Mg-Zn-Al��Ԫϵ��þ��335 ��C���½���
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2014���11��
�������ߣ�����ƽ ������ ������ �� �� ����� �� �� �ظ���
����ҳ�룺3405 - 3412
�ؼ��ʣ�Mg-Zn-Alϵ�����½��棻�ӽ����仯������ܶ�
Key words��Mg-Zn-Al ternary system; isothermal section; �� intermetallic compound; solubility
ժ Ҫ������ƽ��Ͻ�����X�������䡢ɨ��羵����������ϵͳ���о���Mg-Zn-Al��Ԫϵ��þ��335 ��C��ƽ������ɼ���ɷ֡���ʵ����֤ʵ����-Mg�����岢����Mg32(Al,Zn)49 (��)��Ԫ�����仯�����q����ƽ�⣬������һ����Ԫ������Al5Mg11Zn4 (��)��ƽ�⡣����˦�����335 ��C�������ɷַ�Χ������52.5%~56.4% Mg��13.6%~24.0% Al��19.6%~33.9% Zn(Ħ������)��Al��MgZn���еĹ��ܶ�Զ������Mg7Zn3���еĹ��ܶȣ������ֵ�ɴ�8.6%Al(Ħ������)��Al��Zn����ͬʱ�����ڦ�-Mg�������С�
Abstract: The phase equilibria and compositions at the Mg-rich corner of the Mg-Zn-Al ternary system at 335 ��C were systemically investigated through the equilibrated alloy method by using X-ray diffraction (XRD) and scanning electron microscopy (SEM) assisted with energy dispersive spectroscopy of X-ray (EDS). It is experimentally testified that the ��-Mg solid solution is not in equilibrium with the Mg32(Al, Zn)49 (��) ternary intermetallic compound or q quasicrystalline phase, but only in equilibrium with one ternary intermetallic compound Al5Mg11Zn4 (��). The whole composition range of the �� phase was also obtained at 335 ��C, i.e., 52.5%-56.4% Mg, 13.6%-24.0% Al, 19.6%-33.9% Zn (mole fraction). The solubility of Al in the MgZn phase is remarkably more than that in the Mg7Zn3 phase, and the maximum is about 8.6% Al. Aluminum and zinc are simultaneously soluble in the ��-Mg solid solution.
Trans. Nonferrous Met. Soc. China 24(2014) 3405-3412
Yu-ping REN, Shi-neng SUN, Li-qing WANG, Yun GUO, Hong-xiao LI, Song LI, Gao-wu QIN
Key Laboratory for Anisotropy and Texture of Materials, Ministry of Education, Northeastern University, Shenyang 110819, China
Received 24 November 2013; accepted 14 July 2014
Abstract: The phase equilibria and compositions at the Mg-rich corner of the Mg-Zn-Al ternary system at 335 ��C were systemically investigated through the equilibrated alloy method by using X-ray diffraction (XRD) and scanning electron microscopy (SEM) assisted with energy dispersive spectroscopy of X-ray (EDS). It is experimentally testified that the ��-Mg solid solution is not in equilibrium with the Mg32(Al, Zn)49 (��) ternary intermetallic compound or q quasicrystalline phase, but only in equilibrium with one ternary intermetallic compound Al5Mg11Zn4 (��). The whole composition range of the �� phase was also obtained at 335 ��C, i.e., 52.5%-56.4% Mg, 13.6%-24.0% Al, 19.6%-33.9% Zn (mole fraction). The solubility of Al in the MgZn phase is remarkably more than that in the Mg7Zn3 phase, and the maximum is about 8.6% Al. Aluminum and zinc are simultaneously soluble in the ��-Mg solid solution.
