ. Technol. (2007)06-0750-03
DOI: 10.1007/s11771-007-0142-9
Determination of isothermal section of Ag-Ti-Zr ternary system at 1 023 K
ZHOU Guo-jun(�ܹ���), LIU Hua-shan(����ɽ), LIU Li-bin(������), JIN Zhan-peng(��չ��)
(School of Materials Science and Engineering, Central South University, Changsha 410083, China)
Abstract��The isothermal section of Ag-Ti-Zr ternary system at 1023 K was determined by diffusion triple and electron probe microanalysis. The results indicate that four binary intermetallic phases of AgTi, AgTi2, AgZr and AgZr2 are found in Ag-Ti-Zr ternary system at 1 023 K. AgZr2 and AgTi2 form a continuous solid solution, namely Ag(Ti,Zr)2. Four three-phase regions: AgTi +AgZr + Ag, AgTi +AgZr + Ag (Ti, Zr)2, ��-Zr +��(Ti, Zr) + Ag (Ti, Zr) 2 and ��-Ti +��(Ti, Zr) + Ag (Ti, Zr)2 exist in the isothermal section. No ternary compound is observed.
Key words: Ag-Ti-Zr; phase diagram; diffusion triple; isothermal section
1 Introduction
The silver-based alloys have been widely used in industry for many years. For example, the traditional silver solders have been employed in joining ceramics and glass with metallic materials. Recently, the Ag- and Ti-alloys have been applied in semiconductor and metal jointing. Additionally, the IV group transition metal Zr as an important micro-alloying element, can be added in Ag-based alloy to modify the bulk activities and thus to increase its metallicity and wetting [1-7].
The development of new materials requires knowledge of thermodynamics and phase diagram. Very little work has been done about the phase equilibria in Ag-Ti-Zr ternary system. For this purpose, knowledge about the ternary system is helpful for the research on metallic semiconductor contiguity materials. It is also important and necessary to exploit new Ag-alloys, and design reasonable production technique as well as improve materials properties.
The boundary binary systems of the Ag-Ti-Zr ternary system have been well studied. The Ti-Zr is a simple binary system which consists of two solid solutions, ��- and ��-phases, and liquid with a congruent minimum at Ti-50%Zr[8-9]. While the other two binary systems have intermetallic compounds. The Ag-Ti binary system was comprehensively studied[10-11]. There is a wide two-phase region, ��-Ti+L, and two intermediate phases, AgTi and AgTi2[12-13]. Meanwhile, it is generally accepted that there are two intermediate compounds in the Ag-Zr binary system, AgZr and AgZr2, and AgZr has a certain homogeneity region[14-18].
In the present work, phase equilibria in the Ag-Ti-Zr system at 1 023 K were investigated by means of diffusion triple approach and electron microprobe analysis, details about this approach was referred to Ref.[19].
2 Experimental
The Ag-Ti-Zr diffusion triple specimens were prepared from blocks of pure metals: Ag(99.99%), Zr (99.90%) and Ti (99.50%, mass fraction). Firstly, to make a Ti-Zr diffusion couple, the Ti and Zr blocks were diffusionally welded under 4 MPa in argon flow at 1 023 K for 10 min, then cooled to ambient temperature. Subsequently, the Ag-Ti-Zr diffusion triple was assembled from the obtained Ti-Zr couple and the pure Ag block by diffusion welding under 3 MPa at 1 023 K in argon flow for 10 min. Finally, the triple was encapsulated in evacuated quartz tube backfilled with pure argon and annealed at (1 023 ��2) K for 1 440 h. After the heat treatment, the diffusion triple was taken out from the furnace and quenched in water before breaking the quartz tube. The fabricated diffusion triple is shown in Fig.1.
The obtained diffusion triple was ground, polished and examined by means of electron probe microanalysis (EPMA, JX-8800R, Japan, Electron Optics Ltd, Tokyo) under the operation condition of 20 kV, 2��10-8 A and a 40? take-off angle.
