Ca对碳质孕育含Fe的Mg-3%Al合金晶粒细化的“免毒化”作用
来源期刊:中国有色金属学报(英文版)2013年第2期
论文作者:杜 军 王海蕾 周明川 李文芳
文章页码:307 - 314
关键词:Mg-Al合金;Fe;Ca;碳质孕育;形核衬底;免毒化
Key words:Mg-Al alloy; iron; calcium; carbon inoculation; nucleating substrate; poisoning-free effect
摘 要:利用Ca和碳质孕育对Mg-3%Al合金进行复合变质处理,并研究了Fe及其Fe的添加顺序对细化效果的影响。与碳质孕育相比,Ca和碳复合孕育后的晶粒细化效果更为显著,Fe及其添加顺序对复合孕育细化效果无显著影响。Ca的添加可有效避免碳质孕育Mg-Al合金中因Fe所导致的晶粒粗化作用。在Ca和碳质复合孕育试样中可观察到大量的Al-C-O颗粒,这些颗粒实际应为Al4C3,并作为a-Mg的形核核心。然而,在熔体中添加Fe后,试样中可观察到大量在Al-Fe或Al-C-Fe表面吸附Al4C3的双相粒子,该双相粒子可作为a-Mg的形核核心并导致晶粒细化。Ca有利于该双相粒子的生存,并与工艺条件无关,从而使得Fe对晶粒细化的“毒化”作用得到有效抑制,即Ca对Fe有“免毒化”效果。
Abstract: Mg-3%Al alloy was modified by combining Ca addition with carbon inoculation. The effects of Fe addition and addition sequence on the grain refinement were investigated. A higher grain refining efficiency could be obtained for the Mg-Al alloy modified by combining Ca addition with carbon inoculation. Fe addition and addition sequence had no obvious effect on the grain refinement. Ca addition could effectively avoid grain-coarsening resulting from Fe in the carbon-inoculated Mg-Al alloy. The Al-C-O particles, actually being Al4C3, should act as potent substrates for a-Mg grains in the sample treated by combining Ca addition with carbon inoculation. However, the duplex-phase particles of Al4C3 coated on Al-Fe or Al-C-Fe should be the potent substrates for a-Mg grains if Fe existed in the Mg-Al melt. Ca addition can contribute to the formation of the particles of Al4C3 coated on Al-Fe or Al-C-Fe, regardless of the Fe addition sequence. The poisoning effect of Fe was effectively inhibited in the carbon-inoculated of Mg-Al alloy due to Ca addition, namely, Ca has a poisoning-free effect.
Trans. Nonferrous Met. Soc. China 23(2013) 307-314
Jun DU, Hai-lei WANG, Ming-chuan ZHOU, Wen-fang LI
School of Materials Science and Engineering, South China University of Technology, Guangzhou 510640, China
Received 29 November 2011; accepted 20 July 2012
Abstract: Mg-3%Al alloy was modified by combining Ca addition with carbon inoculation. The effects of Fe addition and addition sequence on the grain refinement were investigated. A higher grain refining efficiency could be obtained for the Mg-Al alloy modified by combining Ca addition with carbon inoculation. Fe addition and addition sequence had no obvious effect on the grain refinement. Ca addition could effectively avoid grain-coarsening resulting from Fe in the carbon-inoculated Mg-Al alloy. The Al-C-O particles, actually being Al4C3, should act as potent substrates for a-Mg grains in the sample treated by combining Ca addition with carbon inoculation. However, the duplex-phase particles of Al4C3 coated on Al-Fe or Al-C-Fe should be the potent substrates for a-Mg grains if Fe existed in the Mg-Al melt. Ca addition can contribute to the formation of the particles of Al4C3 coated on Al-Fe or Al-C-Fe, regardless of the Fe addition sequence. The poisoning effect of Fe was effectively inhibited in the carbon-inoculated of Mg-Al alloy due to Ca addition, namely, Ca has a poisoning-free effect.