Key words: Mg-Zn-Al ternary system; isothermal section; �� intermetallic compound; solubility
1 Introduction
The Mg-Zn-Al ternary phase diagram is of theoretical and technical importance for the Al-Zn-Mg-Cu series Al alloys (7XXX) and AZ or AM series Mg alloys. The phase equilibria of Al-rich corner have been mainly focused in the earlier studies, and successfully applied in the optimization of composition and process of 7XXX series Al alloys [1]. However, there are only three isothermal sections at 300-335 ��C at Mg-rich corner of Mg-Zn-Al ternary system [2-6]. It was believed that there exist two ternary compounds of Mg32(Al,Zn)49 (��) and Al5Mg11Zn4 (��) in equilibrium with the ��-Mg solid solution [2-5]. However, it is worthwhile to note that the �� compound is completely surrounded by the MgZn or �� phase in the microstructures of the treated alloys consisting of ��-Mg+ MgZn+�� or ��-Mg+��+�� phases, according to the results obtained at 300 or 320 ��C by REN et al [3,4]. Because there is no contiguity between the �� compound and ��-Mg phase in these treated alloys, it is doubtful whether the relationship is in equilibrium. In addition, the ��-Mg/�� phase equilibrium relationship at 335 ��C is determined by LIANG et al [5], only using one alloy treated for 240 h. It is thought that the annealing time is too short to achieve the phase equilibrium relationship in the treated alloy because the �� compound is surrounded by the MgZn or �� phase by the peritectic reaction [3,4]. On the other hand, it is believed by PETROV [6] that there is quasicrystal phase (q), not �� intermetallic compound in equilibrium with the ��-Mg solid solution at 335 ��C except for these compounds mentioned above, according to the results of TAKEUCHI and MIZUTANI [7]. The quasicrystal phase is stable in the Al15Mg44Zn41 alloy held at 360 ��C for 5 h or heated from room temperature to 380 ��C by means of differential scanning calorimeter at the rate of 10 ��C/min. Unfortunately, the kinetics of phase transformation is neglected because the annealing time is so much short or the heating rate is relatively fast. Therefore, it is necessary to take longer annealing time at certain temperature to certify the stability of the quasicrystal phase in the Mg-Zn-Al ternary system.
Recently, the ZA series Mg alloys have been increasingly focused due to better heat resistant property than AZ series Mg alloys [8-14]. Moreover, it is believed that the strengthening phases mainly include ��, quasi-crystalline, �� and MgZn compounds [11,13]. However, it is worth noting that the amount and size of the �� phase gradually decrease with the extent of the annealing time in the ZA73 Mg alloy treated at 325 ��C [12]. It thus implies that the �� intermetallic compound is possibly not in equilibrium with the ��-Mg solid solution at this temperature in the Mg-Zn-Al ternary system. On the other hand, the quasicrystal phase is metastable and transforms into the �� intermetallic compound when annealed at 220 ��C [14]. Therefore, it is necessary to systematically determine the phase equilibria at the Mg-rich corner of Mg-Zn-Al ternary system, particularly the phase equilibrium relationship with the ��-Mg solid solution. Considering the kinetics of phase transformation and the liquid phase formed at 340 ��C in the Mg-Zn binary system [15,16], the isothermal section at the Mg-rich corner of the Mg-Zn-Al ternary system at 335 ��C was detailly investigated by means of the equilibrated alloy method, in order to better understand the relationship between the composition, microstructure, process and mechanical property in the ZA series Mg alloys.
2 Experimental
Twenty-two desired alloys with nominal compositions of Mg80Al20, Mg70Al25Zn5, Mg70Al20Zn10, Mg60Al25Zn15, Mg70Al15Zn15, Mg70Al12Zn18, Mg60Al13- Zn27, Mg60Al8Zn32, Mg60Al10Zn30, Mg60Al1Zn39, Mg60Al2Zn38, Mg60Al4Zn36, Mg60Al6Zn34, Mg70Al1Zn29, Mg70Al2Zn28, Mg80Zn20, Mg70Zn30, Mg60Zn40, Mg50Al25Zn25, Mg50Al20Zn30, Mg50Al33Zn17, Mg55Al30- Zn15 were prepared by using high purity Mg (99.99%, mass fraction), Al(99.99%), and Zn (99.999%). These alloys were melted in a high purity graphite crucible in the argon atmosphere by using an induction furnace, then the samples were cut from the ingots and held at (335��2) ��C for 1200 h or 3648 h, and finally quenched by water.
The phase constituents of the treated alloys were determined by X-ray diffraction (XRD) on a Philips PW3040/60 diffractometer with Cu K�� irradiation, with a voltage of 40 kV and a current of 40 mA as well as a scanning rate of 3 (��)/min. The microstructures and compositions of the treated Mg-Zn-Al alloys were analyzed by scanning electron microscopy with the assistance of energy dispersive spectroscopy of X-ray (SEM-EDS) on a HITACHI S3400N under an accelerating voltage of 20 kV.