3 Results and discussion
The back-scattered electron image and schematic diagram of the Ag-Ti-Zr triple annealed at 1 023 K are shown in Fig.2. The chemical compositions of the
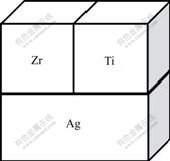
Fig.1 Schematic diagram of Ag-Ti-Zr diffusion triple

Fig.2 Schematic diagram of Ag-Ti-Zr diffusion triple at 1 023 K(a) and back-scattered electron image of A area(b) and back-scattered electron image of B area(c)
equilibrium phases at the tie lines data are listed in Table 1[13]. It is clear from Fig.2 and Table 1 that there are two diffusion layers of intermetallic phases of AgTi and AgTi2 in the intersection of Ag-Ti side, and two diffusion layers of AgZr and AgZr2 in the intersection of Ag-Zr side. However, when the diffusion triple is ground and polished, the phases of AgTi and AgZr are easy to be desquamated because the two phases are very fragile, which has no influence on analyzing the phase relation of the ternary system. No diffusion phases are formed in the intersection of Ti-Zr side, while it is found that parts of ��-Ti and ��-Zr transform to ��-Ti and ��-Zr, which form a continuous solid solution ��(Ti, Zr). All these phases are in good agreement with the Ti-Zr[9], Ag-Ti[13] and Ag-Zr[13] binary phase diagrams. No ternary compound is found.
Table 1 Equilibrium data determined by EPMA in Ag-Ti-Zr system at 1 023 K (Molar fraction)
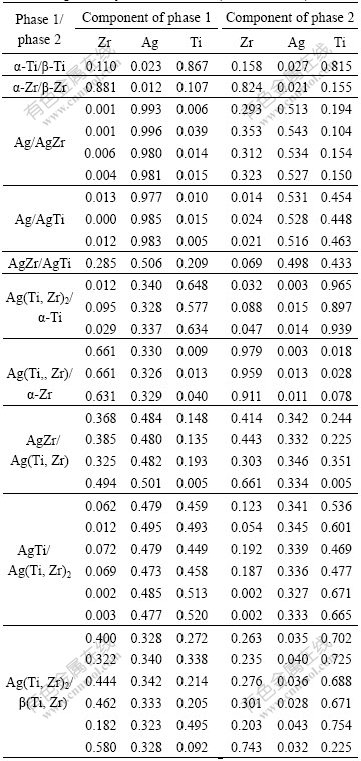
Additionally, according to the data by EPMA in Table 1, the largest solubility of Ti in AgZr is about 20.9% and that of Zr in AgTi is about 7.2%. The intermetallic phase AgTi has a solubility range from 47.3% to 53.1% (molar fraction) Ag and the intermetallic phase AgZr has a solubility range from 48.0% to 54.3%Ag. It can also be seen from Table 2 that AgTi2 and AgZr2 are of the same crystal structures, referring to the experimental data, the binary compounds AgTi2 and AgZr2 are nearly linear compounds and form a continuous solid solution, namely Ag (Ti, Zr)2.
Table 2 AgTi2 and AgZr2 crystal structures

There are not mixing area of two-phase or three-phase but single-phase in the triple diffusion sample because of two direction of concentration gradients. Each dot in single-phase area represents specifical alloy composition. According to the theory of interphase local equilibria and the terminal composition of tie-lines measured by EPMA, all two-phase regions were determined, and then the single-phase regions were ascertained correspondingly. The three-phase equilibria were estimated by extrapolating the related tri-junction joint of 3 two-phase equilibria. Hence the tie-triangles for three-phase equilibria were shown in dashed lines. Combining the data listed in Table 1 with Fig.2, and taking into account the previous published binary systems, the isothermal section of the Ag-Ti-Zr system at 1 023 K was established by connecting the boundary lines of different equilibrium phase regions, as shown in Fig.3. there are 7 single-phase, 10 two-phase and 4 three-phase regions in this ternary section, namely AgTi +AgZr+Ag, AgTi +AgZr+Ag(Ti, Zr)2, ��-Zr+��(Ti, Zr)+ Ag(Ti, Zr)2 and ��-Ti +��(Ti, Zr)+Ag(Ti, Zr)2.
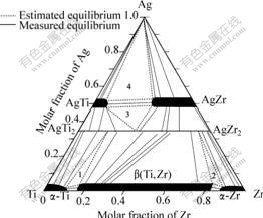
Fig.3 Isothermal section of Ag-Ti-Zr ternary system at 1 023 K
1����-Ti+��(Ti,Zr)+Ag(Ti, Zr)2; 2����-Zr+��(Ti,Zr)+Ag(Ti, Zr)2; 3��AgTi+AgZr+Ag(Ti, Zr)2; 4��AgTi+AgZr+Ag
4 Conclusions
1) The isothermal section of the Ag-Ti-Zr ternary system at 1 023 K is determined, which consists of 4 three-phase regions, AgTi +AgZr + Ag, AgTi +AgZr + Ag (Ti, Zr)2, ��-Zr +��(Ti, Zr) + Ag (Ti, Zr) 2 and ��-Ti +��(Ti, Zr) + Ag (Ti, Zr) 2. No ternary compound is found. The largest solubility of Ti in AgZr is about 20.9% (molar fraction) and that of Zr in AgTi is about 7.2%.
