Microstructural evolution during solution treatment of thixoformed AM60B Mg alloy
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2011���4��
�������ߣ��ƺ��� ����� ��ӱ ��Զ
����ҳ�룺745 - 753
�ؼ��ʣ�AM60Bþ�Ͻ�; �������; ���ܴ���; ��֯; ����ѧ
Key words��AM60B Mg alloy; thixoforming; solution heat treatment; microstructure; kinetics
ժ Ҫ���о��������AM60Bþ�Ͻ��ڹ��ܴ��������е���֯�ݱ估���ȴ�������ѧ������������������AM60Bþ�Ͻ��ڹ��ܴ�������������֯�ݱ�ɷ�Ϊ�����Σ���һ��Ϊ������ (Mg17Al12) ���Ѹ���ܽ⡢���ʿ��پ��Ȼ��������ֻ����ڶ���Ϊ�������������ξ�������������Ρ��ڵ�һ�Σ�����ϸС�������������С�Ķ��ξ�������ɸ��ܽ���̽϶̣�������AlԪ�صľ��Ȼ�����Ҫ��Խϳ���ʱ�䣬ͬʱ����֯��ò�������ߴ��кܴ�仯����395 ��C����ʱ����һ�γ���Լ1 h�����һ����ȵڶ�����Ҫ�ϳ���ʱ�䣬�ڱ���ʵ��������Լ60 h����һ�εĿ��پ��Ȼ�ʹAl��Ũ���ݶ��ڵڶ��δ���ȼ�С������ڸý���֯�仯�������ԡ��ڵ�һ�Σ�����������ij���ģʽ�����ݺ�����ϵ�������ξ�������Ӵ�ͳ�ij��̣�Ȼ�����ڵڶ������ߵij���ʽ��ͬ��
Abstract: The microstructural evolution and kinetic characteristics were studied during solution treatment of AM60B Mg alloy prepared by thixoforming. The results indicate that the microstructural evolution includes two stages: the first stage involves rapid dissolution of eutectic �� (Mg17Al12) phase, homogenization and coarsening, and the second stage is regarded as normal grain growth consisting of primary ��-Mg particles (primary particles) and secondary ��-Mg grains (secondary grains). In the first stage, the dissolution completes in a quite short time because the fine �� phase can quickly dissolve into the small-sized secondary grains. The homogenization of Al element needs relatively long time. Simultaneously, the microstructure morphology and average grain size obviously change. The first stage sustains approximately 1 h when it is solutionized at 395 ��C. Comparatively, the second stage needs very long time and the microstructure evolves quite slowly as a result of low Al content gradient and thus low diffusivity of Al element after the homogenization of the first stage. The growth model of primary particles obeys power function while that of the secondary grains follows the traditional growth equation in the first stage. In the second stage, both of the primary particles and secondary grains behave a same model controlled by diffusion along grain boundaries and through crystal lattice.

HUANG Hai-jun, CHEN Ti-jun , MA Ying, HAO Yuan
Key Laboratory of Gansu Advanced Non-ferrous Metal Materials,
Lanzhou University of Technology, Lanzhou 730050, China
Received 25 September 2010; accepted 24 December 2010
Abstract: The microstructural evolution and kinetic characteristics were studied during solution treatment of AM60B Mg alloy prepared by thixoforming. The results indicate that the microstructural evolution includes two stages: the first stage involves rapid dissolution of eutectic �� (Mg17Al12) phase, homogenization and coarsening, and the second stage is regarded as normal grain growth consisting of primary ��-Mg particles (primary particles) and secondary ��-Mg grains (secondary grains). In the first stage, the dissolution completes in a quite short time because the fine �� phase can quickly dissolve into the small-sized secondary grains. The homogenization of Al element needs relatively long time. Simultaneously, the microstructure morphology and average grain size obviously change. The first stage sustains approximately 1 h when it is solutionized at 395 ��C. Comparatively, the second stage needs very long time and the microstructure evolves quite slowly as a result of low Al content gradient and thus low diffusivity of Al element after the homogenization of the first stage. The growth model of primary particles obeys power function while that of the secondary grains follows the traditional growth equation in the first stage. In the second stage, both of the primary particles and secondary grains behave a same model controlled by diffusion along grain boundaries and through crystal lattice.
