淋溶条件下赤泥盐分离子的迁移与分布
来源期刊:中国有色金属学报(英文版)2018年第3期
论文作者:孔祥峰 江星星 薛生国 黄玲 William Hartley 吴川 李晓飞
文章页码:534 - 541
关键词:赤泥;盐分;离子迁移;土柱模拟;淋溶
Key words:bauxite residue; salinity; ion migration; column stimulation; water leaching
摘 要:赤泥是氧化铝工业生产过程产生的强碱性固体废物,其盐分含量高,对环境有害且制约着赤泥堆场的植被重建。通过土柱模拟淋溶实验,研究赤泥盐离子的动态迁移及垂直分布情况。结果表明:淋溶不同程度地将赤泥中的Na+、K+、Ca2+、 、 和 等盐分离子浸出到渗滤液中,显著降低了赤泥的盐度。赤泥中Na+和K+呈现出较高的迁移能力,从柱体的40~50 cm处向上迁移至20~30 cm处;而Ca2+的迁移能力较弱,主要分布在柱体的30~40 cm处。淋溶刚结束时, 主要分布在柱体的20~30 cm处,随后向下迁移至30~40 cm处,最终受蒸发作用迁移至20~30 cm处。 最初主要分布在柱体的40~50 cm处,最终向上迁移至20~30 cm处。 主要停留在柱体的下部,其迁移特性受蒸发的影响较小。
Abstract: Bauxite residue, a highly saline solid waste produced from digestion of bauxite for alumina production, is hazardous to the environment and restricts vegetation establishment in bauxite residue disposal areas. A novel water leaching process proposed here was used to investigate the dynamic migration and vertical distribution of saline ions in bauxite residue. The results show that water leaching significantly reduced the salinity of bauxite residue, leaching both saline cations Na+, K+, Ca2+ and anions , , . Na+ and K+ migrated from 40-50 to 20-30 cm of the column, presenting a high migration capacity. The migration capacity of Ca2+ was lower and accumulated at 30-40 cm of the column. initially distributed at 20-30 cm of the column, subsequently transported to 30-40 cm of the column, and finally returned to 20-30 cm of the column along with evaporation. was originally distributed at 40-50 cm, but finally migrated to 20-30 cm of the column. Nevertheless, remained at the bottom of the column, and its migratory was less affected by evaporation.
Trans. Nonferrous Met. Soc. China 28(2018) 534-541
Xiang-feng KONG1,2, Xing-xing JIANG1,2, Sheng-guo XUE1,2, Ling HUANG1,2, William HARTLEY3, Chuan WU1,2, Xiao-fei LI1,2
1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. Chinese National Engineering Research Center for Control & Treatment of Heavy Metal Pollution, Central South University, Changsha 410083, China;
3. Crop and Environment Sciences Department, Harper Adams University, Newport, Shropshire, TF10 8NB, United Kingdom
Received 3 January 2017; accepted 24 April 2017
Abstract: Bauxite residue, a highly saline solid waste produced from digestion of bauxite for alumina production, is hazardous to the environment and restricts vegetation establishment in bauxite residue disposal areas. A novel water leaching process proposed here was used to investigate the dynamic migration and vertical distribution of saline ions in bauxite residue. The results show that water leaching significantly reduced the salinity of bauxite residue, leaching both saline cations Na+, K+, Ca2+ and anions 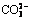 ,
, 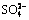 ,
,  . Na+ and K+ migrated from 40-50 to 20-30 cm of the column, presenting a high migration capacity. The migration capacity of Ca2+ was lower and accumulated at 30-40 cm of the column.
. Na+ and K+ migrated from 40-50 to 20-30 cm of the column, presenting a high migration capacity. The migration capacity of Ca2+ was lower and accumulated at 30-40 cm of the column. 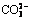 initially distributed at 20-30 cm of the column, subsequently transported to 30-40 cm of the column, and finally returned to 20-30 cm of the column along with evaporation.
initially distributed at 20-30 cm of the column, subsequently transported to 30-40 cm of the column, and finally returned to 20-30 cm of the column along with evaporation. 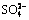 was originally distributed at 40-50 cm, but finally migrated to 20-30 cm of the column. Nevertheless,
was originally distributed at 40-50 cm, but finally migrated to 20-30 cm of the column. Nevertheless,  remained at the bottom of the column, and its migratory was less affected by evaporation.
remained at the bottom of the column, and its migratory was less affected by evaporation.
Key words: bauxite residue; salinity; ion migration; column stimulation; water leaching
1 Introduction
The alumina industry has developed rapidly due to the requirement for aluminum. Unfortunately, such rapid development has brought about multiple environmental problems that profoundly influence the sustainable development [1-3]. Bauxite residue (BR, or red mud) is a highly saline solid byproduct generated during alumina extraction from bauxite using Bayer, sintering and combined Bayer-sintering processes in alumina refineries. The volume of BR generated while producing 1 t of alumina is typically 0.5-2 t [4-7]. The cumulative global inventory of BR has reached an estimated 4 billion tons, and is still increasing [8-10]. Many efforts have been made to find suitable uses for BR, but as yet no economic method has been extensively applied [11-13]. Therefore, almost all BR continues to be disposed in BRDAs [14,15], which requires sustainable efforts to manage the waste [16,17]. Furthermore, freshly formed saline dusts on the surface of BRDA containing large concentrations of salts are harmful to the surrounding environment [18-20]. The elevated salts mean that BR is a hazardous waste, which limit its safe disposal and revegetation.