Fig. 1 Back scattering electron image (a) and XRD pattern (b) of Mg80Zn20 alloy held at 335 ��C for 1200 h

Fig. 2 Back scattering electron image (a) and XRD pattern (b) of Mg60Zn40 alloy held at 335 ��C for 1200 h
3 Results and discussion
Because the Mg7Zn3 phase is stable only at the temperatures of 325-340 ��C [15,16], three Mg-Zn binary alloys were prepared and treated at 335 ��C. Figures 1 and 2 show the microstructures and XRD patterns of the Mg80Zn20 and Mg60Zn40 alloys treated at 335 ��C for 1200 h, respectively. By the SEM-EDS and XRD analysis, the treated Mg80Zn20 alloy is composed of the ��-Mg solid solution and Mg7Zn3 compound (Fig. 1). Similarly, the Mg60Zn40 alloy consists of the MgZn and Mg7Zn3 compounds (Fig. 2). The corresponding equilibrium phase constituents and compositions of the two treated alloys are listed in Table 1. The composition range of the Mg7Zn3 phase is 30.7%-32.5% Zn (mole fraction) at 335 ��C. Therefore, it is experimentally testified that there does exist the Mg7Zn3 phase rather than the Mg51Zn20 phase in the Mg�CZn binary system [15,16]. At the same time, it also implies that the annealing temperature should remain at 335 ��C.
Table 1 Equilibrium phase constituents and compositions in Mg-Zn-Al system at 335 ��C
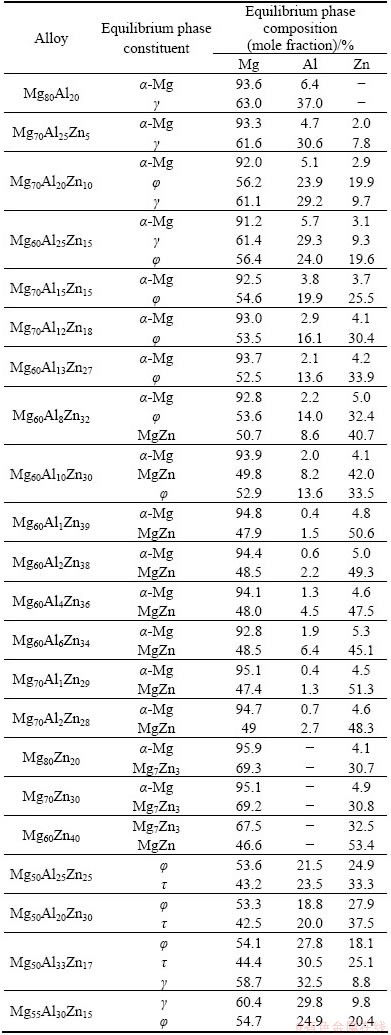
As one of the most important strengthening phases, the �� intermetallic compound or quasicrystalline phase (q) in equilibrium with the ��-Mg solid solution is of technical and theoretical importance for the ZA series Mg alloys. If the �� or q phase is in equilibrium with the ��-Mg solid solution, there exist two three-phase regions consisting of ��-Mg+��+��(q) and ��-Mg+��(q)+ MgZn at the Mg-Zn side [2-6]. Figure 3 shows the backscatter electron (BSE) images and X-ray diffraction patterns of the Mg60Al10Zn30 alloy treated at 335 ��C for 1200 and 3648 h. By SEM-EDS analysis, the dark, gray and light phases correspond to the ��-Mg solid solution, �� and MgZn compounds (Figs. 3(a) and (b)), respectively, which are also confirmed by XRD analysis results (Fig. 3(c)). The compositions of the corresponding equilibrium phases are listed in Table 1. It shows that there exists a three-phase region composed of ��-Mg, �� and MgZn phases at the Mg-rich corner of the Mg-Zn-Al ternary system. Therefore, it suggests that the �� or q phase is not practically in equilibrium with the ��-Mg solid solution, i.e., metastable in the as-cast ZA series Mg alloys.