Key words: AM60B Mg alloy; thixoforming; solution heat treatment; microstructure; kinetics
1 Introduction
Magnesium alloys, as the lightest structural alloys commercially used, have wide applications in various fields such as automotive, aircraft and electronics industries[1]. Energy conservation is the main factor which drives drastically the latent consumption demand of magnesium alloys. AM60B alloy is one of the most widely used commercial magnesium alloys and most of its components are fabricated by high pressure diecasting (HPDC) at present[2]. However, this forming technology has the shortcomings like gas poles in the resulting components. So the mechanical properties of the components are relatively low and they cannot be heat treated, which limits its applications. The promising technology, semisolid forming or semisolid processing (SSF/SSP), combined the advantages of both casting and forging, has been concerned extensively on the view of low porosity, reduction of macrosegregation, fine microstructure, especially low gas entrapment and further improvement of mechanical properties by following heat treatment[2-3].
The reported heat treatment procedures of SSF products are mostly those that are adopted to conventional dendritic castings and are deemed to be not suitable to SSF castings due to their different solidification histories and microstructures[4]. The dissolution and precipitation of �� (Mg17Al12) phase for the conventional casting Mg-Al-based alloys and the related mechanical properties have been extensively investigated in previous works[5-10]. During solutionizing, the dissolution of hard �� phase into the ��-Mg matrix causes the decrease of hardness and ductility and the increase of tensile strength. Subsequent aging induces significant precipitation hardening[11]. However, the �� phase without Guinier�CPreston (GP) zones preferentially precipitates along grain boundaries in a discontinuous form, which results in relatively poor strengthening response[12]. Favorable continuous precipitation (CP) that competes with discontinuous precipitation (DP) is related to the Al content in solidsolution[13]. However, few studies have focused on the microstructural evolution during solid solution treatment of SSF alloys, especially in the early stage, which often exerts a huge influence on the final mechanical properties. Furthermore, the solution kinetics has rarely involved.
Therefore, in this study, the microstructural evolution process of AM60B magnesium alloy prepared by thixo-diecasting (TDC) was investigated during solid solution treatment, and the kinetics of grain growth was also discussed. The variation characteristics of �� phase and other phases during the solution process, especially in the early stage of solution treatment, were particularly discussed.
2 Experimental
The AM60B alloy used in this work was prepared by TDC process. The preparation details have been reported elsewhere[14-15]. Here, the brief process was only described. A quantity of commercial AM60B alloy was first remelted at 790 ��C in a resistance furnace. The melt then was held for 10 min after 1.2% MgCO3 (mass fraction) was added for grain refinement, and finally at (720��5) ��C poured into a permanent mould with a cavity of 70 mm in diameter and 160 mm in length. Following, the cast rods were machined into small rods with dimensions of d 68 mm��110 mm. The small rods were heated in a resistance furnace at 610 ��C for 130 min under protection of argon gas and then transferred into the chamber of a HPDC machine and formed. The chamber temperature of 400 ��C, mould temperature of 300 ��C and injection speed of 3 m/s were employed. A photograph of the formed product is shown in Fig.1.

Fig.1 Photograph of thixoformed AM60B alloy
The samples used for heat treatment with dimensions of 8 mm��8 mm��10 mm were machined from the cross gate of a formed product. These samples were solutionized at 395 ��C for different time, ranging from 2 min to 60 h, and then were quickly water-quenched. The temperature fluctuation was kept within ��1 ��C.
A cross-section of the heated samples was ground and polished by standard metallographic technique to 0.25 ��m-diamond finish, followed by etching using an aqueous solution of 60% ethylene glycol, 20% acetic acid and 5% HNO3 (volume fraction) to visualize grain boundaries and examine grain sizes. Subsequently, part of the samples were ground and polished again and then etched by an alcohol solution with 4% HNO3 (volume fraction) to show microstructure details and thus deduce the microstructural evolution. A JSM6700F electron scanning microscope (SEM) equipped with energy- dispersive spectroscope (EDS) was applied to observing the detailed microstructures and examining the contents of Al in ��-Mg phases in the different microstructures. The related quantitative analysis of �� phase was carried out by IMAGE-PRO PLUS software. The linear intercept method described in the ASTM standard E112-96 was used to examine grain sizes.