High salinity is a result of using sodium hydroxide for extracting alumina from the ore and the formation of a complex-saline mineral. The residual sodium carbonate (Na2CO3) and sodium bicarbonate (NaHCO3) that can not be completely separated by counter-current decantation (CCD) water washing before disposal partly remain in BR, and are responsible for the soluble-sodic salinity [21,22]. Additionally, potash (K2CO3) and Na2SO4 commonly exist in BR. Furthermore, The formed solid phase of tri-calcium aluminate (TCA, Ca3Al2(OH)12), cancrinite (Na6Al6Si6O24・2CaCO3), sodalite (Na6Al6Si6O24・Na2CO3), and hydrogarnet (Ca3Al2(SiO4)x(OH)12-4x) are typical buffering minerals [23,24], which act as another source of salinity.
The removal of BR salinity is therefore critical. Nevertheless, many efforts are to remove or transform the alkalinity of BR, which are commonly applied by alumina refinery to treat with BR prior to store and ameliorate for revegetation at BRDAs [25,26]. The addition of gypsum, organic matter and fertilizer, seawater neutralization, waste acid interaction and carbon dioxide sequestration have been attempted to reduce and/or remove the high alkalinity [27,28]. Nevertheless, removal and separation of salinity have only been minimal. Water washing is a promising way forward in an attempt to reduce the strong alkalinity and remove the high salinity; this may leach approximately 70% alkaline Na whilst also reducing a large proportion of salts [29]. However, information on migration behavior of saline compounds and their subsequent distribution is absent, but is significant if a well-founded transportation-migration theory is to be understood. Furthermore, less concern has been paid to saline migration in BRDAs. Indeed, lack of understanding of dynamic migration and vertical distribution of saline ions in BR during water leaching is a knowledge gap that requires attention.
The objectives of the present study are to investigate leaching and migration behaviors of saline cations and anions in bauxite residue using a column leaching study, and to research the vertical distribution of saline ions in residue during long-term leaching.
2 Experimental
2.1 Field sampling and sample preparation
In October 2015, a raw BR sample, used throughout this study, was collected from an Aluminum Refinery Corporation of China. The freshly stored BR was collected from the surface of BRDA’s. Three sub-samples were collected with a distance of 10 m from each other to obtain a uniform sample (Fig. 1(a)). Samples were stored in polyethylene bags, returned to the laboratory and subsequently air-dried for 4 d, and then sieved to retain the <2 mm fraction. SEM image of the sample (Fig. 1(b)) showed that it consisted of 0.1-0.5 μm particles in 2-10 μm aggregates, which was poorly-crystallized. Compositions of BR are presented in Table 1.
2.2 Long-term leaching experiment
The leaching experiment was conducted using a column (Fig. 2) to simulate long-term leaching by rainfall in a BRDA. Two half-cylinders were bonded to create the column with an outer diameter of 14 cm, an inner diameter of 12.6 cm and a height of 95 cm, and 36 sample holes were pre-drilled and arranged on the outer wall for convenient sampling at various time during the investigation. The column base was mesh, being supported with a 5 cm depth of sand. Subsequently, the column was filled with BR, and Milli-Q water was supplied slowly from the bottom of the column. After 2 d of saturation, Mill-Q water was then supplied slowly from the top, controlling the water level 5 cm above the surface of the packed residue. Leachate was immediately collected from the bottom until no further liquor was produced, whilst the mass of each leachate was measured. After leaching, the leached residues were collected at depths of 15, 25, 35, 45, 55 and 65 cm at regular intervals.

Fig. 1 Bauxite residue sample collected from surface of BRDA (a) and SEM image of bauxite residue (b)
Table 1 Initially saline composition of bauxite residue in water leaching column experiment

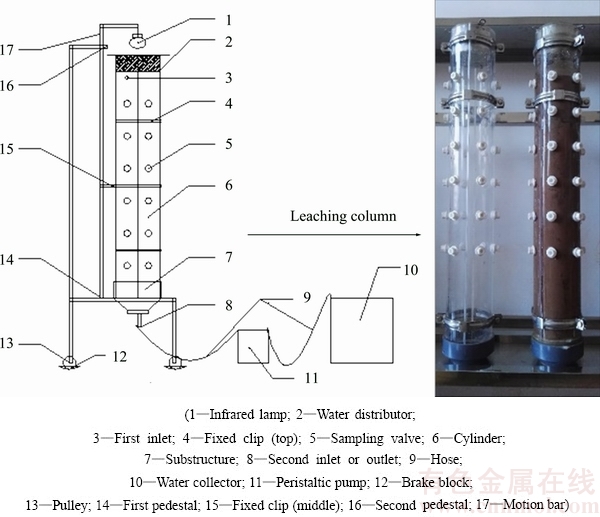
Fig. 2 Leaching column of bauxite residue for simulating long-term leaching of BRDA
2.3 Sample analysis
The leachate was centrifuged at 4000 r/min for 10 min and then analyzed for pH with a probe calibrated to a slope >99%, meanwhile determining electric conductivity (EC) with a probe calibrated to electrode constant K=1.00 (±0.05) in a 2.76 mS/cm solution. Soluble Na+, K+ and Ca2+ concentrations in leachate were analyzed by a PerkinElmer Optima 5300 ICP-AES. Saline 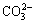 and
and  were analyzed by titration using 0.02 mol/L H2SO4 standardized solution [24], and
were analyzed by titration using 0.02 mol/L H2SO4 standardized solution [24], and 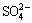 was determined on a Daian ICS-90A HPIC. Residual solids collected from the column were oven-dried at 65 °C and sieved to retain the <0.38 μm fraction. Subsequently, soluble ions were extracted by water from the residues and analyzed.
was determined on a Daian ICS-90A HPIC. Residual solids collected from the column were oven-dried at 65 °C and sieved to retain the <0.38 μm fraction. Subsequently, soluble ions were extracted by water from the residues and analyzed.
