Trans. Nonferrous Met. Soc. China 24(2014) 2314-2319
Effect of surface treatment for aluminum foils on discharge properties of lithium-ion battery
Shigeki NAKANISHI1, Takashi SUZUKI1, Qi CUI1, Jun AKIKUSA2, Kenzo NAKAMURA2
1. Products Research and Development Department, Mitsubishi Aluminum Co., Ltd., 85 Hiramatsu, Susono-city, Shizuoka-pref., 411-1127, Japan;
2. Central Research Institute, Electronic Components Department, Mitsubishi Materials Corporation, 1002-14 Mukohyama, Naka-city, Ibaraki-pref., 311-0102, Japan
Received 17 October 2013; accepted 4 April 2014
Abstract: Aluminum foils having thicknesses of 10-20 μm are commonly employed as current collectors for cathode electrodes in Li-ion batteries. The effects of the surface morphology of the foil on battery performance were investigated by using a foil with roughened surface by chemical etching and a plain foil with smooth surface on both sides. For high-conductivity LiCoO2 active materials with large particle size, there are no significant differences in battery performance between the two types of foils. But for low-conductivity LiFePO4 active materials with small particle size, high-rate discharge properties are significantly different. The possibility shows that optimizing both the surface morphology of the aluminum foil and particle size of active material leads to improvement of the battery performance.
Key words: lithium-ion battery; battery performance; surface treatment; conductivity; plain foil; roughened foil
1 Introduction
Li-ion secondary batteries are now considered the most promising power sources for mobile products. In the future, hybrid and electric vehicles will be supplied with power storage devices that store more energy in order to drive a motor [1], and both wind and solar energy are researched to power household objects.
Figure 1 shows that a battery cell consists of two electrodes: one electrode consists of a coated cathode active material (such as LiCoO2, LiMnO2, and LiFePO4) laminated to an aluminum foil current collector; the other consists of a coated anode active material (such as natural graphite) laminated to a copper foil current collector. The sandwiched layer between the two electrodes is a separator (such as polyethylene or polypropylene). Therefore, the electrodes act as a secondary battery owing to the movement of electrons and Li-ions in the organic electrolyte (such as ethylene carbonate (EC) and diethylene carbonate (DEC)).
Studies on the active material of the Li-ion battery have been conducted for the LiCoO2 commonly used in mobile devices [2,3], but O atoms of this active material are released under conditions that exceed 200 °C, so it has poor thermal stability [4]. Therefore, it may cause many problems when used in transportation equipment such as automobiles. Meanwhile, LiFePO4 has been reported as one of the candidate materials for which raw material costs are not expensive [5]. This compound has olivine structures, compared with formula (1) of LiCoO2 which has layered rock salt structures, and can be seen from formula (2) that LiFePO4 acts as a battery by the movement of electrons and Li-ions. In this case, O atoms possess excellent thermal stability because there is a strong coupling force with PO4 compounds. It has been reported that there are no mass changes in the TGA measurement at 350 °C [6].
On the other hand, the LiFePO4 compound itself has low electronic conductivity [7]. Many studies have been done concerning to improve the conductivity by carbon coating on the material surface [8-10]. Further, an invention for doping the active material with other conductive powders has been reported [11]. Similarly, in studies of the cathode electrode aluminum foil, some inventions to improve the adhesion of the active material by coating carbon on the foil surface have been reported [12,13].

Fig. 1 Structure of Li-ion battery cell and contact with conductive material (LiFePO4 active materials are smaller than LiCoO2)
 (1)
(1)
 (2)
(2)
Thin foils having thicknesses of 10-20 μm are used as the cathode electrode current collector. Cell manufacturers select different alloys according to the desired battery performance (e.g., high capacity, cycle properties). Generally, the foils are required to have high conductivity, strength and less thickness in order to increase the cell capacity per unit mass, and to prevent breakage of the electrode material during coating. Therefore, in some cases, high-conductivity AA1085 (99.85% Al) is used, and in other cases, high-strength AA3003 (Al-1%Mn) is selected. Two thin aluminum foils are usually put together for the final rolling pass, and after being separated, one foil has different surface appearance on both sides (bright and matte). Since these variations in surface roughness are considered to affect battery performance and productivity, a foil with both sides having bright surfaces is used.