3 Results and discussion
3.1 As-cast microstructure
To verify the microstructural evolution during solutionizing, the as-cast microstructure of the TDC AM60B alloy should be first clarified. Figure 2 shows the typical microstructures of the TDC AM60B alloy. It can be seen that the alloy has a homogeneous microstructure with nearly globular primary ��-Mg particles uniformly distributed in the secondarily solidified matrix (Fig.2(a)). There are often several small-sized secondarily solidified particles (liquid phase pools within the primary particles in the semisolid microstructure prior to forming) within each primary particle (Fig.2(b)). The secondarily solidified structure is composed of fine secondary �� dendrites (to differentiate from the primary �� phase, the primary �� phase solidified from the liquid phase in semisolid sate is named as secondary ��-Mg phase) and discontinuous interdendritic �� phase (Fig.2(c)). As shown in Fig.2(d), the eutectic �� phase distributes in skeleton shapes within the eutectic �� phase.
The solidification of semisolid slurry in die cavity is named secondary solidification and plays an important role in determining the final microstructure and mechanical properties[16]. Three stages can be considered for the secondary solidification process. First, some of the secondary primary �� phase directly grows up on the primary �� particles without nucleation in order to rapidly decrease the Mg content in the liquid phase close to the primary particles, forming the serrated or annular structures. Then, nucleation occurs and lots of fine equiaxed �� dendrites form in the whole liquid phase. Finally, the solidification completes by eutectic reaction. Because the size of residual liquid layers or pools is very small when the eutectic reaction occurs, the eutectic �� preferentially attaches the secondary primary dendrites to directly grow up without nucleation and only the eutectic �� phase is left between the dendrites (Fig.2(d)), that is to say, the resulting eutectic structures belong to divorced eutectic.

Fig.2 As-thixodiecasted microstructures of AM60B alloy: (a) OM image; (b-d) SEM images of secondarily solidified particles within primary particles, details of secondarily sodified structure and eutectic phase, respectively
3.2 Microstructural evolution during solution heat treatment
According to Mg-Al equilibrium binary phase diagram[11], �� phase will dissolve into �� phase to form Al supersaturated ��-Mg solid solution during solution heat treatment. Figure 3 shows the microstructures of the TDC AM60B treated at 395 ��C for different durations. As the holding time increases, an obvious characteristic is that the �� phase existed in the secondarily solidified structure disappears gradually. It can be found that when the alloy is heated for 2 min, more than half of the �� phase has dissolved (comparing with Fig.2(b) and Fig.3(a)). With increasing the time to 6 min, most of the �� phase disappears (Fig.3(b)). When the time is prolonged to 10 min, only small-sized �� phase particles are left (Fig.3(c)). From Figs.3(c)-(f), it can be suggested that the dissolution basically completes in 1 h, and further treatment only makes the difference between the primary particles and secondarily solidified structures unclear. Figure 4 quantitatively exhibits the change of volume fraction of �� phase. It shows that the volume fraction of �� phase decreases sharply within 10 min and then the rate becomes slow in the following 20 min. As shown in Fig.3(e) and Fig.4, the �� phase basically disappears after being heated for 1 h. Simultaneously, the morphologies of �� phase significantly change with the variation of its volume fraction. Figure 5 implies that the irregular and discontinuous �� phase becomes small and diffuses gradually.
It is also found that the contrast difference between the primary particles and secondarily solidified structures becomes indistinct as the volume fraction of �� phase decreases (comparing with Figs.3(a)-(e)) and completely disappears after 60 h. The microstructure becomes into a uniform gray matrix (Fig.3(f)). But it should be noted that some white particles always exist regardless of the solution progress (Figs.3(a)-(f)). EDS analysis indicates that these particles are AlxMny phases. Previous investigation indicates that AlxMny phases are very stable during the solution treatment and cannot dissolve[17].
As discussed above, the small-sized Al-rich �� phase particles dispersedly distribute in the Mg-rich ��-Mg phase including secondary �� phase and eutectic �� phase. So the dissolution of �� phase towards the ��-Mg phase is very quick during the early stage of solution treatment (Fig.3(a)). As the Al content in the ��-Mg phase increases, the content gradient between the �� phase and ��-Mg phase decreases, which makes an obvious decrease in dissolution rate after 2 min. All of the �� phases do not disappear until the treatment is conducted for 1 h (Fig.4). The results from line scanning analysis show that the difference of Al content between the primary ��phase and secondarily solidified structures is still large after being treated for 1 h (Fig.6). This means that the homogenization is far behind the dissolution of �� phase and the homogenization with long-distance diffusion is the main phenomenon after 1 h.