3 Results and discussion
3.1 Change of saline cation concentration in leachate
The changes of Na+, K+ and Ca2+ concentrations in leachate presented in Fig. 3 show that they changed slightly in 76 min of leaching. However, after 76 min, soluble Na+ increased with leaching time, but soluble Ca2+ decreased sharply, whilst soluble K+ remained stable. Soluble Na+ concentration increased from 14.13 mmol/L at 76 min to 43.74 mmol/L at 467 min, subsequently, reaching equilibrium. Soluble Ca2+ concentration decreased from 1.90 mmol/L at 76 min to 0.38 mmol/L at 400 min.
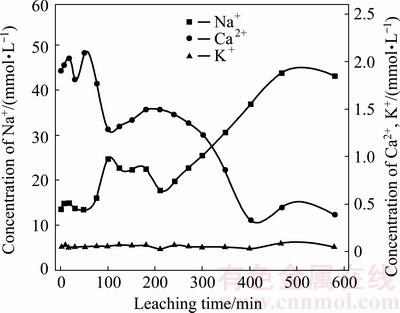
Fig. 3 Concentrations of saline cations Na+, K+ and Ca2+ in leachate
Soluble saline cation results (Fig. 3) indicated that the changes in their concentrations were relatively consistent at 76 min of leaching. During 76 min leaching, the leachate did not penetrate and just moved to the bottom of the column. The leachate collected from the column was a saturated solution of BR and the saline Na+, K+ and Ca2+ only dissolved in pore water of BR, which did not start to migrate. After 76 min leaching, the concentration of soluble Na+ increased in leachate, showing that a large amount of Na+ was leached, further confirming that soluble Na+ dominated the soluble cations in the leachate. Furthermore, soluble Na+ indicated that the colloidal adsorption of Na+ was weak, being affected by ion concentration, ionic radius and charge, further suggesting that its leaching capacity with water was strong and the leaching process may promote its removal. However, soluble Ca2+ in leachate decreased, and colloidal adsorption would profoundly influence its mobility due to its ionic charge and larger ionic radius. This suggested that the Ca2+ migration capacity with water was limited and did not improve its removal. The leaching of K+, however, was relatively stable. Therefore, it may be summarized that the leaching capacity of various saline cations in BR were in the following decreasing order: Na+>K+>Ca2+.
3.2 Change of saline anion concentration in leachate
The concentration changes of 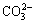 ,
,  and
and 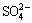 in leachate are presented in Fig. 4, which revealed that they increased during the leaching process. Nevertheless, the concentration of
in leachate are presented in Fig. 4, which revealed that they increased during the leaching process. Nevertheless, the concentration of 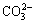 was approximately 1.0 mmol/L within 240 min, subsequently increasing to 7.16 mmol/L at 400 min. The concentration of
was approximately 1.0 mmol/L within 240 min, subsequently increasing to 7.16 mmol/L at 400 min. The concentration of 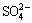 increased from 5.10 mmol/L at 100 min to 8.01 mmol/L at 400 min, whilst
increased from 5.10 mmol/L at 100 min to 8.01 mmol/L at 400 min, whilst  in leachate increased rapidly after 76 min, reaching approximately 14.0 mmol/L at 500 min.
in leachate increased rapidly after 76 min, reaching approximately 14.0 mmol/L at 500 min.
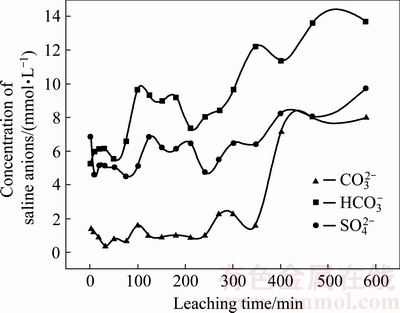
Fig. 4 Concentrations of saline anions 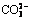 ,
,  and
and 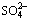 in leachate
in leachate
Soluble saline anion results (Fig. 4) indicated that the changes in the concentrations of 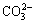 ,
,  and
and 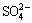 in leachate were uniform within 76 min. The leachate also did not penetrate and just moved to the bottom of the column and the saline
in leachate were uniform within 76 min. The leachate also did not penetrate and just moved to the bottom of the column and the saline 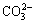 ,
,  and
and 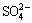 only existed in the saturated solution from the BR. After 76 min leaching,
only existed in the saturated solution from the BR. After 76 min leaching,  concentration in leachate clearly increased, and a large of
concentration in leachate clearly increased, and a large of  was leached. Additionally, the concentration of
was leached. Additionally, the concentration of  was greater than that in the original BR (Table 1), indicating colloid adsorption of
was greater than that in the original BR (Table 1), indicating colloid adsorption of  was weak, suggesting that the leaching capacity of
was weak, suggesting that the leaching capacity of  with water was strong, promoting
with water was strong, promoting  removal. A similar change for
removal. A similar change for 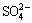 concentration was observed, but the change was delayed by approximately 30 min. However, the concentration of
concentration was observed, but the change was delayed by approximately 30 min. However, the concentration of 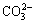 later increased, which was even lower than that in initial BR (Table 1). Less
later increased, which was even lower than that in initial BR (Table 1). Less 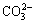 was leached into the leachate, which was deeply affected by colloid adsorption as a result of its multiple ionic charge similar to Ca2+. Thus, the leaching capacities of saline anions in BR were in the following decreasing order:
was leached into the leachate, which was deeply affected by colloid adsorption as a result of its multiple ionic charge similar to Ca2+. Thus, the leaching capacities of saline anions in BR were in the following decreasing order:  >
> 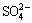 >
>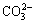 .