Key words: Mg-Al alloy; iron; calcium; carbon inoculation; nucleating substrate; poisoning-free effect
1 Introduction
Grain refinement has been proved to be an effective route to improve the mechanical properties of the Mg-Al alloys which are known as the lightest structural metallic materials [1-3]. Among many grain refining methods, the carbon inoculation was considered the hopeful grain refining route [4,5]. The refining mechanism of Al4C3 acting as nuclei for a-Mg grains has been widely appreciated to date by many researchers [4-9]. Fe was proved to be one of the important elements to determine the nucleating potency of Al4C3 [4,5,10-13]. HAITANI et al [10] concluded firstly that Fe poisoned the potency of the Al4C3 nucleating particles by transforming them into Al-C-Fe-rich intermetallic compounds. However, Fe was proved to play a positive role in the formation of the nucleating particles rather than being an inhibiting element in other studies [11,12]. Recently, DU et al [13] found that whether Fe inhibited the grain refinement or not was closely associated with the operating sequence of carbon inoculation and Fe addition. Obvious poisoning effect of Fe mainly occurred under the condition that the Mg-Al melt was fully inoculated by carbon before Fe addition.
In addition to carbon inoculation, the addition of alkaline-earth elements (Ca and Sr) is another important grain refining method for the Mg-Al alloys [4,5,14-17]. Due to the strong segregating power in the magnesium melt, the significant grain refinement potential of Ca or Sr is associated with higher constitutional undercooling at the advancing solid/liquid (S/L) interface during solidification [4,5]. This refining mechanism is completely different from that of the carbon inoculation. In previous studies [15,16], it has been proved that a higher grain refining efficiency could be obtained for the Mg-Al alloys modified by combining Ca(or Sr) addition with carbon inoculation due to the synergistic action of more potent nuclei and higher constitutional undercooling.
In the present study, a relatively high-purity Mg-3%Al alloy was used and the following research works have been carried out to clarify 1) how Fe addition and addition sequence influence the grain refinement of Mg-Al alloy which has been modified by combining Ca addition with carbon inoculation; 2) whether Fe inhibits the grain refinement of the Mg-Al alloy by combining Ca addition with carbon inoculation or not. Fe is an inevitable impurity element in the commercial Mg-Al alloys. From the viewpoint of commercial industries, the present investigation has an obvious practical significance.
2 Experimental
The raw materials used in the present study included relatively high purity magnesium (99.95%), high purity aluminum (99.99%), Al-15%Fe and Mg-10%Ca master alloys. The pellets containing carbon (Mg-40%Al-10%C) were prepared from Mg, Al and graphite powders by cold isostatic press (CIP). The designed Mg-3%Al alloy was used in this study. The addition amounts of Fe, Ca and carbon were 0.1 %, 0.2% and 0.2% (mass fraction) of the experimental material, respectively. To exactly control the Al content in Mg-Al melt,the amounts of Al in the pellets and Al-15%Fe master alloy were taken into consideration.
About 20 g Mg-3%Al alloy was melted with MgO crucible in an electric resistance furnace at 760 °C. The melt was covered by a protective flux. Eight samples were prepared in the present study. The preparation details of the eight samples, corresponding to the process No. 1 to 8, are described in Table 1.
Table 1 Mg-3%Al melt treated under different process conditions

The pure Mg and pure Al were firstly melted together for all samples. The Ca element was added into the Mg-3%Al melt by Mg-10%Ca master alloy. Carbon-inoculation was operated by plunging the carbon-containing pellets into the melt. The Al-15%Fe master alloy was used to add Fe into the melt. The addition sequence of Fe addition and carbon-inoculation was adjusted for the processes No. 5 to 8. In all processes, the melt was stirred by magnesia rod. More details about the process can be seen in Ref. [13].
