碘-碘化物浸金湿法冶金的热力学和动力学
来源期刊:中国有色金属学报(英文版)2015年第11期
论文作者:S. S. KONYRATBEKOVA A. BAIKONUROVA G.A. USSOLTSEVA C. ERUST A. AKCIL
文章页码:3774 - 3783
关键词:金;湿法冶金;碘-碘化物浸出;无氰浸出剂
Key words:gold; hydrometallurgy; iodine-iodide leaching; non-cyanide lixiviants
摘 要:研究金在碘-碘化物浸出液中的溶解热力学平衡和动力学,着重研究pH值和温度对体系的影响。水溶液中碘的热力学分析结果表明,多种形式的碘化物主要存在于pH值为酸性的区间。体系电势的增加导致碘化物种类的增加,碘元素的添加能提高体系的氧化电位。IO3-离子能稳定存在于电势范围为–2.0至–0.75 V和pH值大于12.1的区间。提高温度会使各种碘化物在酸性pH区间存在的边界线发生移动,其中一些碘化物变得不稳定。扩散系数和扩散边界层厚度的测量值以及圆盘表面的溶剂质量浓度(14 mg/L)分析表明,浸出过程在外层扩散区域进行。因此,当选择碘化物浸出含金物料时,浸出过程必须在pH值为酸性区间内进行,在这些区间I-、I3–和IO4–离子能和金属形成配合物。
Abstract: The thermodynamic equilibria and kinetic aspect of gold dissolution in iodine-iodide leaching were studied with emphasis on the effects of pH value and temperature on the system. The results of thermodynamic analysis of iodine in aqueous solution were given and numerous forms of iodine exist mainly in the acid region of pH values. An increase of the potential of the system results in an increase of iodine speciation. The oxidizing potential of the system will increase by the addition of element iodine. The IO3- anions are stable in the potential range from -2.0 to -0.75 V and at рН value greater than 12.1. An increase of the temperature shifts boundaries of existence of various iodine species in the acid region of pH values. Some of them become unstable. The determined values of the diffusion coefficients and the thickness of the diffusion boundary layer, as well as the solvent concentration on the disc surface (14 mg/L) indicate that the process proceeds in the external diffusion region. Thus, while choosing the conditions of leaching from gold-containing materials of different origins of iodide solvents, it is necessary to carry out the process within the acidic region of pH values, where I-, I3- and IO4- ions are capable to form complex compounds with metals.
Trans. Nonferrous Met. Soc. China 25(2015) 3774-3783
S. S. KONYRATBEKOVA1, A. BAIKONUROVA1, G. A. USSOLTSEVA1, C. ERUST2, A. AKCIL2
1. Mining and Smelting Institute after O.A. Baikonurov, Kazakh National Technical University named after K.I. Satpaev, Satpaev Street, 22, Almaty 050013, Republic of Kazakhstan;
2. Mineral-Metal Recovery and Recycling Research Group, Mineral Processing Division, Department of Mining Engineering, Suleyman Demirel University, Isparta TR32260, Turkey
Received 1 November 2014; accepted 17 June 2015
Abstract: The thermodynamic equilibria and kinetic aspect of gold dissolution in iodine-iodide leaching were studied with emphasis on the effects of pH value and temperature on the system. The results of thermodynamic analysis of iodine in aqueous solution were given and numerous forms of iodine exist mainly in the acid region of pH values. An increase of the potential of the system results in an increase of iodine speciation. The oxidizing potential of the system will increase by the addition of element iodine. The IO3- anions are stable in the potential range from -2.0 to -0.75 V and at рН value greater than 12.1. An increase of the temperature shifts boundaries of existence of various iodine species in the acid region of pH values. Some of them become unstable. The determined values of the diffusion coefficients and the thickness of the diffusion boundary layer, as well as the solvent concentration on the disc surface (14 mg/L) indicate that the process proceeds in the external diffusion region. Thus, while choosing the conditions of leaching from gold-containing materials of different origins of iodide solvents, it is necessary to carry out the process within the acidic region of pH values, where I-, I3- and IO4- ions are capable to form complex compounds with metals.
