ʯú�ᷰ����������Ⱦ��������Ӧ����
��Դ�ڿ����й���ɫ����ѧ��2015���4��
�������ߣ�ФϸԪ �� �� ������ ���dz� ������ �� ϼ
����ҳ�룺1271 - 1278
Key words��vanadium; soil pollution; mobility; microbial response; stone coal smelting district
ժ Ҫ��ͨ��Ұ������о�����ʡijʯú�ᷰұ�����ܱ������з��ĺ�������̬�������Լ�������Ӧ���������������������������������ΧΪ168~1538 mg/kg�����������ô��������������������ֵ(130 mg/kg)������������ƽ���������ߴ�1421 mg/kg��β������ԭ������ұ��������������ƽ���������ֱ�Ϊ380��260��225 mg/kg��BCR˳����ȡ�����������������̬���(��������ȡ̬���ɻ�ԭ̬�Ϳ�����̬)�з�����Ϊ19.2~637.0 mg/kg��ռ�ܷ�������7.4%~42.3%����������۷�����Ϊ21.9~534.0 mg/kg�����⣬�����ߺ���������������ø���Ժͻ��������ʡ�ʯú�ᷰ����������Ⱦ��DZ����̬����ֵ�ù�ע��
Abstract: A field investigation was performed to study the content, speciation and mobility of vanadium, as well as microbial response in soil from a stone coal smelting district in Hunan province of China. The results showed that the contents of soil V ranged from 168 to 1538 mg/kg, which exceeded the maximum permissible value of Canadian soil quality for V. The mean soil V content from wasteland area reached 1421 mg/kg, and those from the areas related with slag heap, ore pile and smelting center were 380, 260 and 225 mg/kg, respectively. Based on the results of the modified BCR sequential extraction procedure, V contents in the mobile fractions varied from 19.2 to 637 mg/kg accounting for 7.4%-42.3% of total V, and those of V(+5) species were between 21.9 and 534.0 mg/kg. Soil enzyme activity and microbial basal respiration were adversely affected by high level of soil V. More attention should be paid to soil V pollution and potential hazardous surrounding the stone coal smelting district.
Trans. Nonferrous Met. Soc. China 25(2015) 1271-1278
Xi-yuan XIAO, Miao YANG, Zhao-hui GUO, Zhi-chao JIANG, Ya-nan LIU, Xia CAO
Institute of Environmental Engineering, School of Metallurgy and Environment, Central South University, Changsha 410083, China
Received 23 April 2014; accepted 13 July 2014
Abstract: A field investigation was performed to study the content, speciation and mobility of vanadium, as well as microbial response in soil from a stone coal smelting district in Hunan province of China. The results showed that the contents of soil V ranged from 168 to 1538 mg/kg, which exceeded the maximum permissible value of Canadian soil quality for V. The mean soil V content from wasteland area reached 1421 mg/kg, and those from the areas related with slag heap, ore pile and smelting center were 380, 260 and 225 mg/kg, respectively. Based on the results of the modified BCR sequential extraction procedure, V contents in the mobile fractions varied from 19.2 to 637 mg/kg accounting for 7.4%-42.3% of total V, and those of V(+5) species were between 21.9 and 534.0 mg/kg. Soil enzyme activity and microbial basal respiration were adversely affected by high level of soil V. More attention should be paid to soil V pollution and potential hazardous surrounding the stone coal smelting district.
Key words: vanadium; soil pollution; mobility; microbial response; stone coal smelting district
1 Introduction
Vanadium (V) is a ubiquitous trace element and the average content of V in soil of the world has been established at 90 mg/kg [1]. However, high content of V can be accumulated in soil due to anthropogenic sources, such as fossil fuel combustion, tailing leakage and metal plants [2]. Mining and smelting activities, especially for vanadiferous magnetite, cause V pollution widespread in many countries [3,4]. For instance, the total contents of V in soil from the vicinity of V mine in South Africa varied from 1570 to 3600 mg/kg [5], and the ranges of V contents in soil are 208-938 mg/kg from smelting area and 112-591 mg/kg from mining area in Panzhihua region of China [4].