Fig.3 SEM images of TDC AM60B alloys heated at 395 ��C for 2 min (a), 6 min (b), 10 min (c), 30 min (d), 1 h (e) and 60 h (f)
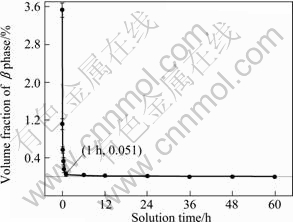
Fig.4 Variation of volume fraction of �� phase with solution time at 395 ��C
To illustrate the homogenization process during solutionizing, the Al contents of the two structures, primary �� particles and the secondly solidified structure, were measured. As shown in Fig.7, the Al content of the secondly solidified structure decreases rapidly while that of the primary �� particle increases quickly within 1 h of treatment, and then both of them change slowly. It can be found that this change tendency is consistent to that of the volume fraction of �� phase discussed above. Because all of the Al-rich �� phase structures are in the secondly solidified structures, there is a large Al content gradient between the primary particles and the secondly solidified structures. So Al atoms intensively diffuse from the later structure to the former structure, resulting in the rapid increase of Al content of the former structure and quick decrease of the latter structure, namely, the Al content gradient is decreased. Thus, the driving force of Al atom diffusion is decreased and the changes of these two Al contents become slow after 1 h. Even after being solutionized for 60 h, a large content difference still exists between these two structures (Fig.7).
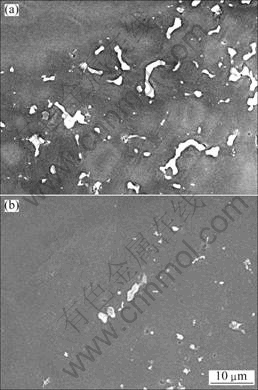
Fig.5 SEM images of �� phase treated at 395 ��C for 6 min (a) and 10 min (b)
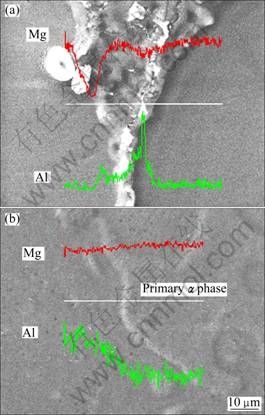
Fig.6 Line scanning analysis maps across primary particles and secondly solidification structures of alloys treated at 395 ��C for 0 h (a) and 1 h (b)
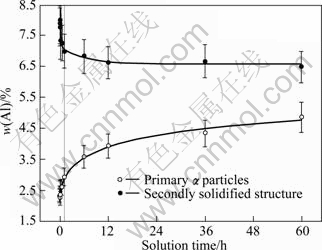
Fig.7 Variation of Al content in primary particles and secondly solidified structures with solution time
With the dissolution of �� phase, rapid coarsening occurs for the primary particles and the secondary ��-Mg grains. Figures 8 and 9 show that both the primary particles and secondary ��-Mg grains rapidly coarsen during the initial stage of 1 h, and then the coarsening rates of these two structures all obviously decrease. To differentiate from the rapid coarsening prior to 1 h, the slow coarsening after 1 h is named normal growth.
Summarizing the above discussion, it can be found that the change tendencies of the coarsening rates are completely consistent to those of the volume fraction of �� phase and the Al contents. All of the changes are divided into two stages by a critical value of 1 h. So the microstructural evolution of the TDC AM60B during solution treatment can be divided into two stages. The first stage is the quick dissolution of �� phase, rapid homogenization and rapid coarsening during the initial period of 1 h, and the second stage is the normal growth of the primary particles and the secondary ��-Mg grains which starts from 1 h.
3.3 Kinetic characteristics of solution treatment
Both the primary particles and secondary grains grow rapidly in the first stage (within 1 h), but their growing modes are quite different. For the secondary grains, the growth is mainly controlled by the dissolution of �� phase while that of the primary particles depends on swallowing the small secondary grains around them. The dissolution causes two influences. The first is that it results in the rapid growth of secondary grains; the second is that it produces obvious difference in Al content between these two types of particles. It can be expected that the second influence is helpful for primary particles to swallow the secondary grains due to the diffusion of Al atoms. After being solutionized for1 h (in the second stage), either the primary particles or the secondary grains grow through the mergence of neighboring grains because all of the �� phases have almost completely dissolved. As shown in Fig.10, a primary particle has swallowed a small secondary grain around it in region A and a relatively large-sized secondary grain has also swallowed a small-sized secondary grain in region B.