Key words: gold; hydrometallurgy; iodine-iodide leaching; non-cyanide lixiviants
1 Introduction
Hydrometallurgical process has been utilized more often than the pyro-metallurgical process because of its ability of complex and low-grade ores extraction. Pyro-metallurgical processing, including incineration and smelting in a furnace or blast furnace at high temperatures, has become a traditional method to recover gold from ores and electronic wastes in the past two decades [1]. Compared with these processes, the hydrometallurgical method is more exact and predictable, and more easily controlled [2]. Gold has mostly been leached out from ores, secondary sources and gold-containing spent materials by cyanidation [3-6]. Biooxidation of refractory gold-bearing arsenopyrite/ pyrite concentrates as a pretreatment step prior to cyanidation has already been proved to be an economically viable and competitive process with reduced environmental impact and lower capital costs [7-10]. Pretreatment of ores and concentrates via biooxidation process is achieved by acidophilic chemolithotrophic microorganisms, which oxidizes the gold-bearing sulfidic minerals, mainly pyrite and arsenopyrite, thereby liberating gold and silver from the sulfide matrix. The liberated gold is then leached by different methods, mainly by cyanidation [11-14]. Cyanide consumers include iron, copper, zinc, cobalt and nickel ions, etc. [5]. It has also been reasoned that cyanide consumption was related to the formation of iron and arsenic precipitates during biooxidation and their subsequent carry-over to cyanidation [15]. Moreover, KOMNITSAS and POOLEY [11] suggested that high cyanide consumption was related to the formation and precipitation of iron and silver jarosites, particularly at low pulp densities. Considering the increase of recent environmental accidents at various gold recovery processes around the world causes severe contamination of natural water resources over the use of cyanide as a lixiviant. Many leaching agents are used to overcome this serious issue [16]. The application of alternative lixiviants, such as thiourea, chloride, bromide, iodine and ammonium thiosulfate, for gold recovery has been extensively researched in the last few years. These lixiviants would be particularly attractive if they improve the gold recovery or result in savings in reagent cost [17]. It can be seen from the discussed literature data that halogenide leaching of gold, and in particular iodide leaching, from gold-containing materials is an alternative to the cyanidation process [18,19]. Ideally, the lixiviant should be cheap or recyclable, selective, nontoxic and compatible with downstream recovery processes [20]. In practice, meeting all these criteria is difficult [21]. The formed various complexes with chloride, bromide and iodide and the stability of formed complex Iˉ> Brˉ> Clˉ were determined depending on the ambient conditions [22,23]. The use of a solution containing iodine–iodide as a lixiviant for gold was initially reported in various studies for recovering gold. Later fundamental studies on this leaching system have been carried out by a number of researchers [24-29]. It is understood that, under general conditions, iodine dissolves in the presence of iodide to form triiodide ion, which acts as the oxidant for the oxidation of element gold to gold(I)–iodide complex. Gold(I, III) also forms with iodide ions binary (AuI, AuI3) and complex compounds (M[AuI2], M[AuI4]). The solubility products of gold iodide (AuI) and gold triiodide (AuI3) in water
are 1.6×10ˉ23 and 1×10ˉ46, respectively. Upon the introduction of potassium iodide (iodide ion) into a solution, the solubility of AuI increases because of the formation of a complex ion, AuI2 [30]. The relevant reactions are shown below along with their thermodynamic data [31,32]:
I2(aq)+I-→I3-, K=714 (1)
I3-+2e→3I-, φ=0.536 V (2)
AuI2-+e→Au+2I-, φ=0.578 V (3)
where K is the equilibrium constant, φ is the potential with respect to standard hydrogen electrode (SHE).
The rate of gold dissolution in iodine–iodide solutions was found to be unaffected by pH value over wide ranges of 2-7 [27] and 2-10 [25]. Recently, in situ or in place leaching has been proposed as a method for the extraction of gold from stranded oxidised resources which cannot be mined economically using conventional technologies [33], and preliminary laboratory work using iodine-iodide as one of the lixiviant systems has returned very promising results [34].
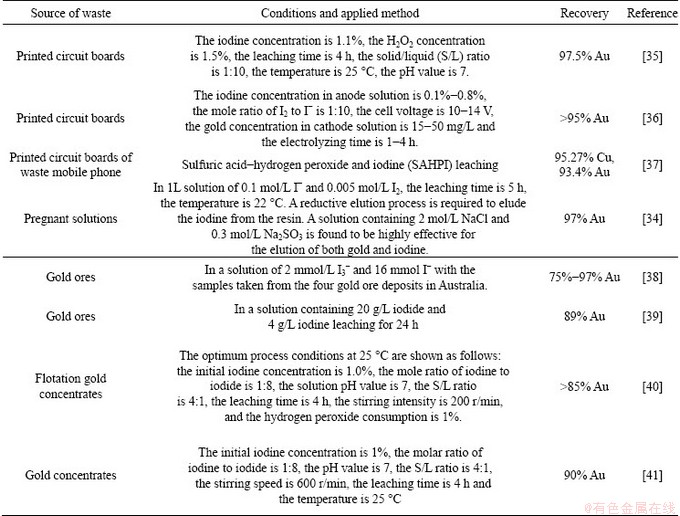
The studies on gold recovery with solution of iodine-iodide are summarized in Table 1.
Table 1 Research and development works on iodine-iodide leaching
2 Experimental
2.1 Material
The eltra electronic scrap consists of 0.45% Au, 0.008% Fe, 35.2% Zn, 63% Cu, 0.0002% Ag, 0.0054% Mn, 1.154% Ni, 0.05% Sn, 0.002% As, 0.008% Bi, 0.0016% Ga, 0.0007% Co and 0.002% Cr. The low- frequency rectangular connectors consists of 2.22% Au, 0.075% Ag, 0.007% As, 0.6% Ni, 7.5% Sn, 0.78% Bi, 49% Cu, 0.39% Fe and 30% Zn. The thickness of the gold covering is 24 μm.