Environmental increasing levels of soil V have raised many concerns [3]. Although the total content of V is still useful in many areas, the knowledge of speciation is of primary importance because the toxicity, mobility, bioavailability, and bioaccumulation depend on the chemical species [4]. The biological and physiological characteristics of V also depend on its oxidation states which primarily are +4 and +5 valent cations in minerals [6]. Generally, V(+5) is more mobile and more toxic to both plants and animals than V(+4) compounds [5], and it is essential to quantify V(+5) species in soil [7]. Furthermore, soil enzymatic activities are highly sensitive to trace metals and can reflect the structure and functions of microbial communities [8]. The high levels of V have an adverse effect on the soil microbial biomass and enzyme activities [9].
Vanadium compounds exist in over 50 different mineral ores and in association with fossil fuels, particularly coal and crude oil, which is mainly mined in South Africa, Russia and China [10]. Stone coal is an important source of V [11], and its resource has accounted for most of the domestic reserve of V resource in China, especially in western region of Hunan province [12], where there are intensive industrial activities of V extraction from stone coal. Soil V contamination from the vicinity of vanadiferous magnetite smelting areas in China was serious [4]. However, there is little information about soil V pollution, mobility as well as microorganism biochemical characteristics affected by industrial activities of V extraction from stone coal. In this study, the main aims were: 1) to study soil V pollution characteristics in different areas surrounding a stone coal smelting district; 2) to elucidate V speciation, mobility and V(+5) species in soils; and 3) to determine the effect of V pollution on soil enzyme activities and basal respiration.
2 Materials and methods
2.1 Study area and soil samples collection
The study was conducted in the district surrounding a plant for V extraction from stone coal, which locates in southern Chenxi county, western Hunan province, China (Latitude of 28��10��12.40��-28��10��23.42��, Longitude of 110��13��18.81��-110��13��36.34��). The V smelting plant was set up and manufactured V2O5 product in 2005.
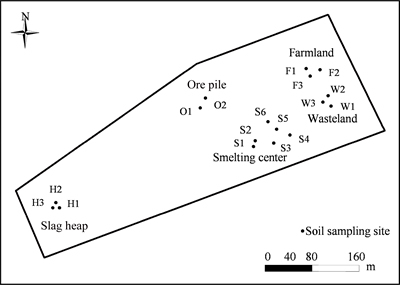
Fig. 1 Sampling sites surrounding V smelting district
Table 1 Description of soil samples collected from different areas surrounding V smelting district
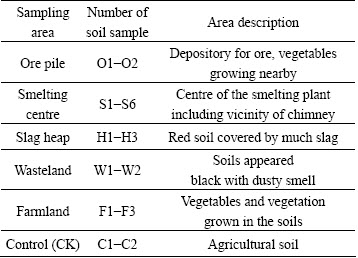
A total of 17 soil samples in the depth of 0-20 cm were collected from five areas around the V smelting plant in March, 2012, including ore pile area, smelting centre area, slag heap area, farmland area and wasteland area (Fig. 1). Area description and number of soil sample are summarized in Table 1. As a control, two soil samples from agricultural field, which is 1 km far away from the smelting district, were collected. Each soil sample consisted of five homogenized subsamples weighing about 1.5 kg, was placed in polyethylene bags and transported to the laboratory. After large pieces of plant material and stones were removed, soils were sieved through 2 mm mesh. Part of samples were kept moist in the dark at 4 ��C for soil microbial activities analysis. The remaining soil samples were air-dried, crushed and sieved through 0.2 mm mesh for analysis.
2.2 Sample analysis
2.2.1 Soil physico-chemical properties
The physico-chemical properties of soil samples were analyzed according to the general methods described by LU [13]. The pH was determined using pH meter with a ratio of soil to deionized water of 1:2.5 (w/v). The content of organic matter (OM) was determined by a volumetric method of K2Cr2O7-heating, and cation exchange capacity (CEC) was determined by extracting with a 1.0 mol/L NH4OAc solution (pH 7.0). Free Fe2O3 and free Al2O3 in soil were extracted with a dithionite citrate system buffered with sodium bicarbonate, and amorphous Fe2O3 and amorphous Al2O3 were extracted with ammonium and oxalate under dark place, respectively. Then, they were determinated by spectrophotometric method.