The melts prepared through different process routes were poured into a cylindric iron-mould with the dimensions of d20 mm×25 mm, and the iron-mould was preheated at 500 °C. Metallographic samples were cut in the horizontal direction at the position of 10 mm from the bottom of the samples. The grain microstructures were observed using a Leica DFC320 type optical microscope. Linear intercept method described in ASTM standard E112-88 was used to evaluate the grain size. The microstructural characteristics and quantitative analysis of nucleating particles in the samples were conducted using an EPMA-1600 electron probe microanalyzer (EPMA).
3 Results
3.1 Grain refining efficiency
Figure 1 shows the grain morphologies of the Mg-3%Al alloy treated through different process routes. For the sample without any treatment, the grain was coarse with a size of about 610 μm (Fig. 1(a)). The grain was significantly refined for the sample treated by carbon inoculation. Its grain size was decreased to about 210 μm (Fig. 1(b)). For the sample treated by combining Ca addition with carbon inoculation, its grain size was obviously further refined to about 180 μm (Fig. 1(c)). A slight grain refining effect could be obtained for the sample treated by Fe and Ca addition. Its grain size was about 550 μm. However, for the rest four samples, the grain sizes were almost the same with the sample treated by combining Ca addition with carbon inoculation. Their grain sizes were all about 170 μm. Fe addition and addition sequence had no obvious effect on grain refinement of the Mg-3%Al alloy which was modified by combining Ca addition with carbon inoculation. A conclusion could be drawn that Fe addition and addition sequence had no obvious effect on grain refinement of the Mg-Al alloy modified by combining Ca addition with carbon inoculation. However, obvious grain- coarsening resulting from Fe was found in the authors’ previous study under the condition that the Mg-3%Al melt was fully inoculated by 0.2%C firstly and then 0.1%Fe was added into the melt [13], as shown in Fig. 2. The holding times after Fe addition were 10 min (Fig. 2(a)) and 20 min (Fig. 2(b)). Their grain sizes were 580 and 710 μm, respectively. Consequently, it seems that Ca could effectively avoid grain-coarsening resulting from Fe in the carbon-inoculated Mg-Al alloy.

Fig. 1 Grain morphologies of Mg-3% Al alloy without treatment (a) and with treatment through different processes of No. 2 (b), No. 3 (c), No. 4 (d), No. 5 (e), No. 6 (f), No.7 (g) and No. 8 (h)
3.2 EPMA observation
Figure 3 shows the EPMA back-scattered electron (BSE) image and EPMA point analyses of elements C, O, Mg, Al and Ca for the sample treated by combining Ca addition with carbon inoculation. Compared with EPMA analysis results measured from the particle A and area B (Figs. 3(b) and (c)), the particle A should be Al-C-O particles with small content of Ca, since the C, O, Al and Ca contents of the particle A were significantly higher than those in the Mg-matrix. The C and O measured from Mg matrix (area B) should be the result of contamination during sample preparation, since the solutions of C and O in the solid Mg are almost zero.

Fig. 2 Grain-coarsening of carbon-inoculated Mg-3%Al alloy resulting from Fe addition [13]
As for the samples treated through processes No. 5 to 8, many particles containing carbon could be easily observed, as shown in Fig. 4 and Fig. 5(a). These particles were further studied under high magnification. Figure 5 illustrates the detailed features of these particles in the sample treated through process No.6 that the Al-15%Fe alloy and carbon-containing pellets were simultaneously added into the Mg-3%Al-0.2%Ca melt.
Judged from the EMPA observation and point analysis, three types of particles existed in the sample treated through process No.6, as shown in Fig. 5. The white particle denoted by A in Fig. 5(b) mainly consisted of Al and Fe with low contents of C and O. The dark-grey particle denoted by B in Fig. 5(b) mainly consists of Al, Fe, C and O. However, a particularly interesting structure with duplex phases could be found under high magnification, as the particle denoted by C in Fig. 5(b). Judged from the EPMA point analysis measured from the core and outer phase of the particle, the central white particle was the Al-C-O-Fe phase with high Fe content. However, its outside area should be Al-C-O phase without Fe. Likewise, these three types of particles could be also observed in the samples treated through processes No. 5, 7 and 8 by magnification from EPMA-BSE images shown in Fig. 4. Moreover, it should be noted that the Ca content in all particles was slightly higher than that in the Mg-matrix (Fig. 3(c)).