2.2 Methods
The leaching of the raw materials was carried out at room temperature in a heat-resistant glass flask of 0.2 L. The polyions solute was added with stirring to the flask with the portion of gold material (the S/L ratio is 1:10 or 1:5, the pH value is 3, the particle size is ~150 μm and the time is 45 min). Filtrate was analyzed for the containing of gold.
In order to provide thermodynamic substantiation of leaching of gold-containing materials with iodide solutions, a comparative analysis was undertaken to assess possible reactions for gold dissolution in different solvents used extensively for this purpose. The analysis was carried out with the aid of the HSC Chemistry 5.11 software program for thermodynamic calculations developed by the Outokumpu Technology Engineering Research Stainless Steel Copper Zinc Metals. The results of kinetics studies of gold dissolution in iodide solutions using the rotating disc method were presented.
3 Results and discussion
3.1 Thermodynamic conditions for gold recovery from gold-containing materials with iodide solutions
The thermodynamic calculations developed by the Outokumpu Technology Engineering Research Stainless Steel Copper Zinc Metals are given in Table 2.
The leaching of gold-containing materials with cyanides and bromides becomes thermodynamically feasible because soluble complex gold compounds (NaAu(CN)2 and H(AuBr4)) are formed, and the isobaric-isothermal potentials of Reactions (5) and (6) are negative and reach 99.1 and 943.4 kJ/mol, respectively.
Table 2 Isobaric-isothermal potentials of gold dissolution reactions with different complex-forming reagents and oxidants
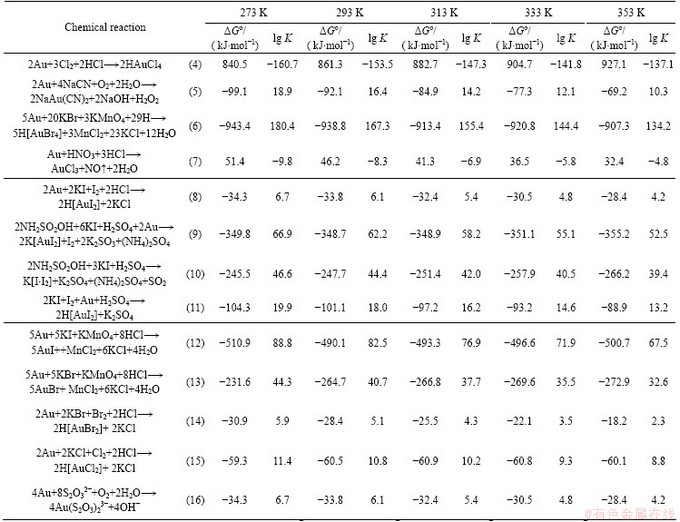
The use of oxidants (Cl2, Br2, I2 and KMnO4), along with iodide ion, increases significantly the probability of dissolution (Reactions (11)-(15) in Table 2) of both gold and metals sulfides, in particular iron sulfide. The results are in good agreement with the literature data [42,43], in which the authors demonstrated the feasibility of gold dissolution with potassium iodide solutions in the presence of an oxidant (H2О2, I2, OCI-). In a thermodynamic study of DAVIS et al [28], I3- was shown as the predominant oxidants species in both systems. However, if the concentrations of OCl- and I- equal, the solid iodine forms. In these systems, iodide (I-) is used to form I3- which is responsible for the gold oxidation and more free iodide needed for the gold complexation is destroyed in the I--OCl- system than that in the I--I2 system. The formation of solid AuI also explains the lower rate of gold dissolution determined for certain conditions in the kinetic study.
BONDARENKO and KULIKOV [44] pointed out good prospects for application of iodine-iodide leaching of gold. It was demonstrated that it was feasible to form complex haloid anions in the process of treatment of gold-containing materials with aqueous solutions of cyclic type with alkyl or haloid radicals [42]. The thermodynamic calculations presented in Table 2 (Reactions (8)-(11)) confirm the probability of iodine-iodide leaching of gold from gold-containing materials. Iodide is required in this case as a complex-forming agent ensuring the formation of a stable complex with gold, and iodine is needed as an oxidant preventing disproportioning of the formed gold complex and gold precipitation [45].
The gold dissolution reaction in an iodine-iodide system is expressed with Reaction (17).
2Au+I-+I3-→2AuI2- (17)
As shown in Table 3, chloride and thiocyanate leaching of gold-containing minerals does not result in the formation of insoluble metal compounds, which is confirmed by the values of the isobaric-isothermal potentials (△Gо>>0) and equilibrium constant of Reaction (4). The thermodynamic probability of gold dissolution in aqua regia (Reaction (7)) increases substantially in comparison with the previous solvents. In a poly-iodide solution, the formation of complex AuI4- is also possible [46]. MURPHY [47] pointed out that by selecting proper concentrations of iodide ion and molecular iodine, it was possible to obtain a redox potential sufficient for gold dissolution (Table 3).
To summarize, an iodine-iodide system has a number of advantages: lower toxicity, higher stability of formed complexes and lower redox potential as compared with other non-cyanide leaching systems.