2.2.2 Soil V species
Soil samples were digested with the mixture of HNO3-H2O2 to determine the total V content [14]. The modified three-step Community Bureau of Reference (BCR) sequential extraction was described by RAURET et al [15] and applied to assessing V fractions in the samples. It basically consists of three extraction steps, i.e., extraction with 0.11 mol/L acetic acid (acid- extractable fractions), extraction with 0.5 mol/L hydroxylamine hydrochloride at pH 1.5 (reducible fraction), and reaction with 8.8 mol/L H2O2 followed by extraction with 1.0 mol/L ammonium acetate at pH 2 (oxidizable fraction). Additionally, residual fraction, which consisted of dissolving the final residue, was digested using the same digestion procedure described for total V determination [14].
Vanadium leachability from soil was determined using standard toxicity characteristic leaching procedure (TCLP) [16]. V(+5) species in soils were extracted with 0.1 mol/L Na2CO3 described by MANDIWANA and PANICHEV [17].
Concentrations of V in extractable and digested solutions were determined with an inductively coupled plasma optical emission spectrometer (ICP-OES, IRIS Intrepid II XSP, USA). The accuracy of the digestion procedure and analytical method was checked with standard reference material for soil (GBW-08303) obtained from China National Center, yielding analytical error <10%.
2.2.3 Soil enzyme activity and basal respiration
Activities of urease and dehydrogenase in soils were determined as described by TABATABAI et al [18] and expressed with NH4+-N/soil (mg/g) and triphencyl formazan (TPF)/soil (mg/g) in 24 h, respectively. Catalase activity was measured by titration method [19] and the results were expressed as mL/g (mL is the volume of 0.1 mol/L KMnO4). Soil basal respiration rate was determined using the alkali-trap method described by ANDERSON [20].
2.3 Statistics analysis
Statistical analyses were performed using Microsoft Excel 2003 and SPSS 15.0. Analysis of variance (ANOVA) was used to examine the statistical significant difference among the V contents of soil from different areas surrounding the V smelting district. Spearman��s correlation coefficient was used to measure the degree of relation between soil physico-chemical parameters and V content. A probability level of 0.05 was significant.
3 Results and discussion
3.1 Soil V content and speciation
There was a large variation of V contents in soil from investigated areas (Fig. 2). The ranges of V contents in soil from ore pile and smelting areas were 257-262 mg/kg and 173-317 mg/kg, respectively. Soil V content from slag heap area ranged from 258 to 576 mg/kg, while that from farmland area (169-227 mg/kg) changed slightly. The soils from wasteland area contained high content of V (1268-1538 mg/kg), indicating that the land was heavily contaminated by smelting wastewater (Table 1). In general, the highest average content of V in soil was found in wasteland area (1421 mg/kg), and followed by slag heap area (380 mg/kg), ore pile area (260 mg/kg), smelting area (225 mg/kg) and farmland area (189 mg/kg), which was mainly attributed to the anthropogenic sources, such as dust deposition from V excavation of stone coal, slag dump waste, fly ash of smelting smoke dust from chimney (Table 2). The results were in good agreement with the research that the contents of V in soils both from smelting area and mining area are higher than those from agricultural area [5]. Vanadium contents higher than 600 mg/kg in urban soils in Mexico were also related to the activities of refinery, thermoelectric plant, and chemical industry [21].

Fig. 2 Vanadium content in soils from different areas surrounding V smelting district
V contents in all soil samples from the surrounding areas of V smelting district exceeded the soil V background value of 86 mg/kg in Hunan province [22] and the maximum permissible value (MPV) of 130 mg/kg of Canadian soil quality for V [23] (Fig. 2). Especially, V content in soil from wasteland area and soil H2 from slag heap area was 9.75-11.8 and 4.43 times as much as MPV, respectively. The exceeding percentage was 64.7% compared with the intervention value (250 mg/kg) of V in soils regulated in Netherlands [24].
Generally, the fraction of V in soil mainly existed in residual fraction, and then followed by oxidizable, reducible and acid-extractable fractions (Fig. 3). The acid-soluble fractions are considered to represent the most mobile forms of V [4,25], and be useful for assessment of the available (potentially hazardous) fraction to environmental and biological systems. The content of acid-extractable V in soils from the investigated areas, except wasteland area, ranged from 0.83 to 6.33 mg/kg accounting for 0.32%-1.88% of total V, which represented a much lower contribution to total V content. However, the acid-extractable V from wasteland area highly reached up to 8.60% with mean content of 132 mg/kg (Fig. 3), indicating that soil V from wasteland area was more mobile. The content of reducible V in the wide range of 15.1-343 mg/kg accounted for 5.63%-34.4% of total V, which was also relatively high in slag heap area, wasteland area and farmland area, and may initially cause a release of V into the environment [26]. The fraction of oxidizable V varied from 2.1 to 160 mg/kg and represented 0.81%- 22.9% of total V, which was also high in slag heap, wasteland and farmland area. The residual fraction of V for all samples accounted for 57.7%-58.8% of total V content, which was in good agreement with previous studies that soil V was usually associated with residual matter [4,25].