Fig. 3 EPMA-BSE image (a) and EPMA point analyses of C, O, Mg, Al and Ca elements from particle A (b) and area B (c) for sample treated by combining Ca addition with carbon inoculation
4 Discussion
Known from Figs. 1(a), (b) and (c), a higher grain refining efficiency could be obtained for the Mg-3%Al alloy refined by the combination of Ca and carbon inoculation. The synergetic refining mechanisms had been discussed in the authors’ previous study [14]. The Al4C3 particles formed in the Mg-Al melt should be the potent nucleating substrates for a-Mg grains [4-9]. The addition of Ca can restrict grain growth due to segregation of Ca atom to the advancing S/L interface, since Ca has high segregating power in Mg melt [4,5,14]. In the present study, the Al-C-O particles with low Ca content could be observed, as shown in Fig. 3(b). The similar Al-C-O particles have been always found in many studies [6-8,11-15]. These Al-C-O particles should actually be the hydrolyzate of Al4C3 particles during specimen polishing through the chemical reaction [6]: Al4C3+12H2O=4Al(OH)3+3CH4↑. The Al-C-O phase was reasonably called Al4C3 compound to avoid being confused in the following discussion, since Al4C3 is the only compound formed by reaction between Al and C known from the Al-C phase diagram [18].

Fig. 4 EPMA-BSE images of samples treated through different process routes
Moreover, many small particles could be observed in the samples treated through processes No. 4 to 8 by EPMA-BSE observation, as shown in Figs. 4 and 5. These particles could be mainly classified into three kinds for every sample, regardless of the process condition. To avoid repeating, high magnification EPMA-BSE images and the EDS analysis were given only for the sample treated through process No. 6 in the present work, as shown in Fig. 5. The white particle denoted by A should be the Al-Fe-rich or Al-Fe-C particles with relatively small contents of C and O. This kind of white particles also existed in the sample treated through process No. 4 although Ca was also added. The Al-Ca-rich or Mg-Ca-rich particles could hardly be found in the samples prepared in the present study due to the low content of Ca addition. Judged from the coarse grain size shown in Fig. 1(d), the Al-Fe-rich particles should not be potent nucleating substrates for a-Mg grains under the present process condition. The second type was the dark-grey particle denoted by B in Fig. 5(b). This kind of dark-grey particles should be Al-C-Fe-rich intermetallic compounds, the oxygen should also be the result of contamination during sample preparation. The third type particle has duplex-phase structure, i.e, white core with dark-grey film, as the particle denoted by C in Fig. 5(b). The central white core should be the Al-Fe-rich or Al-Fe-C intermetallic compounds with high Fe content. However, the outside area of the dark-grey film should be Al-C (Al4C3) phase without obvious Fe.
After being inoculated by carbon, the formation of Al4C3 should be possibly interrupted by Al-Fe intermetallic compounds and solute Fe in the Mg-Al melt. Firstly, direct reaction among Al, C and Fe can occur, resulting in the formation Al-C-Fe intermetallic compounds. Secondly, the Al-C compounds may be formed by a reaction between Al and C firstly, and then the Al-C compounds are adsorbed on the surface of Al-Fe or Al-C-Fe compounds. Thirdly, the Al-C-Fe compounds can be directly generated by a reaction between C and Al-Fe compounds on the surface. These processes may be competing with each other, resulting in the formation of Al-C-Fe compounds with different structures. As for the latter two ways, an Al-C-rich phase (Al4C3) coating film can be formed on Al-Fe-rich or Al-Fe-C-rich compounds, as the particle C shown in Fig. 5(c).