The Gibbs energy of Reactions (9) and (12) is significantly lower than zero, which indicates a thermodynamic probability of these reactions even at room temperature.
The feasibility of the gold leaching process with iodine-containing solutions at low temperatures can be confirmed in an indirect way with the aid of Pourbaix diagrams showing the thermodynamically stable states of iodine in aqueous solutions within the iodine concentrations studies, as well as the potentials and pH values of the medium. The phase diagrams of φ-pH (Pourbaix diagrams) are given in Figs. 1, 2 and 3 for iodine-containing systems at 25, 50 and 80 °C, respectively.
The Pourbaix diagrams of I-H2O system are plotted with the aid of the above mentioned software for thermodynamic calculations of initial iodine concentration of c(I)initial=1×10-2 mol/L within the pH value interval of 0-14 and the potential range from -2.0 to 2.0 V (vs SHE).
At the potential range from -2.0 to -0.6 V and pH value interval of 0-12.1 and 25 °C, iodine is in the form of iodide ion I- (Figs. 1(a), 2(a) and 3(a)). Along with this ion, it is possible that IO3- ion is also present in the solution, which at the given potential and pH values is less stable (Figs. 1(b), 2(b) and 3(b)) and becomes stable in the potential range from -2.0 to -0.75 V at pH value of 12.1 and higher value.
At the potential range from -0.5 to -1.0 V, iodine exists predominantly in the form of hypoiodous acid (Figs. 1(a), 2(a) and 3(a)). Under the same conditions, the formation of unstable iodine ions such as H2IO+, I–, H3IO62-, I3– and IO3– is possible (Figs. 1(b), 2(b) and 3(b)).
With increasing the potential above 0.25 V, IO4– ions become stable at the pH value interval of 0-8.5, and H3IO62- ions become stable at the pH value greater than 8.5, and the formation of IO3– and I3– ions is possible.
Table 3 Standard oxidation potential and formation heat of haloid gold complexes

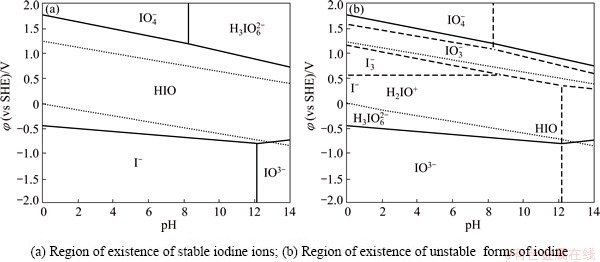
Fig. 1 Pourbaix diagrams of I–H2O system at 25 °C

Fig. 2 Pourbaix diagrams of I–H2O system at 50 °C
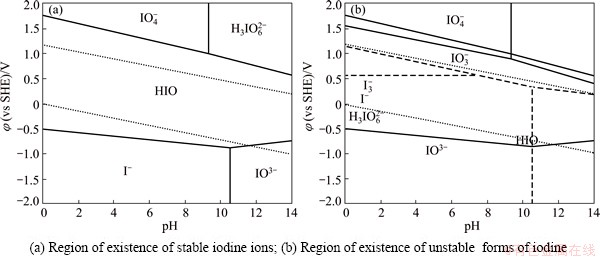
Fig. 3 Pourbaix diagrams of I–H2O system at 80 °C
An increase of the temperature causes a shift of regions of existence of both stable and unstable iodine ions. With an increase of the temperature, the region of IO3- existence shifts to a more acidic region at 50 °C with the pH value down to about 11.3 (Fig. 2(a)) and at 80 °C with the pH value down to 10.5 (Fig. 3(a)). At the same time, the region of H3IO62- existence moves slightly toward the alkaline region at 50 °C with the pH value up to about 8.75 and at 80 °C with the pH value up to about 9.0.
An increase of the temperature results in not only a shift of the boundaries regions of existence of various iodine forms, but also a disturbance of their stability, e.g., the formation of H2IO+ cation becomes thermodynamically impossible. Thus, a thermodynamic analysis of the I-H2O system suggests a conclusion that numerous iodine forms (I-, H3IO62-, IO3–, I3– and IO4–) exist mainly in the acidic region of pH values. An increase of the potential of the system (more than –0.5 V) results in an increase of the number of iodine forms of the aquatic medium. An increase of the temperature causes a shift of the boundaries of the iodine form existence region towards the acidic region of the pH values. At the same time, some of them become unstable, in particular H2IO+ ion.
On the basis of the conducted studies, it can be concluded that the gold leaching process should be carried out within the acidic region of pH values, where I-, I3– and IO4– ions are present, which are capable to form complex compounds with gold. The oxidizing potential of the system will increase in this case by the addition of element iodine.
3.2 Kinetics of gold dissolution in iodide solutions
The determined values of the diffusion coefficients using the rotating disc method and the thickness of the diffusion boundary layer, as well as the solvent concentration on the disc surface indicate that the process proceeds in the external diffusion region.