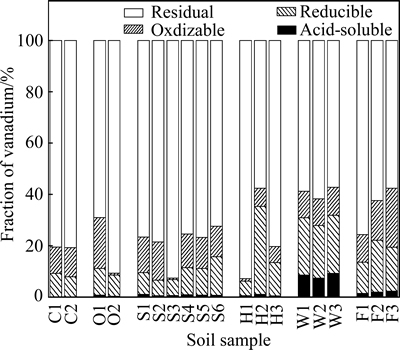
Fig. 3 Chemical speciation of V in soils according to BCR sequential extraction procedure
The potential bioavailability of metals in soil was characterized by the sum of the first three fractions and its ratio to total content of heavy metals extracted by modified BCR [27]. High content and percentage of the first three fractions for heavy metal can reflect not only a great tendency of bioavailability, but also a high level of anthropogenic contamination [28,29]. In this study, the sum of the first three fractions for V contained 7.4%-42.3% of total V content in soils from stone coal smelting district with an average value of 25.5%, and most of them were higher than that from control area (19.6%) (Fig. 3). Previous research has been reported that the first three steps of the BCR sequential extraction scheme accounted for 15%-42% of total V from soils near an oil-fired power plant [25]. The high sum of the first three extractable fractions of V indicated that contents of soil V were greatly affected by intensive smelting activity [21].
3.2 Relationship between soil V content and physico- chemical properties
The pH values of soil samples collected from the areas of V smelting district ranged from 3.12 to 6.23 (Table 2), and those from smelting area and wasteland area were very lower than the mean soil background pH value of 5.6 in Hunan province [22]. The content of OM ranged from 10.3 to 28.6 g/kg, and that of CEC was between 11.0 and 42.2 cmol/kg. The relatively high OM content and elevated CEC in some soil samples could indicate a good natural capacity of soils for immobilizing V. The contents of free Fe2O3 and amorphous Fe2O3 varied in the ranges of 5.21-11.1 g/kg and 1.03-8.20 g/kg, and the average values were 7.55 and 3.48 g/kg, respectively. The content of free Al2O3 was between 35.9 and 41.3 g/kg, while that of amorphous Al2O3 varied in the range of 1.94-9.52 g/kg.
Table 2 Basic physico-chemical properties of soil samples
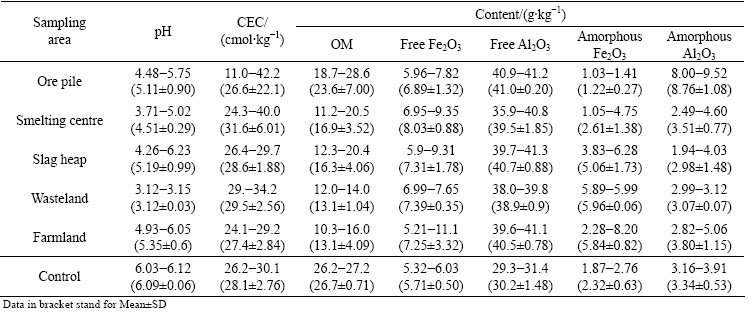
Table 3 Relationship between content of V and soil basic properties
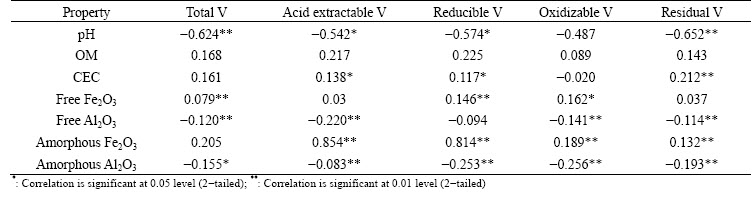
Soil properties are important factors modifying metal bioavailability to ecological receptors [30]. The pH value was negatively correlated with total and all fractions of V except oxidizable fraction, indicating that its mobility increased in acidic soils (Table 3). CEC was positively correlated with acid soluble, reducible and residual V, which was confirmed by the other results [31]. There was no significant correlation between contents of V and OM in soils, which is not in agreement with the report that a significantly negative correlation between soil V and OM content was found [32].