Fig.8 Optical images showing size change of primary ��-Mg particles and secondary ��-Mg grains solutionized at 395 ��C for different durations: (a) 2 min; (b) 6 min; (c) 10 min; (d) 30 min; (e) 1 h; (f) 60 h

Fig.9 Variations of primary particle size and secondary ��-Mg grain size with solution time

Fig.10 Microstructure of AM60B alloy solutionized at 395 ��C for 6 h
To compare the growth speeds of these two kinds of grains, growth rates were calculated by the following formula:
![]() (1)
(1)
where �� is the growth rate, Dn and Dn-1 are the mean grain sizes at time tn and tn-1, respectively. The results shown in Fig.11 clearly indicate that both the growth rates of both the primary particles and the secondary grains are considerably high during the first stage (within 1 h), but the rate of the secondary grains is higher than that of the primary �� particles (Fig.11(a)). This can be attributed to their different growth modes. As discussed above, the dissolution rate of �� phase is more rapid than that of Al atoms, so the growth rate of the secondary grains depending on the dissolution of �� phase is higher than that of the primary particles controlled by the interface diffusion of Al atoms. About 10 min later, both the rates, especially that of the secondary grains, sharply decrease due to the decrease of �� phase. Even so, the growth rates during the first stage are still higher than those in the second stage (comparing Fig.11(a) with 11(b)). After 1 h, both the growth rates keep at a low level below 0.2 h-1, but the growth rate of the secondary grains is still higher than that of the primary particles. The difference between the two rates decreases as the solution time increases. Three reasons can be proposed. Firstly, all of the �� phases have completely dissolved and the growth mode of the secondary grains turns to the mergence, and thus their growth rate sharply decreases. Secondly, the Al content gradient between these two types of grains slowly decreases, so the growth rate of the primary particles slowly decreases. Thirdly, the interface energy of the secondary grains occupied regions should be always higher than that of the primary particles occupied regions due to grain size difference, so the growth rate of the secondary grains is higher than that of the primary particles driven by decreasing the interface energy. But due to the decrease of the grain size difference between these two kinds of grains with the solution time, the difference of the growth rates also decreases.
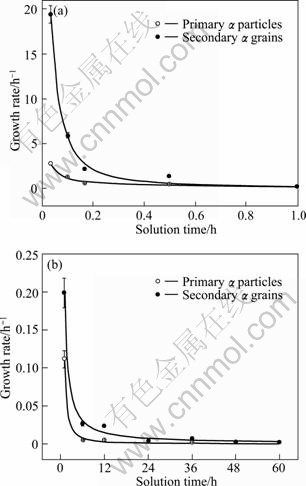
Fig.11 Growth rate variations of primary particles and secondary grains with solution time: (a) Within 1 h; (b) After 1 h
To clarify the growth models of these two kinds of grains, the grain size data are fitted to estimate which function is obeyed (Fig.12). The results reveal that the growth model of the secondary grains obeys the traditional growth equation well in the first stage:
![]() (2)
(2)
where ![]() is the grain size; k is the constant of growth; D is the diffusion coefficient, which can be obtained by
is the grain size; k is the constant of growth; D is the diffusion coefficient, which can be obtained by
![]() (3)
(3)
where D0 is the diffusion constant related to the temperature T and solute concentration; Q is the activation energy, 160.5 kJ/mol[17].