QI and HISKEY [26] used a glassy carbon rotating disk electrode to investigate the electrochemistry of iodide and a gold rotating disk electrode to explore the oxidation behavior of gold in iodide solutions. The effects of iodide concentration, electrode rotation and sweep rate on the electrochemical behavior of gold were examined. In addition, the reduction of iodine species on the gold electrode was also investigated. Two oxidation peaks, which represented the oxidations of Au to Au(I) and Au(III), were observed. It was confirmed that iodide was oxidized sequentially to tri-iodide and then to iodine and both of these reactions were reversible. At high concentrations of iodide and/or a slow scan rate, passivation, which was caused by the formation of solid iodine on the gold electrode surface, was found.
The rotating disc method was used to study the kinetics of gold dissolution from gold-containing material [48]. The test was carried out under hydrodynamic conditions at an installation comprising a reaction vessel with a capacity of 0.5 L, a magnetic mixer and a support stand with a disc fixed on it. A disk having a diameter of 20 mm had a copper base plated with gold (the mass of gold on the disc was 0.0711 g).
The disc was installed at the vessel center, without touching its bottom. Then the vessel was filled with iodide-containing solution and the mixing was started. Gold dissolution from the disc was conducted at room temperature (18-22 °C). The iodine concentration of the solvent varied from 40 to 250 mg/L. After the end of a preset mixing time, the gold-containing disc was removed from the solution, washed with distilled water, dried and weighed. The mass loss of the disc was determined to assess the gold dissolution in time, and accordingly the rate of metal transition to the solvent from a unit surface of the specimen.
The study of gold dissolution versus time was conducted using iodide solutions with a concentration of 40 mg/L and at a rotational speed of not more than 80 r/min to prevent the dissolution of the base material. The effect of stirring speed and iodide solution concentrations on the gold dissolution was performed at approximately 30 min.
In all cases, the copper content of the solution was monitored to make sure that the disc material did not dissolve and the test results obtained were within the permissible error.
Since the system was closed, the condition of the stationarity of the diffusion stream made it possible to solve the differential equations of the 1st and 2nd Fick laws and obtain an integral expression, which is used for calculating heterogeneous diffusion processes.
In accordance with the 1st Fick law, the mass of a substance (dmi), diffusing through a flat surface with an area S and perpendicular to the diffusion flow density during a time (dτ), is proportional to S·dτ and the concentration gradient dρi/dx:
dmi=-DiSdτ(dρi/dx) (18)
where Di is the diffusion coefficient.
The minus sign indicates the diffusion process toward a decreasing concentration gradient. If the diffusion process rate is assumed to be J=Ji=dmi/(dτ·S), called the diffusion flow density, then the equation of the 1st Fick law has a simple form:
J=-Di(dρi/dx) (19)
The reaction equation was recreated with the boundary conditions for any t on the reaction surface x=0 and ρi=ρn, within the solution volume x=△, ρi=ρо. Equation (2.3) has the following form:
Ji=△mi/(60τS) (20)
The calculation results are given in Table 4.
The repeatability of the dissolution rate in the case of different reaction time is very good as the points only slightly deviate from a straight line. A deviation from a straight line becomes noticeable at τ>150 min, therefore, further kinetics studies were conducted at τ=150 min (Fig. 4).
The dissolution rate is dependent on the mixing intensity, which is the main feature of the diffusion regime. The preliminary studies (Fig. 5) indicate that with increasing the mixing intensity above 100 r/min, the kinetics of the process changes (the slope angles of the straight lines), and the gold dissolution rate remains virtually constant.
Table 4 Test conditions and calculation results
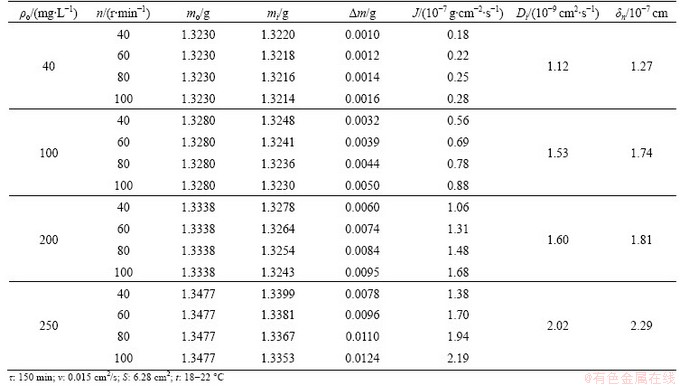
Fig. 4 Effect of process duration on changes of gold mass

Fig. 5 Effect of mixing intensity on gold dissolution rate with different iodine concentrations
Taking into account the slope angle of the straight line:
tan β=J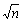 =0.2Di2/3·△ρ·ν–1/6 (21)
=0.2Di2/3·△ρ·ν–1/6 (21)
Assuming △ρ=ρо on the basis of previous calculations, the diffusion coefficients are calculated (Table 4).