.
3.3 Vertical distribution of saline cations in residue
The vertical distributions of Na+, K+ and Ca2+ in the residue of the leaching column at different time are shown in Fig. 5. The Na+ distribution at 0-60 cm (Fig. 5(a)) shows that the peaks of Na+ concentrations in the vertical distribution were located at 45.33 cm at 2 d following the leaching episode, 34.69 cm at 14 d following the leaching episode, and 28.89 cm at 26 d following the leaching episode, respectively. At the beginning of stage following the leaching episode, the liquors continuously penetrated downward under the function of capillary pressure and gravity, meanwhile the intermolecular force interaction between Na+ and hydrone was stronger than the force between Na+ and the colloidal particle of BR, which resulted in the high migration of Na+ at a column depth of 40-50 cm. In later period of leaching, capillary pressure disappeared whilst the leached liquors were affected by evaporation, which changed the distribution of liquor and further promoted the upward migration of Na+, resulting in a concentration peak of Na+ re-distribution located at 20-30 cm.
The distribution of Ca2+ in the column at depths of 0-60 cm (Fig. 5(b)) revealed that the peaks of Ca2+ concentration were situated at 33.17 cm after 2 d of leaching, 25.84 cm, after 14 d of leaching, and 26.66 cm after 26 d of leaching, respectively. At the initial stage following the leaching episode, the intermolecular force interaction between Ca2+ and hydrone was higher than the force between Ca2+ and the colloidal particles of BR. However, Ca2+ was distributed at 30-40 cm of the column, which demonstrated a relatively weak migration compared to the Na+ distribution at the depths of 40-50 cm. This may be due to the multiple ionic charge and large ionic radius (Fig. 2). Later, with a reduction in liquor infiltration and intermolecular force interaction between Ca2+ and hydrone, the migration of Ca2+ became more difficult, resulting in the peak of Ca2+ fixed at approximately 26 cm. Besides, the peak value of Ca2+ concentration was 0.26 mmol/kg in the initial stage following leaching; however, it decreased to approximately 0.10 mmol/L which was reduced by 60%. Meanwhile, the peak value of Na+ concentration was slightly reduced compared to Ca2+. This distribution characteristic suggested that Na+ was replaced with Ca2+ in the initial period following leaching.
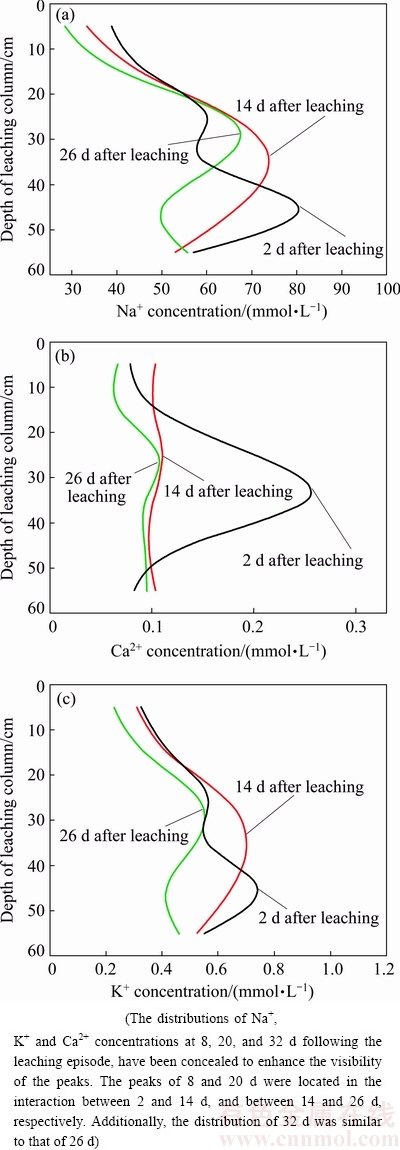
Fig. 5 Vertical distribution of saline cations Na+ (a), Ca2+ (b) and K+ (c) in residue after leaching
The K+ distribution at depths of 0-60 cm (Fig. 5(c)) proves that the peaks of K+ concentration were located at 45.46 cm and 2 d following the leaching episode, 35.24 cm at 14 d following the leaching episode, and 29.00 cm at 26 d following the leaching episode, respectively. The K+ distribution characteristic was similar to that of Na+. At the front-end after leaching, K+ had a high migration rate and concentrated at 40-50 cm of the column. In the later period after leaching, evaporation changed the distribution of liquor and improved the upward migration of K+, which resulted in a concentration peak of K+ re-distributed to 20-30 cm.