In order to further confirm the result mentioned above, both Mg60Al13Zn27 and Mg60Al8Zn32 alloys were also held at 335 ��C for 1200 and 3648 h, respectively. Their microstructures and X-ray diffraction patterns are illustrated in Figs. 4 and 5, respectively. The treated Mg60Al13Zn27 alloy consists of ��, �� compounds and ��-Mg solid solution according to the SEM-EDS analysis result. However, the �� intermetallic compound is not identified by XRD analysis in the Mg60Al13Zn27 alloy treated for 1200 or 3648 h, as shown in Fig. 4(e), since the volume fraction of the �� phase is lower than 5% (Figs. 4(a) and (c)). The �� phase is completely surrounded by ��, and does not contact with ��-Mg solid solution (Figs. 4(a)- (d)), which is similar to the results obtained at 300 or 320 ��C [3,4]. Moreover, the �� phase almost disappears and no new phase forms with the increase of the annealing time from 1200 h to 3648 h, as shown in Figs. 4(a)-(d). It means that the �� phase may be completely eliminated when the annealing time is much more than 3648 h, and then, it is also believed that the �� phase is not in equilibrium when treated at 300 or 320 ��C for 720 h [3,4]. Therefore, it is experimentally certified that the �� compound should be metastable, and the equilibrium phase constituents are the ��-Mg solid solution and �� compound in the treated Mg60Al13Zn27 alloy. This also indirectly supports the result obtained in the treated Mg60Al10Zn30 alloy. Finally, the equilibrium compositions of the ��-Mg and �� phases are listed in Table 1.
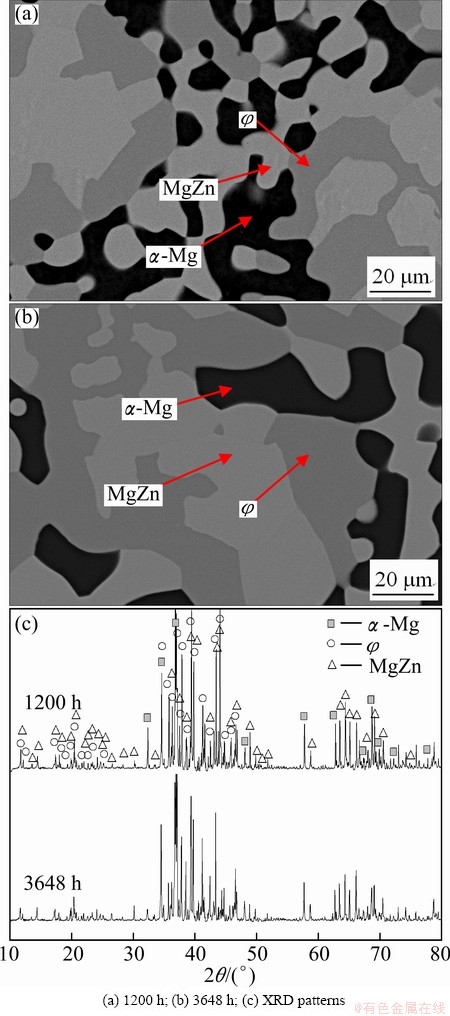
Fig. 3 Back scattering electron images and XRD patterns of Mg60Al10Zn30 alloy held at 335 ��C for different time
The Mg60Al8Zn32 alloy consists of the dark and light phases when held for 1200 h (Fig. 5(a)). A very small amount of new gray phase forms with the extension of the annealing time to 3648 h (Fig. 5(b)), so that it may not be detected by XRD analysis, as shown in Fig. 5(c). By SEM-EDS analysis, the dark, light and new gray phases are the ��-Mg solid solution, MgZn and �� intermetallic compounds, respectively. It implies that there does exist a three-phase region consisting of ��-Mg, MgZn and �� phases. Their equilibrium compositions are also listed in Table 1. The result directly confirms that the �� or q phase should be metastable in the ZA series Mg alloys, which is consistent with the result obtained in the treated Mg60Al10Zn30 alloy. Therefore, it can be concluded that �� or q phase is indeed not in equilibrium with the ��-Mg solid solution at 335 ��C. Thereby, it is also necessary to recognize the role of the �� or q phase in improving the heat resistant property of the as-cast ZA series Mg alloys.