Di=(tan β·ν1/6/0.2△ρ)3/2 (22)
Based on the experimental results, a graph in coordinates J-ρo is ploted (Fig. 6) corresponding to Eq. (23):
Ji=±Di(ρo-ρn)·τ/△i (23)
from which based on the slope angle tangent of the straight line in relation to the abscissa axis, the value of D/△ can be found as
tan α=J/ρo=D/△ (24)
In the heterogeneous diffusion process, the changes of concentration take place only within the diffusion boundary layer (△). Within the rest of the liquid volume, the concentration remains constant.
From the obtained equation, the thickness of the diffusion boundary layer is calculate as (Table 4)
△=D/tan α (25)
The intercept of the straight line on the abscissa axis corresponds to the iodine concentration on the disc surface (ρn), which is calculated on the basis of the value of tan α according to Eq. (26):
ρn=ρo-J/tan α (26)
ρn=250-2.19×10-7/(0.0088×10-7)=250-248.96=1.14 mg/L
The obtained values of the diffusion coefficient and the thickness of the diffusion boundary layer, as well as the solvent concentration on the disc surface indicate that the process takes place in the external diffusion region, which is in good agreement with the well-known literature data [49].
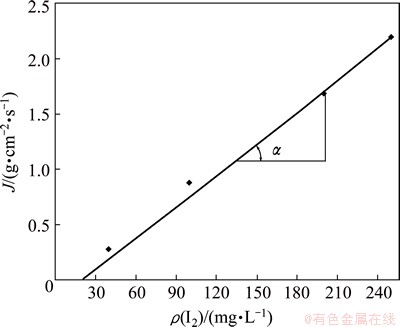
Fig. 6 Effect of iodine concentration in iodide solution on gold dissolution rate
4 Conclusions
1) In the potential range from -2.0 to -0.6) V (vs SHE) and рН value interval of 0-12.1, iodine is in the form of iodide-ion I-. In the potential range from -2.0 to -0.75 V (vs SHE) and рН value greater than 12.1, anion IO3- becomes stable. In the potential range from -0.5 to -1.0 V (vs SHE), iodine exists mainly in the form of iodine acid. At the potential greater than 0.25 V, ions IO4–at рН value interval of 0-8.5 and H3IO62- at рН value greater than 8.5 become stable.
2) An increase of the temperature causes a shift of regions of existence of both stable and unstable iodine ions. With increasing the temperature, the region of IO3– existence shifts to a more acid region and the region of H3IO62- existence moves towards alkaline region. Thus, the gold leaching process should be carried out within the acidic region of pH values, where I-, I3– and IO4– ions are present, which are capable of forming complex compounds with gold.
3) The iodine concentration of the solvent varied from 40 to 250 mg/L are calculated under kinetics process: the value of diffusion coefficient is (1.12-2.02)×10-9 сm2/s, the thickness of diffusion bordering layer is (1.27-2.29)×10-7 cm, the iodine concentration on the surface of the disc is 14 mg/L. Consequently, an increase of stirring intensity is limited by the iodine evaporation from the surface of the solvent.
Acknowledgement
This collaborative research was based on the results of bilateral contacts of Mineral-Metal Recovery and Recycling (MMR&R) Research Group, Mineral Processing Division, Department of Mining Engineering, Suleyman Demirel University, Isparta, Turkey, and Mining and Smelting Institute after O.A. Baikonurov, Kazakh National Technical University named after K.I. Satpaev, Almaty, Kazakhstan.
References
[1] CUI J, ZHANG L. Metallurgical recovery of metals from electronic waste: A review [J]. J Hazard Mater, 2008, 158: 228-256.
[2] ANDREWS D, RAYCHAUDHURI A, FRIAS C. Environmentally sound technologies for recycling secondary lead [J]. J Power Sources, 2000, 88: 124-129.
[3] FRICKER A G. Recovery of cyanide in the extraction of gold [J]. J Cleaner Prod, 1993, 1: 77-80.
[4] TRAPP S, LARSEN M, PIRANDELLO A, DANQUAH-BOAKYE J. Feasibility of cyanide elimination using plants [J]. Eur J Mine Process Environ Prot, 2003, 3: 128-137.
[5] MUDDER T I, BOTZ M M. Cyanide and society: A critical review [J]. Eur J Mineral Process Environ Prot, 2004, 4: 62-74.
[6] BOTZ M, MUDDER T, AKCIL A. Cyanide treatment: Physical, chemical and biological processes [M]//ADAMS M D. Advances in gold ore processing. Amsterdam: Elsevier Ltd., 2005: 672-700.
[7] RAWLINGS D E, DEW D, DU PLESSIS C. Biomineralization of metal containing ores and concentrates [J]. Trend in Biotechnol, 2003, 21: 38-44.
[8] JOHNSON D B. Biohydrometallurgy and environment: Intimate and important interplay [J]. Hydrometallurgy, 2006, 1-4: 153-166.
[9] BRIERLEY J A. Biohydrometallurgy—This microbiologist's perspective [J]. Adv Mat Res, 2007, 20-21: 3-10.
[10] BRIERLEY C L. How will biomining be applied in future? [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(6): 1302-1310.
[11] KOMNITSAS C, POOLEY F D. Bacterial oxidation of an arsenical gold sulphide concentrate from Olympias [J]. Greece Miner Eng, 1990, 3(3-4): 295-306.