High electric potential of ~4 V is characteristic of these battery cells; therefore, the aluminum current collector should have insulating properties to withstand high voltage and conductivity in order to encourage electron pathways. For insulation, as shown in formula (3), the first charge forms an insulating layer on the foil surface that can maintain high corrosion resistance without breaking the aluminum current collector [14].
Al+3PF6-→AlF3+3PF5+3e- (3)
Regarding the conductivity, conductive materials (acetylene black) are described as those that form an electron conductive path in contact with a point on the foil surface [14]. In this study, to increase contact area between the conductive material and aluminum foil, we employed a etching method in which a large number of pits are formed on the surface. The present study was undertaken in order to evaluate the discharge properties of roughened foil surfaces treated by chemical etching and to compare them with those of commonly used plain foil with smooth surface.
2 Experimental
Two types of AA3003 aluminum foils of 15 μm thickness were used in this experiment. The first type, manufactured by foil rolling, had a smooth surface on both sides; the other type, manufactured by chemical etching, had a roughened surface covered by fine pits. Figure 2 shows the foil surfaces observed with a scanning electron microscope. As shown in Fig. 2, the roughened foil surface has fine etching pits of ~1 μm. The capacitances of the foils were measured by LCR meter as 5 and 17 μF/cm2, and the substantial surface area was increased by etching.
Table 1 shows the components of the battery cells. Two active materials were used: LiCoO2 (conductivity 1 S/m at RT) and LiFePO4 (low conductivity ~10-6 S/m at RT). To produce the cathode electrode sheet, a mixed slurry was added at a ratio of 89.5:5.5:5.0 for the active material (LCO and LFP), conductive materials (acetylene black), and binder (polyvinylidene fluoride (PVdF)), respectively. Subsequently, the slurry was coated onto an aluminum foil, pressed, and heat dried. Then, to confirm the adhesion to the cathode electrode sheet, a 90° peel test was performed corresponding to JIS K6854-1.

Fig. 2 SEM images of foil surfaces
Table 1 Cell configurations
Laminate-type cells were assembled by sandwiching the separator (polyethylene) between the lithium metal anode and cathode sheets, and then dipped into the electrolyte (1 mol/L LiPF6 EC/DEC). We chose half-cells so as to observe the influence of only the cathode electrode. After repeating the low-rate charge/discharge several times, a fully charged state was achieved. Rate property tests were performed a discharge cycle at a constant current corresponding to 0.2C-5C (1.5-37.5 mA). Charge/discharge cycle tests were repeatedly (over 200 times) performed at 1C (7.5 mA) for the cells with the LFP active material.
3 Results and discussion
Figure 3 shows the results of the rate property tests. With the LCO active material, no significant difference was confirmed on the characteristics between the plain and roughened foil cells. Meanwhile, for the cells with the LFP active material, the type of foil was confirmed to affect battery performance. Roughened foil has high performance even at discharges of 2C and 5C, but in the case of using plain foil such a high performance was not shown. It was caused from the particle size and conductivity of the active material itself (LCO) with the large particle size; also, LCO has higher conductivity than LFP. It was considered that movement of electrons and energizing the collector are facilitated. However, since the conductivity of LFP is low, it can be presumed to be affected by the distribution of the conductive material.
For confirmation, the cathode electrode sheet was cut with a cross-section polisher (CP) before the rate property test. The results are shown in Fig. 4. First, for plain foil with LCO, the active material itself is embedded in the foil because of compression. Also, for roughened foil with LCO, active and conductive materials are distributed to bite into its surface. Next, the roughened foil with LFP is in the high-adhesion state because slurry enters the foil surface. On the other hand, the interface between the plain foil with LFP is separated by the heat generated by CP cutting (~70 °C). It was observed several times, and the results were all similar. It is believed that such separation affects the cycle properties when subjected to thermal stresses due to the adhesion between the foil and slurry, such as repeated expansion/contraction due to charge/discharge. Therefore, we performed a peeling test to confirm the adhesion between the foil surface and the cathode electrode sheet coated with LFP. Test results are shown in Fig. 5, but regardless of the amount of active material, roughened foil was more advantageous than plain foil of any slurry thickness. Adhesion was improved by the anchor effect of the roughened foil.