3.4 Vertical distribution of saline anion in residue
The distribution characteristics of saline anions in the residue depict a discrepant change with that of saline cations, as presented in Fig. 6. The  distribution of the residue in the leaching column at depths of 0-60 cm (Fig. 6(a)) shows that the peaks of
distribution of the residue in the leaching column at depths of 0-60 cm (Fig. 6(a)) shows that the peaks of  concentrations in the vertical distribution were located at 24.33 cm at 2 d after leaching, 33.58 cm at 14 d after leaching, and 26.12 cm at 26 d after leaching, respectively. In the initial period of leaching,
concentrations in the vertical distribution were located at 24.33 cm at 2 d after leaching, 33.58 cm at 14 d after leaching, and 26.12 cm at 26 d after leaching, respectively. In the initial period of leaching,  ions at the top of the column penetrated downward along with the liquor. Nevertheless, the migration capacity of
ions at the top of the column penetrated downward along with the liquor. Nevertheless, the migration capacity of 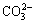 is weak due to its multiple ionic charge and larger ionic radius, which resulted in the reduced migration rate of
is weak due to its multiple ionic charge and larger ionic radius, which resulted in the reduced migration rate of 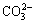 at depths of 20-30 cm. At 14 d following the leaching episode, the peak of
at depths of 20-30 cm. At 14 d following the leaching episode, the peak of 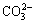 gradually migrated to depths of 30-40 cm. However, in the final period of leaching, the infiltrated pressure was reduced, whilst the leached liquors were influenced by evaporation, which returned to depths of 20-30 cm.
gradually migrated to depths of 30-40 cm. However, in the final period of leaching, the infiltrated pressure was reduced, whilst the leached liquors were influenced by evaporation, which returned to depths of 20-30 cm.
The  distribution of the residue in the leaching column at depths of 0-60 cm (Fig. 6(b)) shows that the peaks of
distribution of the residue in the leaching column at depths of 0-60 cm (Fig. 6(b)) shows that the peaks of  concentration in the vertical distribution were located at 32.76 cm after leaching for 2 d, 55.55 cm after leaching for 14 d, and 42.98 cm after leaching for 26 d following the leaching episode, respectively. At the beginning of stage following the leaching episode, the peak of
concentration in the vertical distribution were located at 32.76 cm after leaching for 2 d, 55.55 cm after leaching for 14 d, and 42.98 cm after leaching for 26 d following the leaching episode, respectively. At the beginning of stage following the leaching episode, the peak of  concentration moved from depths of 32.76 to 55.55 cm, which manifested a high capacity of migration which is in agreement with its leaching capacity. In the medium period, with the capillary pressure and penetrated pressure becoming weak, the migration slowed down. Subsequently, the effect of evaporation was higher than that of the pressure, which resulted in the
concentration moved from depths of 32.76 to 55.55 cm, which manifested a high capacity of migration which is in agreement with its leaching capacity. In the medium period, with the capillary pressure and penetrated pressure becoming weak, the migration slowed down. Subsequently, the effect of evaporation was higher than that of the pressure, which resulted in the  migrating upwards. However, the peak of
migrating upwards. However, the peak of  concentration was fixed at 40-50 cm instead of higher depths of 30-40 cm or 20-30 cm, although the
concentration was fixed at 40-50 cm instead of higher depths of 30-40 cm or 20-30 cm, although the  has a strong capacity of migration. Bicarbonate ions showed a reverse change, suggesting that the migration of
has a strong capacity of migration. Bicarbonate ions showed a reverse change, suggesting that the migration of  was less affected by evaporation. At the final stage, the major
was less affected by evaporation. At the final stage, the major  remained at the bottom of the leaching column rather than on the surface. The migration characteristics of
remained at the bottom of the leaching column rather than on the surface. The migration characteristics of  may avoid the occurrence of its efflorescence, further implying that leaching is helpful to control the salinity of BR.
may avoid the occurrence of its efflorescence, further implying that leaching is helpful to control the salinity of BR.
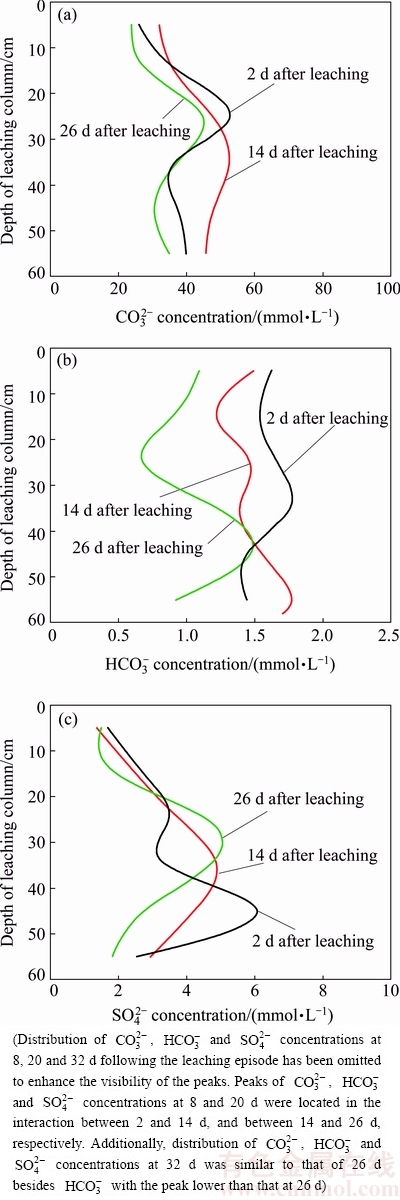
Fig. 6 Vertical distribution of saline anions 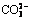 (a),
(a),  (b) and
(b) and 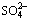 (c) in residue after leaching at different time
(c) in residue after leaching at different time
The 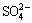 distribution of the residue at depths of 0-60 cm (Fig. 6(c)) shows that the peaks of
distribution of the residue at depths of 0-60 cm (Fig. 6(c)) shows that the peaks of 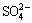 concentration in the vertical distribution were located at 45.19 cm and 2 d, 36.07 cm at 14 d, and 29.85 cm at 26 d following the leaching episode, respectively. At the initial stage after leaching, the peak of
concentration in the vertical distribution were located at 45.19 cm and 2 d, 36.07 cm at 14 d, and 29.85 cm at 26 d following the leaching episode, respectively. At the initial stage after leaching, the peak of 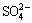 concentration rapidly moved up. In the medium period, migration was controlled by capillary pressure and penetrated pressure, the multiple ionic charge of