As for the Al-C-Fe-rich particles, the most likely intermetallic compounds are AlFe3C0.5 and AlFe6C [19]. They have a face-centered cubic structure with almost the same lattice parameter of 0.3770 nm, which means that they have a planar disregisty mismatch with Mg of almost 15% for the close-packed planes. Obvious grain-coarsening shown in Fig. 2 could also prove that the Al-C-Fe-rich particles should not be the effective nucleating substrates for a-Mg grains, since the Al-C-Fe-rich particles could mainly be observed under the condition that Mg-Al melt was firstly inoculated by carbon and then Fe addition [20]. Therefore, only the particles with duplex-phase structure, namely, Al4C3 coated on Al-Fe or Al-C-Fe should be the potent substrates for a-Mg grains. The real potent nucleating substrates should also be Al4C3.
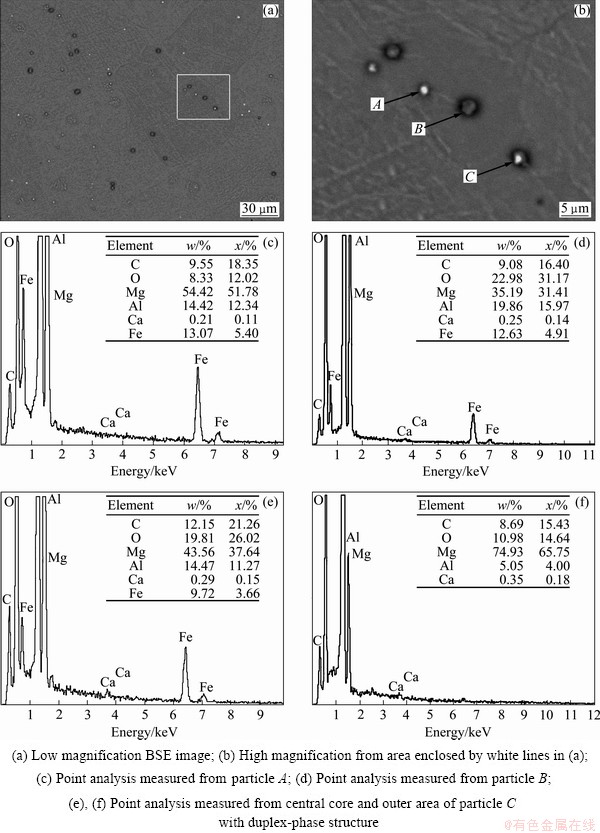
Fig. 5 EPMA-BSE images and EPMA point analysis of C, O, Mg, Al and Ca elements for sample treated through process No. 6
In the authors’ other studies [20], the particles with duplex-phase structure of Al4C3 coated on Al-Fe or Al-C-Fe were found only under the condition that Fe pre-existed in Mg-Al melt and then the melt was inoculated by carbon. However, it is very interesting that this type of particles with duplex-phase structure could be formed in all samples treated by combining Ca addition with carbon inoculation, regardless of the Fe addition sequence. Consequently, it can be reasonably inferred that Ca addition can contribute to the formation of the particle of Al4C3 coated on Al-Fe or Al-C-Fe. Ca can effectively avoid the poisoning effect of Fe on the carbon-inoculated Mg-Al alloy by inhibiting the formation of Al-C-Fe-rich intermetallic compounds. Ca has a poisoning-free effect on the carbon-inoculated Mg-Al melt contained trace Fe.
It is difficult to disclose the clear poisoning-free mechanism of Ca to date. It should be noted that Ca is apt to segregate the interface between different phases or the surface of some phases since it is an active element. Therefore, the Ca content in all particles was slightly higher than that in the Mg-matrix (Fig. 3(c)). The surface tension could reduce due to the segregation of Ca. In addition, the enrichment of Ca atoms on the surfaces can inhibit reaction among different phases. Consequently, the particle with duplex-phase structure of Al4C3 coated on Al-Fe or Al-C-Fe could be easily formed under the present process condition. However, more careful in-depth microscopic works are still needed to uncover the formation of duplex-phase structure and disclose the poisoning-free mechanism of Ca in the carbon- inoculated Mg-Al melt contained trace Fe.