Figure 6 shows the results of over 200 cycle tests using LFP cells, and battery performance is greatly improved with the roughened foil. When using plain foils, battery capacity is reduced after about 100 cycles, but when using roughened foils, the capacity degradation is small. Moreover, battery performance is maintained also in the cycle test at 1C.
Figure 7 shows SEM images of plain and roughened foils using LFP. Since the conductive materials enter three-dimensionally into between the foil and active materials, the movement route of electrons can be secured. So, high-rate discharge was possible in the low- conductivity active material. Furthermore, the anchor effect is presumed to work effectively, as cycle properties have been maintained. In porous form such a structure becomes more enhanced [15], so it is understandable from the results that the performance is excellent.

Fig. 3 Rate discharge curves of LiCoO2 with plain foil (a) and roughened foil (b), LiFePO4 with plain foil (c) and roughened foil (d)

Fig. 4 Cross-sectional structure of cathode sheet of LiCoO2 with plain foil (a) and roughened foil (b), LiFePO4 with plain foil (c) and roughened foil (d)
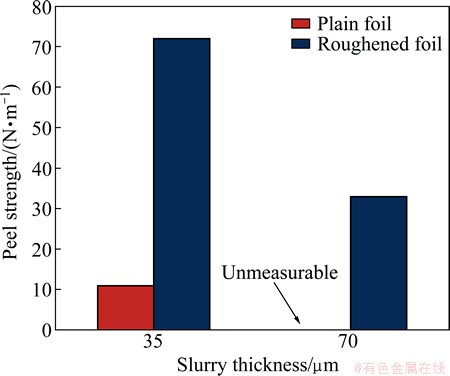
Fig. 5 Adhesive performance in 90° peel test using LiFePO4 cathode sheets

Fig. 6 Cycle performance test using LiFePO4 cells

Fig. 7 FE-SEM images of plain (a) and roughened (b) foil cathode sheet using LiFePO4 cells
The LFP has a lower potential than the LCO, but its thermal stability is high because LFP has an olivine structure with PO4, as the O atom is not detached even if heated or short circuited. The results from this study indicate that such foils can be used in hybrid vehicles and other high-power devices.
4 Conclusions
A chemical etching surface treatment was used to prepare roughened foil for Li-ion battery cells and investigate the effect of surface morphology on battery performance. Evaluation of the cell performance with two active materials (LCO and LFP) was conducted, and there is no significant difference between two types of aluminum foils with LCO because the particles are large and become embedded on the foil by compression. On the other hand, for the cells with the LFP active material and roughened foils, significant difference was confirmed to affect high-rate cycle properties. It is believed that the cathode electrode sheet with roughened foil exhibits improved performance because contact with the surface over the active and conductive material is increased. In particular, if it is possible to create an optimal pit size for the active and conductive materials, the roughened foil was suggested to exert a positive effect on the battery performance.
References
[1] TERADA N, YANAGI T, ARAI S, YOSHIKAWA M, OHTA K, NAKAJIMA N, YANAI A, ARAI N. Development of lithium batteries for energy storage and EV applications [J]. J Power Sources, 2001, 100: 80-92.
[2] NAGAURA T, TOZAWA K. Lithium ion rechargeable battery [J]. Prog Batteries Solar Cells, 1990, 9: 209-217.
[3] OZAWA K. Lithium-ion rechargeable batteries with LiCoO2 and carbon electrodes: The LiCoO2/C system [J]. Solid State Ionics, 1994, 69: 212-221.