[12] UBALDINI S,  F, TORO L, ABBRUZZESE C. Biooxidation of arsenopyrite to improve gold cyanidation: Study of some parameters and comparison with grinding [J]. Int J Miner Process, 1997, 52: 65-80.
F, TORO L, ABBRUZZESE C. Biooxidation of arsenopyrite to improve gold cyanidation: Study of some parameters and comparison with grinding [J]. Int J Miner Process, 1997, 52: 65-80.
[13] OLSON G J, BRIERLEY J A, BRIERLEY C L. Bioleaching review part B: Progress in bioleaching: Applications of microbial processes by the minerals industries [J]. Appl Microbiol Biotechnol, 2003, 63: 249-257.
[14] CIFTCI H, AKCIL A. Effect of biooxidation conditions on cyanide consumption and gold recovery from a refractory gold concentrate [J]. Hydrometallurgy, 2010, 104: 142-149.
[15] MILLER P, BROWN A. Bacterial oxidation of refractory gold concentrates [M]//ADAMS M D. Developments in mineral processing, advances in gold ore processing. Amsterdam: Elsevier Ltd., 2005: 371-402.
[16] SYED S. Recovery of gold from secondary sources—A review [J]. Hydrometallurgy, 2012, 115-116: 30-51.
[17] von MICHAELIS H. Gold processing update. The prospects for alternative leach reagents, can precious metals producers get along without cyanide? [J]. Engineering and Mining Journal, 1987, 188: 42-47.
[18] KONYRATBEKOVAA S S, BAIKONUROVAA A, AKCIL A. Non-cyanide leaching processes in gold hydrometallurgy and iodine-iodide applications: A review [J]. Mineral Processing and Extractive Metallurgy Review, 2015, 36(3): 198-212.
[19] AKCIL A, ERUST C, GAHAN C S, OZGUN M, SAHIN M, TUNCUK A. Precious metal recovery from waste printed circuit boards using cyanide and non-cyanide lixiviants—A review [J]. Waste Management, 2015, 45: 258-271.
[20] AVRAAMIDES J. Prospects for alternative leaching systems for gold: A review [C]//Symposia Series—Carbon-in-pulp Technology for the Extraction of Gold. Australasian Institute of Mining and Metallurgy, Parkville, Victoria, Australia, 1982: 369-391.
[21] MOLLEMAN E, DREISINGER D. The treatment of copper–gold ores by ammonium thiosulfate leaching [J]. Hydrometallurgy, 2002, 66: 1-21.
[22] HISKEY J B, ATLURI V P. Dissolution chemistry of gold and silver in different lixiviants [J]. Mineral Processing and Extractive Metallurgy Review, 1988, 4: 95-134.
[23] ULUSOY U,
[24] DAVIS A, TRAN T. Gold dissolution in iodide electrolytes [J]. Hydrometallurgy, 1991, 26: 163-177.
[25] QI P H, HISKEY J B. Dissolution kinetics of gold in iodide solutions [J]. Hydrometallurgy, 1991, 27: 47-62.
[26] QI P H, HISKEY J B. Electrochemical behavior of gold in iodide solutions [J]. Hydrometallurgy, 1993, 32: 161-179.
[27] ANGELIDIS T N, KYDROS K A, MATIS K A. A fundamental rotating disc study of gold dissolution in iodide–iodine solutions [J]. Hydrometallurgy, 1993, 34: 49-64.
[28] DAVIS A, TRAN T, YOUNG D R. Solution chemistry of iodide leaching of gold [J]. Hydrometallurgy, 1993, 32: 143-159.
[29] MARUN J N, MEISSL R J, LARA R F, GARCIA R A. Gold bearing ore processing with iodine-iodide solutions [C]//Proceedings of the XX International Mineral Processing Congress. Aachen, Germany: GMDB (Gesellschaft fur Bergball, Metallurgie), 1997, 21-26(4): 381-391.
[30] KOZIN L F, MELEKHIN V T. Extraction of gold from ores and concentrates by leaching with the use of cyanides and alternative reagents [J]. Russian Journal of Applied Chemistry, 2004, 77(10): 1573-1592.
[31] BARD A J, PARSONS R, JORDAN J. Standard potentials in aqueous solution [M]. New York and Basel: Marcel Dekker, Inc., IUPAC, 1985: 84-96.
[32] SAHIN M, AKCIL A, ERUST C, ALTYNBEK S, GAHAN C S, TUNCUK A. A potential alternative for precious metal recovery from E-waste: Iodine leaching [J]. Separation Science and Technology, 2015, 50: 2587-2595.
[33] ROBERTS P A, ZHANG H, PROMMER H, JOHNSTON C D, JEFFREY M I, BENVIE B, JEFFREY R G, GIPPS I D, ANAND R R. In place leaching of oxidized gold deposits: A new method for recovering stranded gold resources? [C]//World Gold Conference 2009—The Southern African Institute of Mining and Metallurgy. SAIMM, Jhannesburg, South Africa, 2009: 43-50.