Fig. 4 Back scattering electron images and XRD patterns of Mg60Al13Zn27 alloy held at 335 ��C for different time
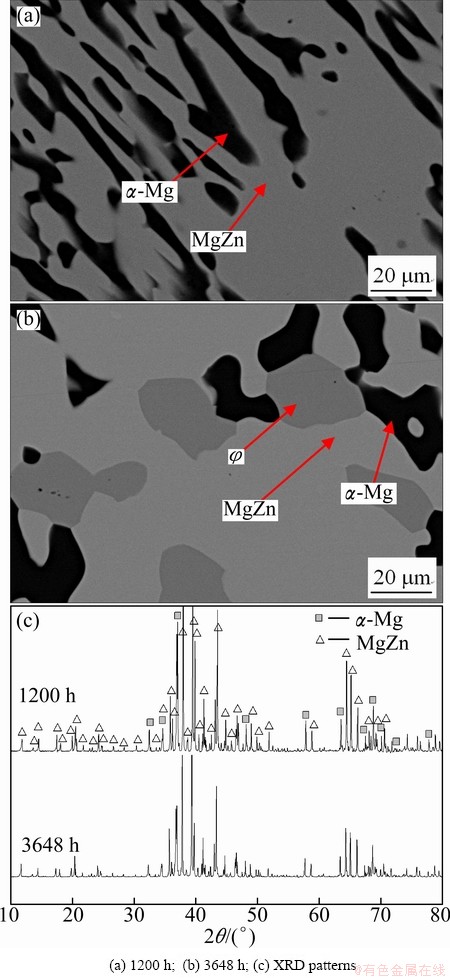
Fig. 5 Back scattering electron images and XRD patterns of Mg60Al8Zn32 alloy held at 335 ��C for different time
The rest of the Mg-Zn-Al alloys treated at 335 ��C for 1200 h are also analyzed by XRD and SEM-EDS. There are two kinds of alloys consisting of two or three phases. The BSE image and XRD pattern of the treated Mg60Al1Zn39 alloy are shown in Fig. 6. The equilibrium phase constituents are composed of ��-Mg and MgZn phases. At the same time, there also exist ��-Mg and MgZn phases in the treated Mg60Al2Zn38, Mg60Al4Zn36, Mg60Al6Zn34, Mg70Al1Zn29 and Mg70Al2Zn28 alloys. It can be also determined that the equilibrium phase constituents are ��-Mg+�� phases in the Mg70Al12Zn18 and Mg70Al15Zn15 alloys, ��-Mg+�� phases in the Mg70Al25Zn5 alloy, ��+�� phases in the Mg50Al20Zn30 and Mg50Al25Zn25 alloys and ��+�� phases in the Mg55Al30Zn15 alloy, respectively. Figure 7 shows typical microstructure and pattern of the treated Mg70Al20Zn10 alloy, which is located in three-phase region composed of ��-Mg, �� and �� phases. Moreover, the equilibrium phase constituents of the treated Mg60Al25Zn15 alloy are also composed of the ��-Mg, �� and �� phases. Similarly, it can be obtained that the equilibrium phase constituents are ��+��+�� in the treated Mg50Al33Zn17 alloys. The equilibrium phase compositions in these treated alloys mentioned above were all determined by SEM-EDS, as listed in Table 1.