Content of free Fe2O3 was positively correlated with total V, fractions of reducible and oxidizable V, and amorphous Fe2O3 was also positively correlated with all fractions of V by BCR extraction (P<0.05) (Table 3). These correlations indicate the important contribution of amorphous Fe oxides in soil V immobilization, suggesting that contents of Fe oxide significantly affected the absorption and adsorption of soil V. These positive correlations which can be explained by the (hydr)oxides of Fe are good adsorbers for V ions because of the occurrence of the element in clay minerals [33]. In contrast, content of free Al2O3 was negatively correlated with total V, fractions of acid-extractable, oxidizable and residual V, and there was also a significantly negative relationship between amorphous Al2O3 and total, all kinds of V fractions. Other result has been confirmed that high contents of urban soil V mostly due to anthropogenic activity showed a poor relation with this geochemically stable mineral of Al2O3 [21].
3.3 Contents of TCLP V and V(+5) species
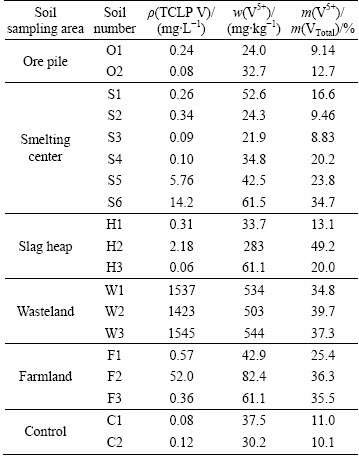
The TCLP V contents in soil from wasteland area were higher than 1423 mg/L (Table 4), and those in soils S5 and S6 from smelting area and soil F2 of farmland area were 5.76, 14.2 and 52.0 mg/L, respectively, and which were far above the universal treatment standard for non-wastewater of 1.6 mg/L TCLP set by USEPA [34]. The result indicates that V could be leached from the soils and thus be released to the surface and groundwater as well as accumulated in the food chain of the study area.
The pentavalent cation of V is considerably more soluble than the trivalent cation, which is readily dissolved by groundwater, easily leached from soils into water, and can be transported over long distance [35]. The contents of V(+5) species in soil samples ranged from 21.9 to 544 mg/kg, accounting for 8.83%-49.2% of total V (Table 4). Especially, the contents of V(+5) speciation in soil H2 of slag heap area and soil from wasteland area exceeded 283 mg/kg, and were much higher than MPV value of 130 mg/kg [23]. The percentage of V(+5) accounted for about 11.0% of total V for control soil samples, while those from smelting centre, slag heap, wasteland and farmland area were almost higher than 20.0%. V(+5) content in soil H2 of slag heap area constituted 49.2% of total V, and those observed in soil W1-W3 of wasteland and F2-F3 of farmland were 34.8%-39.7% and 35.5%-36.3%, respectively. Previous results showed that the contents of V(+5) species were found to be in the range of 620�C 1680 mg/kg accounting for about 50% of total V in soils from V mine of South African [6]. The amount of V(+5) in agricultural soil collected from the vicinity of thermal power plant was 32.6% of the total V, while that from non-industrial field was only 8.9% [7]. The results show that soils from industrial areas can have higher content of V(+5) species due to intensive V-related anthropogenic activities such as emission of particulate matter containing V, metal plants and phosphorite treating factories [36]. Notwithstanding, our present results showed that higher V(+5) species occurred at smelting centre area, slag heap area and wasteland area, and potential bioavailability of metals in soil characterized by the sum of the first three fractions of V by BCR extraction procedure as well as TCLP V content was also higher in the same above areas (Fig. 3 and Table 4). Therefore, the risk of soil V pollution surrounding the stone coal smelting district is serious.