2) The intermetallic phase AgTi has a solubility range from 47.3%Ag to 53.1%Ag and the intermetallic phase AgZr has a solubility range from 48.0%Ag to 54.3%Ag at 1 023 K. AgZr2 and AgTi2 form a continuous solid solution as Ag (Ti, Zr) 2.
References
[1] Benko E, Bielanska E, Pereverteilo V M, et al. Formation peculiarites of the interfacial structure during cBN wetting with Ag-Ti, Ag-Zr and Ag-Hf alloys[J]. Diamond and Related Materials, 1997, 6(8): 931-936.
[2] Pan Wei, LI Rong-ti, Chen Jian, et al. Thermodynamic properties of Ti in Ag�CTi alloys[J]. Mater Sci Eng A, 2000, 287(1): 72-77.
[3] Novakovic R, Muolo M L, Passerone A. Bulk and surface properties of liquid X-Zr (X =Ag, Cu) compound forming alloys[J]. Surface Science, 2004, 549(3): 281-293.
[4] Loehman R E, Hosking F M, Gauntt B, et al. Reactions of Hf-Ag and Zr-Ag alloys with Al2O3 at elevated temperatures[J]. Journal of Materials Science, 2005, 40(9): 2319-2324.
[5] Loehman R E, Gauntt B D, Hosking F M, et al. Reaction and bonding of Hf and Zr containing alloys to alumina and silica[J]. J Europ Ceram Soc, 2003, 23(15): 2805-2811.
[6] Koltsov A, Hodaj F, Eustathopoulos N, et al. Wetting and interfacial reactivity in Ag-Zr/sintered AlN system[J]. Scripta Mater, 2003, 48(4): 351-357.
[7] Muolo M L, Ferrera E, Novakovic R, et al. Wettability of zirconium diboride ceramics by Ag, Cu and their alloys with Zr[J]. Scripta Mater, 2003, 48(2): 191-196.
[8] Ruch M, Arias D, Comments on the equilibrium diagram of the Ti-Zr system[J]. Scripta Metall Mater, 1993, 29(4): 533-538.
[9] Massalski T B, Okamoto H, SUBRAMANIAN P R, et al. Binary Alloy Phase Diagrams[M]. 2nd ed. OH, ASM, Materials Park, 1992, 3: 3502-3503.
[10] Eremenko V N, Buyanov Y I, Panchenko N M. Constitution diagram of the system titanium-silver[J]. Porosh Metall, 1969, 79(7): 55-59.
[11] Plichta M R, Aaronson H I, Perepeko J H. Thermodynamics and kinetics of the beta yields alpha //m transformation three Ti-X systems[J]. Acta Metall, 1978, 26(8): 1293-1305.
[12] FU X L, LI C R, WANG F M, et al. Experimental study on the phase equilibria of the AgTi system[J]. Mater Sci Eng A, 2005, 408(2): 190-194.
[13] Massalski T B, Okamoto H, Subramanian P R et al. Binary Alloy Phase Diagrams[M]. 2nd ed. OH, ASM, Materials Park, 1992, 1: 105-119.
[14] Karakaya I, Thompson W T. The Ag-Zr binary system[J]. Phase Equil, 1992, 13(3): 143-146.
[15] Osamu T, Yoshiaki I. Reaction diffusion in silver-zirconium system[J]. JIM, 1994, 35(10): 673-678.
[16] Zhang K H, ZHao H Z, ZHOU Y H. Investigation of the Ag-Zr phase diagram[J]. J Less-Comm Met, 1988, 138(2): 172-177.
[17] Zhou Y H, Zhao H Z, Zhang K H. Investigation of the Ag-Cu-Zr ternary system[J]. J Less-Comm Met, 1988, 138(2): 7-10.
[18] Zhang K H, LI J H. Partial phase diagram of the Au-Ag-Zr ternary system[J]. Journal of Alloys and Compounds, 2006, 217(2): 155-158.
[19] Jin Zhan-peng. Study of the range of stability of sigma phase in some ternary systems[J]. J Metall, 1981, 10: 279-287.
(Ediyed by YANG Hua)
Foundation item: Projects(50371104; 50401011) supported by the National Natural Science Foundation of China
Received date: 2007-04-10; Accepted date: 2007-06-15
Corresponding author: ZHOU Guo-jun, Doctoral candidate; Tel: +86-731-8877732; E-mail: mikezhgj@163.com