However, the growth model of the primary particles is quite different and follows the following equation:
![]() (4)
(4)
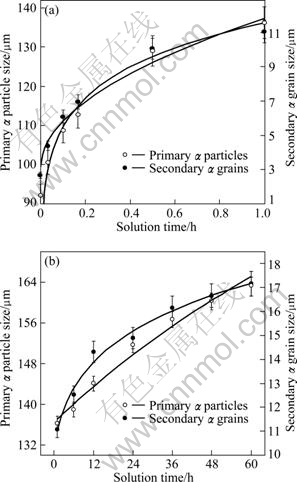
Fig.12 Variations of primary particle size and secondary ��-Mg grain size with solution time: (a) Within 1 h; (b) After 1 h
The fitting results show that the value of n is equal to 0.1, less than 2, indicating that the growth does not result from phase transformation controlled by diffusion. Therefore, two different growth models can be proposed, the growth of primary particles during the first stage is governed by the mergence while that of the secondary grains is controlled by diffusion transformation (the dissolution of �� phase produces Al supersaturated �� solid solution). And in the second stage, the growth models of these two types of grains all obey a same relation:
![]() (5)
(5)
where ![]() and
and ![]() are the initial and final grain sizes, respectively; t is the solution time; and k and n are constants related to diffusivity, interfacial energy and solute concentration. The present results indicate that the n values for the primary particles and secondary grains are 3.66 and 3.98, respectively, larger than 3, but lower than 4. According to the mechanistic models for grain coarsening, if the coarsening is controlled by diffusion via dislocation cores, diffusion along grain boundaries, diffusion through the lattice or diffusion across a particle matrix interface, n can be 5, 4, 3, or 2, respectively[12]. So it can be concluded that the coarsening of the two kinds of grains is a mixture model including Al diffusion along grain boundaries and through crystal lattice, and these two diffusion mechanisms lead to the interface migration and thus the grain coarsening.
are the initial and final grain sizes, respectively; t is the solution time; and k and n are constants related to diffusivity, interfacial energy and solute concentration. The present results indicate that the n values for the primary particles and secondary grains are 3.66 and 3.98, respectively, larger than 3, but lower than 4. According to the mechanistic models for grain coarsening, if the coarsening is controlled by diffusion via dislocation cores, diffusion along grain boundaries, diffusion through the lattice or diffusion across a particle matrix interface, n can be 5, 4, 3, or 2, respectively[12]. So it can be concluded that the coarsening of the two kinds of grains is a mixture model including Al diffusion along grain boundaries and through crystal lattice, and these two diffusion mechanisms lead to the interface migration and thus the grain coarsening.
4 Conclusions
1) The TDC AM60B alloy has homogeneous microstructure with nearly globular primary ��-Mg particles uniformly distributed in the secondly solidified matrix. The secondly solidified structure is composed of fine secondary ��-Mg dendrites and interdendritic devoiced ������ eutectic.
2) The microstructural evolution during solution treatment includes two stages. One is the rapid dissolution of �� phase, homogenization and coarsening; the other is the normal growth of primary ��-Mg particles and secondary ��-Mg grains. The first stage maintains about 1 h, and the second stage lasts very long time when treated at 395 ��C.
3) During the first stage, the rapid growth of the secondary grains is attributed to the quick dissolution of �� phase while that of the primary particles results from their combination with surrounding secondary grains. In the second stage, the coarsening of these two kinds of grains belongs to the mergence of neighboring grains and this regime exhibits a low growth rate.
4) The growth models of the two types of grains are different in the first stage. The growth of the primary particles obeys power function while that of the secondary grains follows the traditional growth equation in the first stage. But in the second stage, they all obey a same model controlled by diffusion along grain boundaries and through crystal lattice.
References
[1] LIU G, WANG Y, FAN Z. A physical approach to the direct recycling of Mg-alloy scrap by the rheo-diecasting process [J]. Mater Sci Eng A, 2008, 472(1-2): 251-257.
[2] FAN Z. Development of the rheo-diecasting process for magnesium alloys [J]. Mater Sci Eng A, 2005, 413: 72-78.
[3] FAN Z, LIU G, WANG Y. Microstructure and mechanical properties of rheo-diecast AZ91D magnesium alloy [J]. J Mater Sci, 2006, 41(12): 3631-3644.
[4] MOLLER H, GOVENDER G, STUMPF W. T6 heat treatment of semi-solid metal processed alloy A356 [J]. Open Materials Science Journal, 2008(2): 6-10.
[5] ZENG R, KE W, HAN E. Influence of load frequency and ageing heat treatment on fatigue crack propagation rate of as-extruded AZ61 alloy [J]. Int J Fatigue, 2009, 31(3): 463-467.
[6] HUANG J, YU H, LI Y, CUI H, HE J, ZHANG J. Precipitation behaviors of spray formed AZ91 magnesium alloy during heat treatment and their strengthening effect [J]. Mater Des, 2009, 30(3): 440-444.
[7] WU H, HSU C, WON J, SUN P, WANG J, LEE S, CHIU C, TORNG S. Effect of heat treatment on the microstructure and mechanical properties of the consolidated Mg alloy AZ91D machined chips [J]. J Mater Process Technol, 2009, 209(8): 4194-4200.