5 Conclusions
1) Higher grain refining efficiency could be obtained for the Mg-Al alloy modified by combining Ca addition with carbon-inoculation. Fe addition and addition sequence had no obvious effect on the grain refinement. Ca addition could effectively avoid grain- coarsening resulting from Fe in the carbon-inoculated Mg-Al alloy.
2) The Al-C-O particles, actually being Al4C3, should act as potent substrates for a-Mg grain in the sample treated by combining Ca addition with carbon inoculation. However, the duplex-phase particles of Al4C3 coated on Al-Fe or Al-C-Fe should be the potent substrates for a-Mg grains if Fe existed in the Mg-Al melt.
3) Ca addition can contribute to the formation of the particles of Al4C3 coated on Al-Fe or Al-C-Fe, regardless of the Fe addition sequence. The poisoning effect of Fe was effectively inhibited in the carbon- inoculated of Mg-Al alloy due to Ca addition, namely, Ca has a poisoning-free effect.
References
[1] SURESH M, SRINIVASAN A, RAVI K R, PILLAI U T S, PAI B C. Microstructural refinement and tensile properties enhancement of Mg-3Al alloy using charcoal additions [J]. Materials Science and Engineering A, 2011, 528: 2502-2508.
[2] DU J, YANG J, KUWABARA M, LI W F, PENG J H. Effects of carbon and/or alkaline earth elements on grain refinement and tensile strength for AZ31 alloy [J]. Materials Transactions, 2008, 49: 2303-2309.
[3] MA Y Q, CHEN R S, HAN E H. Keys to improving the strength and ductility of the AZ64 magnesium alloy [J]. Materials Letters, 2007, 61: 2527-2530.
[4] St. JOHN D H, MA Q, EASTON M A, CAO P. Grain refinement of magnesium alloys [J]. Metallurgical Materials Transactions A, 2005, 36: 1669-1679.
[5] LEE Y C, DAHLE A K, St. JOHN D H. The role of solute in grain refinement of magnesium [J]. Metallurgical Materials Transactions A, 2000, 31: 2895-2906.
[6] LU L, KDAHLE A, St. JOHN D H. Grain refinement efficiency and mechanism of aluminium carbide in Mg-Al alloys [J]. Scripta Materialia, 2005, 53: 517-522.
[7] LIU Sheng-fa, LI Bo, HAN Hui, KANG Liu-gen, WANG Xiao-hu, SU Wei. Refinement effect and mechanism of Mg-Al4C3 master alloy in AZ91D Mg alloy [J]. The Chinese Journal of Nonferrous Metals, 2009, 19(1): 32-37. (in Chinese)
[8] WANG Zhao-hui, KANG Yong-lin, ZHAO Hong-jin, XU Yue. Grain refinement of Mg-Al magnesium alloys by carbon inoculation [J]. Transactions of Nonferrous Metals Society of China, 2006, 16(s): s1851-s1854.
[9] HAN G, LIU X F. Duplex nucleation in Mg-Al-Zn-Mn alloys with carbon inoculation [J]. Journal of Alloys and Compounds, 2009, 487: 194-197.
[10] HAITANI T, TAMURA Y, YANO E, MOTEGI T, KONO N, SATO E. Grain refining mechanism of high-purity Mg-9mass%Al alloy ingot and influence of Fe or Mn addition on cast grain size [J]. Journal of Japan Institute of Light Metals, 2001, 51: 403-408.
[11] PAN Y C, LIU X F, YANG H. Role of C and Fe in grain refinement of an AZ63B magnesium alloy by Al-C master alloy [J]. Journal of Materials Science and Technology, 2005, 21: 822-826.