Table 4 Contents of TCLP V and V(+5) in soils from different areas surrounding V smelting district
3.4 Soil enzyme activity and basal respiration
Soil enzymes were used to estimate the adverse effects of various pollutants on soil health [8,37]. Urease activity in soil samples surrounding the V smelting district was significantly lower than that in control area (P<0.05), and those from wasteland, smelting centre and slag heap area accounted for 36.2%, 48.5% and 52% of urease activity in control soils, respectively (Fig. 4). Dehydrogenase activities from all V polluted soils were obviously lower than that from control area, and only accounted for 24.1%-47.9% of that in CK soils (P<0.05). Similarly, soil catalase activities from ore pile, smelting centre and wasteland area were also significantly inhibited and were 36.7%, 53.7% and 73.4% of that from CK area (P<0.05), respectively. Previous research was also reported that high V content showed negative correlation with urease and dehydrogenase activities [9], indicating that soil enzyme activities were good indicators of soil V pollution and could be useful for monitoring changes in soil health.

Fig. 4 Enzyme activities and basal respiration in soils from different areas surrounding V smelting district
Soil basal respiration has been largely used to estimate heavy metal toxicity [38]. In this study, Soil basal respiration from ore pile, slag heap, and farmland area with high V content was significantly lower than that from control area (P<0.05) (Fig. 4). Especially, soil basal respiration was not detected in wasteland area, showing that high level of V has a serious adverse effect on basal respiration. The result suggested that there is high ecological risk of soil V pollution by stone coal smelting activity.
4 Conclusions
1) The V pollution of soil surrounding the stone coal smelting district was serious. The contents of V in all soil samples substantially exceeded the maximum permissible value of Canadian soil quality for V.
2) Vanadium in soil was predominantly present in residual form and characterized as follows: residual> oxidizable �� reducible > acid-extractable; however, the potential high mobility of V according to the modified BCR procedure, as well as high content of V(+5) species was found in slag heap area, wasteland area and farmland area. Soil enzyme activities and basal respiration were adversely affected by V pollution. Therefore, some countermeasures should be taken into account to prevent future ecological risk.
References
[1] REIMANN C, CARITAT P. Chemical elements in the environment [M]. Berlin: Springer-Verlag, 1998.
[2] NRIAGU J O. Vanadium in the environment, Part 1: Chemistry and biochemistry [M]. New York: John Wiley and Sons, Inc, 1998.
[3] HOPE B K. An assessment of the global impact of anthropogenic vanadium [J]. Biogeochemistry, 1997, 37: 1-13.
[4] TENG Yan-guo, YANG Jie, SUN Zong-jian, WANG Jin-sheng, ZUO Rui, ZHENG Jie-qiong. Environmental vanadium distribution, mobility and bioaccumulation in different land-use districts in Panzhihua region, SW China [J]. Environmental Monitoring and Assessment, 2011, 176: 605-620.
[5] PANICHEV M N, MANDIWANA K, MOEMA D, MOLATLHEGI R, NGOBENI P. Distribution of vanadium(V) species between soil and plants in the vicinity of vanadium mine [J]. Journal of Hazardous Materials, 2006, 137: 649-653.
[6] KHAN S, KAZI T G, KOLACHI N F, BAIG J A, AFRIDI H I, SHAH A Q, KUMAR S, SHAH F. Hazardous impact and translocation of vanadium (V) species from soil to different vegetables and grasses grown in the vicinity of thermal power plant [J]. Journal of Hazardous Materials, 2011, 190: 738-743.
[7] CHEN Z L, OWENS G. Trends in speciation analysis of vanadium in environmental samples and biological fluids��A review [J]. Acta Chimica Sinica, 2008, 607: 1-14.
[8] HUANG Shun-hong, PENG Bing, YANG Zhi-hui, CHAI Li-yuan, ZHOU Li-cheng. Chromium accumulation, microorganism population and enzyme activities in soils around chromium- containing slag heap of steel alloy factory [J]. Transactions of Nonferrous Metals Society of China, 2009, 19(1): 241-248.
[9] XIAO Xiao-yuan, YANG Miao, GUO Zhao-hui, LUO Yue-ping, BI Jun-ping. Permissible value for vanadium in allitic udic ferrisols based on physiological responses of green Chinese cabbage and soil microbes [J]. Biological Trace Element Research, 2012, 145(2): 225-232.
[10] GUMMOW B. Vanadium: Environmental pollution and health effects [M]//Encyclopedia of Environmental Health. Amsterdam, the Netherlands: Elsevier, 2011: 628-636.