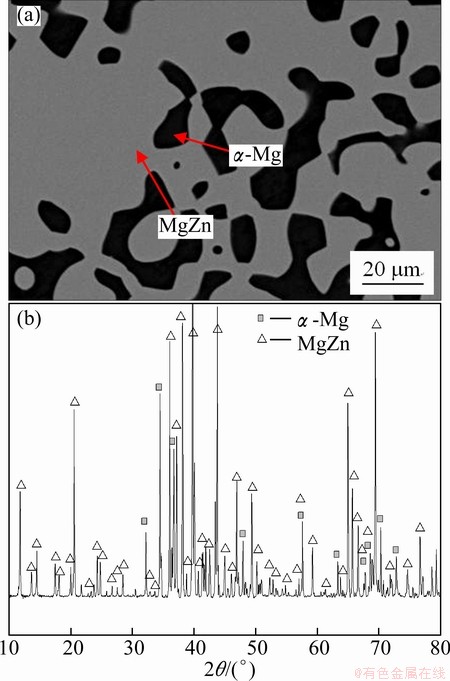
Fig. 6 Back scattering electron image (a) and XRD pattern (b) of Mg60Al1Zn39 alloy held at 335 ��C for 1200 h
Based on the equilibrium phase compositions listed in Table 1, the isothermal section of Mg-rich corner at 335 ��C is consequently constructed in the Mg-Zn-Al system, as shown in Fig. 8. There exist four intermetallic compounds such as ��, ��, MgZn and Mg7Zn3 in equilibrium with the ��-Mg solid solution, while there is not equilibrium relationship between the �� or q phase and ��-Mg phase, which is completely different from the presently accepted results [2-6]. Moreover, the composition range of �� ternary compound is also determined, i.e., 52.5%-56.4% Mg, 13.6%-24.0% Al, and 19.6%-33.9% Zn (mole fraction). It shows that the �� phase is not the linear intermetallic compound, different from the calculated result obtained by LIANG et al [5] or PETROV et al [6]. It is suggested by the calculated result that the Mg contents in the �� compound remain constant, and the Zn or Al compositions change to some extent. It was probably convenient to construct the thermodynamic model of the �� compound and calculate the relative phase equilibria. The solubility of Al in the MgZn phase is remarkably more than that of Mg7Zn3 phase, i.e., the maximum of the former reaches 8.6% Al (mole fraction) at 335 ��C, while the latter is almost zero. This is probably the reason why there is completely the MgZn phase, not the Mg7Zn3 phase in equilibrium with the ��-Mg solid solution in the isothermal section at 335 ��C calculated by LIANG et al or PETROV et al [6]. Moreover, the solubility of Al in the �� phase is around 37% (mole fraction), equilibrated with ��-Mg phase in the Mg-Al binary system, which is obviously different from the earlier result [17], and consistent with the result obtained recently by our group by means of the diffusion couple technique [18]. At the same time, the solubility of Zn in the �� compound is also very large, and the maximum is about 9.7% Zn (mole fraction).

Fig. 7 Back scattering electron image (a) and XRD patter (b) of Mg70Al20Zn10 alloy held at 335 ��C for 1200 h
Al and Zn are simultaneously soluble in the ��-Mg matrix, and the solubility of Zn increases due to the Al addition. It inevitably affects the solid solution strength of the Zn solute in the ��-Mg matrix [19]. On the other hand, the Mg-Zn based Mg alloy is one of the important precipitation strengthening systems, their precipitation behaviors have been extensively studied during the aging, such as Mg-Zn-Cu [20], Mg-Zn-Sn [21], etc. However, the effect of Al on the precipitation behavior of the Mg-Zn based alloys is not still paid attention, such as the sequence, composition and structure of the metastable phases, etc. Most studies were focused on the strengthening role of the second phases such as ��, MgZn, quasicrystalline and �� phases in the ZA series Mg alloys [8-13]. Obviously, the thermal stability of the �� intermetallic compound or q quasicrystalline phase is not better than that of the �� phase, according to our result. Therefore, the strengthening role of Al and Zn in the ��-Mg matrix should be largely noted, in particular for the precipitation strengthening during the aging, in order to better understand the fact that the creep-resistant property of ZA series Mg alloys containing the �� or q phase is better than that of AZ series Mg alloy.
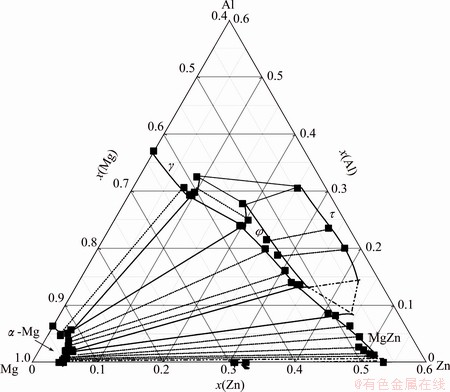
Fig. 8 Isothermal section of Mg-rich corner in Mg-Zn-Al ternary system at 335 ��C
On the other hand, it is necessary to re-assess the thermodynamical data of Mg-Zn-Al ternary system according to the results obtained in this work, including the thermodynamical models, stability and composition ranges of the non-stoichiometric compounds such as �� and ��, and binary compounds such as ��, MgZn and Mg7Zn3, in particular for the thermodynamical description of the ��-Mg solid solution. Finally, it can thermodynamically provide the accurate and reliable data for the composition design, optimization of heat treatment and process of the Mg-Zn-Al based Mg alloys, particularly for the precipitation behavior of the ��-Mg matrix during the aging. At the same time, the role of �� intermetallic compound or q quasicrystalline phase should be recognized in the as-cast ZA series Mg alloys, particularly for the dissolution behavior during the homogeneous or solid solution treatment and the precipitation behavior during the aging at lower temperatures. Finally, it can be revealed why the heat-resistant property of the ZA series Mg alloys is improved, compared to the AZ series Mg alloys, so that the fundamental information can be provided for designing the new type of the creep-resistant Mg-based alloys.