[12] DU J, YANG J, KUWABARA M, LI W F, PENG J H. Effect of iron and/or carbon on the grain refinement of Mg-3Al alloy [J]. Materials Transactions, 2007, 48: 2903-2908.
[13] DU J, WANG M H, LI W F. Effects of Fe addition and addition sequence on carbon inoculation of Mg-3%Al alloy [J]. Journal of Alloys and Compounds, 2005, 502: 74-79.
[14] DU J, YANG J, KUWABARA M, LI W F, PENG J H. Improvement of grain refining efficiency for Mg-Al alloy modified by the combination of carbon and calcium [J]. Journal of Alloys and Compounds, 2009, 470: 134-140.
[15] DU J, YANG J, KUWABARA M, LI W F, PENG J H. Effect of strontium on the grain refining efficiency of Mg-3Al alloy refined by carbon inoculation [J]. Journal of Alloys and Compounds, 2009, 470: 228-232.
[16] BAI J, SUN Y S, XUN S, XUE F, ZHU T B. Microstructure and tensile creep behavior of Mg-4Al based magnesium alloys with alkaline-elements Sr and Ca additions [J]. Materials Science and Engineering A, 2006, 419: 181-188.
[17] ZENG X Q, WANG Y X, DING W J, LUO A A, SACHDEV A K. Effect of strontium on the microstructure, mechanical properties, and fracture behavior of AZ31 magnesium alloy [J]. Metallurgical Materials Transactions A, 2006, 37: 1333-1341.
[18] Massalski T B, Murray J L, Bennett L H, BAKER H. Binary alloy phase diagrams [M]. Metals Park, Ohio: American Society for Metals, 1986.
[19] Villers P, Calvert L D. Pearson’s handbook of crystallographic data for intermetallic phases [M]. 2nd ed. Materials Park, OH: ASM International, 1991.
[20] Du J, Wang M H, Fan T, Li W F. Influence of Fe on the formation of nucleation in carbon-inoculated Mg-3%Al alloy [J]. Advanced Materials Research, 2011, 284-286: 1588-1593.
杜 军,王海蕾,周明川,李文芳
华南理工大学 材料科学与工程学院,广州 510640
摘 要: 利用Ca和碳质孕育对Mg-3%Al合金进行复合变质处理,并研究了Fe及其Fe的添加顺序对细化效果的影响。与碳质孕育相比,Ca和碳复合孕育后的晶粒细化效果更为显著,Fe及其添加顺序对复合孕育细化效果无显著影响。Ca的添加可有效避免碳质孕育Mg-Al合金中因Fe所导致的晶粒粗化作用。在Ca和碳质复合孕育试样中可观察到大量的Al-C-O颗粒,这些颗粒实际应为Al4C3,并作为a-Mg的形核核心。然而,在熔体中添加Fe后,试样中可观察到大量在Al-Fe或Al-C-Fe表面吸附Al4C3的双相粒子,该双相粒子可作为a-Mg的形核核心并导致晶粒细化。Ca有利于该双相粒子的生存,并与工艺条件无关,从而使得Fe对晶粒细化的“毒化”作用得到有效抑制,即Ca对Fe有“免毒化”效果。
关键词: Mg-Al合金;Fe;Ca;碳质孕育;形核衬底;免毒化
(Edited by Hua YANG)
Foundation item: Project (50901034) supported by the National Natural Science Foundation of China (NSFC); Project (2010-1174) supported by Scientific Research Foundation (SRF) for the Returned Overseas Chinese Scholars (ROCS), State Education Ministry (SEM); Project (2012ZZ0005) supported by the Fundamental Research Funds for the Central Universities, South China University of Technology, China
Corresponding author: Jun DU; Tel: +86-20-87113747; E-mail: tandujun@sina.com
DOI: 10.1016/S1003-6326(13)62462-0