[11] WANG Li, SUN Wei, LIU Run-qing, GU Xiao-chuan. Flotation recovery of vanadium from low-grade stone coal [J]. Transactions of Nonferrous Metals Society of China, 2014, 24(4): 1145-1151.
[12] JIANG Kai-qi, GUO Zhao-hui, XIAO Xi-yuan. Regional distribution of vanadium ore resources and extraction processes of vanadium from stone coal in China [J]. Hydrometallurgy of China, 2010, 29(1): 216-219, 224. (in Chinese)
[13] LU Ru-Kun. Soil and agro-chemistry analytical methods [M]. Beijing: Agriculture Science and Technology Press of China, 1999, 15-20: 223-227. (in Chinese)
[14] USEPA. Acid digestion of sediments sludge and soils [EB/OL]. http://www.epa.gov/ sw-846/pdfs/3050b.pdf. 1996: 1-12.
[15] RAURET G,  J, SAHUQUILLO A, RUBIO R, DAVIDSON C, URE A, QUEVAUVILLER P. Improvement of the BCR three step sequential extraction procedure prior to the certification of new sediment and soil reference materials [J]. Journal of Environmental Monitoring, 1999, 1(1): 57-61.
J, SAHUQUILLO A, RUBIO R, DAVIDSON C, URE A, QUEVAUVILLER P. Improvement of the BCR three step sequential extraction procedure prior to the certification of new sediment and soil reference materials [J]. Journal of Environmental Monitoring, 1999, 1(1): 57-61.
[16] Test method for the evaluation of solid waste, physical/chemical methods, Method 1311: Toxicity characteristic leaching procedure [EB/OL]. http://www.epa.gov/wastes/hazard/testmethods/sw846/ pdfs/131. 1992.
[17] MANDIWANA K L, PANICHEV N. Electrothermal atomic absorption spectrometric determination of vanadium (V) in soil after leaching with Na2CO3 [J]. Analytica Chimica Acta, 2004, 517: 201-206.
[18] TABATABAI M A, BEZDICEK D F, SMITH S. Soil enzymes, methods of soil analysis, Part 2: Microbial and biochemical properties [M]. Madison, WI: Soil Sci Soc Am 1994: 775-833.
[19] YAN Chang-sheng. Research method of soil fertility [M]. Beijing: Chinese Agriculture Press, 1988: 277-279. (in Chinese)
[20] ANDERSON J P E. Soil respiration [M]//Methods of soil analysis, Part 2: Chemical and microbial properties. Madison, WI: Soil Science Society of America, 1982: 831-872.
[21] HERNANDEZ H, RODRIGUEZ R. Geochemical evidence for the origin of vanadium in an urban environment [J]. Environmental Monitoring and Assessment, 2012, 184(9): 5327-5342.
[22] China National Environmental Monitoring Center. The background values of soil elements in China [M]. Beijing: China Environmental Science Press, 1990: 364-389. (in Chinese)
[23] Canadian Council of Ministers of the Environment. Canadian soil quality guidelines for the protection of environmental and human health: vanadium [S]. 1999.
[24] LIJZEN J P A, BAARS A J, OTTE P F, RIKKEN M, SWARTIES F A, VERBRUGGEN E M J, van WEZEL A P. Technical evaluation of the intervention values for soil/sediment and groundwater. Human and ecotoxicological risk assessment and derivation of risk limits for soil, aquatic sediment and groundwater [J]. Advance in Earth Sciences, 2001, 16(2): 267-271.
[25]  Y. Speciation of beryllium, nickel, and vanadium in soil samples from Csepel Island, Hungary [J]. Fresenius Journal of Analytical Chemistry, 2001, 370: 768-775.
Y. Speciation of beryllium, nickel, and vanadium in soil samples from Csepel Island, Hungary [J]. Fresenius Journal of Analytical Chemistry, 2001, 370: 768-775.
[26] van ZINDEREN BAKKER E M, JAWORSKI J F. Effects of vanadium in the Canadian environment [M]. Ottawa: National Research Council Pub., 1980: 1-94.
[27]  S, KARTAL S. Comparison of metal fractionation results obtained from single and BCR sequential extractions [J]. Bulletin of Environment Contamination and Toxicology, 2005, 75: 180-188.