[8] ZHANG Z, YU H, WANG S, WANG H, MIN G. Microstructure and tensile properties of ZK60 alloy fabricated by simplified rapid solidification powder metallurgy (S-RS P/M) process [J]. J Mater Sci Technol, 2010, 26(2): 151-155.
[9] WAN D, WANG J, WANG G, LIN L, FENG Z, YANG G. Precipitation and responding damping behavior of heat-treated AZ31 magnesium alloy [J]. Acta Metall Sin, 2009, 22(1): 1-6.
[10] MARYA M, HECTOR L, VERMA R, TONG W. Microstructural effects of AZ31 magnesium alloy on its tensile deformation and failure behaviors [J]. Mater Sci Eng A, 2006, 418(1-2): 341-356.
[11] YANG L, WEI Y, HOU L. Microstructure evolution of thixomolding AZ91D magnesium alloy during heat treatment [J]. J Mater Sci, 2010, 45(13): 3626-3634.
[12] WANG Y, LIU G, FAN Z. Microstructural evolution of rheo-diecast AZ91D magnesium alloy during heat treatment [J]. Acta Mater, 2006, 54(3): 689-699.
[13] DULY D, SIMON J, BRECHET Y. On the competition between continuous and discontinuous precipitations in binary Mg-Al alloys [J]. Acta Metall Mater, 1995, 43(1): 101-106.
[14] STANJU T. Influence of the working technology on Al-alloys in semi-solid state [J]. Mater Technol, 2009, 43(4): 213-217.
[15] SEO P, KANG C, LEE S. A study on reheating characteristics for thixo die casting process with electromagnetic stirring and extruded aluminum alloys and their mechanical properties [J]. Int J Adv Manuf Technol, 2009, 43(5): 482-499.
[16] HITCHCOCK M, WANG Y, FAN Z. Secondary solidification behaviour of the Al-Si-Mg alloy prepared by the rheo-diecasting process [J]. Acta Mater, 2007, 55(5): 1589-1598.
[17] MRVAR P, TER ELJ M, KUGLER G, FAZARINC M, MEDVED J. Characterization of solidification and hot workability of MgAl6Mn foundry alloy [J]. RMZ M&G, 2006, 53(2): 175-192.
�ƺ���, �����, �� ӱ, �� Զ
����������ѧ ����ʡ��ɫ�����²���ʡ�����������ص�ʵ���ң����� 730050
ժ Ҫ���о��������AM60Bþ�Ͻ��ڹ��ܴ��������е���֯�ݱ估���ȴ�������ѧ������������������AM60Bþ�Ͻ��ڹ��ܴ�������������֯�ݱ�ɷ�Ϊ�����Σ���һ��Ϊ������ (Mg17Al12) ���Ѹ���ܽ⡢���ʿ��پ��Ȼ��������ֻ����ڶ���Ϊ�������������ξ�������������Ρ��ڵ�һ�Σ�����ϸС�Ħ����������С�Ķ��ξ�������ɸ��ܽ���̽϶̣�������AlԪ�صľ��Ȼ�����Ҫ��Խϳ���ʱ�䣬ͬʱ����֯��ò�������ߴ��кܴ�仯����395 ��C����ʱ����һ�γ���Լ1 h�����һ����ȵڶ�����Ҫ�ϳ���ʱ�䣬�ڱ���ʵ��������Լ60 h����һ�εĿ��پ��Ȼ�ʹAl��Ũ���ݶ��ڵڶ��δ���ȼ�С������ڸý���֯�仯�������ԡ��ڵ�һ�Σ�����������ij���ģʽ�����ݺ�����ϵ�������ξ�������Ӵ�ͳ�ij��̣�Ȼ�����ڵڶ������ߵij���ʽ��ͬ��
�ؼ���:��AM60Bþ�Ͻ�; �������; ���ܴ���; ��֯; ����ѧ
(Edited by LI Xiang-qun)
Foundation item: Project ((G2007CB613706)) supported by the National Basic Research Program of China; Project supported by the Development Program for Outstanding Young Teachers in Lanzhou University of Technology, China; Project (SKL03004) supported by the Opening Foundation of State Key Laboratory of Advanced Nonferrous Materials, China
Corresponding author: CHEN Ti-jun; Tel: +86-931-2976573; E-mail: chentj@lut.cn, chentj1971@126.com
DOI: 10.1016/S1003-6326(11)60775-9