4 Conclusions
1) The isothermal section of Mg-rich corner in Mg-Zn-Al ternary system at 335 ��C is constructed. There are three-phase regions composed of ��-Mg+��+��, ��-Mg+��+MgZn, ��-Mg+MgZn +Mg7Zn3 at the Mg-rich corner.
2) It is experimentally testified that there indeed does not exist the equilibrium relationship between the �� intermetallic compound or q quasicrystalline phase and ��-Mg solid solution, completely different from the currently accepted result. Therefore, it is necessary to clarify the mechanism of heat resistance in the ZA series Mg alloys containing the �� or q phase.
3) Aluminum and zinc are simultaneously soluble in the ��-Mg solid solution, and the solubility of Zn in the ��-Mg matrix increases due to the Al addition. It suggests that it should be of practical importance to further investigate the effect of Al content on the precipitation behavior in the ZA series Mg alloys.
References
[1] SAUNDERS N, MIODOWNIK A P. CALPHAD (Calculation of phase diagrams): A comprehensive guide [M]. UK: Elsevier Science Ltd, 1998: 349-352.
[2] VILLARS P, PRINCE A, OKAMOTO H. Handbook of ternary alloy phase diagrams [M]. Geauga County OH: ASM International, 1995, CDROM.
[3] REN Y P, QIN G W, PEI W L, GUO Y, ZHAO H D, LI H X, JIANG M, HAO S M. The ��-Mg solvus and isothermal section of Mg-rich corner in the Mg-Zn-Al ternary system at 320 ��C [J]. J Alloys Compd, 2009, 481(1-2): 176-181.
[4] REN Y P, QIN G W, PEI W L, LI S, GUO Y, ZHAO H D. Phase equilibria of Mg-rich corner in the Mg-Zn-Al ternary system at 300 ��C [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(2): 241-245.
[5] LIANG P, TARFA T, ROBINSON J A, WAGNER S, OCHIN P., HARMELIN M G, SEIFERT H J, LUKAS H L, ALDINGER F. Experimental investigation and thermodynamic calculation of the Al-Mg-Zn system [J]. Thermochim Acta, 1998, 314(1-2): 87-110.
[6] PETROV D, WATSON A,  J, ROGL P, TEDENAC J C, BULANOVA M, TURKEVICH V. Ternary alloy systems�Cphase diagrams, crystallographic and thermodynamic data [M]. EFFENBERG G, ILYENKO S, eds. Berlin, Germany: Springer, 2006: 191-209.
J, ROGL P, TEDENAC J C, BULANOVA M, TURKEVICH V. Ternary alloy systems�Cphase diagrams, crystallographic and thermodynamic data [M]. EFFENBERG G, ILYENKO S, eds. Berlin, Germany: Springer, 2006: 191-209.
[7] TAKEUCHI T, MIZUTANI U. Electronic structure, electron transport properties, and relative stability of icosahedral quasicrystals and their 1/1 and 2/1 approximants in the Al-Mg-Zn alloy system [J]. Phys Rev B, 1995, 52(13): 9300-9309.
[8] WAN X F, NI H J, HUANG M Y, ZHANG H L, SUN J H. Microstructure, mechanical properties and creep resistance of Mg- (8%-12%)Zn-(2%-6%) Al alloys [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(4): 896-903.
[9] BALASUBRAMANI N, PILLAI U T S, PAI B C. Optimization of heat treatment parameters in ZA84 magnesium alloy [J]. J Alloys Comp, 2008, 457(1-2): 118-123.
[10] VOGEL M, KRAFT O, ARZT E. Creep behavior of magnesium die-cast alloy ZA85 [J]. Scripta Mater, 2003, 48(8): 985-990.
[11] ZHANG J, GUO Z X, PAN F S, LI Z S, LUO X D. Effect of composition on the microstructure and mechanical properties of Mg-Zn-Al alloys [J]. Mater Sci Eng A, 2007, 456(1-2): 43-51.