S, KARTAL S. Comparison of metal fractionation results obtained from single and BCR sequential extractions [J]. Bulletin of Environment Contamination and Toxicology, 2005, 75: 180-188.
[28] GUEVARA-RIBA A, SAHUQUILLO A, RUBIO R, RAURET G. Assessment of metal mobility in dredged harbour sediments from Barcelona [J]. Science of the Total Environment, 2004, 321: 241-255.
[29] ZHI Ying-biao, WANG Zai-lan, MA Zhong, WANG Zhong-sheng, DENG Zi-fa, LI Hong-li. The speciation and bioavailability of heavy metals pollutants in soil along highway in Erdos [J]. Acta Ecologica Sinica, 2007, 27: 2030-2039. (in Chinese)
[30] BRADHAM K D, DAYTON E A, BASTA N T PAYTON M E, LANNO R P. Effects-plants, soil, invertebrates, microflora: soil invertebrates effect of soil properties on lead bioavailability and toxicity to earthworms [J]. Environmental Toxicology and Chemistry, 2006, 25: 769-775.
[31] CAPPUYNS V, SLABBINCK E. Occurrence of vanadium in Belgian and European alluvial soils [J]. Applied and Environmental Soil Science, 2012, 2012: 1-12.
[32] TUME P, BECH J, TUME L, BECH J, REVERTER F, LONGAN L, CENDOYA P. Concentrations and distributions of Ba, Cr, Sr, V, Al, and Fe in Torrelles soil profiles (Catalonia, Spain) [J]. Journal of Geochemical Exploration, 2008, 96(2-3): 94-105.
[33] NAEEM A, WESTERHOFF P, MUSTAFA S. Vanadium removal by metal (hydr)oxide adsorbents [J]. Water Research, 2007, 41(7): 1596-1602.
[34] USEPA. Best demonstrated available technology (BDAT) background document for inorganic chemical production wastes �� k176, k177, k178[EB/OL]. http://www.epa.gov/osw/inforesources/ pubs/ pub- b.htm. 2001.
[35] USEPA. Ecological soil screening levels for vanadium [EB/OL]. Interim Final. OSWER Directive 9285.7-5. http://www.epa.gov/ nscep/index.html. 2005.
[36] POLEDNIOK J, BUHL F. Speciation of vanadium in soil [J]. Talanta, 2003, 59: 1-8.
[37] DICK R. Soil enzyme activities as integrative indicators of soil health [M]//Biological Indicators of Soil Health. Wallingford, UK: CAB International, 1997: 121-156.
[38] DAI J, BECQUER T, ROUILLER J H, REVERSAT G, BERNHARD-REVERSAT F, LAVELLE P. Influence of heavy metals on C and N mineralization and microbial biomass in Zn-, Pb-, Cu-, and Cd-contaminated soils [J]. Applied Soil Ecology, 2004, 25: 99-109.
ФϸԪ���� ���������ͣ����dz��������У��� ϼ
���ϴ�ѧ ұ���뻷��ѧԺ ��������ϵ����ɳ 410083
ժ Ҫ��ͨ��Ұ������о�����ʡijʯú�ᷰұ�����ܱ������з��ĺ�������̬�������Լ�������Ӧ���������������������������������ΧΪ168~1538 mg/kg�����������ô��������������������ֵ(130 mg/kg)������������ƽ���������ߴ�1421 mg/kg��β������ԭ������ұ��������������ƽ���������ֱ�Ϊ380��260��225 mg/kg��BCR˳����ȡ�����������������̬���(��������ȡ̬���ɻ�ԭ̬�Ϳ�����̬)�з�����Ϊ19.2~637.0 mg/kg��ռ�ܷ�������7.4%~42.3%����������۷�����Ϊ21.9~534.0 mg/kg�����⣬�����ߺ���������������ø���Ժͻ��������ʡ�ʯú�ᷰ����������Ⱦ��DZ����̬����ֵ�ù�ע��
�ؼ��ʣ�����������Ⱦ�����ԣ�������Ӧ��ʯú�ᷰ��
(Edited by Xiang-qun LI)
Foundation item: Project (41201492) supported by the National Natural Science Foundation of China
Corresponding author: Zhao-hui GUO; Tel: +86-731-88836442; E-mail: zhguo@csu.edu.cn
DOI: 10.1016/S1003-6326(15)63727-X