[34] ZHANG H, JEFFERY C A, JEFFREY M I. Ion exchange recovery of gold from iodine–iodide solutions [J]. Hydrometallurgy, 2012, 125-126: 69-75.
[35] XU Qu, CHEN Dong-hui, CHEN Liang, HUANG Man-hong. Iodine leaching process for recovery of gold from waste PCB [J]. Chinese Journal of Environmental Engineering, 2009, 3(5): 911-914. (in Chinese)
[36] XU Qu, CHEN Dong-hui, CHEN Liang, HUANG Man-hong. Electrodepositing gold of iodine leaching solution from waste printed circuit board [J]. The Chinese Journal of Nonferrous Metals, 2009, 19(6): 1130-1135. (in Chinese)
[37] YIN J F, ZHAN S H, XU H. Comparison of leaching processes of gold and copper from printed circuit boards of waste mobile phone [J]. Advanced Materials Research, 2014, 955-959: 2743-2746.
[38] ROBERTS P A, ZHANG H, PROMMER H, BENVIE B, JEFFREY M I, JOHNSON C D, ANAND R R. Ore characterization, hydrometallurgical and reactive transport studies for in-place leaching of oxidized gold deposits [J]. Miner Metall Process, 2010, 27: 72-80.
[39] BAGHALHA M. Gold extraction from an oxide ore in iodine-iodide solutions [J]. ECS Transactions, 2012, 41(17): 53-60.
[40] WANG H X, SUN C B, LI S Y, FU P F, SONG Y G, LI L, XIE W Q. Study on gold concentrate leaching by iodine-iodide [J]. International Journal of Minerals, Metallurgy and Materials, 2013, 20(4): 323-328.
[41] LI Shao-ying, WANG Hai-xia, SUN Chun-bao, ZHAO Liu-cheng, YAN Zhi-qiang. Effects of different iodides on gold concentrates leaching process in iodine-iodide solution [J]. The Chinese Journal of Nonferrous Metals, 2013, 23(5): 1434-1439. (in Chinese)
[42] SERGENT R H, KENNETH T. Process for metal recovery and compositions useful therein: US, 4637865 [P]. 1987-01-20.
[43] TRAN T, DAVIS A. Fundamental aspects on the leaching of gold in nalide media [C]//EPD Congr. 1992 Proc. Symp, TMS Annu. Met. San Diego, Calif, 1991: 99-113.
[44] BONDARENKO S. KULIKOV G V. Industrial underground water [M]. Nedra, Moscow: Nedra Publishing House, 1984, 340.
[45] LASKORIN B N. Hydrometallurgy gold [M]. Moscow: Metallurgy, 1992: 365.
[46] ROBINSON J J. Extraction of gold from sulphidic concentrates by roasting and cyanidation [J]. Journal of the South African Institute of Mining and Metallurgy, 1988, 88(4): 117-130.
[47] MURPHY J W. Electrolytic process for the simultaneous deposition of gold and replenishment of elemental iodine: US, 4734171 [P]. 1988-03-29.
[48] KAKOVSKY I A, POTASHNIKOV Y M. Kinetics of dissolution [M]. Moscow: Metallurgy, 1975: 365.
[49] EMMANUEL N M, KNORRE D G. The rate of chemical kinetics [M]. Moscow: Moscow State University Press, 1969: 285.
S. S. KONYRATBEKOVA1, A. BAIKONUROVA1, G. A. USSOLTSEVA1, C. ERUST2, A. AKCIL2
1. Mining and Smelting Institute after O.A. Baikonurov, Kazakh National Technical University named after K.I. Satpaev, Satpaev Street, 22, Almaty 050013, Republic of Kazakhstan;
2. Mineral-Metal Recovery and Recycling Research Group, Mineral Processing Division, Department of Mining Engineering, Suleyman Demirel University, Isparta TR32260, Turkey
摘 要:研究金在碘-碘化物浸出液中的溶解热力学平衡和动力学,着重研究pH值和温度对体系的影响。水溶液中碘的热力学分析结果表明,多种形式的碘化物主要存在于pH值为酸性的区间。体系电势的增加导致碘化物种类的增加,碘元素的添加能提高体系的氧化电位。IO3-离子能稳定存在于电势范围为–2.0至–0.75 V和pH值大于12.1的区间。提高温度会使各种碘化物在酸性pH区间存在的边界线发生移动,其中一些碘化物变得不稳定。扩散系数和扩散边界层厚度的测量值以及圆盘表面的溶剂质量浓度(14 mg/L)分析表明,浸出过程在外层扩散区域进行。因此,当选择碘化物浸出含金物料时,浸出过程必须在pH值为酸性区间内进行,在这些区间I-、I3–和IO4–离子能和金属形成配合物。
关键词:金;湿法冶金;碘-碘化物浸出;无氰浸出剂
(Edited by Mu-lan QIN)
Corresponding author: A. AKCIL; Tel: +90-246-2111321; E-mail: ataakcil@sdu.edu.tr
DOI: 10.1016/S1003-6326(15)63980-2