[4] DAHN R, FULLER E W, OBROVAC M, von SACKEN U. Thermal stability of LixCoO2, LixNiO2 and LixMnO2 and consequences for the safety of Li-ion cells [J]. Solid State Ionics, 1994, 69: 265-270.
[5] YAMADA A, CHUNG S C, HINOKUMA K. Optimized LiFePO4 for lithium battery cathodes [J]. J Electrochem Soc, 2001, 148(3): A224-A229.
[6] PADHI A K, NANJUNDASWAMY K S, GOODENOUGH J B. Phospho-olivines as positive-electrode materials for rechargeable lithium batteries [J]. J Electrochem Soc, 1997, 144(4): 1188-1194.
[7] SRINIVASAN V, NEWMAN J. Discharge model for the lithium iron-phosphate electrode [J]. J Electrochem Soc, 2004, 151(10): A1517-A1529.
[8] HUANG H, YIN S C, NAZAR L F. Approaching theoretical capacity of LiFePO4 at room temperature at high rates [J]. Electrochem Solid-State Lett, 2001, 4(10): A170-A172.
[9] SIDES C R, CROCE F, YOUNG V Y, MARTIN C R, SCROSATI B. A high-rate, nanocomposite LiFePO4/carbon cathode [J]. Electro- chem Solid-State Lett, 2005, 8(9): A484-A487.
[10] LIN Y, GAO M X, ZHU D, LIU Y F, PAN H G. Effects of carbon coating and iron phosphides on the electrochemical properties of LiFePO4/C [J]. J Power Sources, 2008, 184: 444-448.
[11] CHUNG S Y, BLOKING J T, CHIANG Y M. Electronically conductive phospho-olivines as lithium storage electrodes [J]. Nat Mat, 2002, 1: 123-128.
[12] STRIEBEL K, SHIM J, SIERRA A, YANG H, SONG X, KOSTECKI R, MCCARTHY K. The development of low cost LiFePO4-based high power lithium ion battery [J]. J Power Sources, 2005, 146(1): 33-38.
[13] OMORI M. Current collector for secondary battery, positive electrode for the secondary battery, negative electrode for the secondary battery, the secondary battery, and manufacturing method of them: JP4593488 [P]. 2010.
[14] TACHIBANA K, SATO Y, NISHINA T, ENDO T, MATSUKI K, ONO S. Passivity of aluminum in organic electrolytes for lithium batteries (1) film growing mechanism [J]. Electrochemistry, 2001, 69(9): 670-680.
[15] YAO M, OKUNO K, IWAKI T, KATO M, TANASE S, EMURA K, SAKAI T. LiFePO4-based electrode using micro-porous current collector for high power lithium ion battery [J]. J Power Sources, 2007, 173: 545-549.
锂电池中铝箔表面形貌对电池性能的影响
Shigeki NAKANISHI1, Takashi SUZUKI1, Qi CUI1, Jun AKIKUSA2, Kenzo NAKAMURA2
1. Products Research and Development Department, Mitsubishi Aluminum Co., Ltd., 85 Hiramatsu, Susono-city, Shizuoka-pref., 411-1127, Japan;
2. Central Research Institute, Electronic Components Department, Mitsubishi Materials Corporation, 1002-14 Mukohyama, Naka-city, Ibaraki-pref., 311-0102, Japan
摘 要:10~20 μm厚的铝箔经常应用于锂电池的阴极。将被化学腐蚀的粗糙表面铝箔与光滑平面的铝箔进行对比,以考察铝箔的表面形貌对锂电池的影响。对于高电导率且颗粒较大的LiCoO2锂电池材料,两种铝箔的锂电池性能没有明显不同。但是对于低电导率且颗粒较小的LiFePO4材料,高倍率放电性有很大的差异。通过优化铝箔的表面形貌及电池材料的颗粒尺寸,可使电池的性能得到提高。
关键词:锂电池;电池性能;表面处理;电导率;光滑铝箔;粗糙铝箔
(Edited by Hua YANG)
Corresponding author: Shigeki NAKANISHI; E-mail: sgnakans@malco.co.jp
DOI: 10.1016/S1003-6326(14)63350-1