concentration rapidly moved up. In the medium period, migration was controlled by capillary pressure and penetrated pressure, the multiple ionic charge of 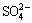 and the evaporation of liquors; when the capillary pressure and penetrated pressure became weak, the migrated direction of
and the evaporation of liquors; when the capillary pressure and penetrated pressure became weak, the migrated direction of 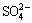 changed. The peak of
changed. The peak of 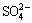 moved up to a depth of 36.07 cm after 14 d. Subsequently, the effect of evaporation became stronger, which further resulted in the continuing migration of
moved up to a depth of 36.07 cm after 14 d. Subsequently, the effect of evaporation became stronger, which further resulted in the continuing migration of 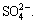 In the final period, the peak of
In the final period, the peak of 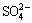 concentration migrated up the upside of the leaching column instead of the bottom.
concentration migrated up the upside of the leaching column instead of the bottom.
3.5 Potential implication for revegetation
Previously, the management practice of BRDAs has concentrated on containment, with less attention to the changes in long-term leaching of the salinities of the residues in BRDAs. Currently, management projects are moving gradually towards amendment and revegetation to ameliorate the salinity and alkalinity of the stored bauxite residue and/or further soil-formation of BRDAs to reduce environmental hazards associated with natural disposal. Subsequently, this will naturally establish a relatively stable ecosystem in the residue disposal areas, finally reverting to occupied land areas to an alternative option. In this work, a self-designed column was used to simulate the saline migration and distribution of BR in BRDAs during water leaching. The migration characteristics of Na+, 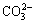 and
and  could hinder the efflorescence occurrence of BRDAs’ surface, and are helpful to control the salinity by water leaching. Leaching process provides a potential means of ameliorating the high saline and sodic conditions on the surface of BRDAs.
could hinder the efflorescence occurrence of BRDAs’ surface, and are helpful to control the salinity by water leaching. Leaching process provides a potential means of ameliorating the high saline and sodic conditions on the surface of BRDAs.
Currently, global inventory of about 4 billion tons of bauxite residue has been stored in BRDAs [30], long-term leaching requires large quantities of water in order to guarantee a steady supply, which may not be practical in consideration of cost implications. Additionally, it is actual that water should be transported over long distances whilst generating enormously secondary leachate in leaching, which would highlight the potential dam-failure. If the supply of water (perhaps taking full advantage of rainfall) and recovery of leachates (returned to alumina extraction process) could be resolved economically, then water leaching of bauxite residue in BRDAs may be a potential option.
Nevertheless, the surface revegetation of freshly disposed bauxite residue in BRDAs following long-term water leaching has many challenges due to its high salinity and sodic conditions. Recent researches in saline-sodic soils [3,18] have implied that the use of salt resistant halophytes (Cynodon dactylon together with Arbuscular mycorrhizal fungi and Arundo donax L.) would be a potential method to further ameliorate the high saline and sodic conditions and improve the physical and chemical properties on BRDAs’ surface. Implication for the revegetation of bauxite residue following long-term leaching that integrates with phytoremediation may be considered as a feasible option.
4 Conclusions
1) Water leaching significantly reduced salinity, and leached both saline cations Na+, K+, Ca2+, and saline anions 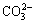 ,
,  ,
, 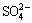 into leachate.
into leachate.
2) The migration capacity of saline cations was in the following decreasing order: Na+>K+>Ca2+. Na+ and K+ initially concentrated at 40-50 cm of the column, and gradually migrated to 20-30 cm at the final stage, whilst Ca2+ accumulated at depths of 30-40 cm.
3) The migration capacity of saline anions was in the following decreasing order:  >
>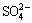 >
>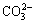 , with low migration rate of
, with low migration rate of 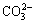 concentrated at 20-30 cm of the column.
concentrated at 20-30 cm of the column. 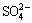 accumulated at depths of 20-30 cm, and
accumulated at depths of 20-30 cm, and  migrated to the bottom.
migrated to the bottom.
4) The migration characteristics of Na+, 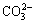 and
and  could impede the efflorescence occurrence of BRDAs’ surface and are helpful to reduce bauxite residue salinity.
could impede the efflorescence occurrence of BRDAs’ surface and are helpful to reduce bauxite residue salinity.
References
[1] KONG Xiang-feng, TIAN Tao, XUE Sheng-guo, HARTLEY W, HUANG Long-bin, WU Chuan, LI Chu-xuan. Development of alkaline electrochemical characteristics demonstrates soil formation in bauxite residue undergoing natural rehabilitation [J]. Land Degradation and Development, 2018, 29: 58-67
[2] XUE Sheng-guo, KONG Xiang-feng, ZHU Feng, HARTLEY W, LI Xiao-fei, LI Yi-wei. Proposal for management and alkalinity transformation of bauxite residue in China [J]. Environmental Science and Pollution Research, 2016, 23: 12822-12834.