[12] ZHANG J, ZUO R L, CHEN Y X, PAN F S, LUO X D. Microstructure evolution during homogenization of a ��-type Mg-Zn-Al alloy [J]. J Alloys Comp, 2008, 448(1-2): 316-320.
[13] ZHANG Z, TREMBLAY R,  D, COUTURE A. Solidification microstructure of ZA102, ZA104 and ZA106 magnesium alloys and its effect on creep deformation [J]. Canadian Metall Quart, 2000, 39(4): 503-512.
D, COUTURE A. Solidification microstructure of ZA102, ZA104 and ZA106 magnesium alloys and its effect on creep deformation [J]. Canadian Metall Quart, 2000, 39(4): 503-512.
[14] VOGEL M, KRAFT O, DEHM G, ARZT E. Quasi-crystalline grain-boundary phase in the magnesium die-cast alloy ZA85 [J]. Scripta Mater, 2001, 45(5): 517-524.
[15] NAYEB-HASHEMI A A, CLARK J B. Phase diagrams of binary magnesium alloys [M]. Geauga County, OH: ASM International, 1988: 353-364.
[16] AGARWAL R, FRIES S G, LUKAS H L, PETZOW G, SOMMEN F, CHART T G, EFFENBERG G. Assessment of the Mg-Zn system [J]. Z Metallkd, 1992, 83(4): 216-223.
[17] MASSALSKI T B. Handbook of binary alloy phase diagrams [M]. Geauga County, OH: ASM International, 1990: 169-171.
[18] REN Y P, QIN G W, LI S, GUO Y, SHU X L, DONG L B, LIU H H, ZHANG B. Re-determination of ��/(��+��-Mg) phase boundary and experimental evidence of R intermetallic compound existing at lower temperatures in the Mg-Al binary system [J]. J Alloys Compd, 2012, 540: 210-214.
[19]  C H, BLAKE A. The strength of concentrated Mg-Zn solid solutions [J]. Phys Stat Sol (a), 2002, 194(1): 147-158.
C H, BLAKE A. The strength of concentrated Mg-Zn solid solutions [J]. Phys Stat Sol (a), 2002, 194(1): 147-158.
[20] BUHA J, OHKUBO T. Natural aging in Mg-Zn(-Cu) alloys [J]. Metall Mater Trans A, 2008, 39(9): 2259-2273.
[21] BAMBERGER M, DEHM G. Trends in the development of new Mg alloys [J]. Annu Rev Mater Res, 2008, 38: 505-533.
����ƽ�������ܣ������䣬�� �ˣ���������� �ɣ��ظ���
������ѧ ���ϸ���������֯���������ص�ʵ���ң����� 110819
ժ Ҫ������ƽ��Ͻ�����X�������䡢ɨ��羵����������ϵͳ���о���Mg-Zn-Al��Ԫϵ��þ��335 ��C��ƽ������ɼ���ɷ֡���ʵ����֤ʵ����-Mg�����岢����Mg32(Al,Zn)49 (��)��Ԫ�����仯�����q����ƽ�⣬������һ����Ԫ������Al5Mg11Zn4 (��)��ƽ�⡣����˦�����335 ��C�������ɷַ�Χ������52.5%~56.4% Mg��13.6%~24.0% Al��19.6%~33.9% Zn(Ħ������)��Al��MgZn���еĹ��ܶ�Զ������Mg7Zn3���еĹ��ܶȣ������ֵ�ɴ�8.6%Al(Ħ������)��Al��Zn����ͬʱ�����ڦ�-Mg�������С�
�ؼ��ʣ�Mg-Zn-Alϵ�����½��棻�ӽ����仯������ܶ�
(Edited by Yun-bin HE)
Foundation item: Projects (50901017, 51171043, 51271053) supported by the National Natural Science Foundation of China, Project (20090042120008) supported by the Doctoral Program Foundation of Institutions of Higher Education of China; Project (2011BAE22B04-2) supported by National Key Technology R&D Program of China during the Twelfth Five-Year Plan Period
Corresponding author: Gao-wu QIN; Tel: +86-24-8369-1565; E-mail: qingw@smm.neu.edu.cn
DOI: 10.1016/S1003-6326(14)63483-X