[3] XUE Sheng-guo, ZHU Feng, KONG Xang-feng, WU Chuan, HUANG Ling, HUANG Nan, HARTLEY W. A review of the characterization and revegetation of bauxite residues (red mud) [J]. Environmental Science and Pollution Research, 2016, 23: 1120-1132.
[4] ZHU Feng, XUE Sheng-guo, HARTLEY W, HUANG Ling, WU Chuan, LI Xiao-fei. Novel predictors of soil genesis following natural weathering processes of bauxite residues [J]. Environmental Science and Pollution Research, 2016, 23: 2856-2863.
[5] WU Chuan, HUANG Liu, XUE Sheng-guo, PAN Wei-song, ZOU Qi, HARTLEY W, WONG Ming-hung. Oxic and anoxic conditions affect arsenic (As) accumulation and arsenite transporter expression in rice [J]. Chemosphere, 2017, 168: 969-975.
[6] XUE Sheng-guo, SHI Li-zheng, WU Chuan, WU Hui, QIN Yan-yan, PAN Wei-song, HARTLEY W, CUI Meng-qian. Cadmium, lead, and arsenic contamination in paddy soils of a mining area and their exposure effects on human HEPG2 and keratinocyte cell-lines [J]. Environmental Research, 2017, 156: 23-30.
[7] SAMAL S, RAY A K, BANDOPADHYAY A. Characterization and microstructure observation of sintered red mud-fly ash mixtures at various elevated temperatures [J]. Journal of Cleaner Production, 2015, 101: 368-376.
[8] KONG Xiang-feng, GUO Ying, XUE Sheng-guo, HARTLEY W, YE Yu-zhen, CHENG Qing-yu. Natural evolution of alkaline characteristics in bauxite residue [J]. Journal of Cleaner Production, 2017, 143: 224-230.
[9] LIU Wan-chao, CHEN Xiang-qing, LI Wang-xing, YU Yan-fen, YAN Kun. Environmental assessment, management and utilization of red mud in China [J]. Journal of Cleaner Production, 2014, 84: 606-610.
[10] ZHU Feng, HOU Jing-tao, XUE Sheng-guo, WU Chuan, WANG Qiong-li, HARTLEY W. Vermicompost and gypsum amendments improve aggregate formation in bauxite residue [J]. Land Degradation and Development, 2017, 28: 2109-2120.
[11] DAVRIS P, BALOMENOS E, PANIAS D, PASPALIARIS I. Selective leaching of rare earth elements from bauxite residue (red mud), using a functionalized hydrophobic ionic liquid [J]. Hydrometallurgy, 2016, 164: 125-135.
[12] ZHU Feng, LIAO Jia-xin, XUE Sheng-guo, HARTLEY W, ZOU Qi, WU Chuan. Evaluation of aggregate microstructures following natural regeneration in bauxite residue as characterized by synchrotron-based X-ray micro-computed tomography [J]. Science of the Total Environment, 2016, 573: 155-163.
[13] ZHOU Qiu-sheng, LI Chuang, LI Xiao-bin, PENG Zhi-hong, LIU Gui-hua, QI Tian-gui. Reaction behavior of ferric oxide in system Fe2O3-SiO2-Al2O3 during reductive sintering process [J]. Transactions of Nonferrous Metals Society of China, 2016, 26: 842-848.
[14] ZHU Feng, LI Xiao-fei, XUE Sheng-guo, HARTLEY W, WU Chuan, HAN Fu-song. Natural plant colonization improves the physical condition of bauxite residue over time [J]. Environmental Science and Pollution Research, 2016, 23: 22897-22905.
[15] GRAFE M, POWER G, KLAUBER C. Bauxite residue issues: III. Alkalinity and associated chemistry [J]. Hydrometallurgy, 2011, 108: 60-79.
[16] ZHU Feng, ZHOU Jia-yi, XUE Sheng-guo, HARTLEY W, WU Chuan, GUO Ying. Aging of bauxite residue in association of regeneration: A comparison of methods to determine aggregate stability & erosion resistance [J]. Ecological Engineering, 2016, 92: 47-54.
[17] ZHU Feng, HUANG Nan, XUE Sheng-guo, HARTLEY W, HAN Fu-song, LI Yi-wei. Effects of binding materials on micro-aggregate size distribution in bauxite residues [J]. Environmental Science and Pollution Research, 2016, 23: 23867-23875.
[18] ALSHAAL T, 
 J, BALOGH P. Phytoremediation of bauxite-derived red mud by giant reed [J]. Environmental Chemistry Letters, 2013, 11: 295-302.
J, BALOGH P. Phytoremediation of bauxite-derived red mud by giant reed [J]. Environmental Chemistry Letters, 2013, 11: 295-302.
[19] ZHU Feng, CHENG Qing-yu, XUE Sheng-guo, LI Chu-xuan, WU Chuan, TIAN Tao. Influence of natural regeneration on fractal features of residue microaggregates in bauxite residue disposal areas [J]. Land Degradation and Development, 2018, 29: 138-149.
[20] ZHU Feng, LI Yu-bing, XUE Sheng-guo, HARTLEY W, WU Hao. Effects of iron-aluminium oxides and organic carbon on aggregate stability of bauxite residues [J]. Environmental Science and Pollution Research, 2016, 23: 9073-9081.
[21] MENZIES N W, FULTON I M, MORRELL W J. Seawater neutralization of alkaline bauxite residue and implications for revegetation [J]. Journal of Environmental Quality, 2004, 33: 1877-1884.
[22] KIRWAN L J, HARTSHORN A, MCMONAGLE J B, FLEMING L, FUNNELL D. Chemistry of bauxite residue neutralisation and aspects to implementation [J]. International Journal of Mineral Processing, 2013, 119: 40-50.
[23] LI Xiao-bin, NIU Fei, TAN Jie, LIU Gui-hua, QI Tian-gui, PENG Zhi-hong, ZHOU Qiu-sheng. Removal of S2- ion from sodium aluminate solutions with sodium ferrite [J]. Transactions of Nonferrous Metals Society of China, 2016, 26: 1419-1424.
[24] CLARK M W, JOHNSTON M, REICHELT-BRUSHETT A J. Comparison of several different neutralisations to a bauxite refinery residue: Potential effectiveness environmental ameliorants [J]. Applied Geochemistry, 2015, 56: 1-10.
[25] CAPPAI G, DE G G, MUNTONI A, SPIGA D, ZIJLSTRA J J. Combined use of a transformed red mud reactive barrier and electrokinetics for remediation of Cr/As contaminated soil [J]. Chemosphere, 2012, 86: 400-408.
[26] COURTNEY R, HARRINGTON T. Growth and nutrition of Holcus lanatus in bauxite residue amended with combinations of spent mushroom compost and gypsum [J]. Land Degradation and Development, 2012, 23: 144-149.
[27] COUPERTHWAITE S J, JOHNSTONE D W, MILLAR G J, FROST R L. Neutralization of acid sulfate solutions using bauxite refinery residues and its derivatives [J]. Industrial & Engineering Chemistry Research, 2013, 52: 1388-1395.
[28] LIANG Wen-tao, COUPERTHWAITE S J, KAUR G, YAN Cheng, JOHNSTONE D W, MILLAR G J. Effect of strong acids on red mud structural and fluoride adsorption properties [J]. Journal of Colloid and Interface Science, 2014, 423: 158-165.
[29] ZHANG Ran, ZHENG Shi-li, MA Shu-hua, ZHANG Yi. Recovery of alumina and alkali in Bayer red mud by the formation of andradite-grossular hydrogarnet in hydrothermal process [J]. Journal of Hazardous Materials, 2011, 189: 827-835.
[30] KONG Xiang-feng, LI Meng, XUE Sheng-guo, HARTLEY W, CHEN Cheng-rong, WU Chuan, LI Xiao-fei, LI Yi-wei. Acid transformation of bauxite residue: conversion of its alkaline characteristics [J]. Journal of Hazardous Materials, 2017, 324: 382-390.
孔祥峰1,2,江星星1,2,薛生国1,2,黄 玲1,2,William HARTLEY3,吴 川1,2,李晓飞1,2
1. 中南大学 冶金与环境学院,长沙 410083;
2. 中南大学 国家重金属污染防治工程技术研究中心,长沙 410083;
3. Crop and Environment Sciences Department, Harper Adams University, Newport, Shropshire, TF10 8NB, United Kingdom
摘 要:赤泥是氧化铝工业生产过程产生的强碱性固体废物,其盐分含量高,对环境有害且制约着赤泥堆场的植被重建。通过土柱模拟淋溶实验,研究赤泥盐离子的动态迁移及垂直分布情况。结果表明:淋溶不同程度地将赤泥中的Na+、K+、Ca2+、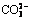 、
、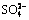 和
和 等盐分离子浸出到渗滤液中,显著降低了赤泥的盐度。赤泥中Na+和K+呈现出较高的迁移能力,从柱体的40~50 cm处向上迁移至20~30 cm处;而Ca2+的迁移能力较弱,主要分布在柱体的30~40 cm处。淋溶刚结束时,
等盐分离子浸出到渗滤液中,显著降低了赤泥的盐度。赤泥中Na+和K+呈现出较高的迁移能力,从柱体的40~50 cm处向上迁移至20~30 cm处;而Ca2+的迁移能力较弱,主要分布在柱体的30~40 cm处。淋溶刚结束时,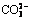 主要分布在柱体的20~30 cm处,随后向下迁移至30~40 cm处,最终受蒸发作用迁移至20~30 cm处。
主要分布在柱体的20~30 cm处,随后向下迁移至30~40 cm处,最终受蒸发作用迁移至20~30 cm处。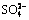 最初主要分布在柱体的40~50 cm处,最终向上迁移至20~30 cm处。
最初主要分布在柱体的40~50 cm处,最终向上迁移至20~30 cm处。 主要停留在柱体的下部,其迁移特性受蒸发的影响较小。
主要停留在柱体的下部,其迁移特性受蒸发的影响较小。
关键词:赤泥;盐分;离子迁移;土柱模拟;淋溶
(Edited by Wei-ping CHEN)
Foundation item: Project (41371475) supported by the National Natural Science Foundation of China; Project (201509048) supported by the Environmental Protection’s Special Scientific Research for Chinese Public Welfare Industry
Corresponding author: Sheng-guo XUE; Tel: +86-13787148441; E-mail: sgxue70@hotmail.com; sgxue@csu.edu.cn
DOI: 10.1016/S1003-6326(18)64686-2

